Natural Product Biosynthesis
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
What is the difference between primary and secondary metabolites?
Primary: products made during exponential phase of growth, used by cells as building blocks for essential macromolecules.
Secondary: produced in nature, serves as survival functions for the organism. Have competitive advantages.
What are the advantages and disadvantages of natural products?
+ves: more complex structures, often with chirality → selectivity. Potent
-ves: complex structures can be costly to prepare on large scale. Can have harmful/toxic properties.
Give an example of how advances in biotechnology are enabling new natural products/speeding up synthesis
E.g. use of fermentation (with genetically engineered yeast) to cut down synthesis time of Artemisinin. Reduces labour required and cuts down time from over a year to a few weeks.
Draw the synthesis of SAM
What is the role of SAM? Give two examples of its use.
CH3+ source. Used in O- and N- alkylation, carbon alkylation (e.g. SEAr to beneze, or alkylation of an enolate species).
Write the short hand for acetyl CoA enzyme
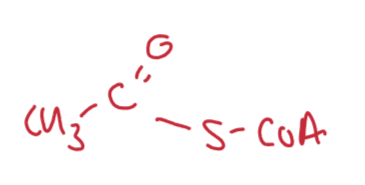
Give two examples of the type of reaction SAM can be used in?
Acts as an electrophile in acylation.
Acts as a nucleophile in enolate chemistry.
Draw the reaction to form DMAPP and IPP from acetyl CoA.
What is used as a hydride source in NPB?
NADH (oxidised to NAD+) OR NADPH (NADP+)
Give the overall name for the synthase used in fatty acid biosynthesis
Mammalian Fatty Acid Synthase
Draw the two building blocks used in fatty acid biosynthesis. Which is used to form the starter unit and which is the extender unit?
Acetyl CoA → starter unit and Malonyl CoA → Extender unit
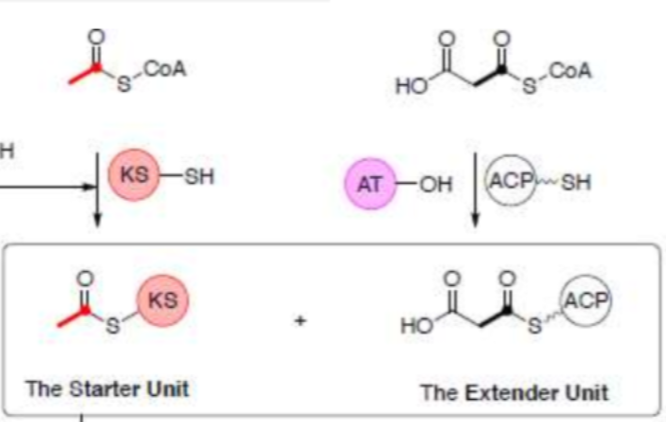
Give the roles of each of these synthase domains: KS, DH, ER, KR, AT, ACP, TE.
KS + ACP: catalyse the chain extension and the condensation reaction between the two building blocks.
AT: used to get all building blocks loaded onto the FAS and to their specific domains.
TE: releases/cleaves the acyl group using a nucleophile (e.g. water via hydrolysis).
KR: reduced ketone to OH. Requires NADPH.
DH: dehydratase: OH → alkene. -H2O
ER: reduces alkene to alkane. Requires NADPH.
What are the two ways a methyl group could be installed? How can we check which route?
Use of a different start unit e.g. methyl malonyl CoA. Would add a methyl for every extender unit if keep using the same extender unit.
SAM mediated methylation - can add a methyl group or a cyclopropane into the unit.
Checked by labelling SAM with 13C isotope.
Give the rest of the chain for a acetate labelled at each of these positions:
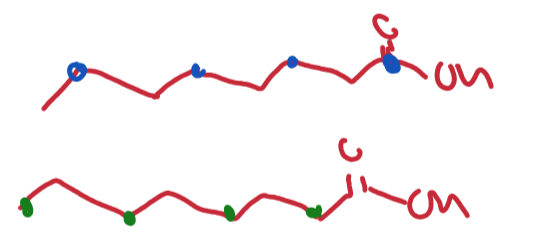
How can we diversify a polyketide from fatty acids?
Change the starter or extender units.
Remove KR, DH, ER etc.
