IMED2001 - Introduction to Viruses and Virology (L10)
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
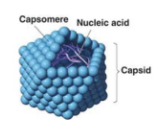
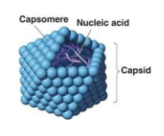
Viruses can be defined by 4 traits
- Viruses are obligate intracellular parasites that have an infectious extracellular stage. Most virologists refer to this extracellular stage as a virion
- All viruses encode at least one capsomere protein. Capsomeres cover and protect the nucleic acid in a virion
- All viruses replicate, not by growing larger and dividing as do cells, but by assembly. That is, cells infected by viruses synthesise the component parts of the virion, and then the parts, once synthesised, spontaneously assemble into new virions
- Viruses have the capacity to evolve. Because the minimum composition of viruses is nucleic acids and protein units called capsomeres, viral populations can change through typical evolutionary processes that alter those nucleic acids in a heritable way
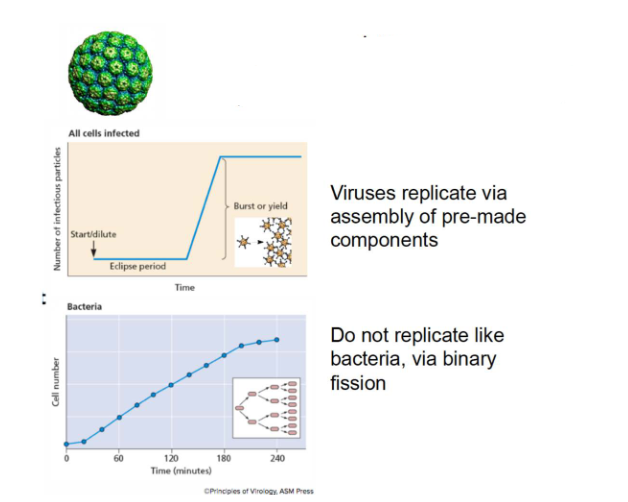
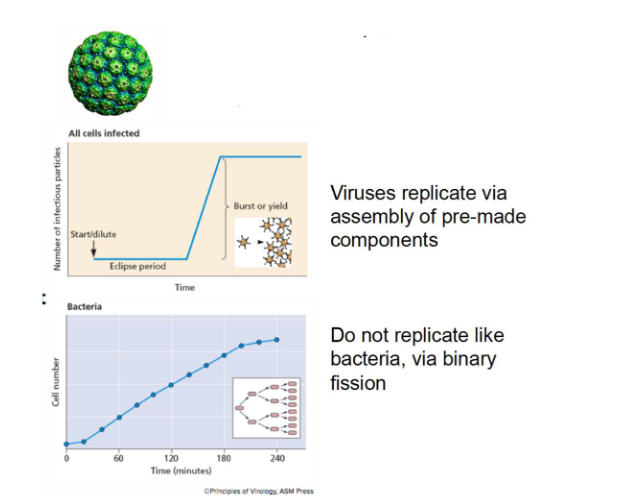
