Nuclear Semester 2
1/64
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
Define open fuel cycle
Fuel is used once in reactor, wet interim stored and ultimately dry stored for disposal.
Define partially closed fuel cycle
Fuel is reprocessed to separate U, Pu, U-Pu made into MOx. Main U not recycled.
Define fully closed fuel cycle
Reprocessed U-Pu made into MOx fuel and U fuel fully recycled.
Chemical formula for yellow cake
U3O8
Explain the process of Uranium mining
Ore is crushed and ground
Acid leaching is predominantly used (H2SO4)
Uranium oxidised to get it into hexavalent form
Leaching solution is fltered and concentrated using solvent extraction
Precipitated as yellow cake 70-80% using ammonia/magnesia/caustic soda
Explain how to get from yellow cake to fuel ready Uranium
Yellow cake (U3O8) to UO3 conversion and purification
Conversion of UO3 to UF4
UF4 to UF6 (high temp)
Enrichment of U235 - leads to significant depleted uranium by-product
Back conversion of UF6 to UO2 granule solid for fuel
Give the advantages and disadvantages of a closed fuel cycle
ADV
Smaller final waste volumes
Lower long term inventories of Pu
Reduced requirement for new U mining
DIS
Cost
U price not increased sufficiently to warrant recycle
Nuclear proliferation risk
Explain why open fuel cycles help nuclear proliferation
Pu stockpiling due to non recycling
If Pu is not separated, fuel cannot be used for nuclear weapons
How long do we have to store spent fuel for?
At reactor cooling for 12 months
Central pond facility - cooling for reprocessing and handling
Long term (decades) usually dry (prior to disposal)
Describe decay heat
Created from beta and gamma emissions from fission products
Amount and type of fission depend on number of factors
Initial enrichment
Irradiation time
Irradiation history
Cooling time
Compare wet and dry storage of fuel
Wet store - active system, requiring engineering systems to maintain environment
Dry store - passive system requiring no intervention
