BC3- lipids, biological membranes
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
lipids
-highly reduced carbon-rich substances including the fats, oils, sterols that are soluble in nonpolar organic solvents (e.g. ether, benzene)
-hydrophobic (nonpolar) or amphipathic (containing both nonpolar and polar regions)
-most of the lipids in food are in the form of triacylglycerols, cholesterol, and phospholipids
common types of lipids: fatty-acids
-Fatty acids are carboxylic acids with a long hydrocarbon chain; they are key constituents of lipids
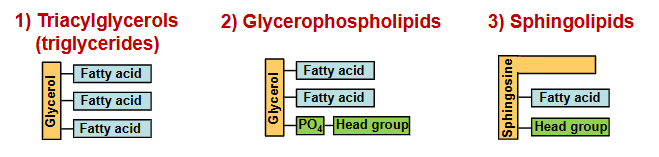
fatty acid-containing lipids: triacylglycerols, glycerophospholipids, sphingolipids
common types of lipids: non-fatty acid-containing lipids
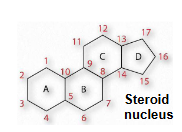
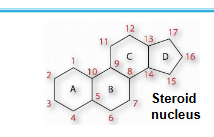
sterols: have four fused rings (steroid nucleus) and a hydroxyl group
lipid functions
Energy storage (e.g. triacylglycerol)
Membrane structure (e.g. glycerophospholipids, sphingolipids, sterols)
Electron carriers (e.g. ubiquinone’s)
Emulsifying agents (e.g. bile salts)
Hormones (e.g. steroid hormones)
fatty acids
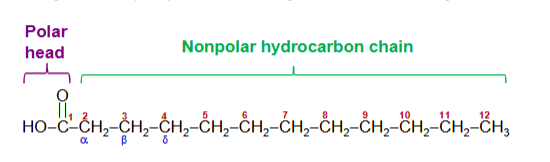
carboxylic acids with hydrocarbon chains
Standard nomenclature assigns the number 1 to the carboxyl carbon
Carbon atoms 2 and 3 are also referred to as α and β, respectively
saturated fatty acid
no C-C double bonds
unsaturated fatty acid
at least one C-C double bond
monounsaturated fatty acid
only one C-C double bond
polyunsaturated fatty acid
two or more C-C double bonds
fatty acids: common name
oleic acid
fatty acids: nomenclature
specifies the chain length (counting starts from the carboxyl carbon) and number of double bonds
fatty acids: position of double bonds indicated by (nomenclature)
delta(n)
n indicates lower numbered carbon of each pair
fatty acid nomenclature: total # C:
# double bonds, delta double bond positions
a 16-carbon saturated fatty acid is abbreviated as
16:0
an 18-carbon fatty acid with one double bond between C-9 and C-10 is designated
18:1^delta9
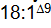
polyunsaturated fatty acids can be numbered from the
methyl carbon (carbon most distant from the carboxyl group) which is called the omega carbon
positions of the double bonds are indicated relative to the omega carbon
polyunsaturated fatty acids with a double bond between C3 and C-4 are called
omega-3 fatty acids
omega-3 fatty acids
essential nutrients
humans cannot synthesize them
include alpha-linolenic acid (ALA), docosahexaenoic acid (DHA), and eicosatetraenoic acid (EPA)
essential fatty acids
cannot be synthesized by the body, must be obtained from diet
melting points of fatty acids
determined by the length and degree of unsaturation of the hydrocarbon chain
the longer the fatty acid chain, the higher the melting point
the fewer the double bonds in a fatty acid, the higher the melting point
the effect of the double bond on the physical properties of the fatty acid is due to the
conformation of the lipid that is caused by the double bond
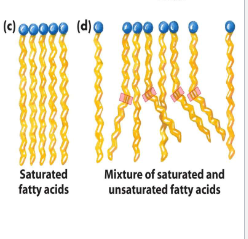
in saturated fatty acids, the most stable arrangement is
very close packing of the side chains of the lipids
lipids assume an almost crystalline array
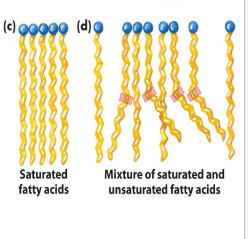
can tight packing of fatty acid chains take place?
