Organic Chemistry Hydrocarbons, Alcohols, and Functional Groups Practice
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
What is a hydrocarbon made up of?
A hydrocarbon is made up of hydrogen and carbon atoms.
Give an example of a hydrocarbon used in everyday life.
An example of a hydrocarbon used in everyday life is gasoline, which primarily consists of octane.
What do you understand by a saturated hydrocarbon?
A saturated hydrocarbon is a hydrocarbon that contains only single bonds between carbon atoms, meaning it cannot add more hydrogen atoms.
How is a saturated hydrocarbon different from an unsaturated hydrocarbon?
An unsaturated hydrocarbon contains at least one double or triple bond between carbon atoms, allowing it to bond with additional hydrogen atoms.
Gasoline is mostly __________.
Gasoline is mostly octane.
Where do hydrocarbons come from?
Hydrocarbons primarily come from fossil fuels, which are formed from the remains of ancient plants and animals buried under layers of sediment over millions of years.
What is the general formula for alkanes?
The general formula for alkanes is CnH2n+2.
What is an example of an alkane?
An example of an alkane is propane (C3H8), which is saturated and contains only single bonds.
What is the general formula for alkenes?
The general formula for alkenes is CnH2n.
What is an example of an alkene?
An example of an alkene is ethylene (C2H4), which contains a double bond.
What is the general formula for alkynes?
The general formula for alkynes is CnH2n-2.
What is an example of an alkyne?
An example of an alkyne is acetylene (C2H2), which contains a triple bond.
What is hydrogenation of an alkene?
Hydrogenation of an alkene is a chemical reaction where hydrogen is added to the alkene, converting it into an alkane.
Give an example of a hydrogenation reaction of any alkene.
An example of a hydrogenation reaction is the conversion of propene (C3H6) to propane (C3H8) using hydrogen gas.
What is the IUPAC name of the continuous chain alkane with six carbon atoms?
The IUPAC name of the continuous chain alkane with six carbon atoms is hexane.
What is the IUPAC name for a one-carbon alkyl group?
The IUPAC name for a one-carbon alkyl group is methyl.
What is the IUPAC name for a two-carbon alkyl group?
The IUPAC name for a two-carbon alkyl group is ethyl.
What is the IUPAC name for a three-carbon alkyl group?
The IUPAC name for a three-carbon alkyl group is propyl.
What is the IUPAC name for the compound CH3-CH2-CH2-CH2-CH3?
The IUPAC name for the compound CH3-CH2-CH2-CH2-CH3 is pentane.
What is the IUPAC name for ethylene?
The IUPAC name for ethylene is ethene.
What is the IUPAC name for 1-butyne?
The IUPAC name for 1-butyne is butyne, indicating it has a triple bond.
What is a cycloalkene?
A cycloalkene is a cyclic hydrocarbon that contains at least one double bond.
What is the IUPAC name for cyclopropane?
The IUPAC name for cyclopropane is cyclopropane, which is a three-carbon ring with single bonds.
What is the IUPAC name for cyclohexane?
The IUPAC name for cyclohexane is cyclohexane, which is a six-carbon ring with single bonds.
What is the IUPAC name for cyclohexene?
The IUPAC name for cyclohexene is cyclohexene, which is a six-carbon ring with one double bond.
What is the IUPAC name for cycloheptyne?
The IUPAC name for cycloheptyne is cycloheptyne, which is a seven-carbon ring with a triple bond.
What is the IUPAC name for cyclooctane?
The IUPAC name for cyclooctane is cyclooctane, which is an eight-carbon ring with single bonds.
What is an alcohol?
An organic compound containing a hydroxyl (-OH) functional group.
What is the compound CH3 - CH2 - SH known as?
It is known as a thiol.
What functional group do alcohols contain?
Hydroxyl group.
What is the IUPAC name for 3-methyl-1-hexanol?
3-methyl-1-hexanol.