Getting fuel completely dry is difficult
New storage racks holding 63 fuel cans in THORP will allow greater storage capacity for wet
Number of routes to longer term dry storage being assessed
Generally, how do we dispose of spent fuel?
GDF (Geological Disposal Facility)
Multiple barriers
Secure against unauthorised removal of nuclear material
Has to be safe for 1000s of years
Advantages and Disadvantages for solvent extraction
ADV
Safety
Mature technology
Low cost
Scale up easy
Selectively good
High purity achievable
Low temp
Re-use solvent
High HM loading capacity
DIS
Degradation of solvent
Entrainment
Not always CHON only
Waste management
What are the general principles of solvent extraction?
Use affinity of solute for specific solvent to concentrate solute
Reverse operation to recover solute
MIX - SETTLE - SEPARATE
What does PUREX stand for?
Plutonium Uranium Reduction EXtraction
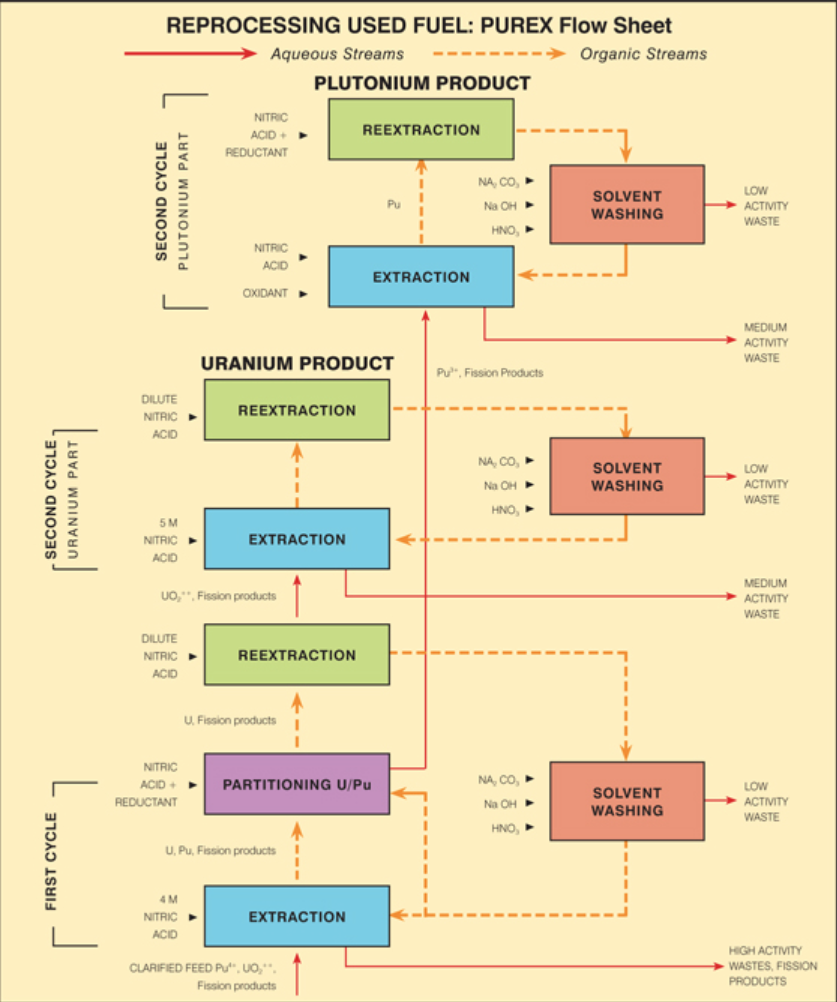
PUREX Flow sheet
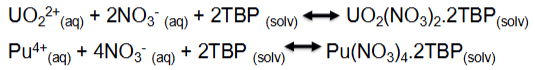
What are the equations for the U and Pu extraction? (reversible)
Equation for equilibrium constant for Uranium extraction
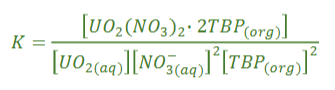
Why is the equilibrium constant of U extraction favoured by high NO3-?
Le Chatelier’s principle, higher nitrate core will push equilibrium to the right, run normally at high acid concentrations 3-5 M, runs with high TBP (20-30%)
Describe the partitioning of U and Pu (PUREX)
Based on reducing Pu(IV) to Pu(III) which is an inextractable in solvent
U remains in the solvent phase, the Pu goes into the aqueous phase
The reductant used is uranous nitrate (U(IV)), stabilised by hydrazine
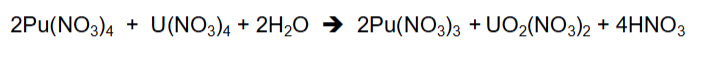
Describe the back extraction of U (PUREX)
No need to change valence state
High back extraction greatly favoured by diltue NO3-
Run back extraction using very dilute acid, will shift the reaction towards the release of U complex, run 0.01-0.1M acid in back extraction
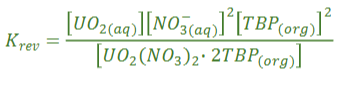
What are the downsides to TBP?
