isomerism and E/Z isomerism
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
what is geometric isomerism?
compounds/molecules w/ the same structural formula
but atoms/bonds/groups arranged differently in space
what is stereoisomerism? give 2 examples:
compounds/molecules w/ the same molecular and same structural formula but a different arrangement of atoms in space:
geometric isomerism
optical isomerism
what are structural isomers? give 3 examples:
compounds/molecules with the same molecular formula but a different structural formula:
chain isomerism
position isomerism
functional group isomerism
what are the 3 types of structural isomerism?
chain isomer
position isomer
functional group isomer
whata chain isomer?
compounds/molecules w/ the same functional group and molecular formula but a different arrangement of the carbon chain
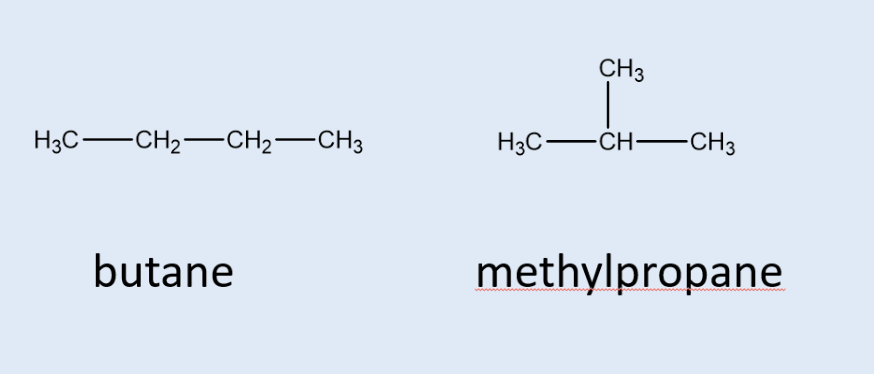
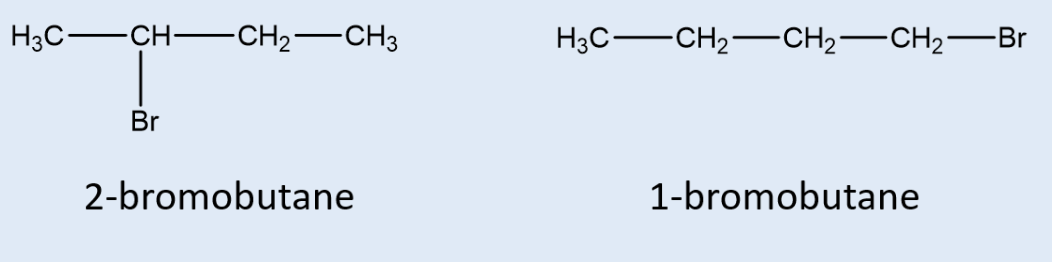
what is a position isomer?
compounds/molecules w/ the same molecular formula and carbon chain but w/ the functional group in a different place
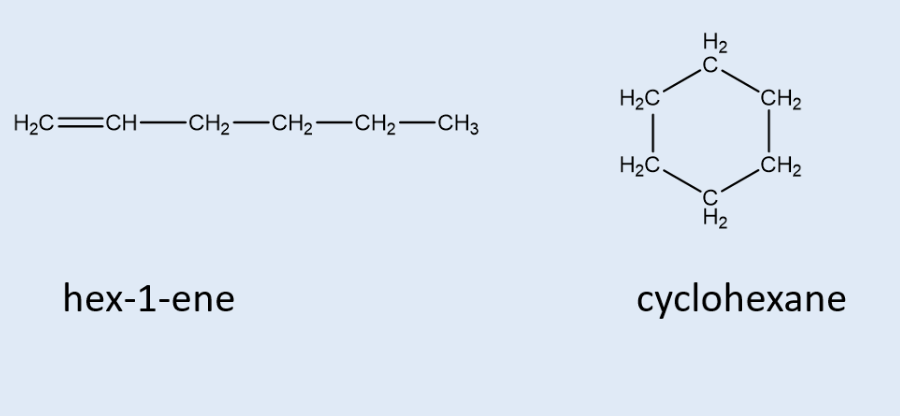
what are functional group isomers?
compounds/molecules w/ the same no. of atoms but arranged to form a diff functional group
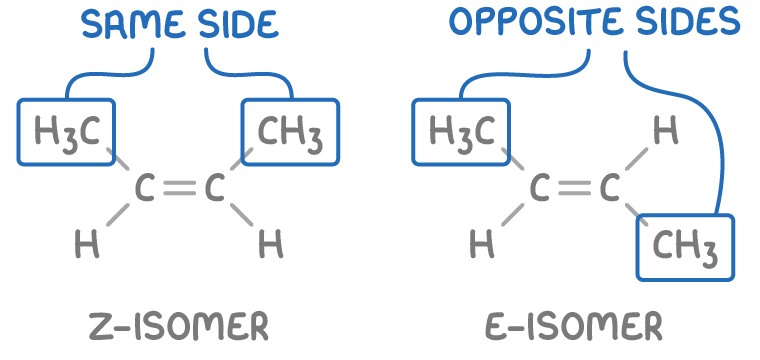
why does E-Z isomerism occur?
restricted rotation around the C=C double bond
give the 2 types of E/Z isomers:
Z isomer (zusammen) = higher priority groups are on the same side of the C=C double bond
E isomer (entgegen) = higher priority groups are on opposite sides of the C=C double bond
summarise the CIP priority rules:
C w/ the groups w/ the highest atomic number = highest priority