Viruses are obligate intracellular parasites
- Require host cells to multiply. No machinery for protein synthesis and energy production
- Viruses replicate via assembly of pre-made components
- Do not replicate like bacteria, via binary fission (bacteria do binary fission, viruses do not)

Viruses are infectious particles minimally made up of nucleic acids and proteins
- Virions are made up of nucleic acids protected by capsomeres. The protien coat of capsomeres that surrounds the nucleic acids is called a capsid
- Virions can be separated into two classes based on whether they have an external layer consisting of a proteinaceous lipid bilayer. Enveloped virions (D) actually have a lipid bilayer, whereas naked virions (A) do not
- capsids may be spherical (A, E) or helical (F). Spherical capsids are actually icosahedrons, which approximate the volume of a sphere yet are constructed from repeated subunits. Spherical capsids are somewhat rigid, whereas helical capsids can be rigid or flexible, depending on the virus
- Some bacteriophages (C) combine icosahedral and helical elements, so that their heads, full of nucleic acids, are icosahedral but their tails are helical assemblages of specialised tail proteins
- In some viruses, the nucleic acid genome and the capsomeres are so intimately associated that their structure is termed a nucleocapsid. Shown here is the SARS-CoV-2 nucleocapsid (N protein)

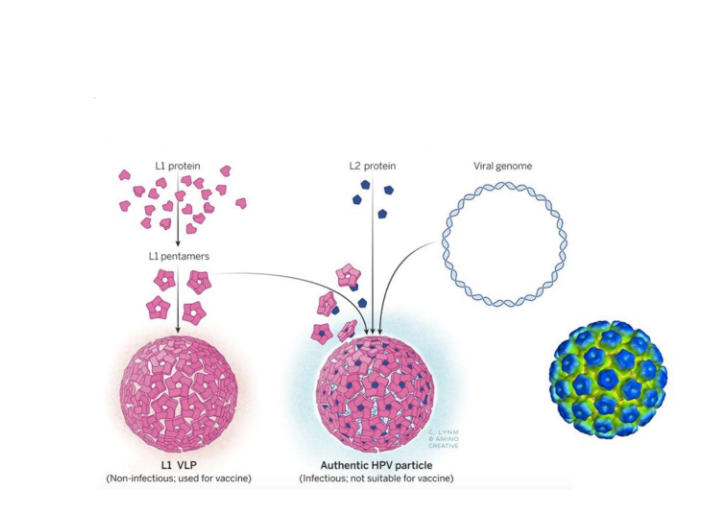
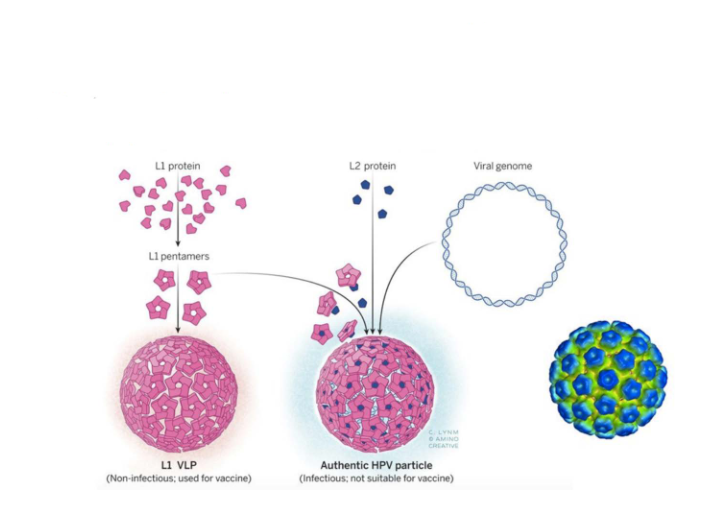
Many capsid proteins can assemble into 'virus-like particles'
- This strategy is used to produce vaccine against human papilloma virus (HPV)
.
- basically we just make a copy of the virus particle but non-pathogenic
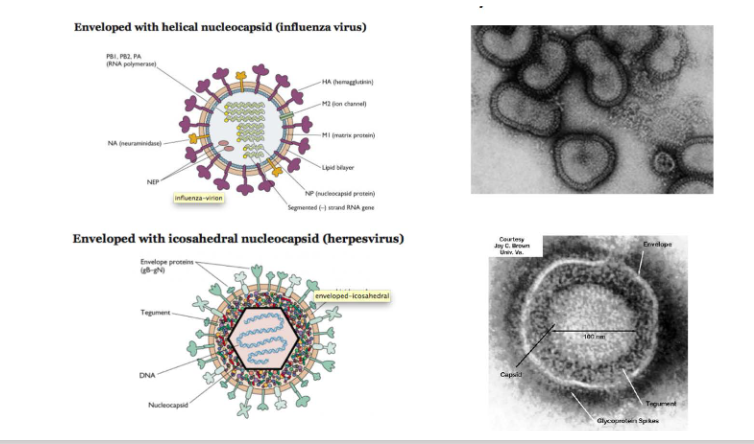
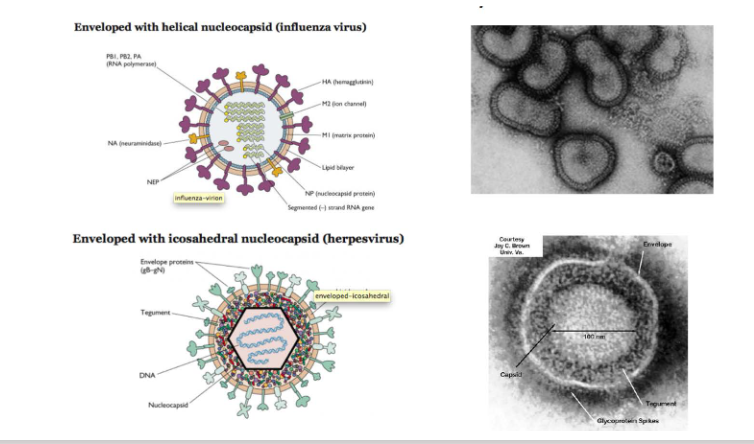
Enveloped viruses may have helical or icosahderal nucleocapsids
DIAGRAM ON SLIDE 10
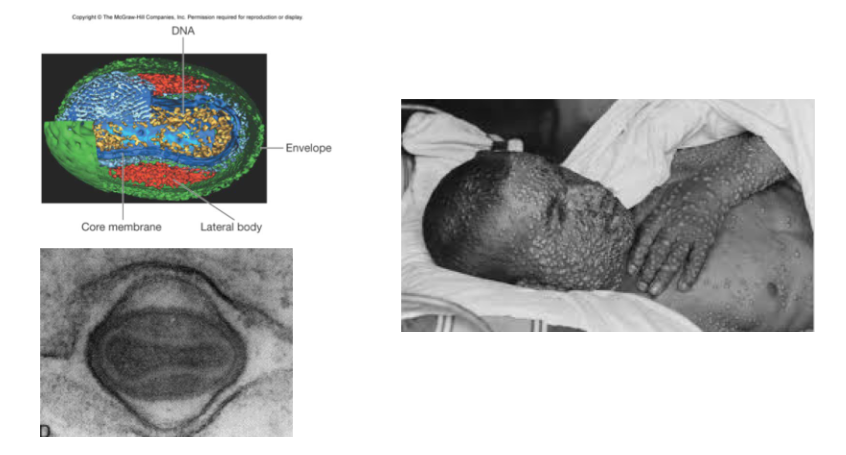
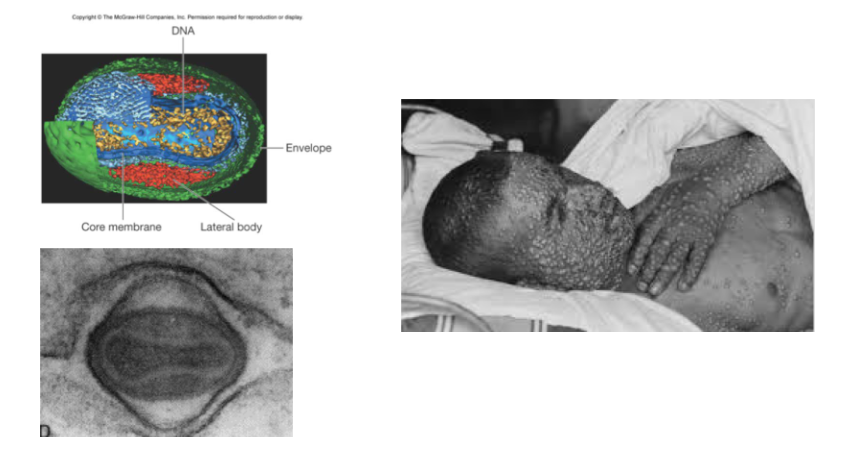
Some viruses lack clearly defined capsids e.g poxviruses
DIAGRAM ON SLIDE 11
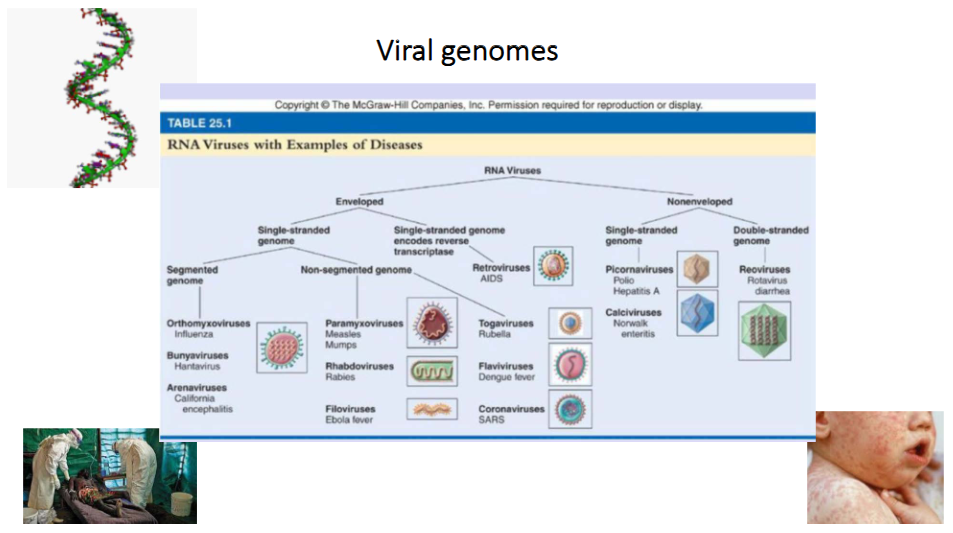
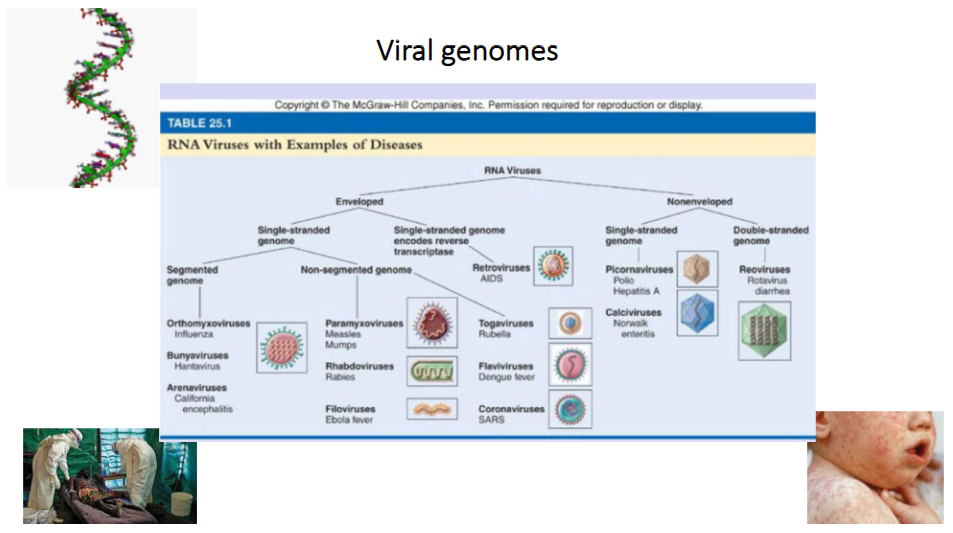
Viral genomes of RNA Viruses
- RNA Viruses can be enveloped or non-enveloped