Very susceptible to radiolitic breakdown, owning to phospohorus atom
Leads to breakdown products that are less efficient, and limits TBP recycle
TBP is not very volatile, good for safety perspective but difficult to recycle via thermal separation
Describe pulsed columns
Approx 1m of column needed for one theoretical stage
Requires large amount of head space but little floor space
Moderate solvent inventory
Long residence times
Describe mixer settlers
Physical and theoretical stages nearly equivalent in efficiency
Simple to design and operate
Large settler volume
long residence time
high process inventory
Poor geometry for critically (high volume hold up)
Tolerant of solids
Describe centrifugal contractors
A process intensified PI technology
Each unit near one theoretical stage
Requires little headroom or floor space, requires remote maintenance capability
Small solvent inventory
Short residence times
Overview of the PUREX process
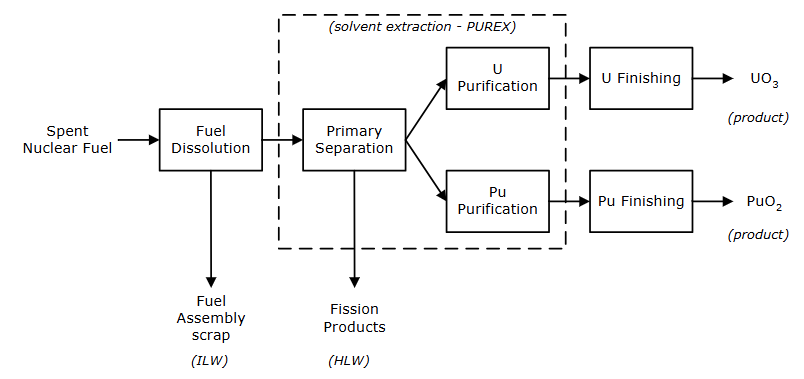
Describe step 1 of the PUREX system (Fuel Dissolution)
Shear assembly to expose fuel
Dissolve fuel in nitric acid
Centrifuge dissolver liquor to remove solids
Store clarified liquor for monitoring and feed to solvent extraction
Describe step 2 of the PUREX system (Solvent Extraction)
Primary separation
Separation of fission products from U/Pu
Separation of Pu from U
Decontamination of solvents
Treatment of fission product stream
Describe primary separation (PUREX)
Extract U and Pu into solvent phase
Split U and Pu, U in solvent, Pu in aqueous
Clean Pu steam of any carried over solvent
Describe Uranium purification (PUREX)
Decontamination of U stream from Np
Decontamination of solvents
Uranium purification
Initial U and Np back extraction to aqueous
Adjust temperature/conc to ensure U and Np in correct oxidation states
Solvent extraction to remove Np from U
Back strip with dilute acid to recover U
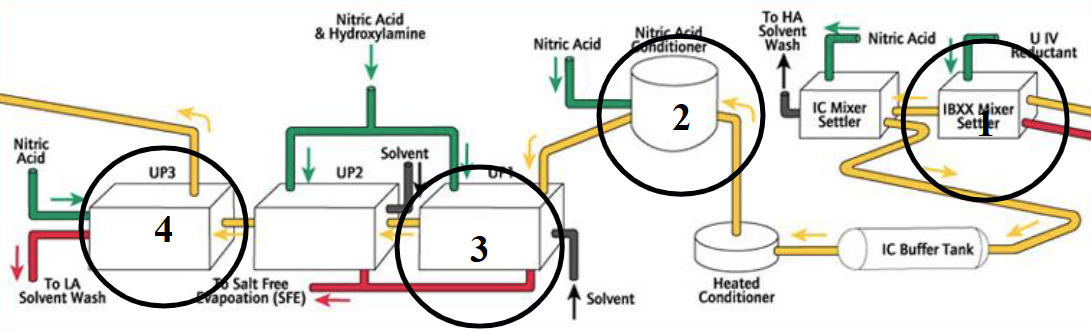
Describe Plutonium purification (PUREX)
Decontamination of Pu stream from Tc
Pu nitrate evaporation
Decontamination of solvents
Plutonium purification
Use NOx to oxidise Pu(III) to Pu(IV)
Solvent extraction to separate Pu(IV) from Tc
Backwash Pu(III) into acid
Clean out any entrained solvent from acid
Evaporate to reduce volume for storage
