Failed Question Bank - Chem
1/31
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
32 Terms
What is the maximum number of electrons that can be held in the third energy level?
18
3s + 3d + 3p or 2e + 10e + 6e = 18e
The third energy level refers to the orbitals with 3 in front of the.
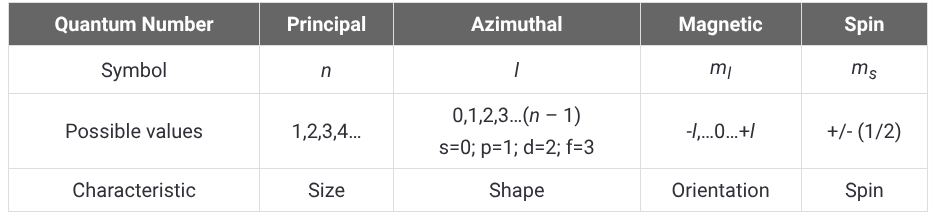
How do you determine a possible set of quantum numbers
What is an Ionic Bond?
formed when electrons are transferred between atoms, resulting in oppositely charged ions that attract one another.
Lattice energy increase as
Charge number increases and ion size decreases
Going down and left on the periodic table
Intramolecular forces
Covalent Bonds
Ionic Bonds
Metallic Bonds
Intermolecular forces
London Dispersion Forces
Dipole-Dipole Forces
Ion-Dipole Forces
Ionization Energy
The amount of energy required to remove an electron.
(1st is one electron, 2nd is two, 3rd is three, ect.)
Binary Acid Trend
The trend in acid strength for binary acids (HX). Increases as you go to the right and down the periodic table.
How is acid strength related to bond dissociation energy?
As acid strength increases, the bond dissociation energy decreases (because acids like to dissociate into ions).
Bond dissociation energy
The amount of energy required to break a bond in a molecule
What groups react with pure oxygen?
Which group reacts more vigorously with oxygen?
Group 1 elements and Group 2 elements
Group 1 elements
Transition metals form ___ compounds due to the presence of partially filled d orbitals.
colorful
atoms with a +1 charge have undergone their
first ionization energy
Ionization energy greatly increase once
the octet is formed
When dealing with isoelectric (Same # of e- ) atoms, the atom with the smallest atomic radius has?
The most amount of protons
Alkali metal properties
Silvery white metals
High conductivity
Low melting points
How to find new bond length given bond lengths of other molecules
divide the bond lengths by 2
Add both (bond length/2) together to find new atom bond length
Characteristics of metals
low ionization energies
low electronegativity
Tend to lose electrons and form positively charged ions
What is electron affinity?
The energy released when an atom gains an electron. Tends to increase going up and to the right of the periodic table
Ionization increases-
going up and to the right of the periodic table

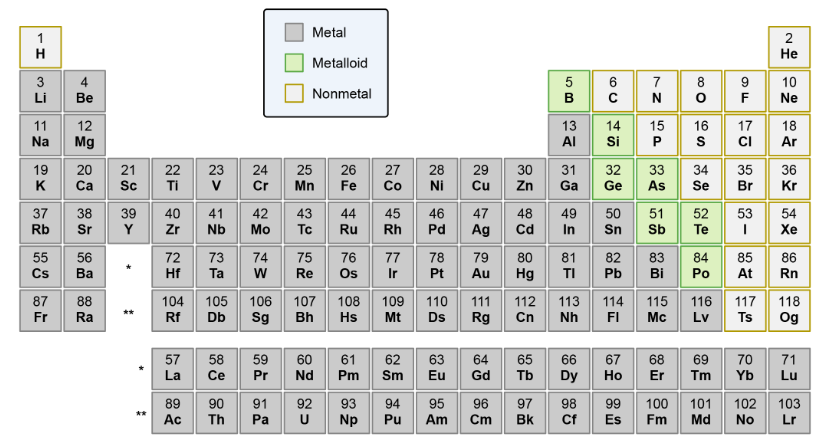
Name the metals, metalloids, and nonmetals

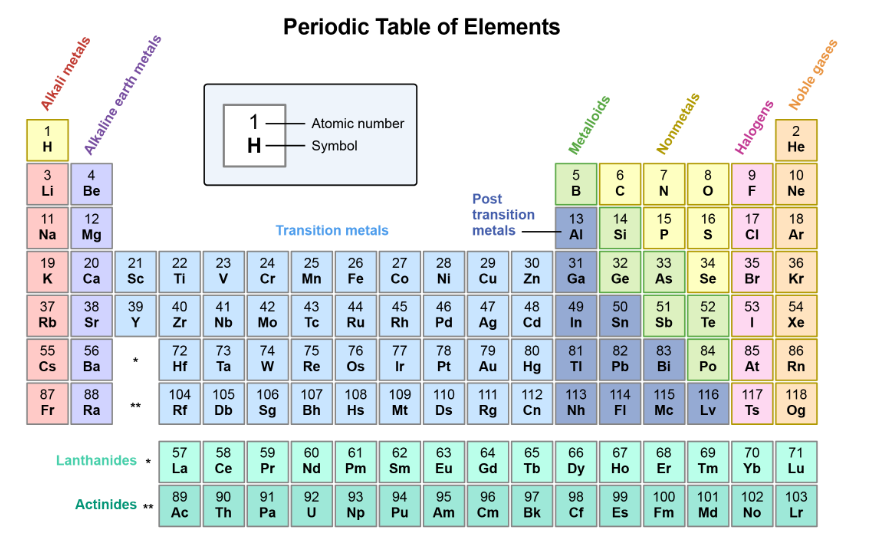
Name the series in the periodic table
Alkali metals
Alkaline earth metals
Transition metals
Post transition metals
Metalloids
Nonmetals
Halogens
Noble gases
Lanthanides
Actinides
What is the trend for elements that are able to have an expanded octet?
What are commons elements that have expanded octets?
Elements that are beyond period 3. Common elements include phosphorus, sulfur, and chlorine.
When do you use the combined gas law?
When a starting set of conditions and ending set of conditions is given for the variables P, V, T, or n
Gases act ideally under five specific assumptions
The volume of individual gas molecules is considered to be negligible.
There are no intermolecular attractions or repulsions between the gas molecules.
Gas particles are always in continuous and random motion.
Collisions between gas particles are elastic. Therefore, there is no net loss or gain of kinetic energy when the particles collide.
The average kinetic energy is always the same for all gases at a specific temperature, regardless of the identity of the gas.
Hydrogen is _-atomic, and Helium is _-atomic
diatomic; monoatomic
1 atm = ? torr = ? mmHg = ? Pa = ? kPa
1 atm = 760 torr = 760 mmHg = 101,325 Pa = 101.325 kPa
How to distinguish R value based on pressure
If the pressure is in atm, r = 0.08206. If the pressure is in kPa, r = 8.314
As the temperature of a gas is lowered, the rate of condensation __.
increases; inversely related
A gas becomes less ideal as-
temperature decreases
What causes the pressure of a gas in a container to increase when the volume is decreased at a constant temperature?
The molecules strike the container more often due toreduced space, increasing pressure.