- each category can have single stranded or double stranded genome
- in single stranded genome of enveloped virus we can have segmented or non-segmented genome
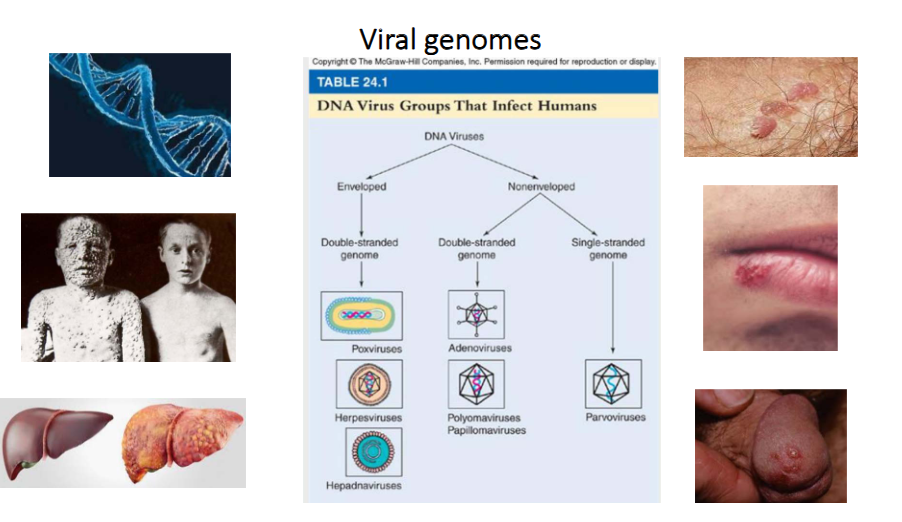
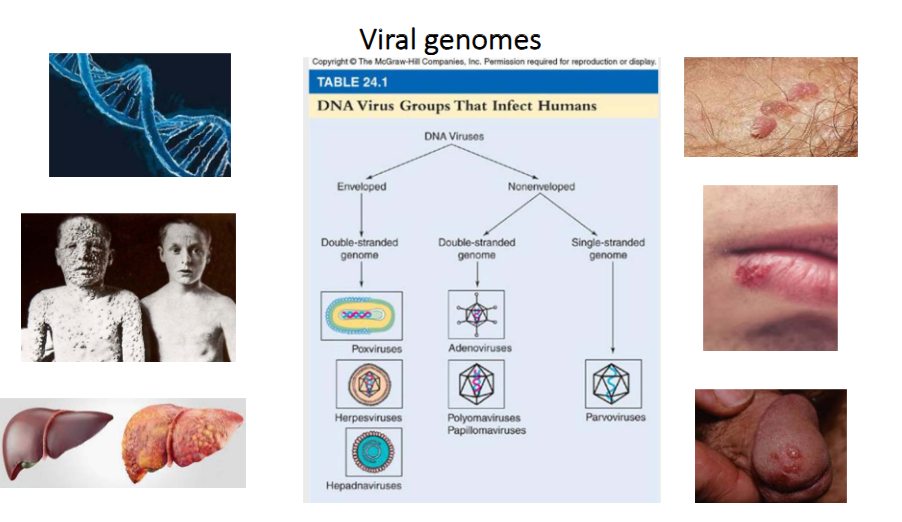
Viral Genomes of DNA Viruses
- DNA Viruses can be enveloped or non-enveloped
- enveloped virus can be double-strnaded genome
- non-enveloped can be double stranded genome or single stranded genome
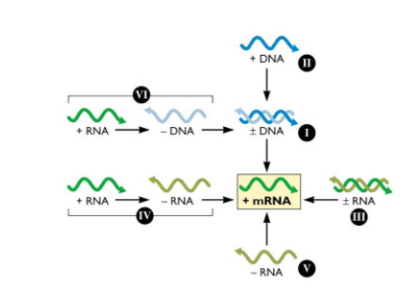
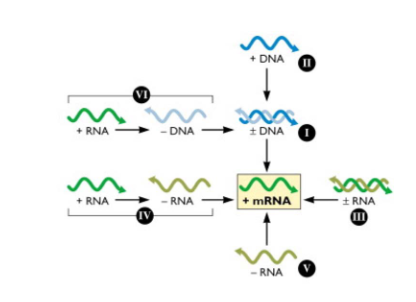
Viruses can be classified according to the ways they synthesise and use mRNA - the Baltimore Classification
- cellular genes are encoded in dsDNA from which mRNAs are produced to direct the synthesis of protein
- Francis Crick conceptualised this flow of information as the central dogma of molecular biology: DNA -> RNA -> protein
- All viruses must direct the synthesis of mRNA to produce proteins. No viral genome encodes a complete system for translating proteins
- therefore all viral protein synthesis is completely depednent upon the translational machinery of the cell
