Exercise Physiology (Exam 3)
1/72
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
73 Terms
key neuroendocrine organs
Two neuroendocrine organs coordinate hormonal control during exercise:
Hypothalamus
Integrates signals from the nervous system
Activates the sympathetic nervous system
Stimulates the pituitary gland → triggers endocrine responses
Initiates hormonal changes needed for exercise (energy mobilization, stress response)
Adrenal Medulla
Inner portion of the adrenal gland
Releases catecholamines: epinephrine and norepinephrine
Increases heart rate, blood pressure, and blood glucose
Helps mobilize energy for working muscles during exercise
Key idea:
Exercise activates the neuroendocrine system, linking the nervous system (fast electrical signals) with the endocrine system (slower hormonal signals) to maintain homeostasis and supply energy to muscles.
Main endocrine glands involved in hormonal control during exercise
The main endocrine glands involved in exercise regulation are:
Hypothalamus
Neuroendocrine control center
Links nervous system → endocrine system
Stimulates the pituitary gland (hypophysis) and activates the sympathetic nervous system
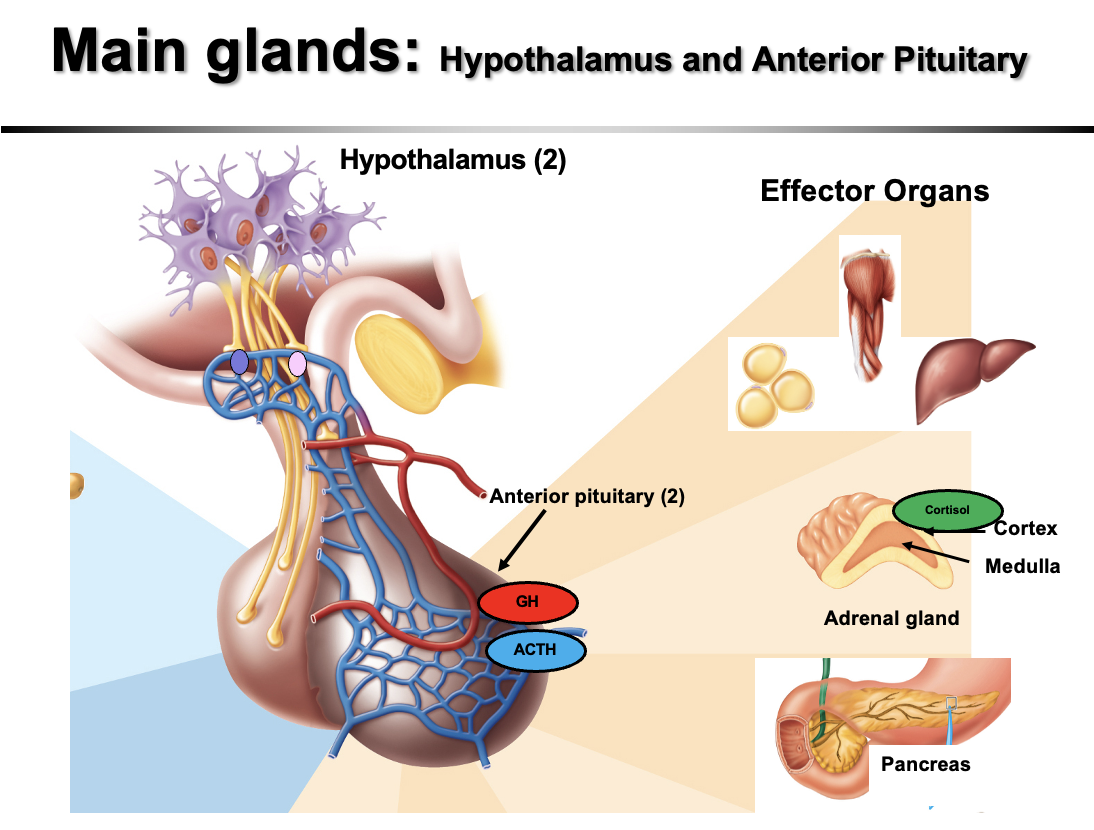
Pituitary gland
Called the “master gland”
Releases hormones that control other endocrine glands
Important for exercise responses (e.g., ACTH, growth hormone)
Adrenal glands
Sit on top of kidneys
Adrenal medulla: releases epinephrine & norepinephrine (fight-or-flight, ↑ HR, ↑ blood glucose)
Adrenal cortex: releases cortisol (mobilizes energy)
Pancreas
Regulates blood glucose during exercise
Insulin: lowers blood glucose
Glucagon: raises blood glucose
Key idea:
During exercise, these glands work together to mobilize energy, regulate blood glucose, and maintain homeostasis.
Two chemical classes of hormones
Hormones are divided into 2 main chemical groups:
1. Steroid hormones
Derived from cholesterol
Lipid-soluble
Can diffuse through the cell membrane
Bind intracellular receptors and act in the nucleus
Directly alter gene transcription
Examples: cortisol, aldosterone, testosterone, estrogen
2. Protein (peptide) and amino acid–derived hormones
Made from amino acids
Water-soluble
Cannot cross the cell membrane
Bind receptors on the cell surface
Use second messengers (ex: cAMP) to trigger cellular responses
Key idea:
The chemical structure of a hormone determines how it acts on cells (nuclear action vs membrane receptor signaling).
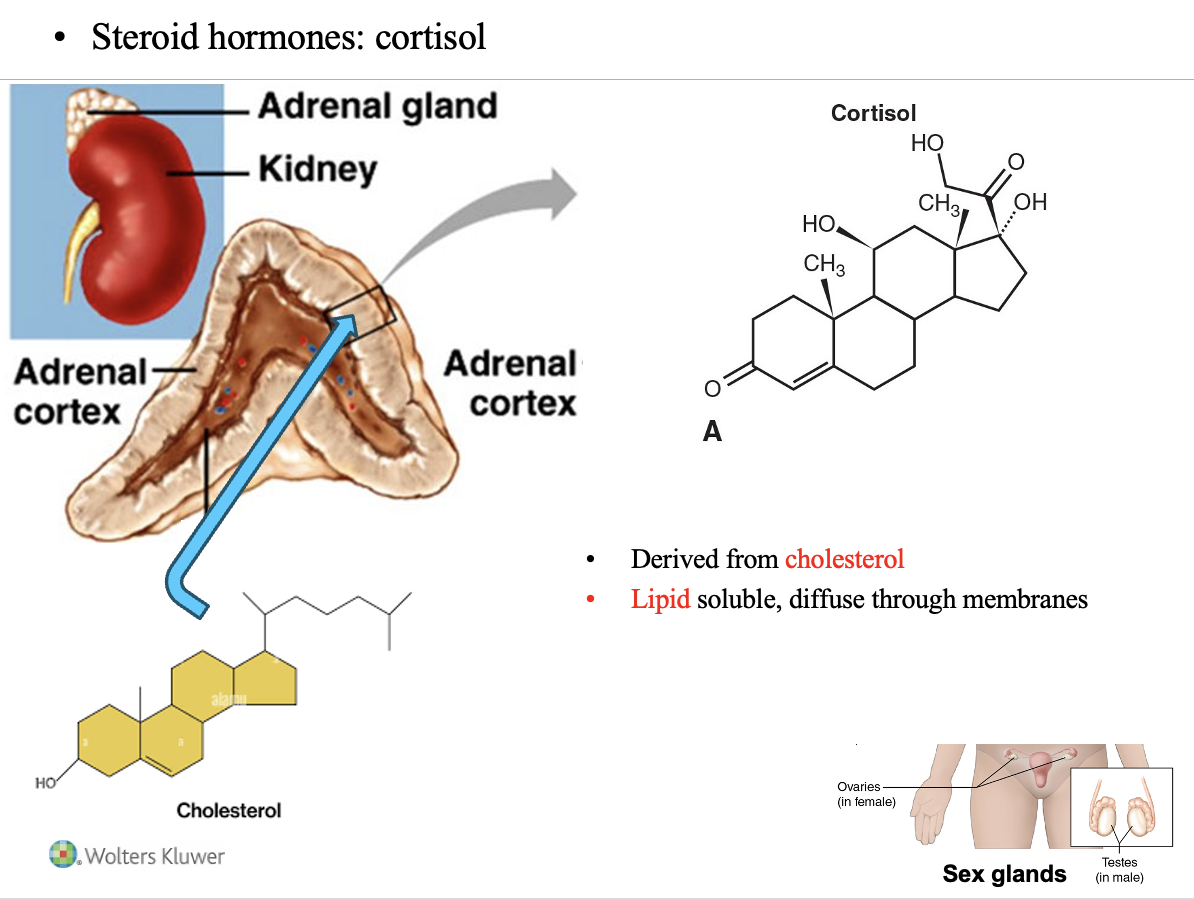
Cortisol and steroid hormones
Cortisol is the main steroid hormone emphasized in this class.
Key features of steroid hormones:
Produced in: adrenal cortex and sex glands
Lipid-soluble → diffuse through the cell membrane
Receptors located in:
Cytoplasm
Nucleus
Hormone–receptor complex enters the nucleus and alters gene transcription
Protein/peptide and amino acid–derived hormones
Peptide/protein hormones
Chains of amino acids synthesized like proteins
Examples:
Pancreas: insulin, glucagon - peptide
Anterior pituitary: growth hormone (GH), ACTH (adrenal cortex - steroid)
Hypothalamus: releasing hormones
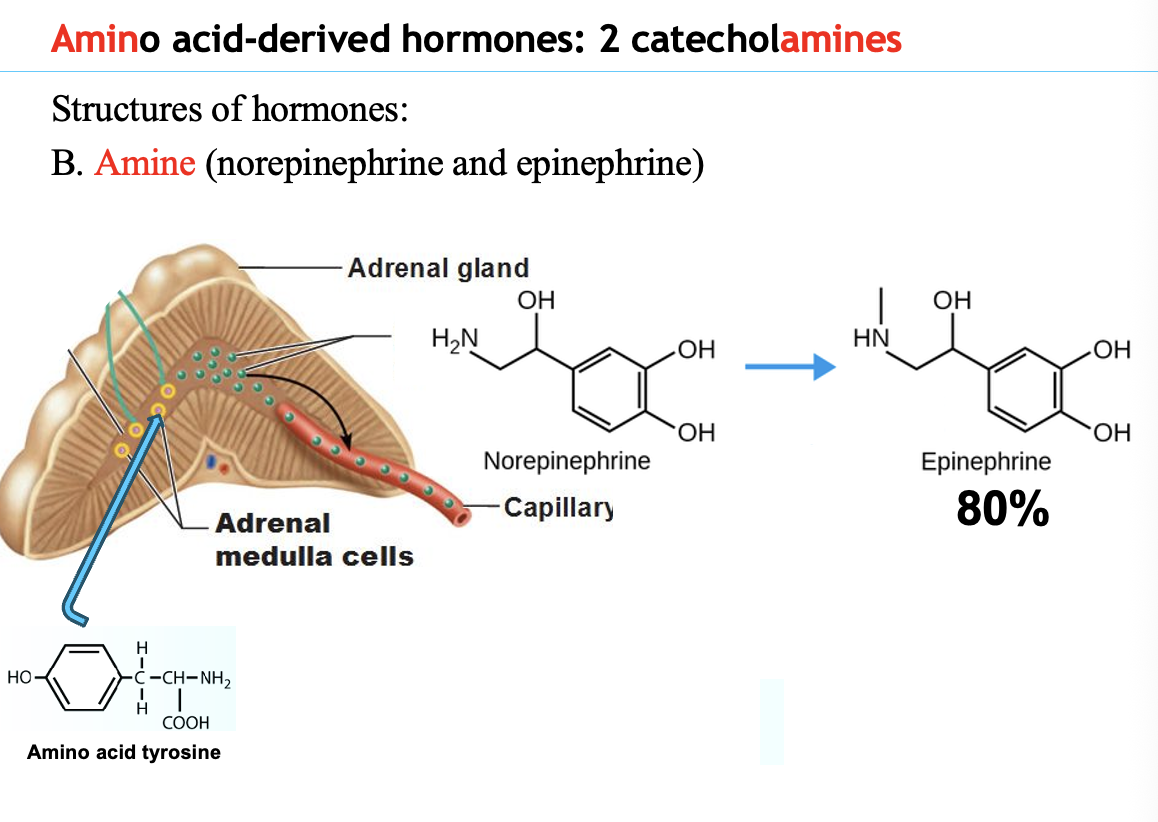
Amino acid–derived hormones (amines)
Small molecules modified from the amino acid tyrosine
Examples: epinephrine and norepinephrine
cortisol = stroid (adrenal coretex)
epinephrine/norepinephrine = amino acid derived
growth hormone = peptide
Pancreatic hormones
The pancreas secretes two protein hormones:
Glucagon
Secreted by alpha (α) cells
Insulin
Secreted by beta (β) cells
Key idea:
Both are protein (peptide) hormones that regulate blood glucose levels
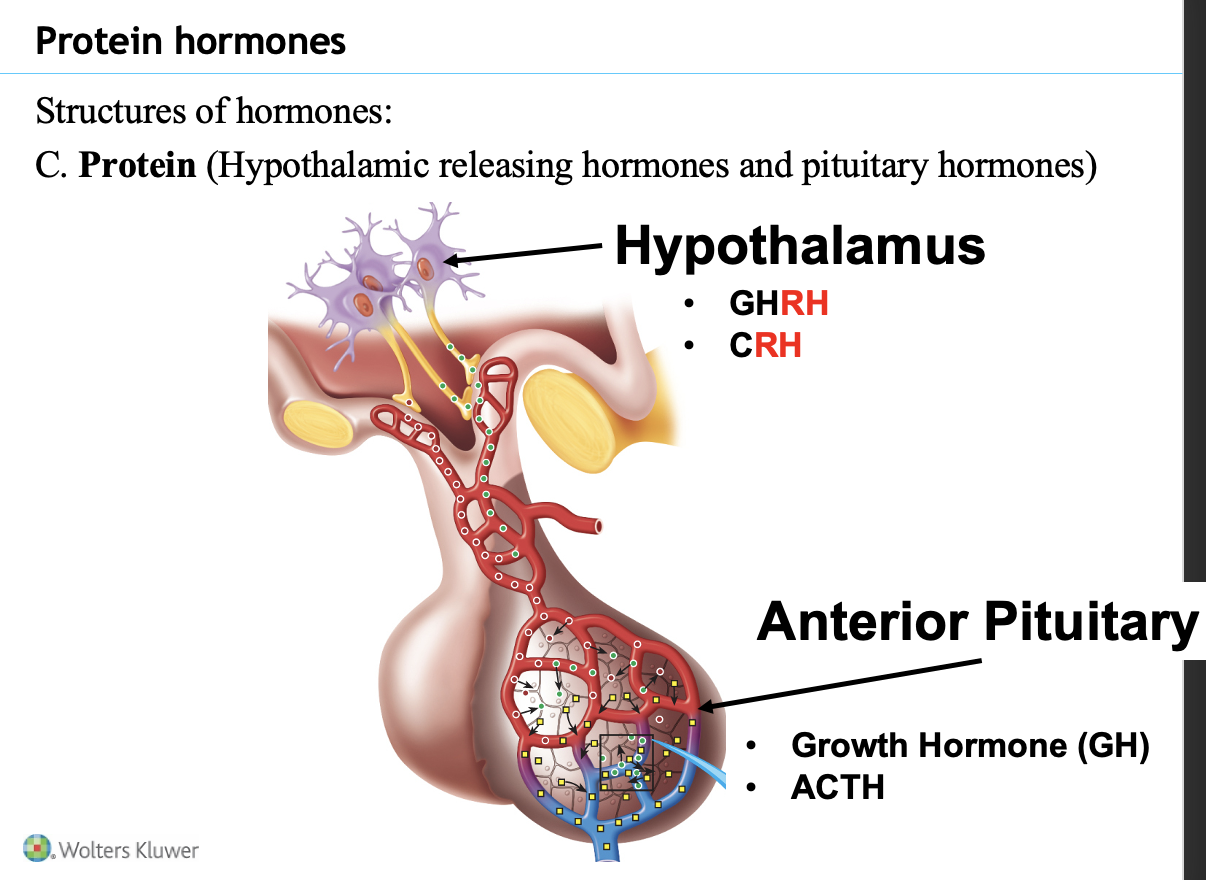
Hypothalamus and anterior pituitary hormones
Hypothalamus secretes releasing hormones that stimulate the pituitary:
GHRH (Growth Hormone Releasing Hormone)
CRH (Corticotropin Releasing Hormone)
Anterior pituitary secretes protein hormones:
Growth hormone (GH)
ACTH (Adrenocorticotropic hormone)
Key idea:
The hypothalamus controls the anterior pituitary, which then releases hormones that act on other endocrine glands.