Describe Uranium finishing (PUREX)
Steam strip acid to remove entrained solvent
Evaporate to remove water and acid
Use hot air to breakdown U nitrate to U oxide
Cool, blend and store U oxide powder in drums
Describe Plutonium finishing (PUREX)
Final conditioning to ensure Pu(IV)
Add oxalic acid to precipitate Pu
Separate liquid and solids
Dry and calcine to convert solids to Pu dioxide
Blend, sample and store in cans
Wet Pu oxalate is fed continuously to a 2-stage drying calcining furnace
Drying/calcination operates at up to 600 degrees celcius in electrically heated furnaces with counter current air flow
Define heterogenous recycling
U, Pu actinides as separate products
Define homogeneous recycling
U, Pu actinides in a single product
Describe homogeneous recycling
GANEX 1st Cycle
Solvent extraction process
Use of ligand that only extracts Uranium
DEHiBA in diluent
Still have nitric acid dissolution = head end is the same
Uranium is still extracted as a UO2 uranyl ion
GANEX 2nd Cycle
EURO-GANEX is a solvent-extraction process aiming to co-separate all transuranic elements
For this, a solvent consisting of TODGA and DMDOHEMA in kerosene is used
Define adsorption
Mass transfer from liquid/gas to solid phase
Define leaching
Mass transfer from solid to liquid phase
Define solvent extraction
Mass transfer from one liquid phase to another
Pros and Cons of solvent extraction (SX)
Pros
Using the correct ligand is generally straight forwards as long as it dissolves in the solvent
High ligand concentration usually okay if solubility/miscibility in solvent/diluent is high
Chemicals are usually recoverable
Superior when the target species conc in water is >2g/L
Cons
Generally unavoidable entrainment of organic and aqueous phases
More prone to degradation
Pros and cons of adsorption
Pros
Adsorbent regeneration is generally straight forward
Less pron to degradation
Generally superior for anion separation
Superior when the target species conc in water is <2g/L
Cons
Attatching the correct ligand to the adsorbent not always easy
Cannot have a very high ligand concentraion or physical properties are affected
Can last long time but no generally recycled
Define ionic bonding
Complete transfer of valence electrons between bonding partners to create a lattice of cations and anions