- Baltimore classification scheme is based on the central role of the translational machinery and importance of viral mRNAs in programming viral protein synthesis
- In this scheme, mRNA is in the centre, with pathwyays to mRNA from DNA or RNA genomes
- This arrangement highlights the obligatory relationship between the viral genome and its mRNA
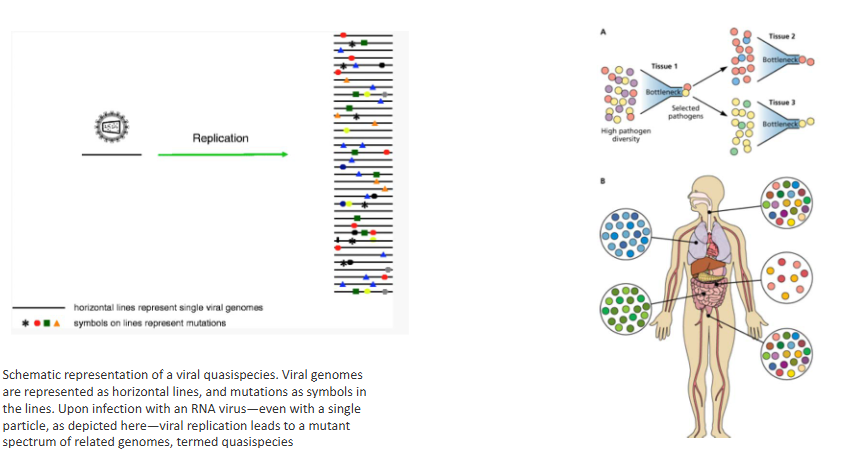
RNA viruses may exist as quasispecies
- Viral genomes are represented as horizontal lines, and mutations as symbols in the lines
- upon infection with an RNA virus, even with a single particle, as depicted, viral replication leads to a mutant spectrum of related genomes, termed quasispecies
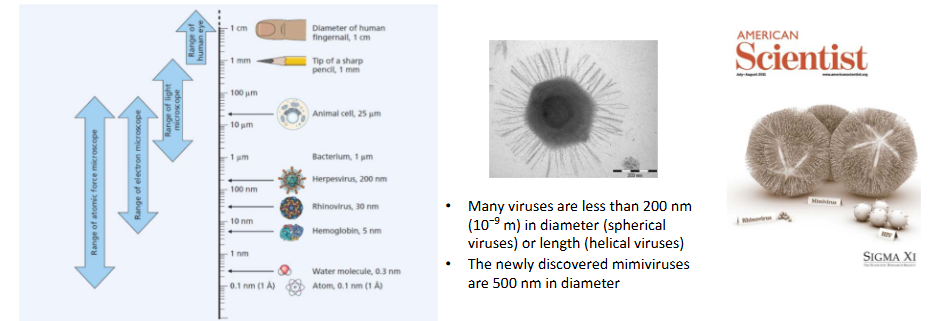
Viruses are too small to be seen with the naked eye
- Viruses are too small to be seen by the human eye, and most are too small to be seen by light microscopy
- There are exceptions to these rules (e.g mimiviruses)
- nevertheless, light microscopy is an important tool in virology because it can be used to visualise the effects of virus infection on host cells
.
