C3.3 - Types of Chemical reactions (I made half AI for 2nd half)
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Define Oxidation: (in terms of oxygen)
Oxygen is added to the element/compound
Define Reduction: (in terms of oxygen)
Oxygen is removed from the element/compound
Define a Redox Reaction:
Reaction where both oxidation and reduction occur at the same time
In the reaction, ZnO + C becomes ZnCO, what has been oxidised and what has been reduced? and which is which agent?
Carbon has been oxidised since it gains oxygen
Zinc oxide compound has been reduced as oxygen is lost
Carbon is the reducing agent as it reduces another substance (ZnO)
Zinc oxide is the oxidising agent as it oxidises another substance (Carbon)
Remember OILRIG for Oxidation and Reduction in terms of electrons and what does it stand for?
Oxidation Is Loss (of electrons) Reduction Is Gain (of electrons)
How to form an ionic equation? and then a half equation?
Write the overall balanced symbol equation:
Zn (s) + CuSO4 (aq)→ ZnSO4 (aq) + Cu (s)
Write out the ions separately:
Zn (s) + Cu2+(aq) + SO42- (aq) →Zn2+(aq) + SO42-(aq) + Cu (s)
Eliminate ions that occur on both sides (spectator ions):
Zn (s) + Cu2+(aq) → Zn2+(aq) + Cu (s)
Identify which species has lost or gained electrons ( u can tell which has gained and lost then u can write out the half equations)
Zinc has lost electrons and been oxidised:
Zn → Zn2+ + 2e-
Copper ions have gained electrons and been reduced:
Cu2+ + 2e- → Cu
Define an Acid:
Acids are substances that form positively charged hydrogen ions (H+)
presence of H+ makes solution acidic
Define an Alkali:
Alkalis are substances that form negatively charged hydroxide ions (OH-)
presence of OH- ions makes aqueous solution alkali
Define the PH scale:
A numerical scale used to show how acidic or alkaline a solution is
measure for amount of Ions present in a solution
What Ph is neutral and what does lower Ph than neutral mean and what does higher Ph than neutral mean?
Ph 7 - Neutral
Lower than 7 means more acidic
Higher than 7 means more alkaline
2 ways to measure PH:
Indicator or Digital pH meter (probe)
How does universal indicator work and what’s colour scheme of the Ph scale like due to this?
Universal indicator is a substance which changes colour depending on Ph of solution to which it’s added
Red at low pH (acidic), moving through orange, yellow, green (neutral, pH 7), then blue to purple at high pH (alkaline).
Synthetic vs natural indicator
Synthetic - Very precise PH ranges
Natural - wide range of indicators
What are the products when an acid reacts with a metal oxide or hydroxide?
A salt and water only. General equation: acid + base → salt + water
What are the products when an acid reacts with a metal carbonate?
A salt, carbon dioxide and water. General equation: acid + metal carbonate → salt + carbon dioxide + water
What are the products when an acid reacts with a metal?
salt and hydrogen gas. General equation: metal + acid → salt + hydrogen. Only metals above hydrogen in the reactivity series react with dilute acids
How do you name a salt?
Two parts:
first part comes from the metal, metal oxide, metal hydroxide or metal carbonate used.
Second part comes from the acid — hydrochloric acid → chloride, sulfuric acid → sulfate, nitric acid → nitrate Give examples of salt naming rules
Give examples of salt naming rules:
Zinc reacts with sulfuric acid → zinc sulfate.
Zinc reacts with hydrochloric acid → zinc chloride.
Magnesium carbonate + nitric acid → magnesium nitrate + carbon dioxide + water
What is the formula for hydrochloric acid and sulfuric acid?
Hydrochloric acid = HCl. Sulfuric acid = H₂SO₄.
These are the two most common acids you will be asked about in exams — make sure you know their formulae
What is a strong acid?
A strong acid fully dissociates in water — all of its molecules split into ions. This means the equilibrium lies completely to the right. Examples: HCl, H₂SO₄, HNO₃
What is a weak acid?
A weak acid only partially dissociates in water — an equilibrium exists between the dissociated ions and the undissociated molecules. The equilibrium lies to the left, meaning most molecules remain undissociated The ⇌ symbol is used. Example: CH₃COOH (ethanoic acid)
What is the difference between a strong acid and a concentrated acid?
Strong/weak refers to the degree of dissociation.
Concentrated/dilute refers to how many moles of acid are dissolved in the solution.
A weak acid can be concentrated and a strong acid can be dilute
How much ion change is there between each scale on the Ph Meter?
The pH scale is logarithmic, meaning that each change of 1 on the scale represents a change in concentration by a factor of 10
What is the relationship between H⁺ concentration and pH?
Higher H⁺ concentration = lower pH (more acidic). Lower H⁺ concentration = higher pH (more alkaline). At pH 7 (neutral) the H⁺ and OH⁻ concentrations are equal
What does a titration curve show?
How the pH changes as a base is added to an acid (or vice versa). The pH increases rapidly at the equivalence point (the point of neutralisation) — shown by a steep vertical section on the curve
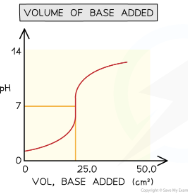
What does the titration curve look like when a base is added to an acid?
Starts at low pH, rises slowly, then rises very steeply at the equivalence point (neutralisation), then levels off at high pH. The curve is S-shaped
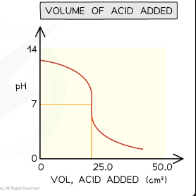
What does the titration curve look like when an acid is added to a base?
Starts at high pH, decreases slowly, then drops very steeply at the equivalence point, then levels off at low pH — mirror image of base added to acid