Catecholamines and metabolism during exercise
Catecholamines are hormones derived from an amino acid (tyrosine) and are secreted by the adrenal medulla.
Epinephrine
~80% of adrenal medulla secretion
Main catecholamine hormone
AS A NEUROTRANSMITTERS - norepinephrine is more abundent. HORMONE = EPINEPRHINE
Affects metabolism and cardiovascular function
Norepinephrine (NE)
Mainly affects cardiovascular function
Does NOT significantly affect metabolism
Key idea:
Epinephrine is the only catecholamine hormone that significantly regulates metabolism during exercise.
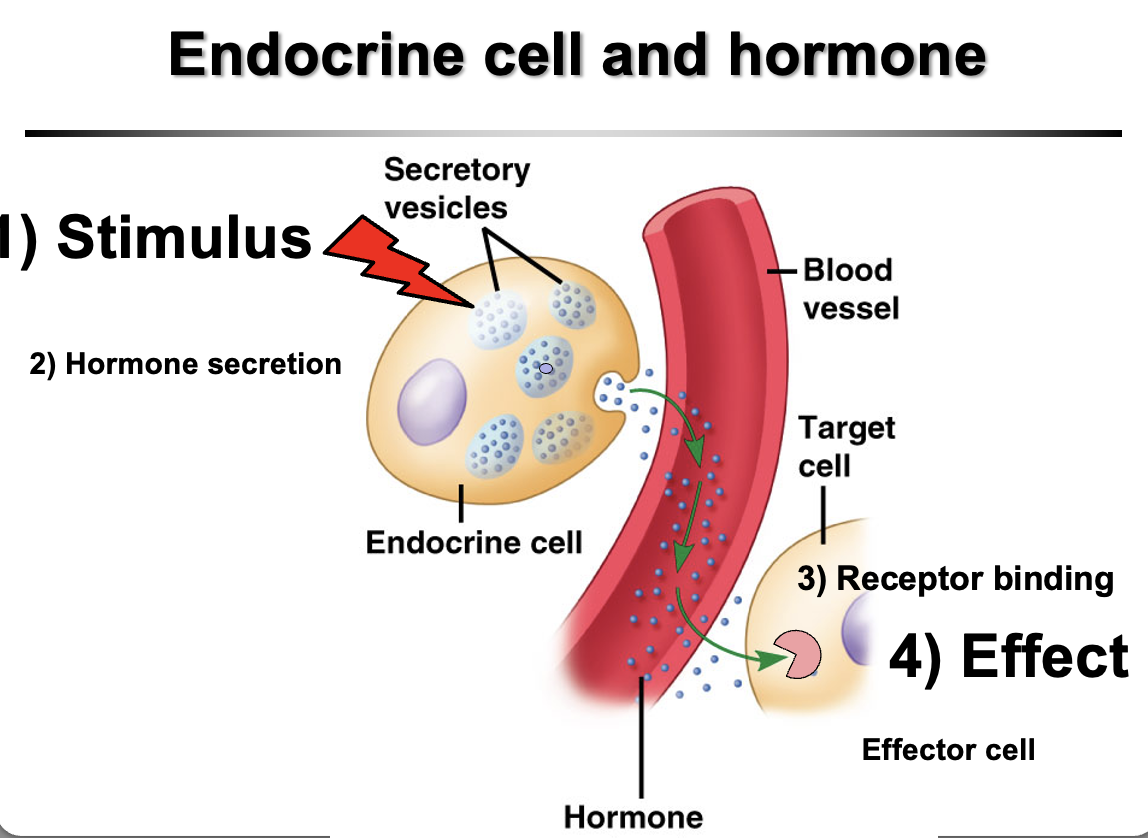
How do hormones work?
Hormones are chemical messengers secreted by endocrine cells directly into the bloodstream.
Process:
Stimulus causes an endocrine cell to release a hormone
Hormone travels through the bloodstream
It reaches target cells with specific receptors
Hormone binds the receptor
Binding causes a physiological effect in the target cell
Key idea:
A hormone only affects cells that have the specific receptor for it.
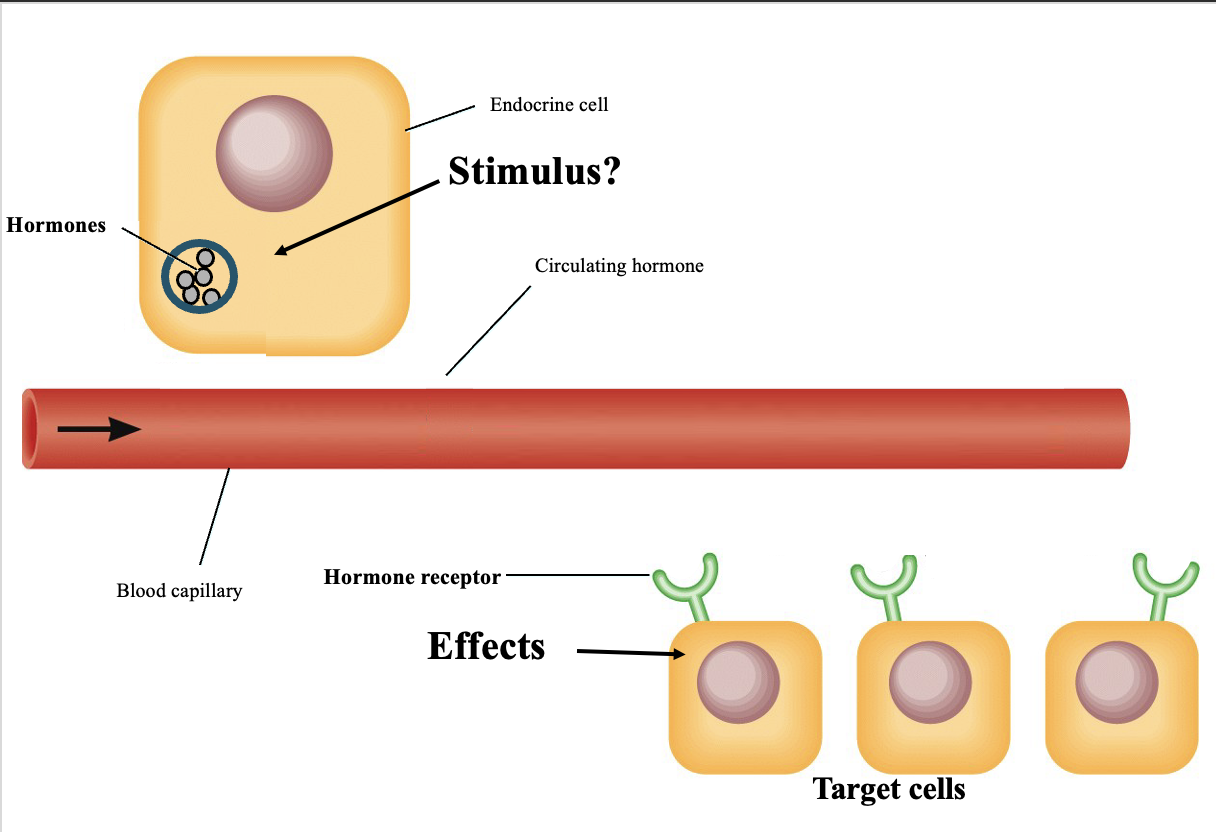
What are the three types of gland stimulation for hormonal secretion?
Endocrine glands require a stimulus to release hormones.
1. Humoral stimulation
Changes in blood glucose levels of nutrients or ions trigger hormone release
2. Neural stimulation
Nerve signals stimulate hormone release
3. Hormonal stimulation
One hormone stimulates another endocrine gland to release a hormone
Key idea:
Hormone secretion is triggered by blood chemistry, nerve signals, or other hormones.
Hormone receptors
Hormones bind to specific receptors on target cells.
Hormone receptors are proteins and fall into two main classes:
1. Intracellular receptors
Located in the cytoplasm or nucleus
Bind lipid-soluble hormones (steroids)
2. Cell membrane receptors
Located on the cell surface
Bind water-soluble hormones (peptide and amino acid–derived)
Key idea:
Only target cells with the specific receptor will respond to a hormone.