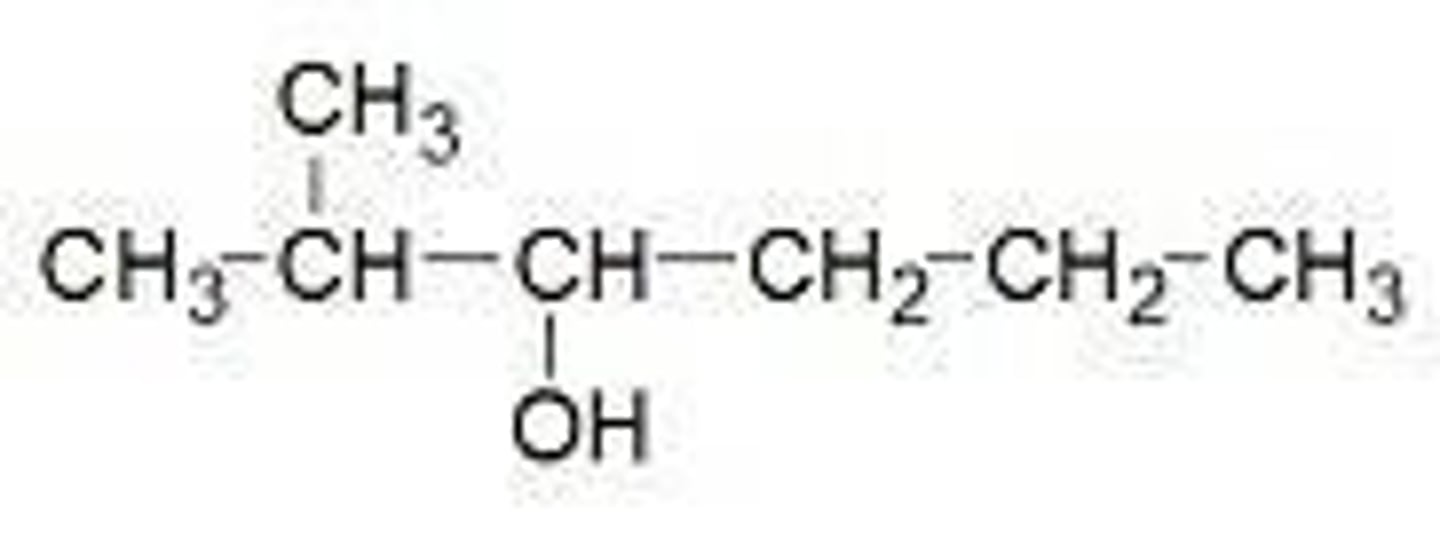
What is the IUPAC name for the compound with the structure CH3 - CH2 - O - CH2 - CH3?
It is known as 5-methyl-4-hexanol.
What is the line angle structure of phenol?
Phenol consists of a benzene ring with a hydroxyl group (-OH) attached.
What is the IUPAC name for the compound CH3 - C(OH)(CH3) - CH3?
2-methyl-2-propanol.
Which compound is a thiol?
CH3 - SH is a thiol.
What is the name for cyclopentanol?
Cyclopentanol.
What do thiols contain in place of oxygen?
They contain sulfur in place of oxygen in the functional group.
What is methanol commonly used for?
Methanol is used in making plastics, racing fuel, solvents, and paint remover.
What is 1,2-ethanediol (ethylene glycol) used for?
It is used as antifreeze, solvent for paint, and in the production of synthetic fibers.
What kind of odors do thiols produce?
Thiols produce skunky or bad-smelling odors.
What is the reason diethyl ether has been replaced as a general anesthetic?
It is very volatile and highly flammable.
Which alcohol is most soluble in water?
Ethanol is the most soluble alcohol in water.
What is a secondary alcohol?
A secondary alcohol has a hydroxyl group bonded to a carbon that is bonded to two alkyl groups.
