3.10 Reaction Rates
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
What is the equation used to calculate rate?
Rate = change in concentration / time
What is the unit for rate of reaction?
mol dm⁻³s⁻¹
What must particles do in order to react?
Collide with sufficient energy (activation energy) and the correct orientation
Do most collisions result in a reaction?
No
What are the factors that affect rate of reaction?
● Temperature
● Pressure
● Concentration
● Surface area
● Catalyst
What is the effect of increasing temperature on rate of reaction and why?
● Increasing temperature → increased rate of reaction
● much higher proportion of particles have energy greater than the activation energy, leading to more successful collisions per second.
What is the effect of increasing concentration/pressure on rate of reaction and why?
● Increased concentration/pressure → increased rate of reaction
● there are more particles in a given volume, leading to more frequent successful collisions.
What are the variables in an experiment that can be monitored to calculate the rate of reaction?
● Concentration of reactant or product
● Gas volume of products
● Mass of substances formed
How to calculate rate from a concentration time graph?
Draw a tangent and work out the gradient of the tangent using the equation: Gradient = change in y / change in x.
How to calculate rate from a concentration time graph?
● Draw a tangent
● Work out the gradient of the tangent using the equation
● Gradient = change in y / change in x
What is a catalyst?
A substance which increases the rate of reaction but is not used up in the reaction.
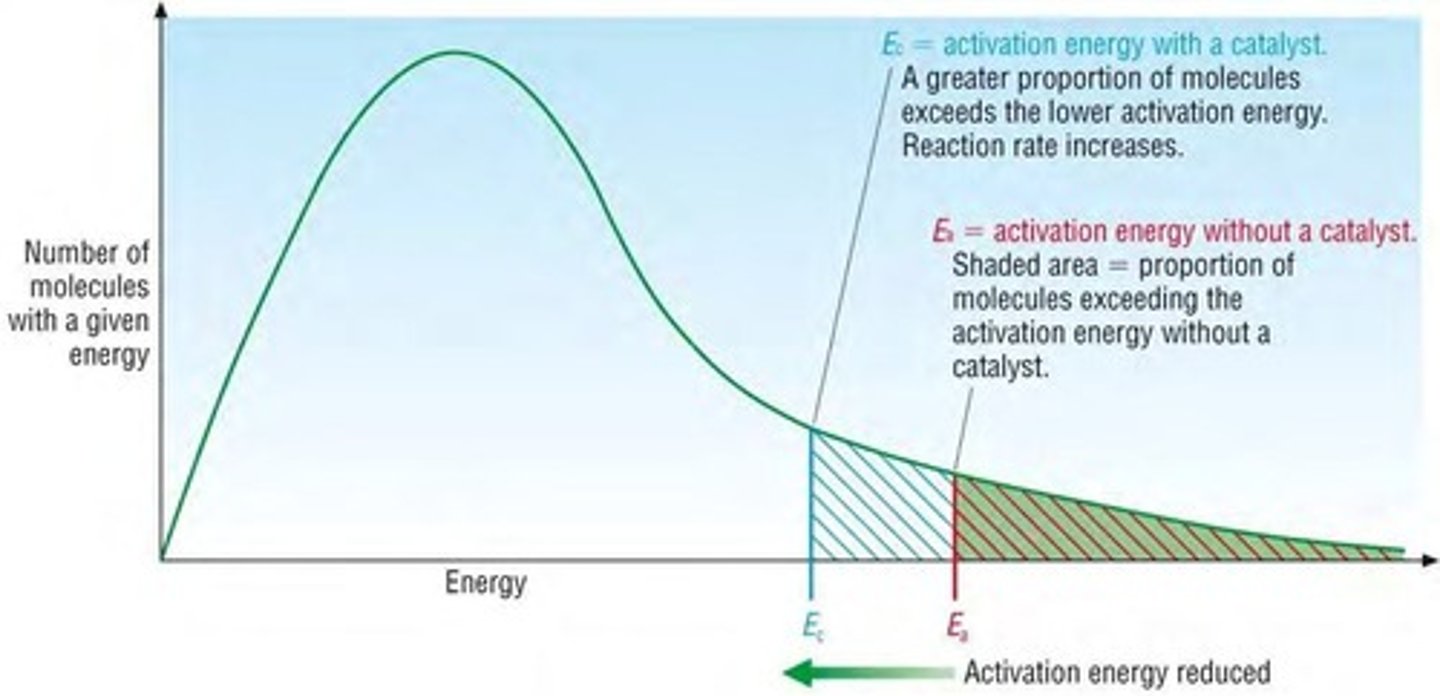
How do catalysts work and how do they increase the rate of reaction?