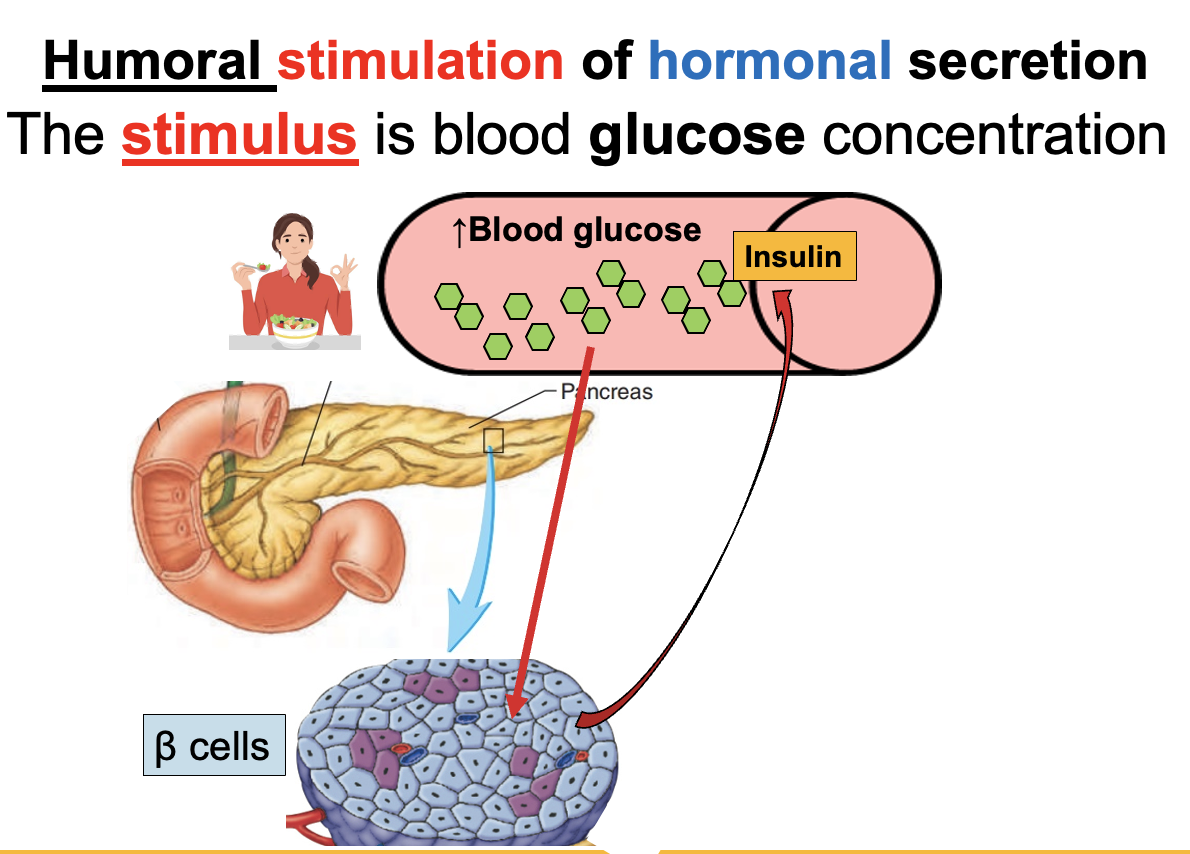
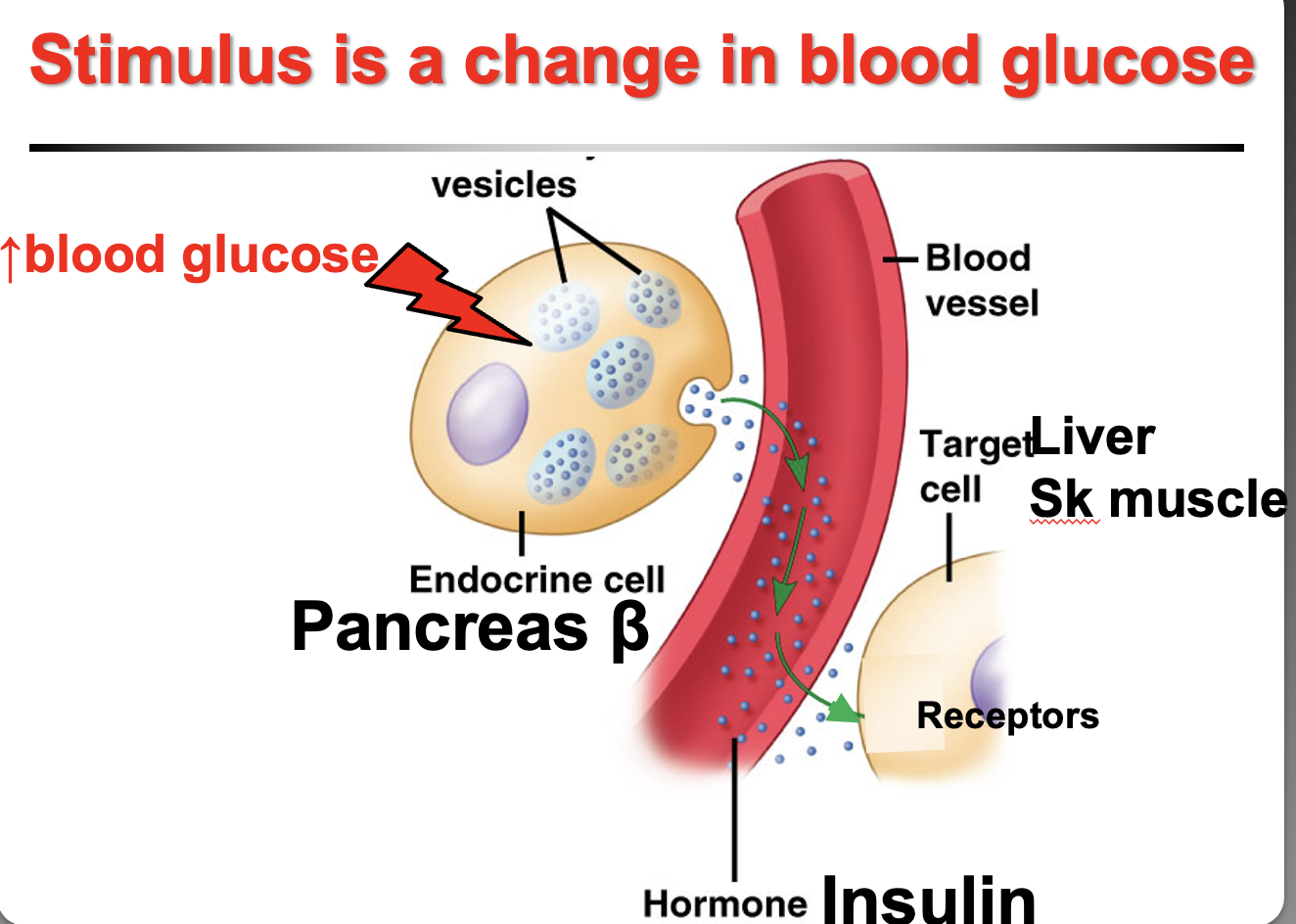
Humoral stimulation of hormone release
Humoral stimulation is the release of hormones in response to changes in blood levels of ions or nutrients (e.g., ↑ or ↓ glucose).
Examples:
↑ Blood glucose (after eating) → pancreas releases insulin
↓ Blood glucose (fasting) → pancreas releases glucagon
Key idea:
Changes in blood chemistry directly stimulate endocrine glands to release hormones.
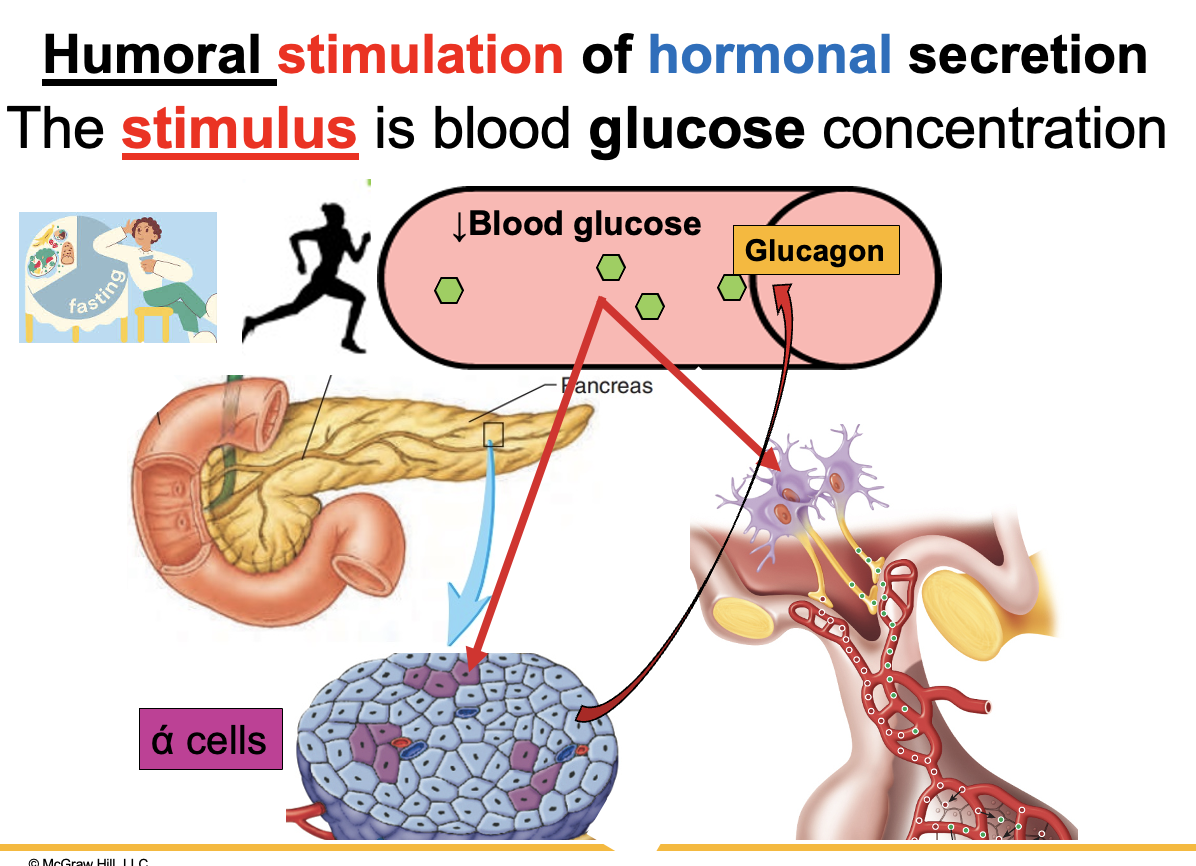
humoral stimulation by low blood glucose
Low blood glucose (hypoglycemia) acts as the stimulus.
Causes:
Fasting
Prolonged aerobic exercise
Response:
Alpha (α) cells of the pancreas release glucagon
Neurons in the hypothalamus are also stimulated
Result:
Glucagon acts to increase blood glucose levels to maintain homeostasis.
How do hyperglycemia and hypoglycemia regulate pancreatic hormone release?
Changes in blood glucose stimulate the pancreas by humoral stimulation (because glucose is a substance in blood, not a hormone).
Hyperglycemia (high blood glucose):
Stimulates β cells of the pancreas and neurons in the hypothalamus
Release insulin (protein hormone)
Insulin receptors are on the plasma membrane
Effect: lowers blood glucose
Hypoglycemia (low blood glucose):
Stimulates α cells of the pancreas
Release glucagon (protein hormone)
Effect: raises blood glucose back to normal
Key idea:
Changes in blood glucose outside the normal range stimulate pancreatic cells to restore homeostasis.
only steroid hormones have neurotransmitter receptors in the nucleus
protein and some amino acid derived have receptors in the plasma membrane
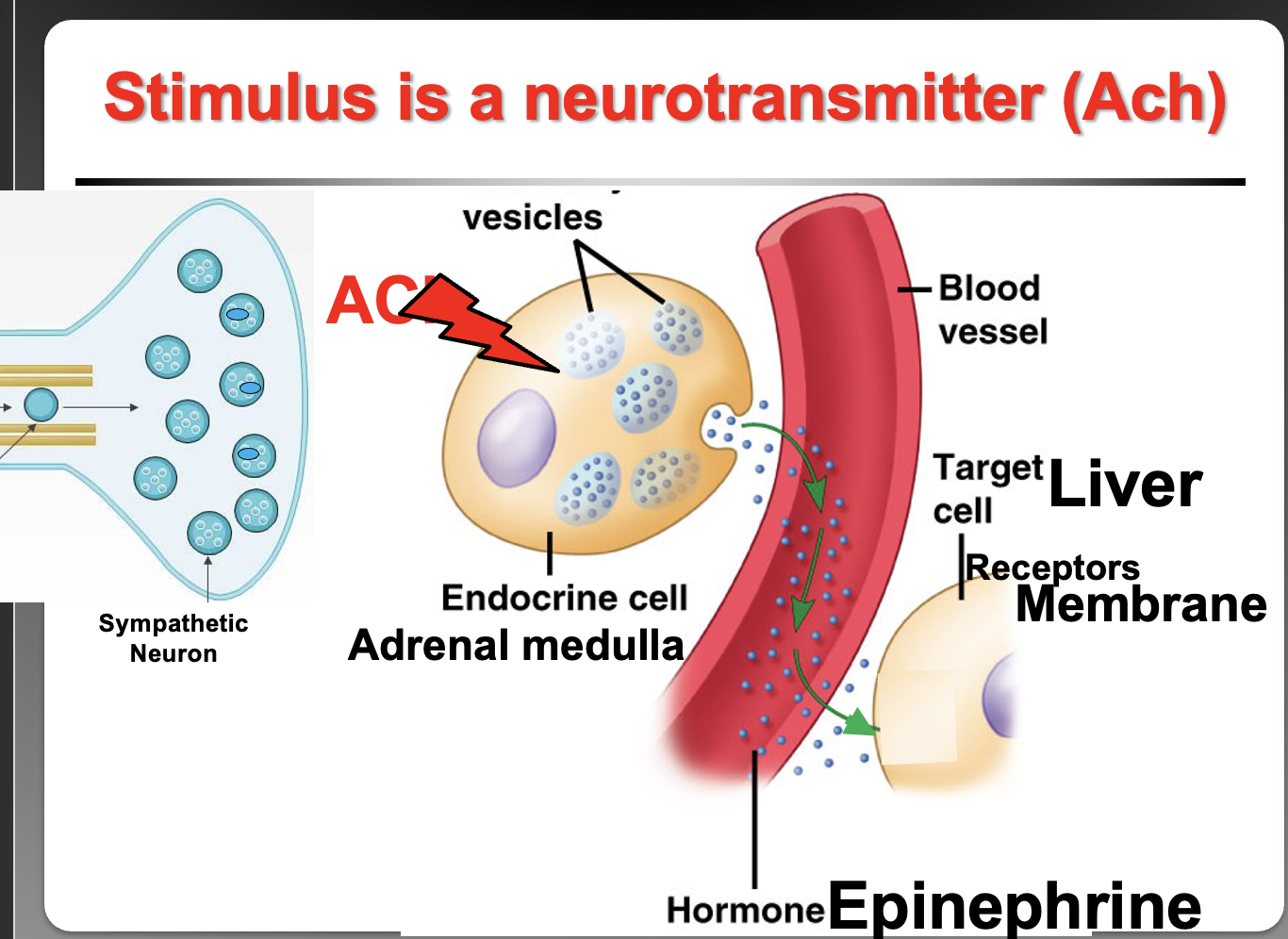
How does the adrenal medulla release catecholamines and where are epinephrine receptors located?
Acetylcholine (ACh) from sympathetic neurons stimulates the adrenal medulla → this is neural control of hormone secretion. The released catecholamines (epinephrine/norepinephrine) bind plasma membrane receptors because epinephrine is an amino acid–derived hormone and cannot diffuse through the lipid bilayer.
What type of endocrine control occurs when ACTH stimulates the adrenal cortex to release cortisol?
Hormonal stimulation. One hormone (ACTH from the anterior pituitary) stimulates another endocrine gland (adrenal cortex) to secrete a hormone (cortisol).