- many viruses are less then 200nm in diameter (spherical viruses) or length (helical viruses)
- the newly discovered mimiviruses are 500nm in diameter
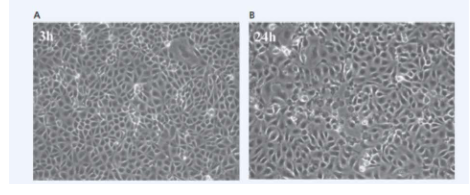
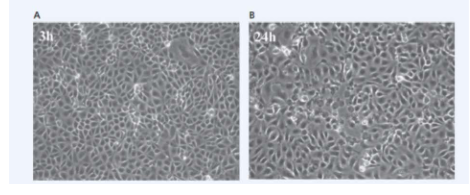
Virus infected cells may display cytopathic effects
- Animal cells in culture typically form confluent carpets in which cells introduced into a tissue culture flask attach to and spread out on the specially prepared plastic. As the cells undergo mitosis, the cell population increases in number until all the cells are touching one another without overlapping (A); at this point, contact inhibition (touching other cells on all sides) prevents further population increase
- When viruses infect these cells, the cells typically display a variety of cytopathic effects that depend on the specific host cell and virus. For example, many viruses cause infected cells to round up and detach from the tissue flask as they die (B)
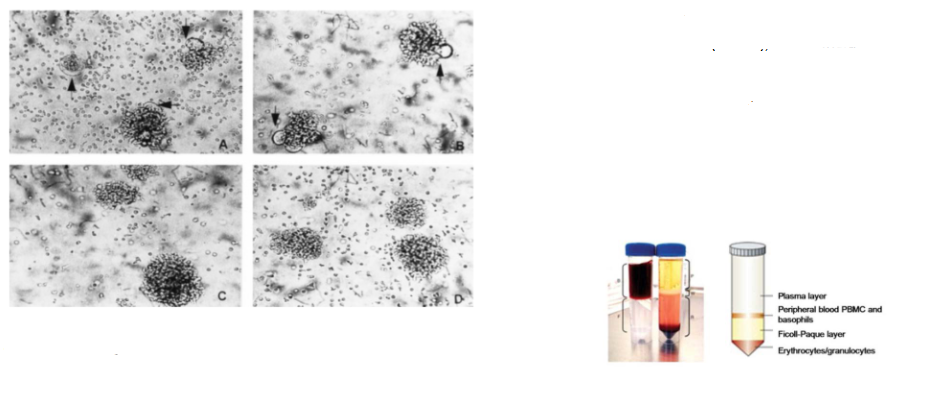
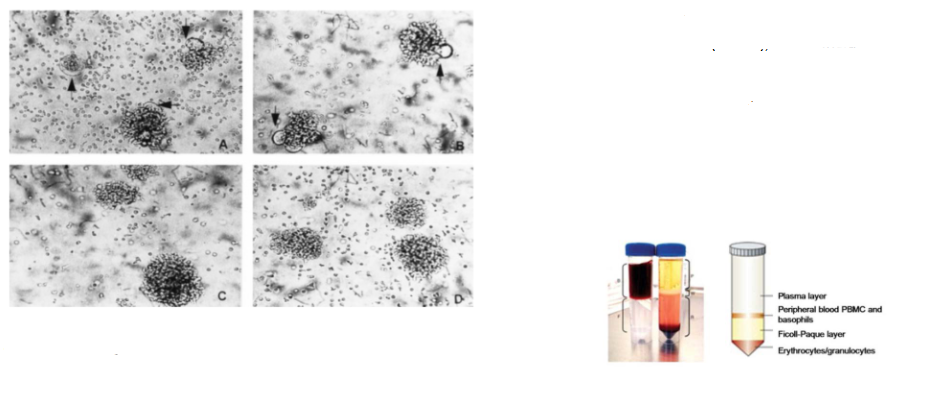
Capacity to induce cytopathic effect is virus strain-specific
- Syncytium formation in HIV-1-infected peripheral blood mononuclear cells (PBMC), which include CD4+ T cells
- Panels A and B: PBMC infected with HIV-1 strains SF2 and LAV display syncytia after 4 days of culture
- Panels C and D: PBMC infected with HIV-1 strains JC and MN show no syncytium formation up to 14 days post-infection
- PBMC are isolated from whole blood by density gradient centrifugation
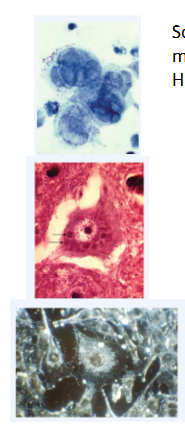
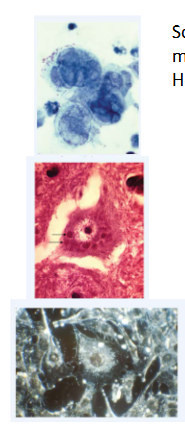
Virus-induced cytopathic effects (CPE)
- Some viruses cause the cells to form syncytia, which are large, multinucleate cells
- Herpes simplex virus-induced syncytia are shown here
.