Define covalent bonding
Chemical bonds are formed from both bonding partners sharing valence electrons
Define coordinate bonding
Chemical bonds are formed from one bonding partner donating valence electrons
Difference between trans and cis ligands
Trans are on opposite diagonals, cis are on the same side of a complex
Explain HSAB theory
Dissolved metal ions in solution are considered lewis acids (capable of accepting a lone pair to form a chemical bond)
Likewise, potential ligands are lewis bases (capable of donating a lone electron pair to form a chemical bond
Uranyl *UO2 (2+) is slightly unusual, being a polyatomic cation
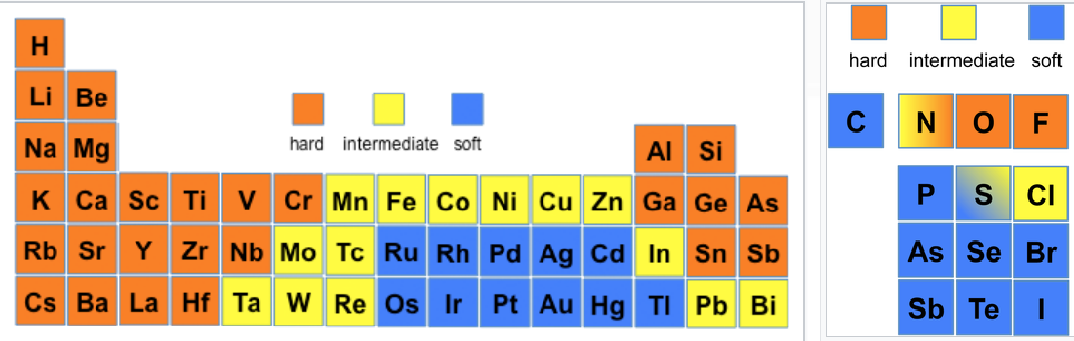
Describe hard acids and bases
High charge density
Not very polarizable
Bonding interactions more ionic in nature
Smaller ionic radius (relative to mass)
Describe soft acids and bases
Low charge density
Highly polarizable
Bonding interactions more covalent in nature
Larger ionic radius
Given some examples of soft hard and intermediate acids/bases
Define and describe chelation
A chelation or cherlating interaction occurs when a single ligand bonds to a metal ion via multiply heteroatoms
Bonding can be very strong, and its sometimes not a helpful for processes such as solvent extraction
Common chelating agent is EDTA
Active ingredient in chemical first-aid sprays found in labs
Describe selective separations
Uses a carefully chosen ligand to target specific chemical species
Applications usually based on removing just the valuable component of a wastestream
Reagents can sometimes be expensive and/or toxic
Example : Leaching of gold mind tailings
Describe Non-selective separations
Large variety of chemical species transferred from one phase to another
Applications usually based on removing hazardous components of a wastestream, prior to discharge to environemnt
Example : ion-exchange column to produce potable water
Equation for the distribution coefficient (AKA partition coefficient) for separation processes

Equation for the separation factor
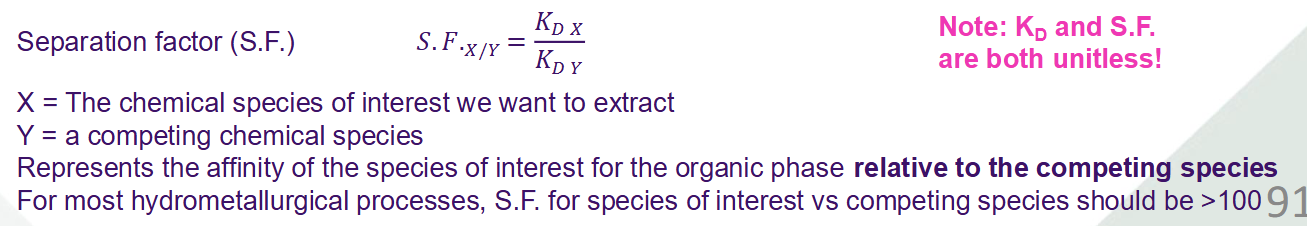
What are the requirements of a good organic phase for nuclear separations?
Able to extract some components to a larger extent than the rest of the components in the aq phase