● They provide an alternate reaction pathway with a lower activation energy
● resulting in more particles having energy greater than activation energy, leading to more frequent successful collisions.
What does homogeneous catalyst mean?
A catalyst that is in the same phase as the reactants
e.g., liquid catalyst mixed with liquid reactants.
What does heterogeneous catalyst mean?
A catalyst used in the reaction that is in a different phase to the reactants
e.g., gaseous reactants passed over solid catalyst.
What are catalytic converters?
They are present in vehicles to reduce toxic emissions and prevent photochemical smog.
Define activation energy.
The minimum energy that particles must collide with for a reaction to occur.
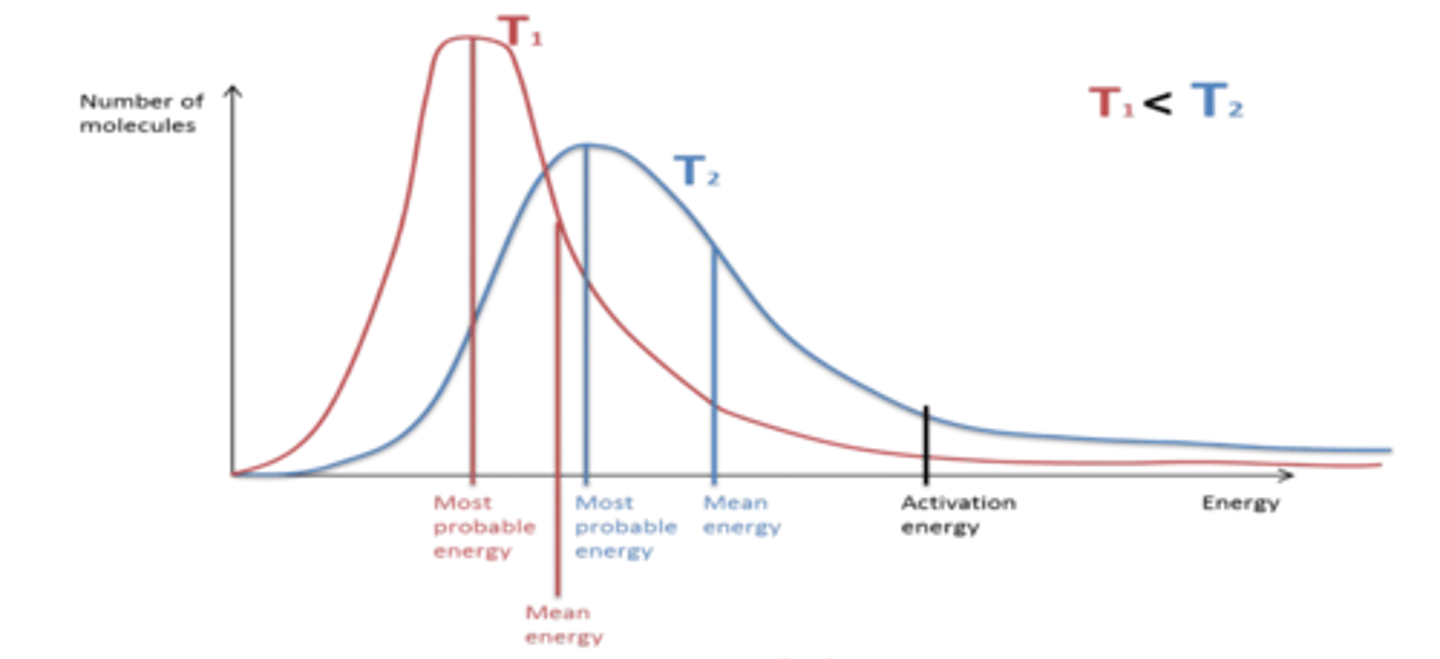
Name some important features of Boltzmann distribution.
● Area under the curve = total number of molecules
● area does not change when conditions alter
● curve starts at the origin
● curve does not touch or cross the energy axis
● only molecules with energy greater than activation energy can react.