no, due to the kink that results from cis double bonds
since the interactions between these arrays are less extensive, it takes less energy to disrupt them, resulting in a lower melting point
triacylglycerols
contain 3 fatty acid molecules esterified to the three hydroxyl groups of glycerol
primary storage form of body fat, many fatty acids in biological systems exist this way
very nonpolar, insoluble in water (since carboxylates are esterified and no longer bear a negative charge)
lipases
catalyze the hydrolysis of the ester bond of triacylglycerols in order for the fatty acid to be used for fuel and other purposes
oils
triacylglycerols rich in unsaturated fatty acids generally liquid at room temperature
fats
triacylglycerols rich in saturated fatty acids, generally semisolids or solids at room temperature
two major classifications of lipids
storage lipids (triacylglycerols)
structural lipids providing structure for membranes
storage lipids (triacylglycerols)
used primarily to store energy, neutral and nonpolar
structural lipids
contain both nonpolar and polar groups, provide structure for membranes
two major types of structural lipids
glycerophospholipids and sphingolipids
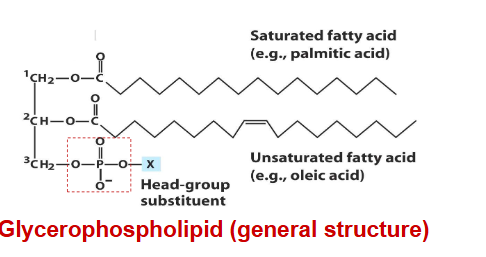
glycerophospholipids
primary constituents of cell membranes
differ from triacylglycerols in that only two of the hydroxyl groups of glycerol are esterified to fatty acids.
third hydroxyl group contains a phosphate group that is connected to a given “head group” via a phosphodiester bond
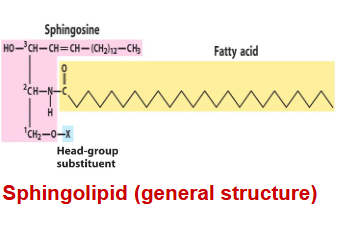
sphingolipids
The most distinct difference between sphingolipids and glycerophospholipids is that the backbone is sphingosine, NOT glycerol
contain sphingosine, which is a long-chain amino alcohol. A fatty acid is joined to sphingosine via an amide linkage rather than an ester linkage as seen in glycerol
sterols
contain the steroid nucleus and a hydroxyl group
steroid nucleus: 17-C four-fused ring structure, three of the rings are six-membered, and one is 5-membered
sterols can be present in the membranes of most eukaryotes (like cholesterol)
fatty acids: summary
[R-COOH (R=hydrocarbon chain)] are components of
triacylglycerols, glycerophospholipids, sphingolipids
Unsaturated fatty acids have lower melting points than their saturated