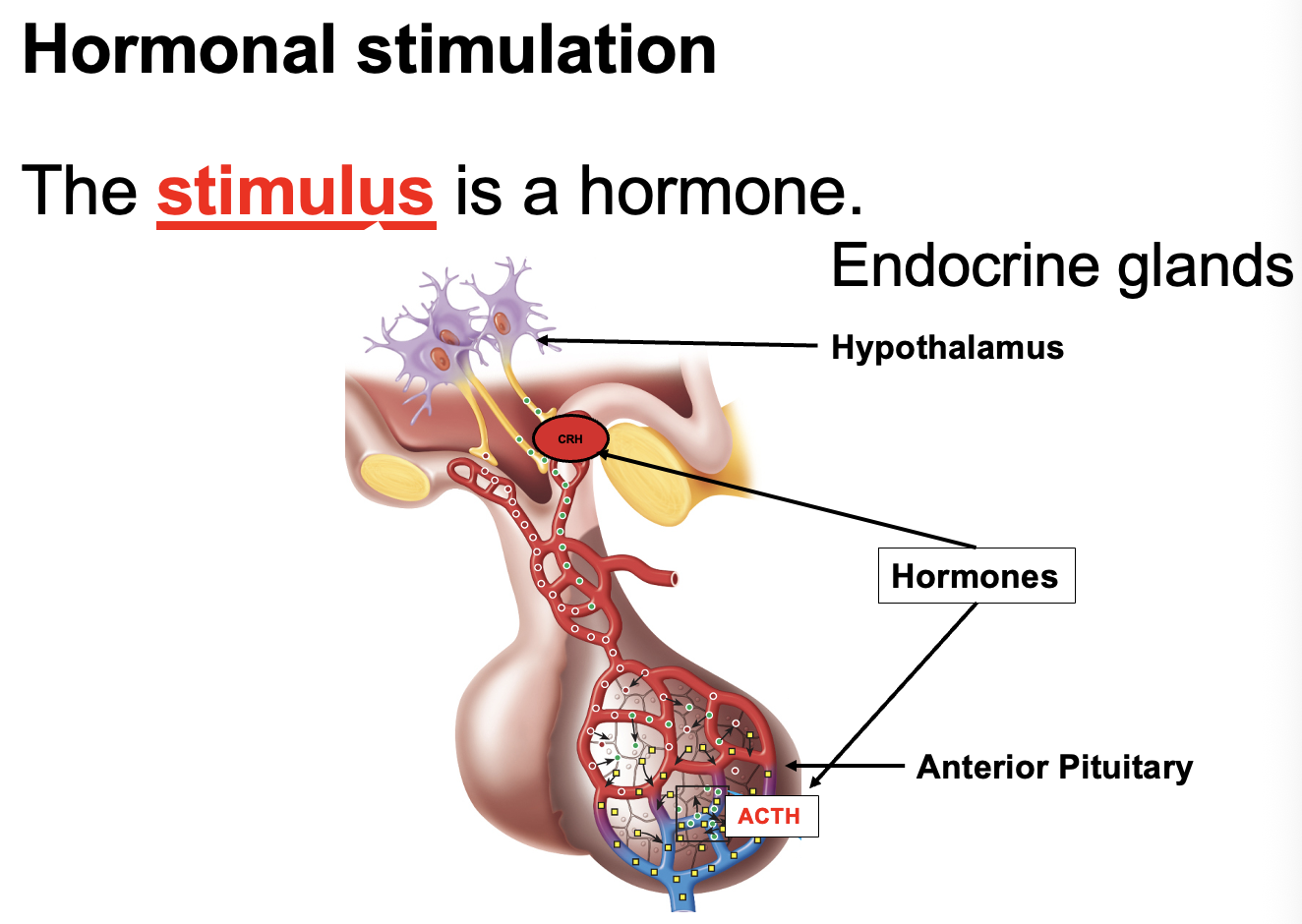
What type of endocrine stimulation occurs when ACTH stimulates the adrenal cortex to release cortisol?
Hormonal stimulation — one hormone (ACTH (Peptide) from the anterior pituitary) stimulates another endocrine gland (adrenal cortex) to secrete a hormone (cortisol) - steroid.
How does ACTH stimulate cortisol release and where are cortisol receptors located?
ACTH from the anterior pituitary stimulates the adrenal cortex to release cortisol → this is hormonal stimulation (one hormone stimulating another gland). Cortisol is a steroid hormone, so it diffuses through the membrane and binds intracellular receptors inside the cell.
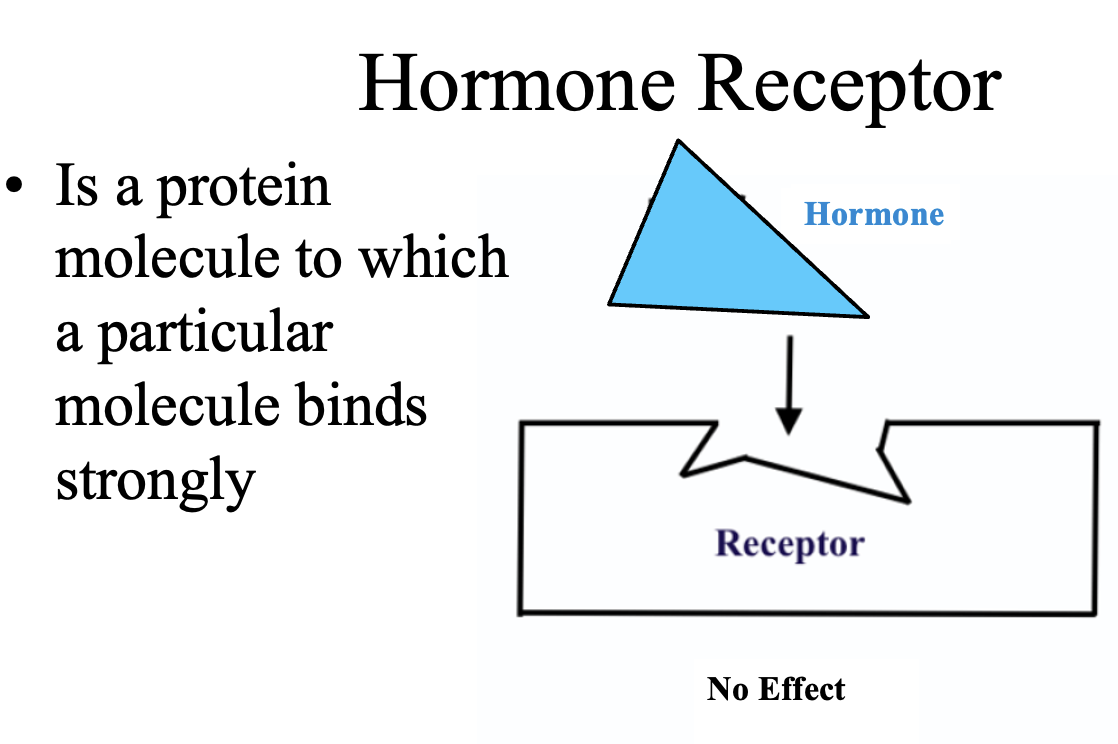
What is a hormone receptor?
A hormone receptor is a protein molecule that specifically binds a hormone with high affinity. Only cells that have the correct receptor for a hormone will respond to it; cells without the receptor will have no effect.
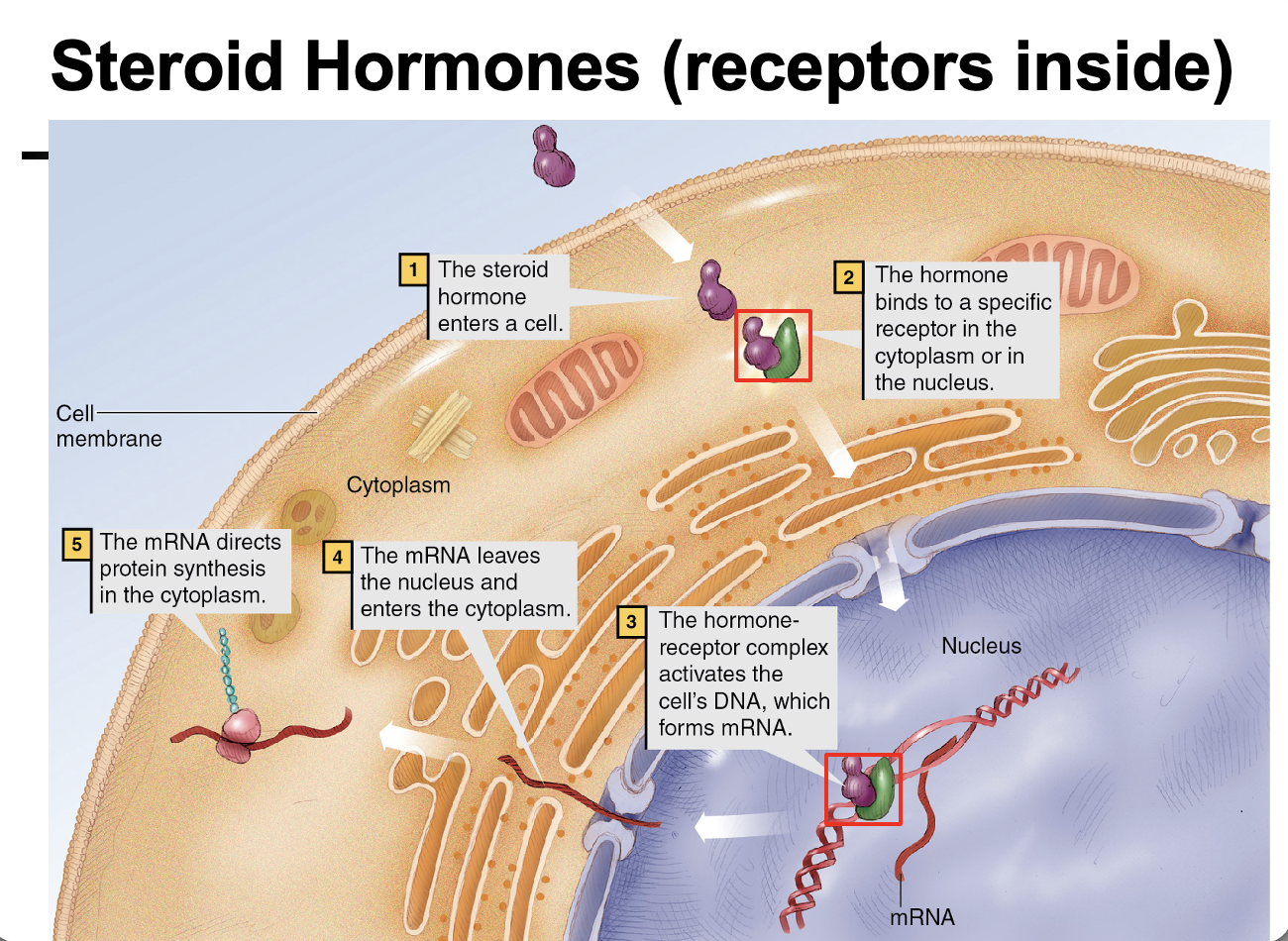
How do steroid hormones exert their effects on target cells?
Steroid hormones are lipid-soluble, so they diffuse through the cell membrane and bind intracellular receptors in the cytoplasm or nucleus. The hormone–receptor complex enters the nucleus, binds DNA, and regulates gene transcription → mRNA synthesis → protein synthesis.
What is the general mechanism of action of steroid hormones?
Steroid hormones are lipid-soluble and diffuse through the cell membrane → bind intracellular receptors in the cytoplasm or nucleus → the hormone–receptor complex enters the nucleus and binds DNA → activates gene transcription → mRNA production → protein synthesis. Example: testosterone binds receptors in skeletal muscle to increase protein synthesis and cause muscle hypertrophy.
What is the mechanism of action of steroid hormones?
Lipid-soluble steroid hormones diffuse through the cell membrane → bind intracellular receptors (cytoplasm or nucleus) → hormone–receptor complex binds DNA → activates gene transcription → mRNA production → protein synthesis (e.g., testosterone increases protein synthesis in skeletal muscle causing hypertrophy).
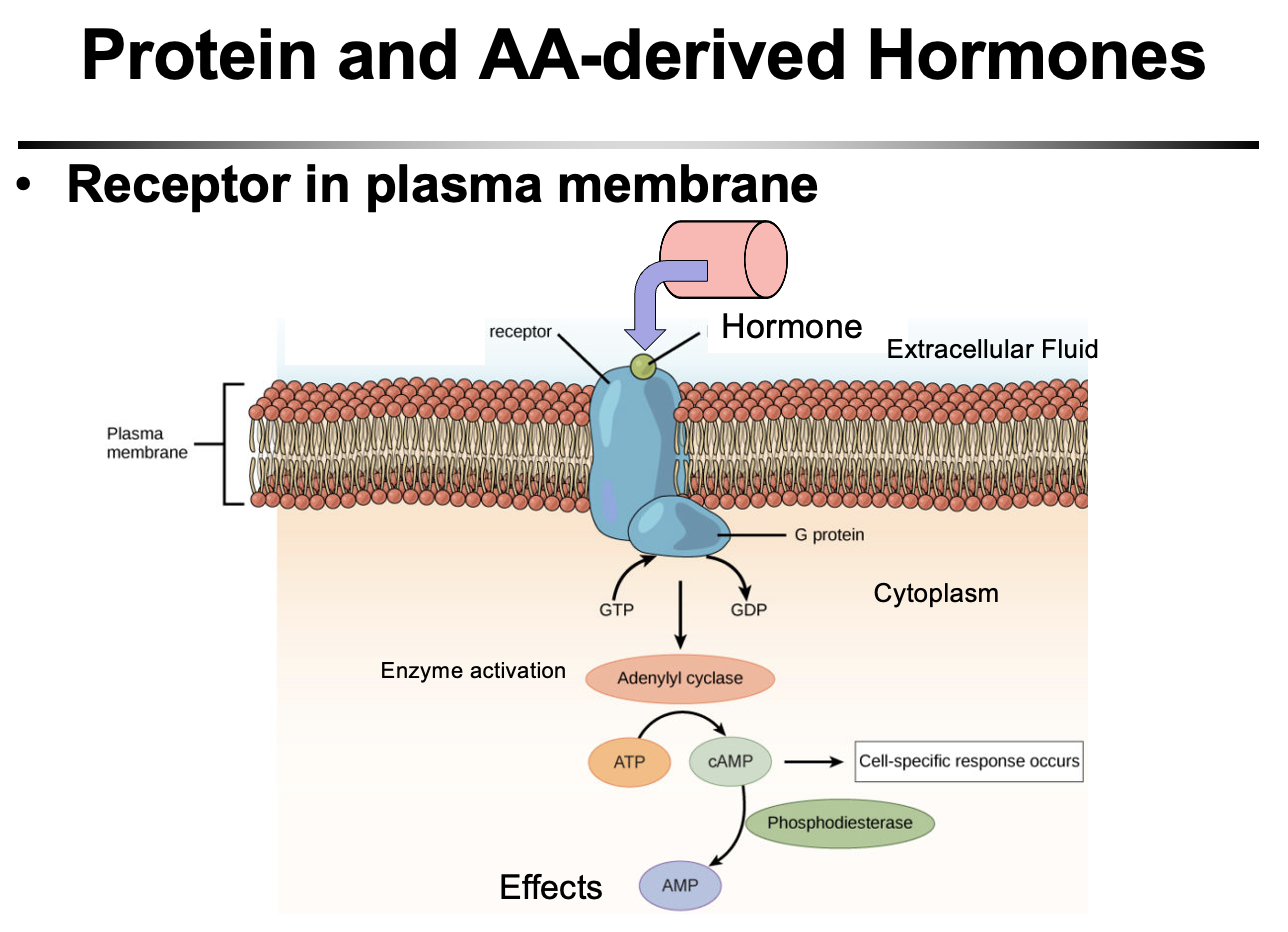
What is the mechanism of action of protein and amino acid–derived hormones?