- Other viruses cause abnormal internal structures visible when the host cells are stained; such inclusion bodies can form in the cytoplasm or nucleus, depending on the virus
- In most cases that have been investigated more closely, these inclusions turn out to be the sites of viral gene expression, genome replication, assembly or some combination of these
- Rabies virus inclusion bodies are visible in the cytoplasm; 2 are pointed out by arrows
- Transformed cancerous cells grow on top of eachother instead of responding to contact inhibitions as normal cells would. In the bottom image, the cells around the central one are piled on top of one another

Virus classification - Taxonomic categories
VIRUS NAMING SCHEMES ARE NOT SYSTEMATIC AND CAN SEEM WHIMSICAL. VIRUSES CAN BE NAMED FOR:
- the diseases they cause. Examples include poliovirus and influenza virus.
- symptoms of the diseases they cause, such as Crimean-Congo hemorrhagic fever virus.
- the parts of the human body they infect, such as the common cold virus rhinovirus (rhino means nose in Greek) or hepatitis (from the Greek word for liver).
- the geographical location where they first emerged, such as Marburg virus and Ebola virus.
- the properties of their virions. For example, the picornaviruses are tiny (pico) and have RNA genomes, whereas the geminiviruses have twin capsids (gemini is Latin for twins). Coronaviruses seem to have 'crowns' in EM images.
- Obvious evolutionary relatedness does exist among some viruses, so they may be classified by order (-virales), family (-viridae), subfamily (-virinae), genus (-virus) and species. Some viruses also have subspecies (see Table).

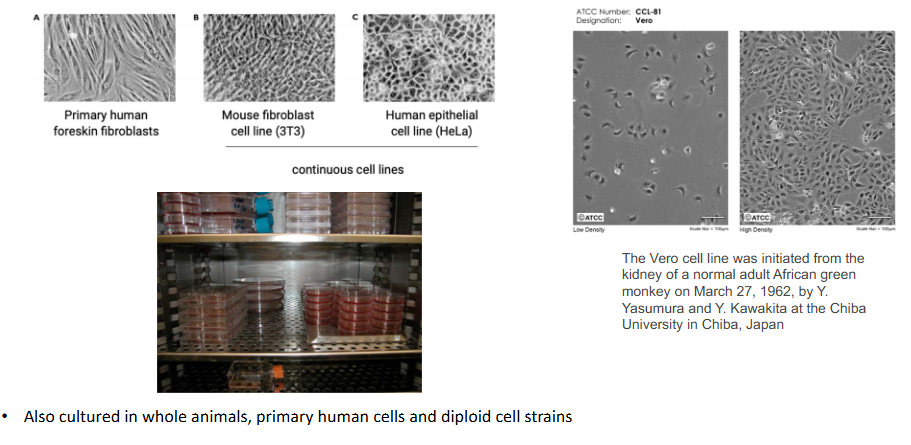
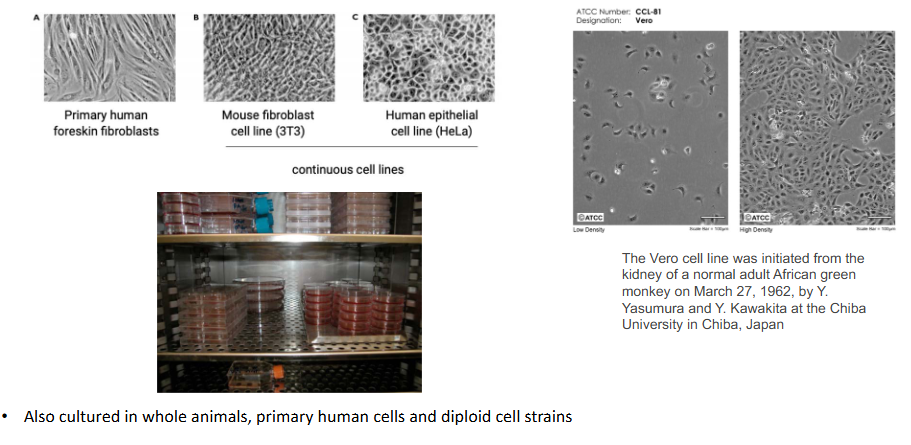
Viruses are cultured in animal and human cells
- also cultured in whole animals, primary human cells and diploid cell strains
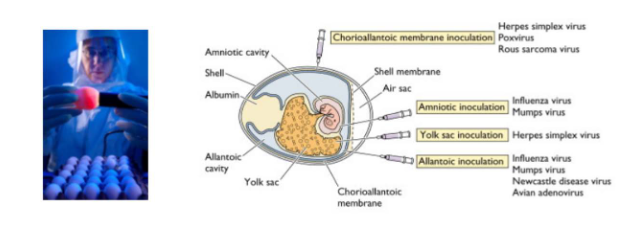