Reaction should be stable but reversible
The compound formation should be easily reversed so that the solvent phase can be reused
Density of solvent phase at all times should be different from the aq phase to effect the separation
Inexpensive and cost effective with more than one supplier
Low toxicity
Low viscosity to aid mixing
High boiling point
Why is TBP a good ligand for PUREX?
P=O bond is strongly polarised meaning electron rich
Hydrophobic alkyl groups allow for good solvation in the organic phase
Hydrophobic parts of the complex interact with the organic phase and keep the hydrophilic parts contained
Describe acid hydrolysis and radiolysis of TBP
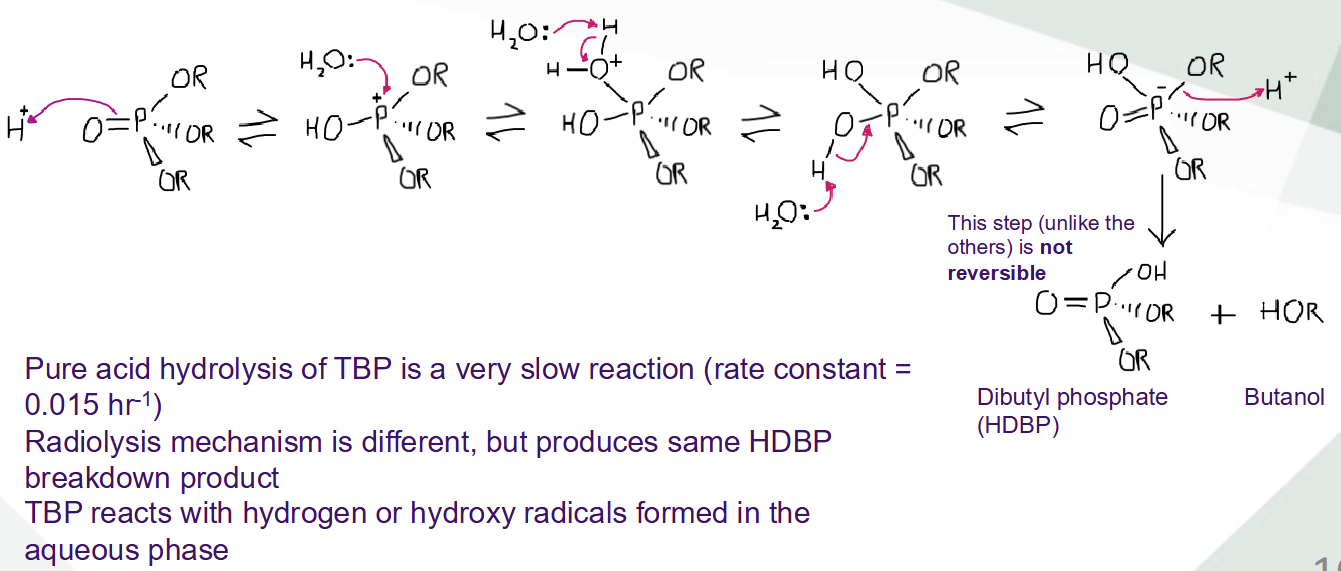
Describe the degradation of TBP by hydrolysis and radiolysis
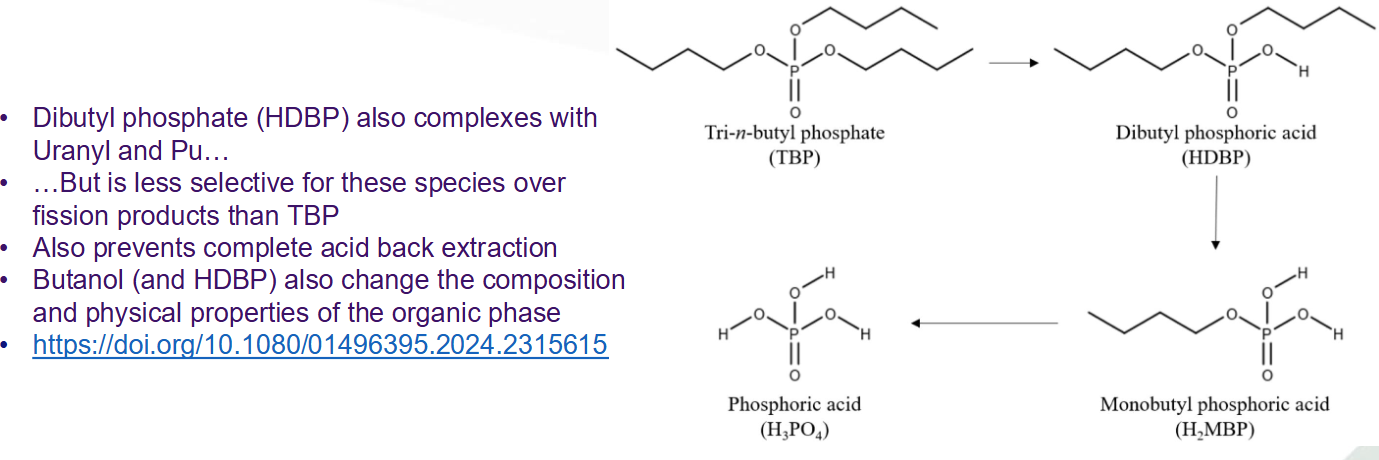
Advantages of CHON ligands
Still prone to radiolysis but
Degradation products have low affinity compared to other fission products
Have relatively low boiling points so no issues residue when spent solvent is incinerated
More selective for U/Pu than TBP, overall lower capacity due to solubility limits of the complexes in the organic phase
What is the importance of relative densities of phases?
In SX, must always be a density difference between the aqueous and organic phase
System must also account for the changes in phase density that occur during mixing, due to mass-transfer
Phases being close in density less problematic for mixer/settlers and pulse columns
Can be very problematic for centrifugal contract set up
Describe the issues with Iodine-129
I129 and I131 are major contaminants in spend nuclear fuel
Complex chemical speciation and volatility means radioiodine is present in most stages of fuel recycke
Gas and aqueous streams require treatment
I129 is most significant dose contributor to plant emissions
What is the solution to I129 issues in separation?
Apply HSAB theory to the radioiodine management problem
Designing a selective solvent extraction process for non-metals is very difficult
Easier to design a solid-phase adsorbent and contact this material with a gaseous or aqueous stream
Want the binding to be close to irreversible
Radioiodine loaded material can then be processed as solid waste and sent for geological disposal