Protein and amino acid–derived hormones cannot cross the cell membrane, so they bind receptors on the plasma membrane → activate second messenger systems (e.g., cAMP) inside the cell → trigger cellular responses. Their effects are faster than steroid hormones because they act through signaling cascades rather than gene transcription.
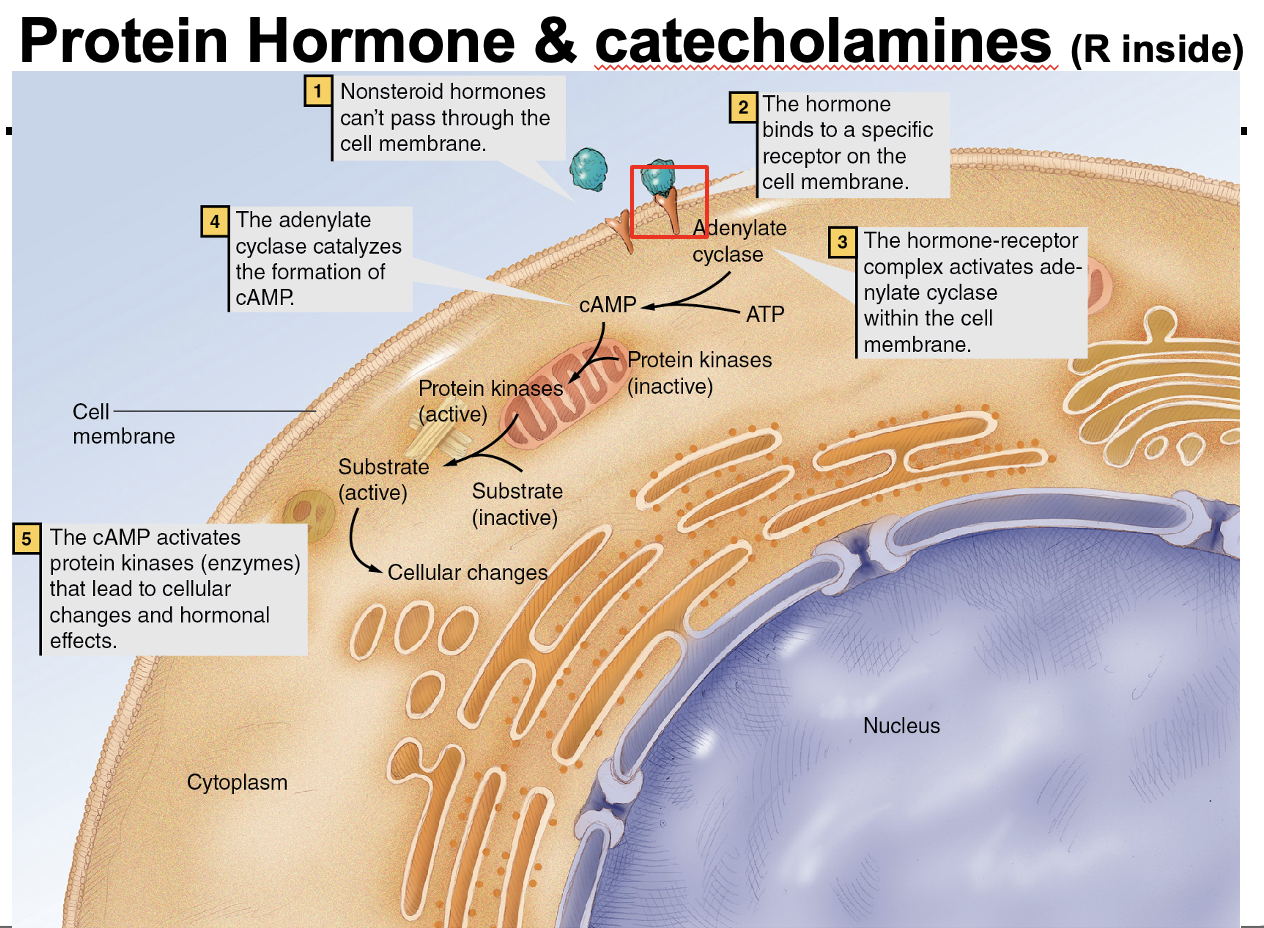
What is the mechanism of action of protein and amino acid–derived hormones?
Protein and amino acid–derived hormones are not lipid soluble, so they cannot cross the cell membrane → they bind receptors on the cell membrane → activate second messengers (e.g., cAMP) inside the cell that carry out the hormone’s effects.
What is the mechanism of action of nonsteroid (protein and amino acid–derived) hormones?
Non-steroid hormones cannot cross the cell membrane, so they bind receptors on the cell membrane → activate second messengers (e.g., cAMP) inside the cell → trigger cellular functions. Their effects are faster than steroid hormones because they act through signaling pathways rather than gene transcription.
What is the difference between synergism and antagonism in hormone action?
Synergism: two or more hormones produce similar or additive effects.
Antagonism: two or more hormones produce opposite effects.

Insulin acts alone no synergism or antagonism
Which hormones have anabolic vs catabolic effects on carbohydrates, proteins, and fats?
Insulin is anabolic → promotes glycogenesis, protein synthesis, and lipogenesis.
Glucagon, epinephrine, cortisol, and growth hormone are catabolic → promote glycogenolysis and lipolysis.
Growth hormone and testosterone promote protein synthesis (anabolic on proteins).
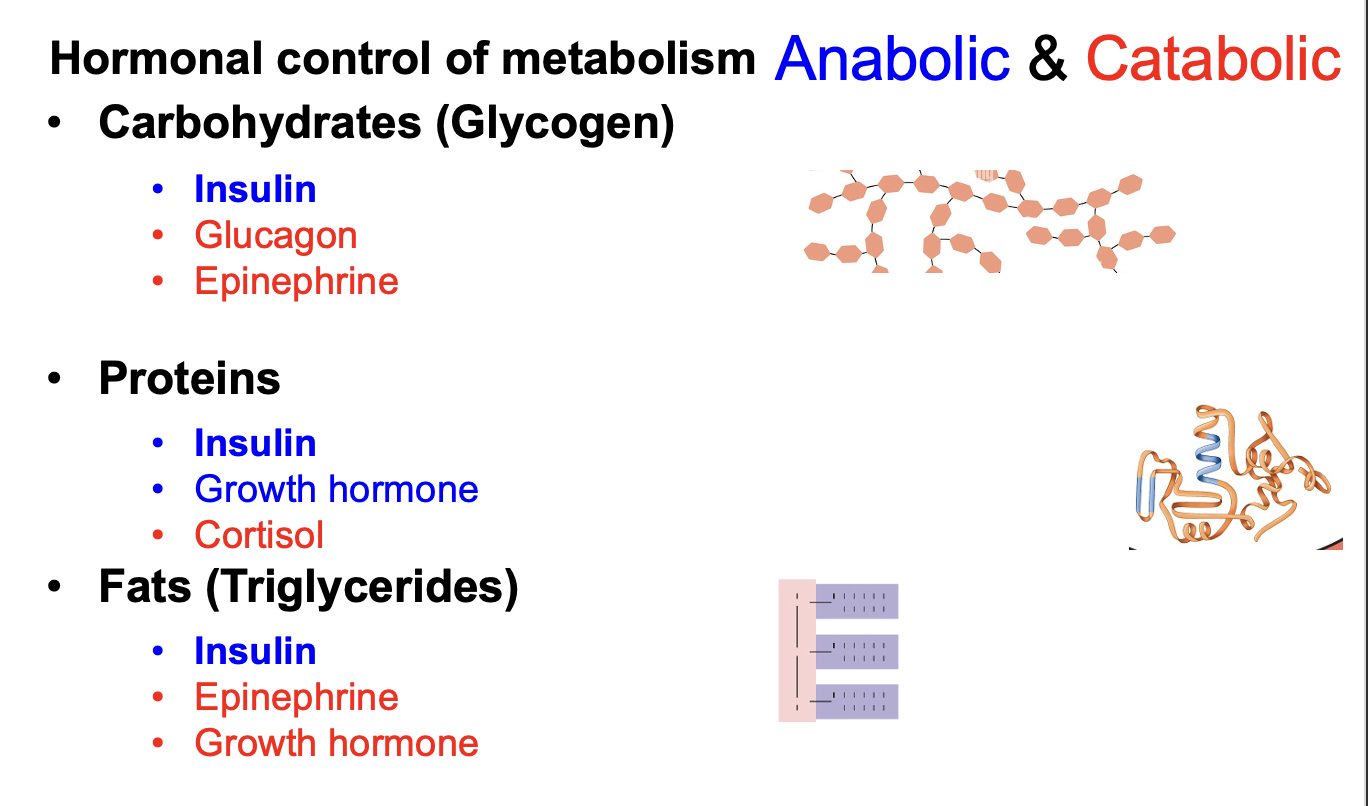
What are the anabolic vs catabolic pathways for glucose, proteins, and fats?
Glucose → Glycogenesis (anabolism) | Glycogenolysis (catabolism)
Amino acids → Proteogenesis / protein synthesis (anabolism) | Proteolysis (catabolism)
Fats → Lipogenesis (anabolism) | Lipolysis (catabolism)

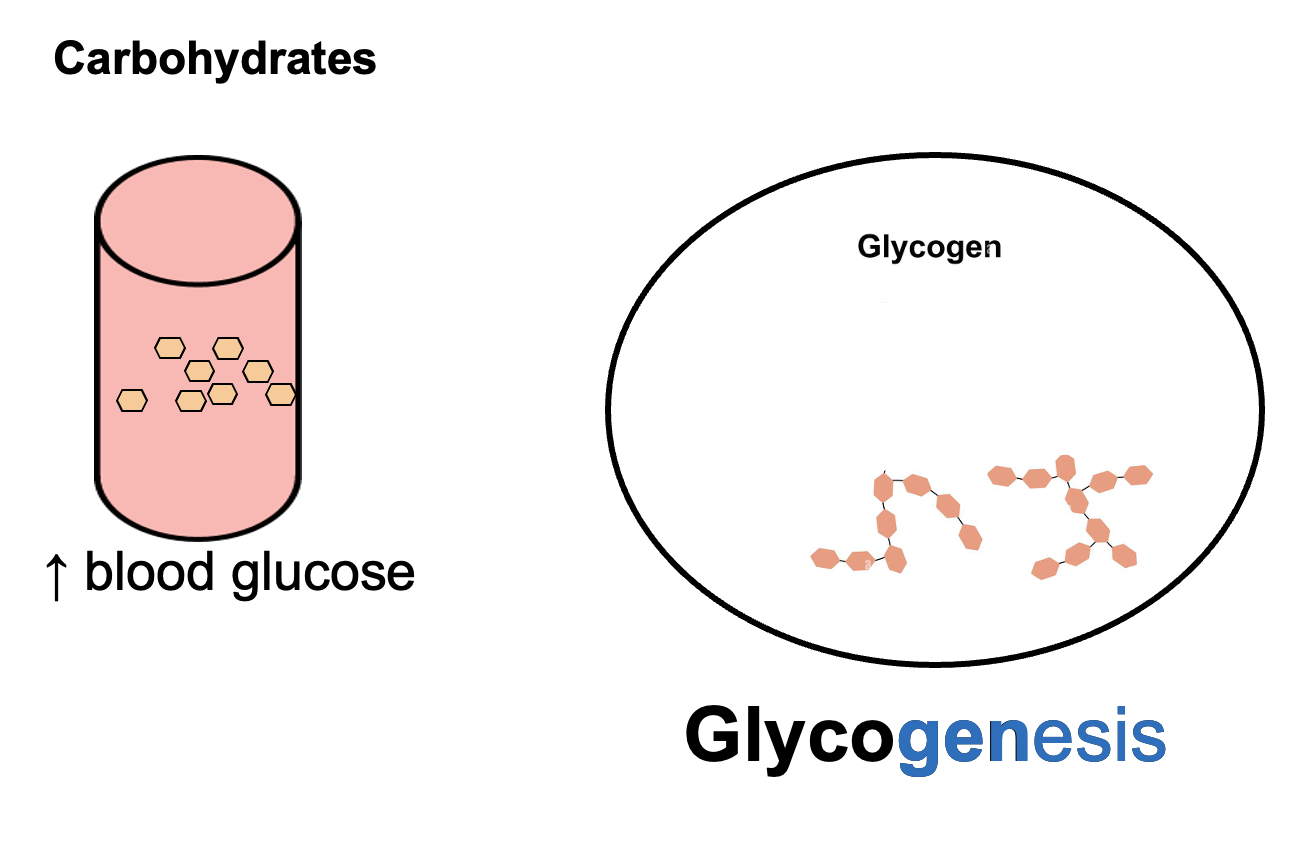
What is glycogenesis?
Glycogenesis is the anabolic process of converting glucose into glycogen for storage, typically in the liver and skeletal muscle, which helps lower blood glucose levels.