counterparts; the greater the degree of unsaturation, the lower the
melting point
Fatty acids are important metabolic fuels
Fatty acids are stored as neutral lipids called triaclyglycerols
triacylglycerols: summary
are composed of 3 fatty acyl residues esterified to a glycerol
Glycerophospholipids and sphingolipids are the major structural components of membranes
Sterols contain four fused rings (steroid nucleus) and a hydroxyl group e.g. cholesterol
lipid bilayers
structural basis for all biological membranes
noncovalent interactions among lipids make them flexible and self-sealing
polar head groups contain aqueous medium
nonpolar tails point to interior

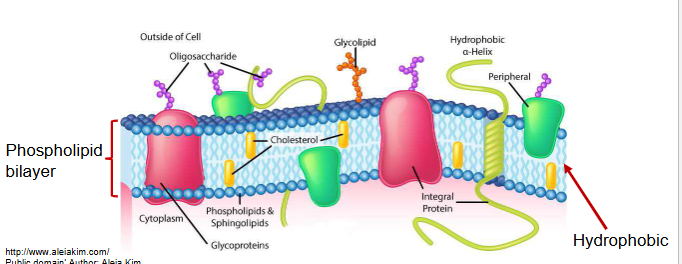
biological membranes
define the external boundaries of cells and separate cell compartments
consist of proteins embedded in or associated with a lipid bilayer
functions of membranes
selective import and export of waste/nutrients
retain metabolites and ions in the cell
sense signals, transmit info to the cell
provide compartmentalization
separate energy-producing reactions from energy-consuming ones
keep proteolytic enzymes away from important cell proteins
store energy as a proton gradient
support ATP synthesis
integral membrane proteins
also called trans-membrane proteins, contain hydrophobic regions embedded in the hydrophobic lipid bilayer
penetrate or span the bilayer
mediate movement of ions and polar molecules across membrane
can generate proton gradients for ATP production
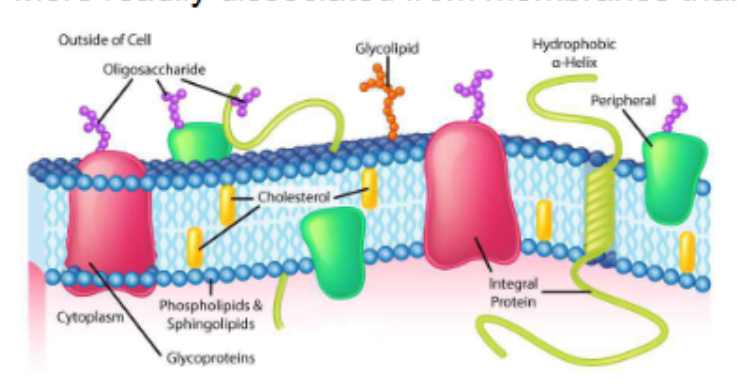
peripheral membrane proteins
associated with membrane through charge-charge or hydrogen bonding interactions to integral proteins or membrane lipids
more readily dissociated from membranes than integral proteins
proteins on peripheral membrane proteins respond to extracellular signals and communicate them to the cell interior (e.g. insulin receptor)
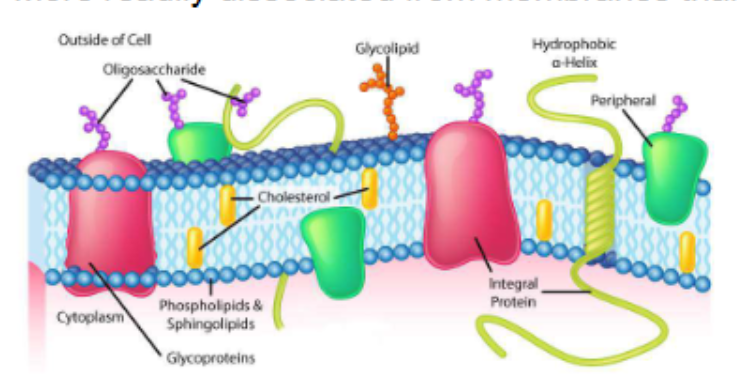
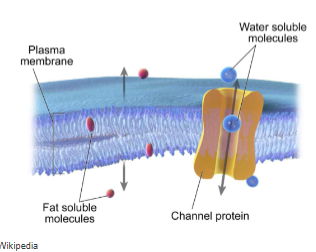
Hydrophobic (fat soluble) molecules can pass through a membrane’s hydrophobic interior by
simple diffusion down their concentration gradient
e.g. the diffusion of O2 and CO2 across membranes
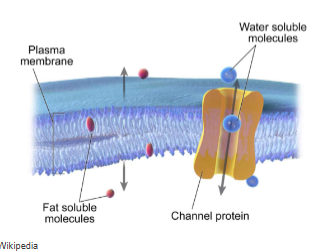
cell membranes are impermeable to ions and large polar molecules. how do they move across the membrane?