Changing the nucleophile at termination.
Addition of a side chain methyl group.
Use of cyclase enzyme (cyclise) followed by KR and an aromatase to form aromatic ring systems.
Define an alkaloid
Any non-peptidic and non-nucleotide nitrogenous secondary metabolite. Can be classified by those derived from: lysine or ornithine, phenylalanine or tyrosine, or tryptophane.
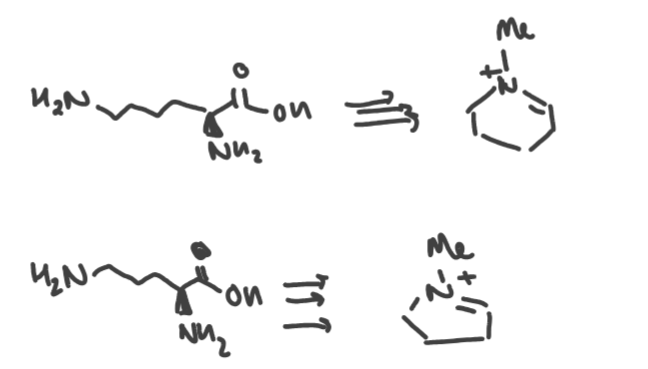
What are lysine and ornithine precursors to? Draw the structure
Lysine → 6 membered piperidine alkaloids.
Ornithine → 5 membered pyrrolidine alkaloids.
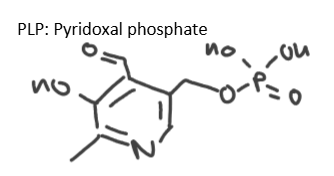
Draw PLP and describe what PLP dependent enzymes are used for.
PLP dependent enzymes can be used for: decarboxylation of amino acids, transamination, racemisation (L→D amino acids). Uses PLP =cofactor that sites in active site for enzyme to function.
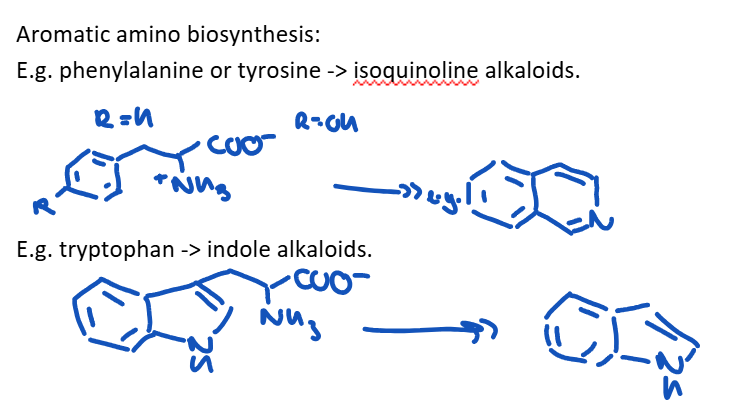
What is phenylalanine and tyrosine , and tryptophan precursors to?
Define the shikimate pathway. Name the two key intermediates
The biosynthetic pathway by which the aromatic acid phenylalanine, tyrosine and tryptophane are produced.
Key intermediates: shikimate and chorismate.
How are terpenes classified?
Terpenes are classified by the number of carbons in their skeletons:
10 carbons = monoterpenes e.g. carvone.
15 carbons = sesquiterpene.
20 carbons = diterpenes.
30 carbons = triterpene.
What is the use of IPP and DMAPP? Which is the electrophile and which is the nucleophile?
Used to build up terpenes. Can combine to form GPP, where DMAPP acts as an electrophile, losing OPP. Then attacked by IPP alkene (nucleophile)
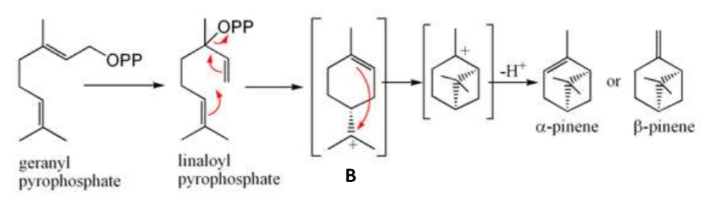
Draw the reaction to form alpha and beta pinene from GPP.