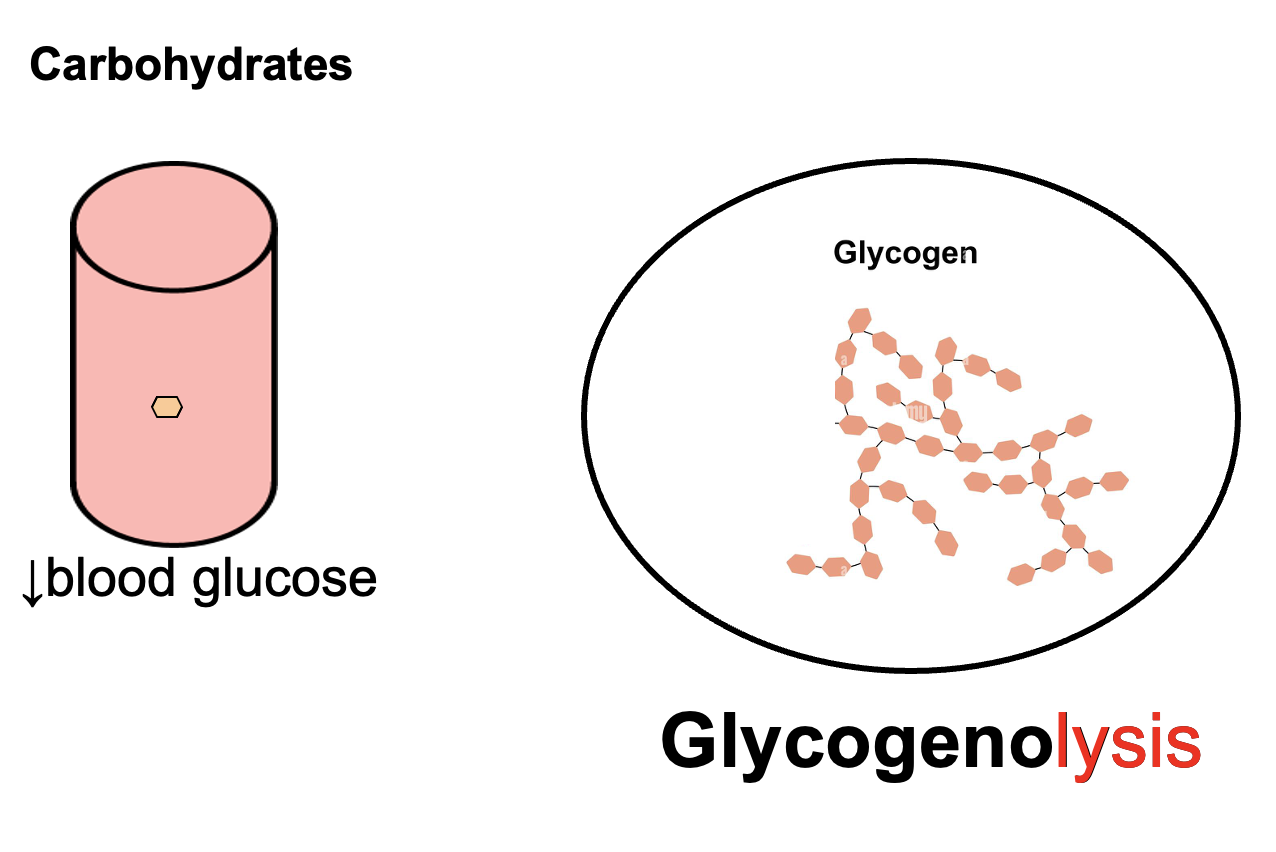
What is glycogenolysis?
Glycogenolysis is the catabolic breakdown of glycogen into glucose, primarily in the liver and skeletal muscle, which helps increase blood glucose levels.
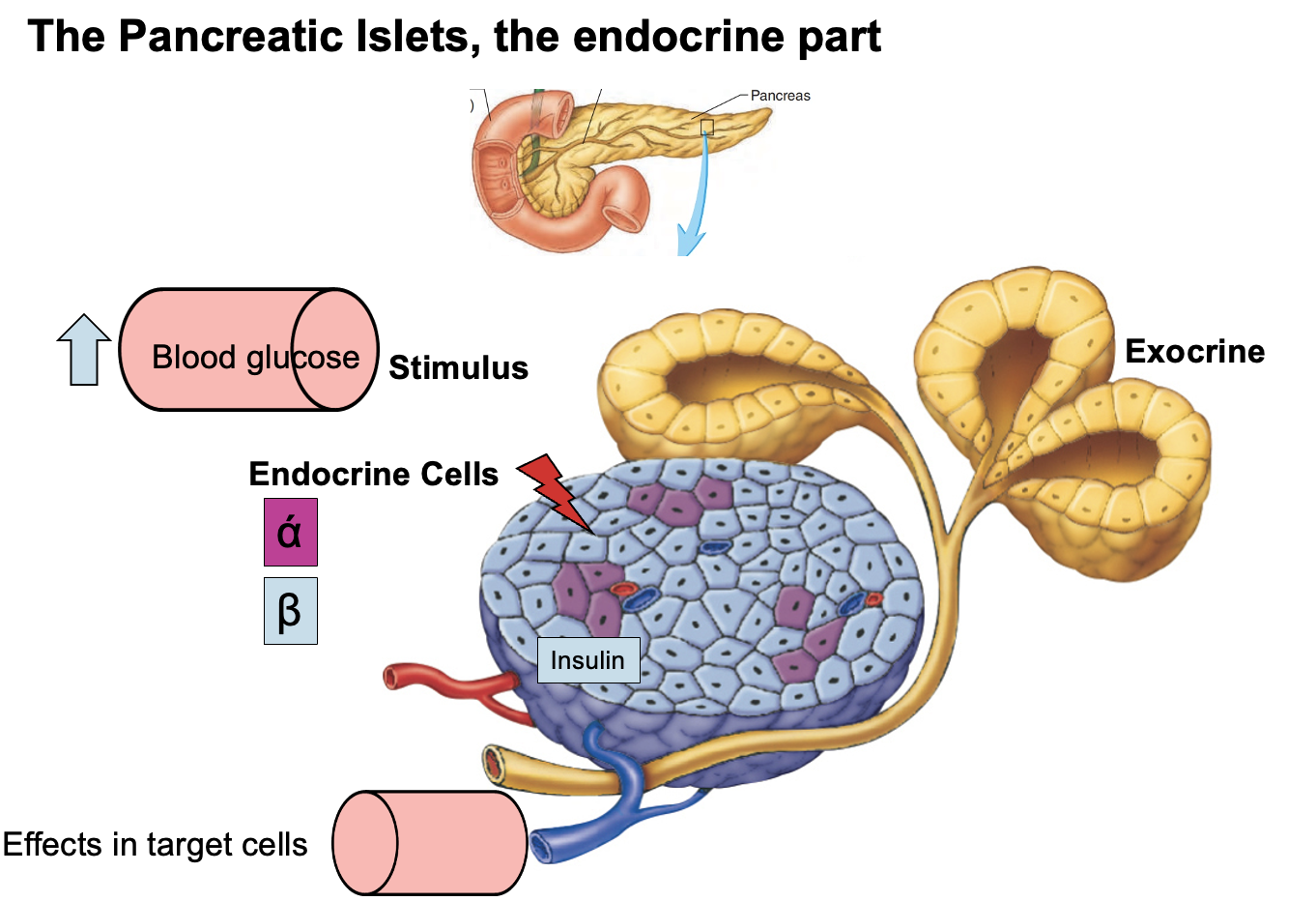
What are the exocrine and endocrine functions of the pancreas, and which pancreatic islet cells secrete insulin and glucagon?
Pancreas location
Retroperitoneal organ located behind the stomach along the duodenum (small intestine).
Exocrine pancreas
Produces pancreatic digestive juices that travel through the pancreatic duct into the duodenum.
These include:
• amylase → carbohydrate digestion
• lipase → fat digestion
• proteases (trypsin, chymotrypsin) → protein digestion
• bicarbonate → neutralizes stomach acid
Endocrine pancreas
Consists of pancreatic islets (Islets of Langerhans) that release hormones directly into the bloodstream.
Islet cell types:
• Alpha (α) cells → secrete glucagon → increase blood glucose
• Beta (β) cells → secrete insulin → decrease blood glucose
High-yield rule:
Beta brings glucose down (insulin)
Alpha adds glucose (glucagon)
What happens after eating when blood glucose rises, and how does the pancreas regulate it?
The pancreas has two functional components.
Exocrine pancreas
Secretes digestive (pancreatic) juices into the duodenum to aid digestion.
Endocrine pancreas (islets of Langerhans)
Secretes hormones into the bloodstream.
After eating:
Elevated blood glucose stimulates beta (β) cells of the pancreatic islets to release insulin.
Insulin travels through the blood to its target tissues:
• Skeletal muscle
• Liver
• Adipose tissue
Effect of insulin:
Promotes glucose uptake and storage, which lowers blood glucose back to normal.
Key principle:
This is negative feedback, where the hormone response opposes the original stimulus (high blood glucose).
What hormone is released during prolonged fasting or prolonged aerobic exercise, and how does it restore blood glucose levels?
During prolonged fasting (> 8 hours) or prolonged aerobic exercise (> 1.5 hours), blood glucose decreases (hypoglycemia).
This stimulates alpha (α) cells of the pancreatic islets to secrete glucagon.
Glucagon travels through the bloodstream to its main target organ:
Liver
Effect of glucagon on liver cells:
• stimulates glycogenolysis (breakdown of glycogen)
• stimulates gluconeogenesis (production of new glucose)
Result:
Blood glucose increases back toward normal levels.
Key principle:
This is negative feedback, where the hormone response opposes the original stimulus (low blood glucose).
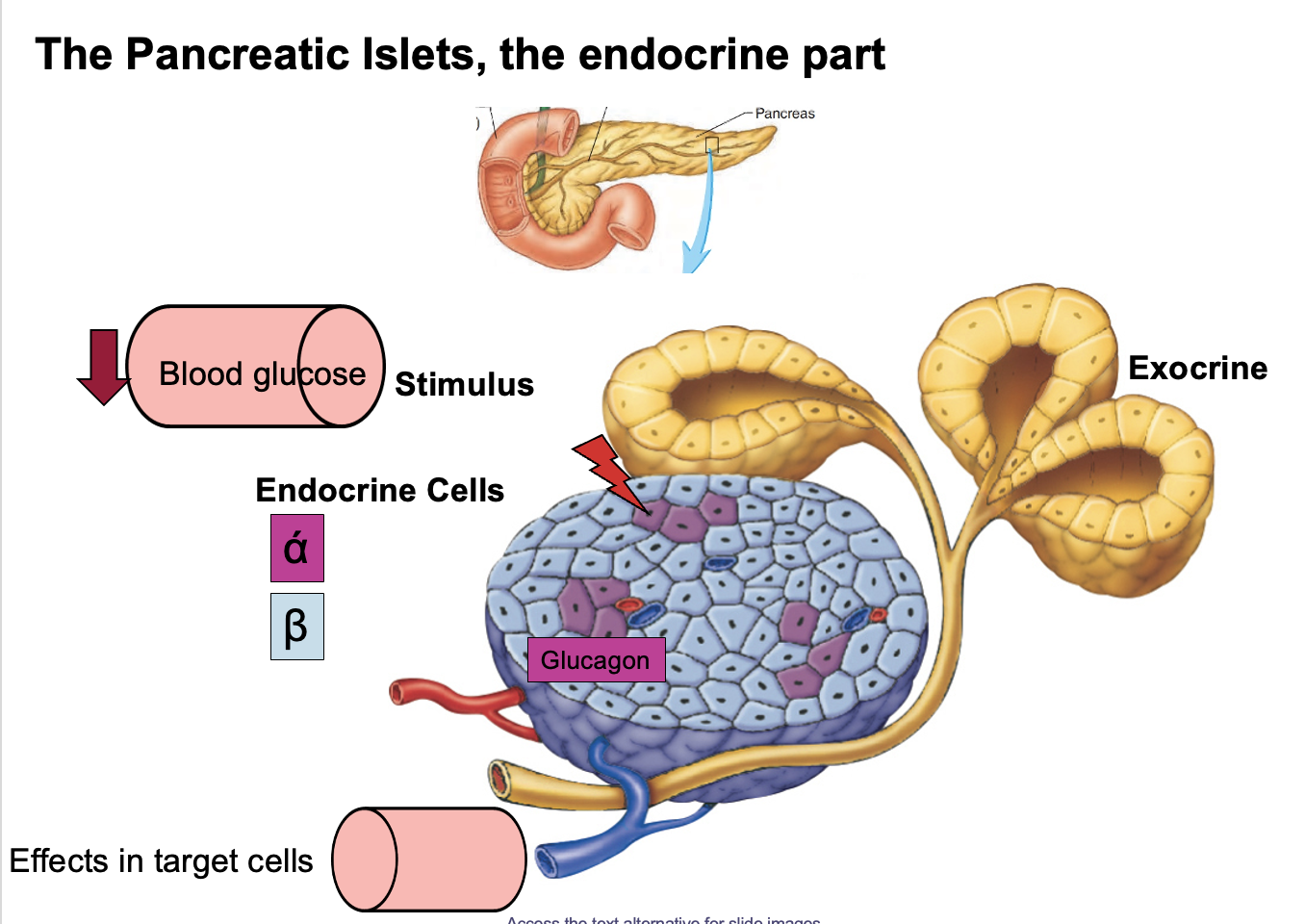
Which hormones oppose insulin in blood glucose regulation, when are they released, and which organ is the primary site of glucose uptake?
Insulin lowers blood glucose, while four hormones increase blood glucose.
Hormones that oppose insulin:
• Glucagon
• Growth hormone
• Cortisol
• Epinephrine
Relationship:
These hormones are antagonistic to insulin because they have opposite effects on blood glucose.
When they are released:
After eating (high blood glucose)
→ Insulin is released to lower blood glucose.
During prolonged fasting or prolonged exercise (low blood glucose)
→ Glucagon, growth hormone, cortisol, and epinephrine are released to increase blood glucose.
Primary organ for glucose uptake:
Skeletal muscle
About 53% of glucose uptake and storage occurs in skeletal muscle, where glucose is stored as glycogen.