require membrane transporter proteins to form passages through the hydrophobic barrier
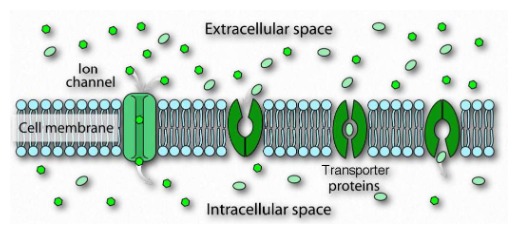
three types of integral membrane transporter proteins
channels
passive transporters
active transporters
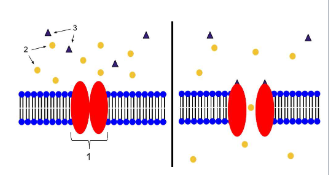
channels
transmembrane proteins with aqueous pores that extend across membrane
when open, pores allow inorganic ions of appropriate size and charge to pass
ions move down concentration gradient
requires no energy
much faster than transport mediated by transporters
usually not saturable
passive transporters
transmembrane proteins
bind the specific solute to be transported and undergo a series of conformational changes to transfer the solute
solute moves down its concentration gradient
requires no energy
carrier proteins are saturable with solute
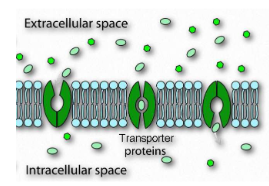
passive transport (facilitated diffusion) overview
• Passive transport (facilitated diffusion) does not require an energy source
• The solute is to be transported move down its concentration gradient across the membrane
• The solute is moved through a specific channel
active transport
requires energy, moves a solute up its concentration gradient (low concentration to high concentration)
can be powered by a direct source of energy as ATP, electron transport, or light
saturable with solute
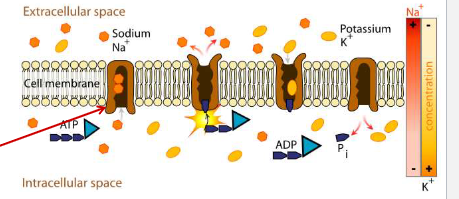
active transport example
The cytochrome b6f complex involved in photosynthesis uses light energy to move H+ across the thylakoid membrane against their concentration gradient
simple diffusion: protein carrier? saturable with solute? movement relative to concentration gradient? energy input required?
protein carrier: no
saturable with solute: no
movement relative to concentration gradient: down
energy input required: no
facilitated diffusion: channel: protein carrier? saturable with solute? movement relative to concentration gradient? energy input required?
protein carrier: channel
saturable with solute: no
movement relative to concentration gradient: down
energy input required: no
facilitated diffusion: passive transporter: protein carrier? saturable with solute? movement relative to concentration gradient? energy input required?
protein carrier: passive transporter
saturable with solute: yes
movement relative to concentration gradient: down
energy input required: no
active transport: protein carrier? saturable with solute? movement relative to concentration gradient? energy input required?
protein carrier: active transporter
saturable with solute: yes
movement relative to concentration gradient: up
energy input required: yes
summary: biological membranes
Lipid bilayers are the structural basis for all biological membranes
Membrane
1) Lipids
2) Proteins
3) Carbohydrates
Membrane proteins
1) Integral membrane proteins
2) Peripheral membrane proteins
summary: biological membranes
Membrane protein functions:
1) transport molecules across membrane (channels, transporters)
2) receive and transduce extracellular signals (receptors)
In passive transport, solutes move across membranes down their concentration gradient
In active transport, solutes move across membranes against their concentration gradient by using energy
Membrane receptors