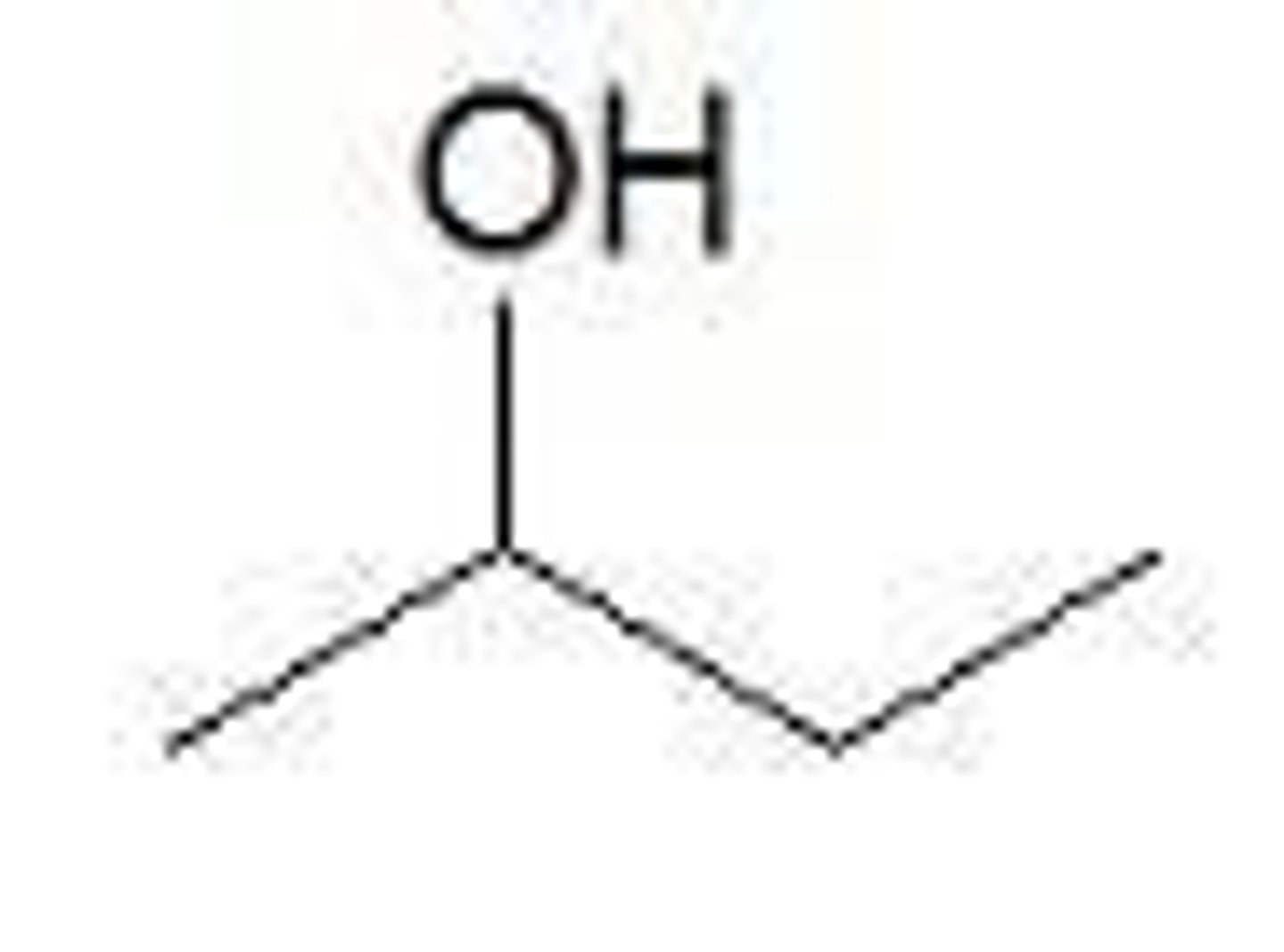
What is a primary alcohol?
A primary alcohol has a hydroxyl group bonded to a carbon that is bonded to one alkyl group.
What is a tertiary alcohol?
A tertiary alcohol has a hydroxyl group bonded to a carbon that is bonded to three alkyl groups.
What type of bonds do alcohols form between individual molecules?
Alcohols form hydrogen bonds between individual molecules.
What is produced when an alcohol undergoes dehydration in the presence of a strong acid?
An alkene is produced.
What is the dehydration product of CH3 - CH2 - CH2 - OH?
The product is propene.
What happens during the oxidation of an alcohol to a ketone?
There is a gain of oxygen.
What type of alcohol can be oxidized to yield a ketone?
Secondary alcohols can be oxidized to yield a ketone.
Why can't tertiary alcohols be oxidized?
There are no hydrogen atoms attached to the alcohol carbon.
What is the product when an alcohol is oxidized?
The product can be a ketone or an aldehyde, depending on the structure.
What is produced in addition to an alkene during the dehydration of an alcohol?
Water is produced in addition to the alkene.
What happens to methanol in the liver?
Methanol is oxidized by the liver to carboxylic acids.
What can thiols be oxidized to?
Thiols can be oxidized to disulfides.