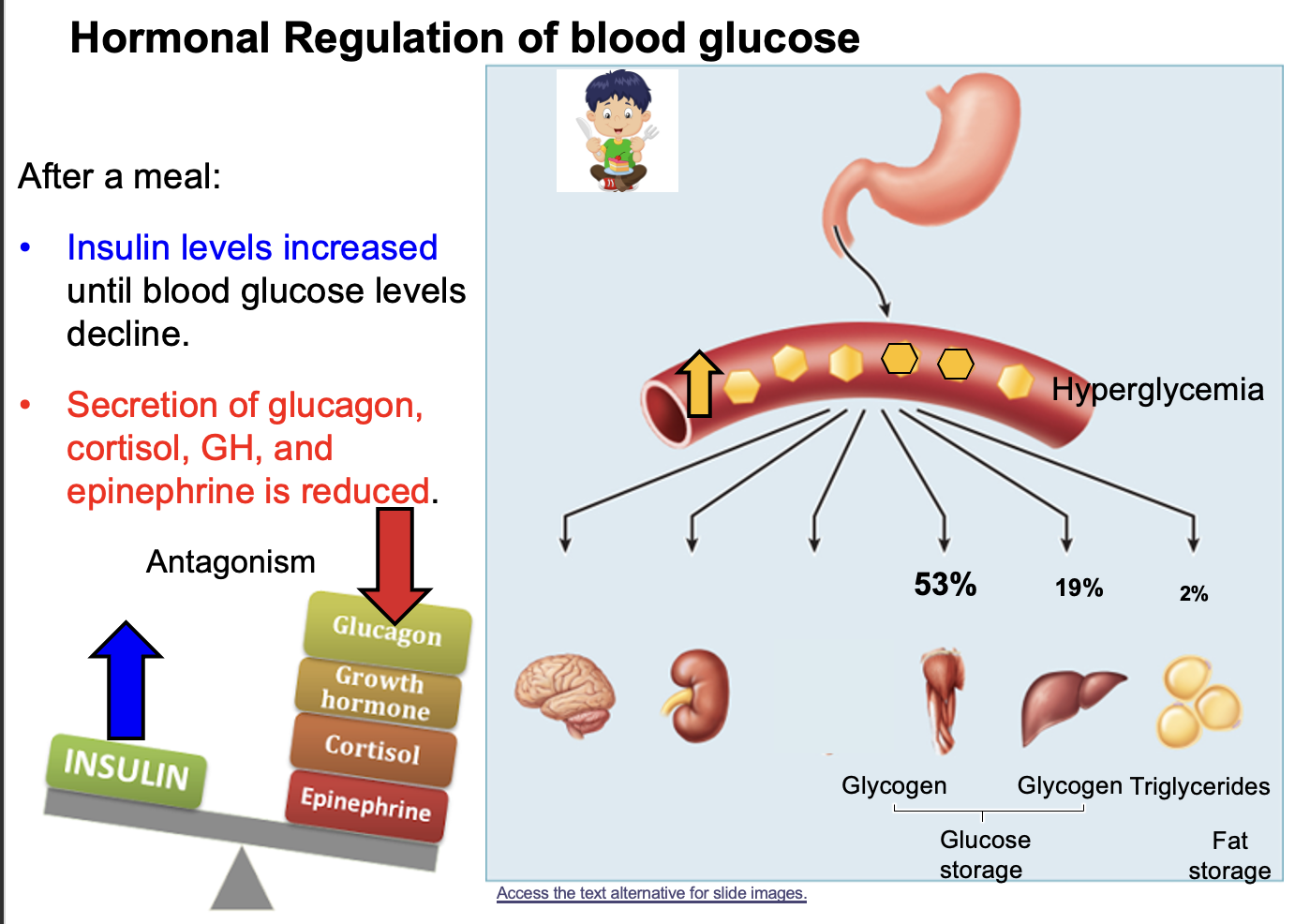
After digestion, where are carbohydrates, proteins, and fats absorbed, and what happens to blood glucose about 30 minutes after eating?
After digestion, carbohydrates, proteins, and fats are absorbed in the small intestine, primarily in the jejunum and ileum.
About 30 minutes after eating, blood glucose levels increase significantly due to the absorption of glucose from digested carbohydrates into the bloodstream.
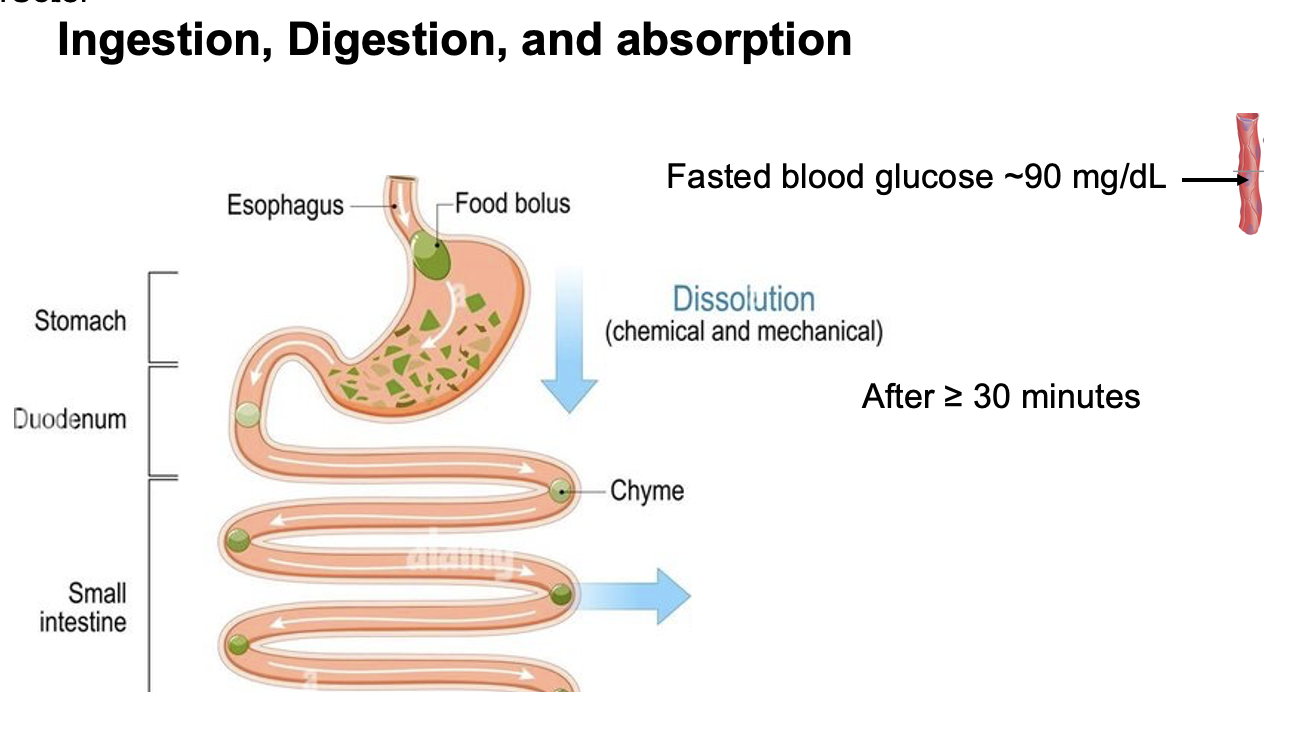
What stimulates pancreatic β-cells, and what hormone do they secrete?
In the pancreatic islets (Islets of Langerhans), β-cells are stimulated by an increase in blood glucose.
In response, β-cells secrete insulin, which acts to lower blood glucose by promoting glucose uptake and storage in target tissues (especially skeletal muscle, liver, and adipose tissue).
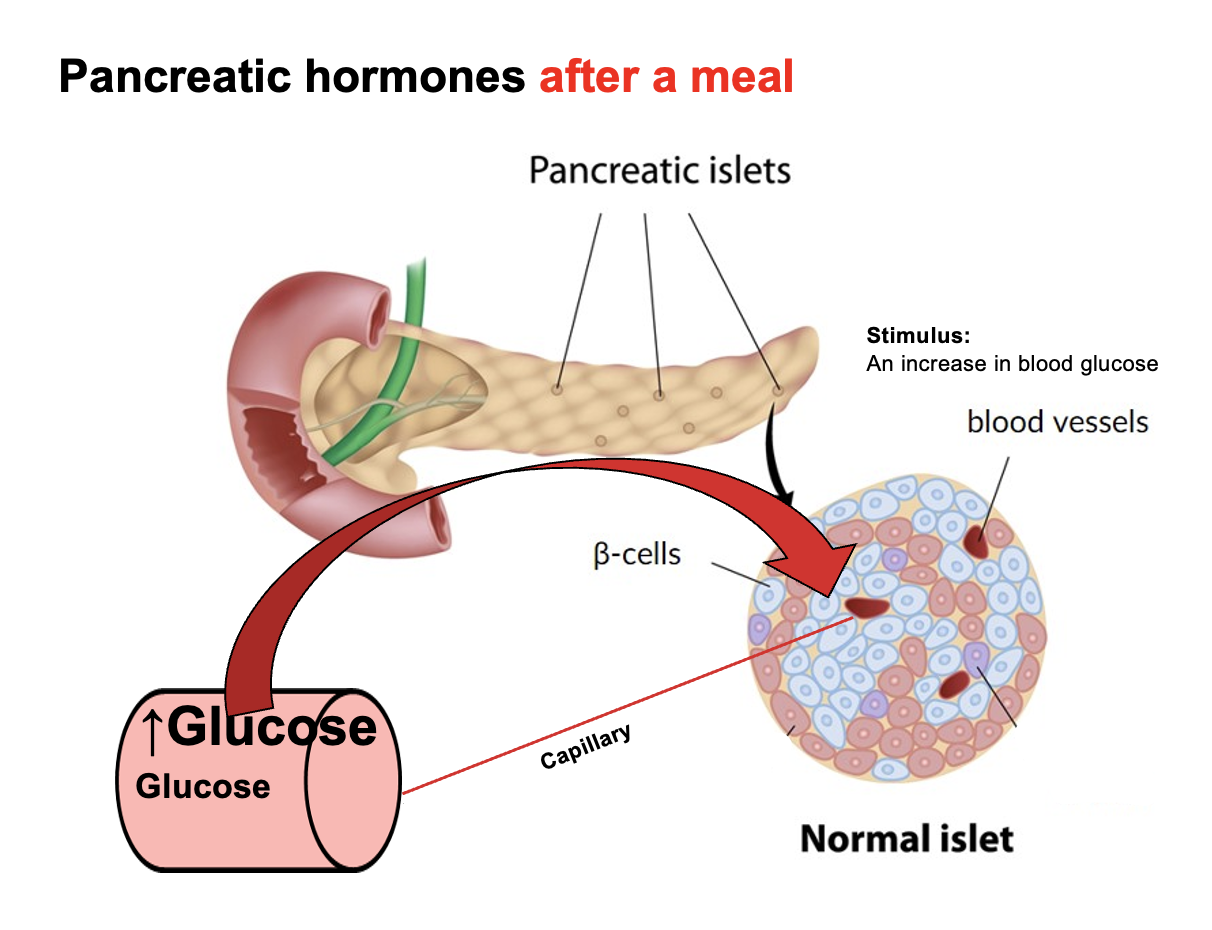
What happens to blood glucose and insulin levels after eating a carbohydrate-containing meal in a healthy person?
After eating a meal containing carbohydrates, blood glucose rises rapidly and peaks at about 30 minutes.
The resulting hyperglycemia stimulates pancreatic β-cells in the islets of Langerhans to secrete insulin.
Insulin then acts on target tissues (especially skeletal muscle, liver, and adipose tissue) to promote glucose uptake and storage.
As insulin works, blood glucose decreases and returns to the pre-meal level within about 1 hour after eating in a healthy person.