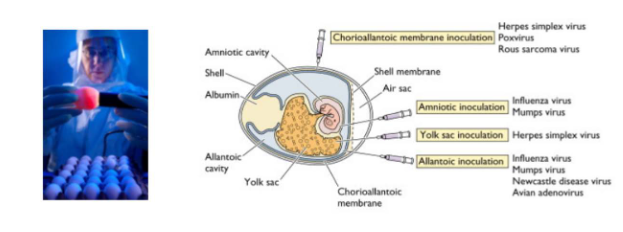
Growth of virus in eggs
- Before the development of cell culture, many viruses were propogated in embyronated chicken eggs
- today this method is most commonly used for growth of influenza virus - for vaccine production
- Egg is placed in front of a light source ("candled") to locate a non-veined are of the allatonic cavity just below the air sac
- Virus is injected into the allatonic cavity where it replicates

Growth of virus in laboratory animals
- In the early 1900s when viruses were first isolated, freezers and cell cultures were not available and it was necessary to maintain virus stocks by continous passage in animals
- Today laboratory animals are mainly used as model systems to study viral pathogenesis

Emergence of a novel Coronavirus, Wuhan, China, December 2019
- seven patients with severe virus-induced pneumonia disease admitted to intensive care; six were sellers or deliverymen from local seafood market
- Samples sent to Wuhan Institute of Virology for diagnosis of causative pathogen
- Pan-CoV PCR primers identified five samples as PCR+ve (outbreak occurred in winter and in same environment as SARS)
- NGS on BALF from one patient identified a 29,891 bp CoV genome with 79.6% sequence identity to SARS-CoV BJO1
- WGS from another four patients: sequences >99.9% identical
- 12 Dec 2019 - 26 Jan 2020: 2,794 lab confirmed infections and 80 deaths
2019-nCoV sufficiently divergent from SARS-CoV to be considered a new human-infecting betacoronavirus
- 2019-nCoV genomes >99.98% identical
- 88% identity to two bat SARS-like CoVcollected Zhoushan 2018
- 79% identical to SARS-CoV
- 50% identical to MERS-CoV
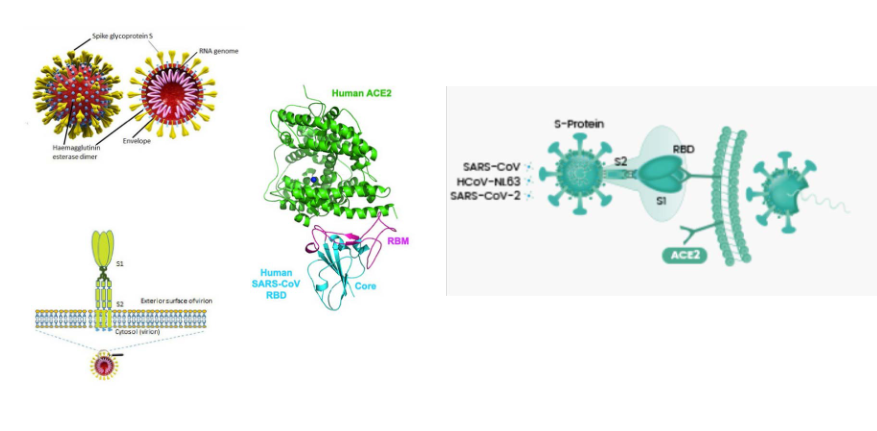
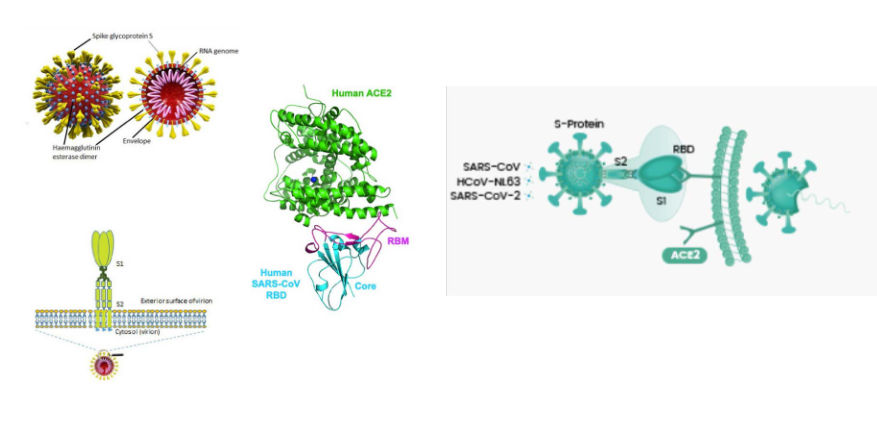
First step in SARS-CoV-2 cell entry: binding of spike protein to receptor, ACE2
DIAGRAM ON SLIDE 26

Summary (Read Carefully)
DIAGRAM ON SLIDE 27