What are the axes in a Boltzmann distribution?
X axis - energy
Y axis - number of molecules with a given energy.
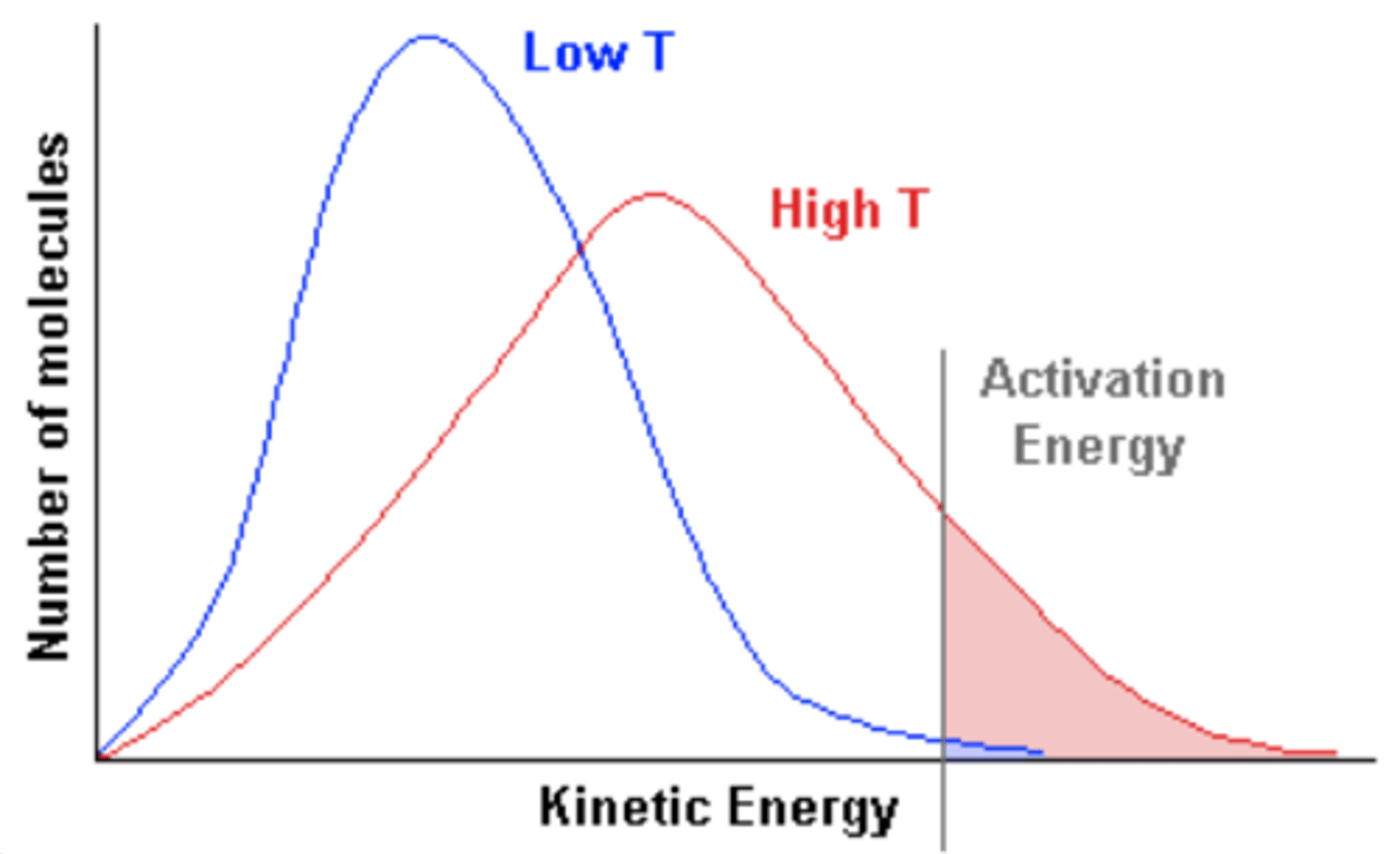
Draw a labelled Boltzmann Curve with labels of average energy, activation energy and most probable energy.
Draw in a different colour the effect of increasing temperature
Draw a labelled Boltzmann Curve showing the effect of catalyst of rate of reaction