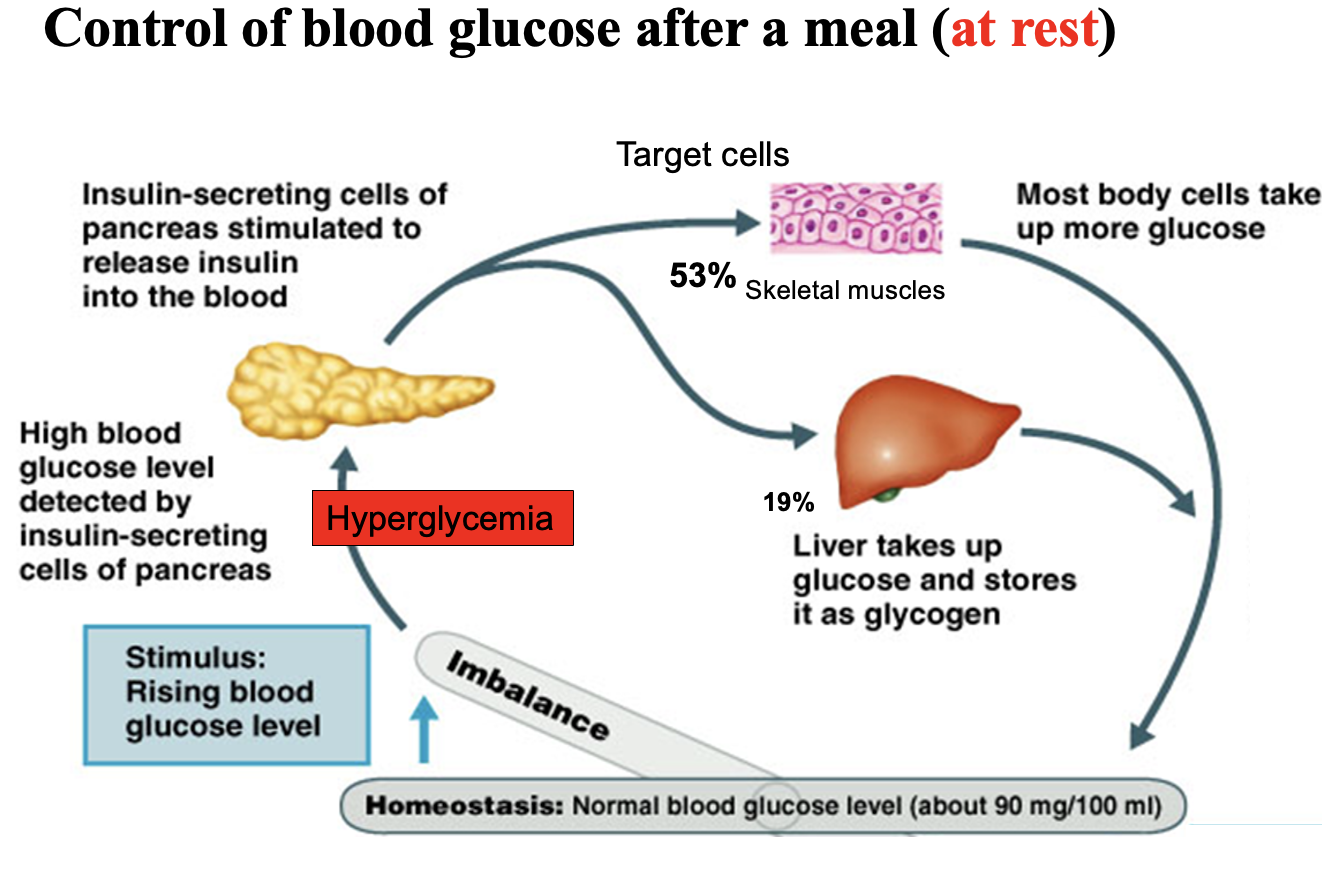
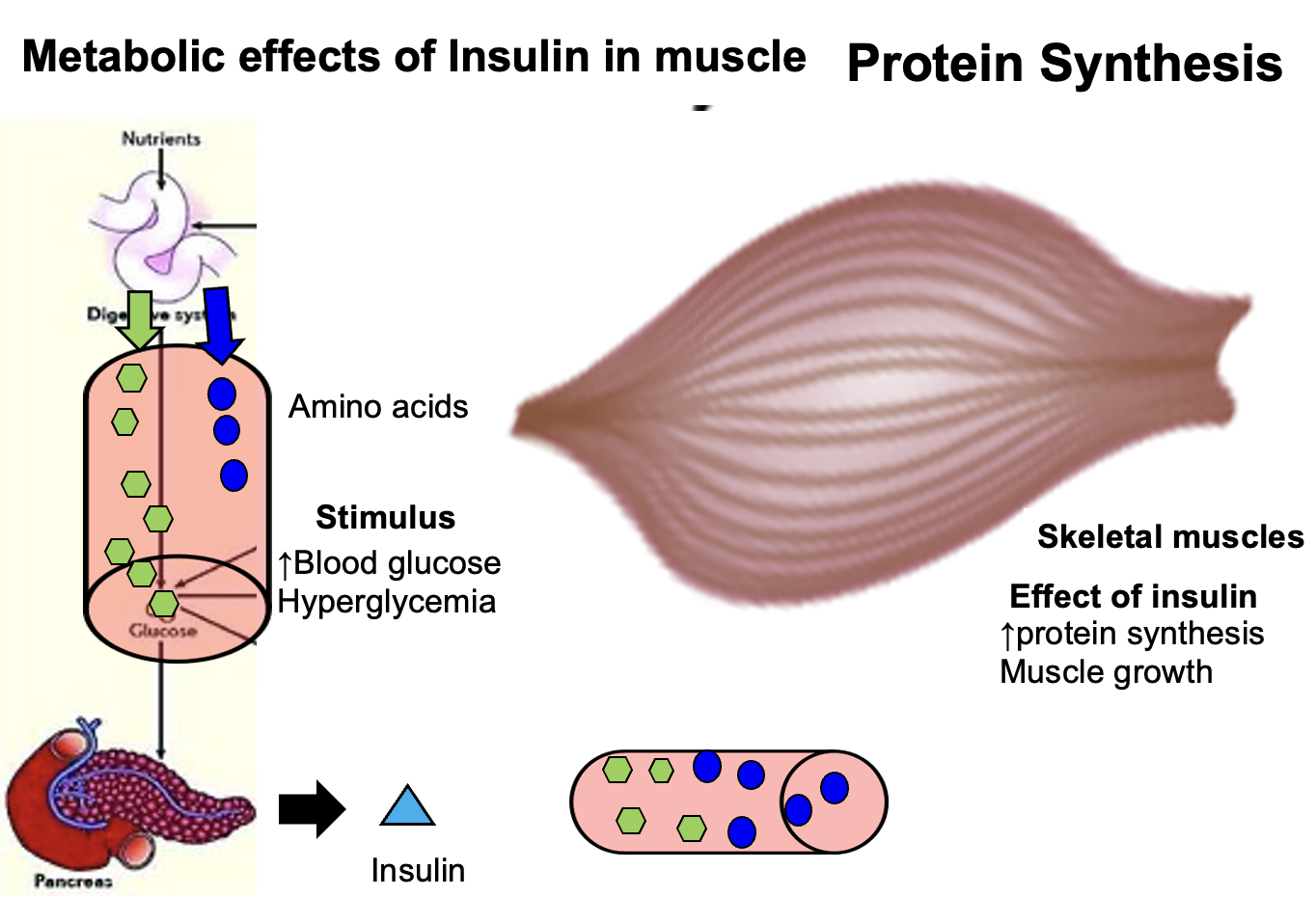
Why is insulin considered an anabolic hormone, and what energy-storage processes does it stimulate?
Insulin is an anabolic hormone because it promotes the uptake of nutrients into cells and their storage as energy.
Insulin facilitates the entry of:
• Glucose
• Fatty acids
• Amino acids
It promotes storage through three main anabolic pathways:
• Glycogenesis → glucose stored as glycogen in liver and skeletal muscle
• Lipogenesis → glucose contributes to triglyceride synthesis (glycerol backbone derived from glucose)
• Protein synthesis → amino acids used to build proteins
Overall effect:
Energy storage and decreased blood glucose.
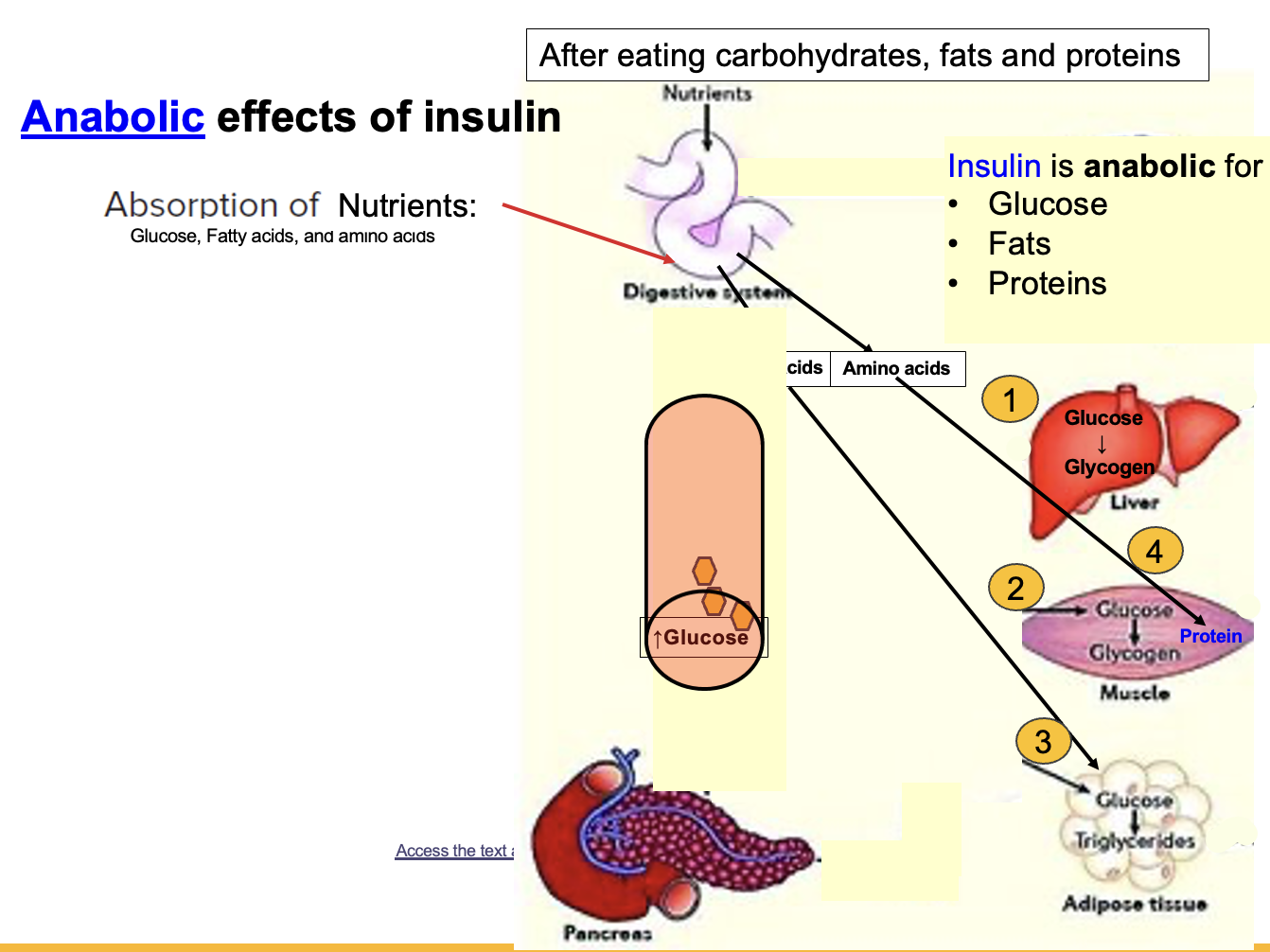
How does insulin stimulate glucose uptake into cells through GLUT-4?
GLUT-4 is a glucose transporter responsible for insulin-dependent glucose uptake in skeletal muscle and adipose tissue.
In unstimulated cells:
• GLUT-4 is stored in the cytoplasm in intracellular vesicles.
After eating:
• Blood glucose increases
• Insulin is released from pancreatic β-cells
Mechanism:
Insulin binds to its receptor on the cell membrane
The receptor sends a signal causing GLUT-4 vesicles to move to the plasma membrane
GLUT-4 inserts into the membrane
Glucose enters the cell via facilitated diffusion
Timing:
This process occurs within minutes after carbohydrate ingestion.
Key idea:
This pathway is insulin-dependent, meaning without insulin, GLUT-4 does not move to the membrane and glucose uptake by this mechanism does not occur.
Additional effect of insulin:
Insulin signaling also stimulates carrier proteins to increase amino acid uptake into muscle fibers, promoting protein synthesis.
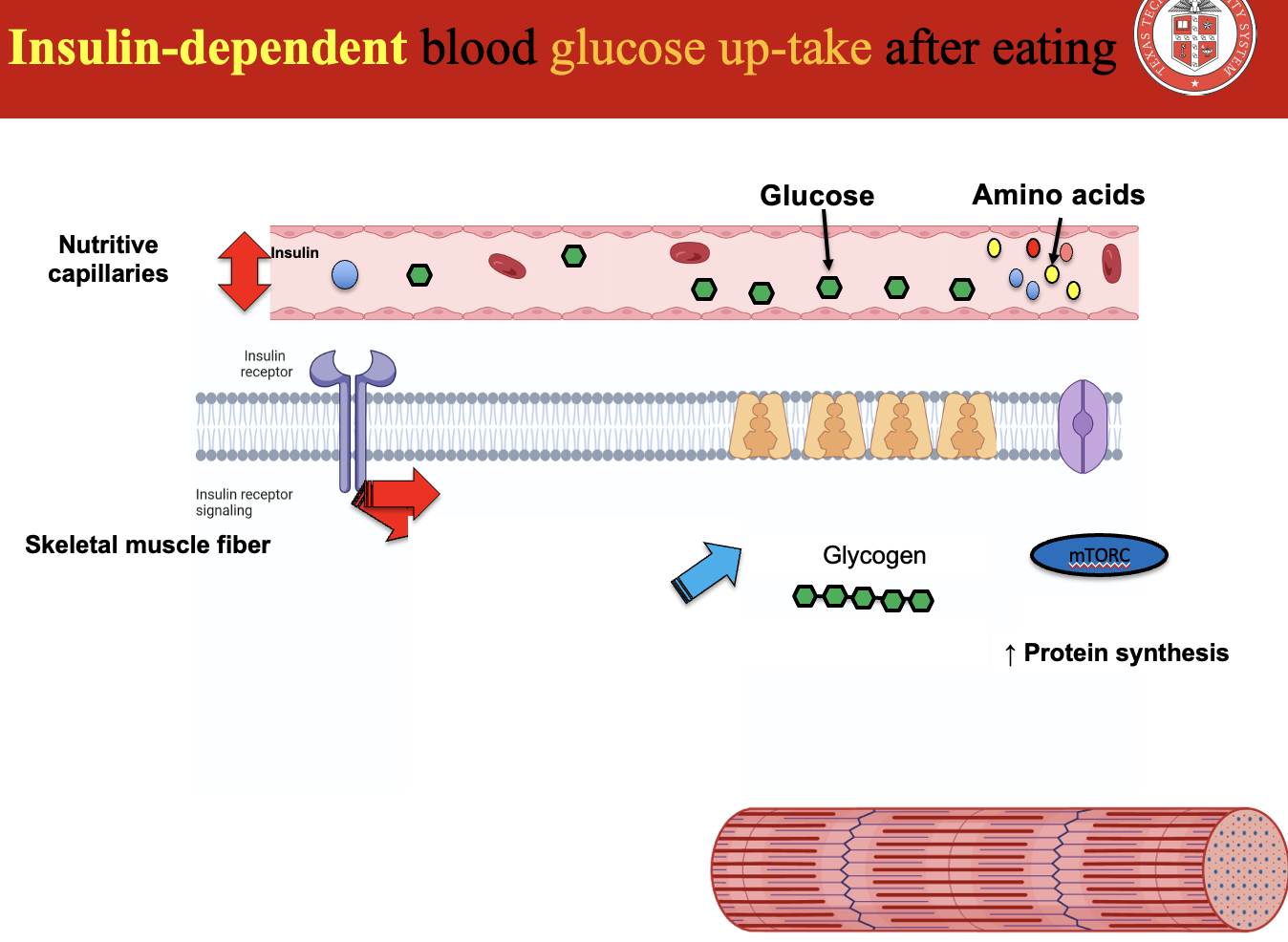
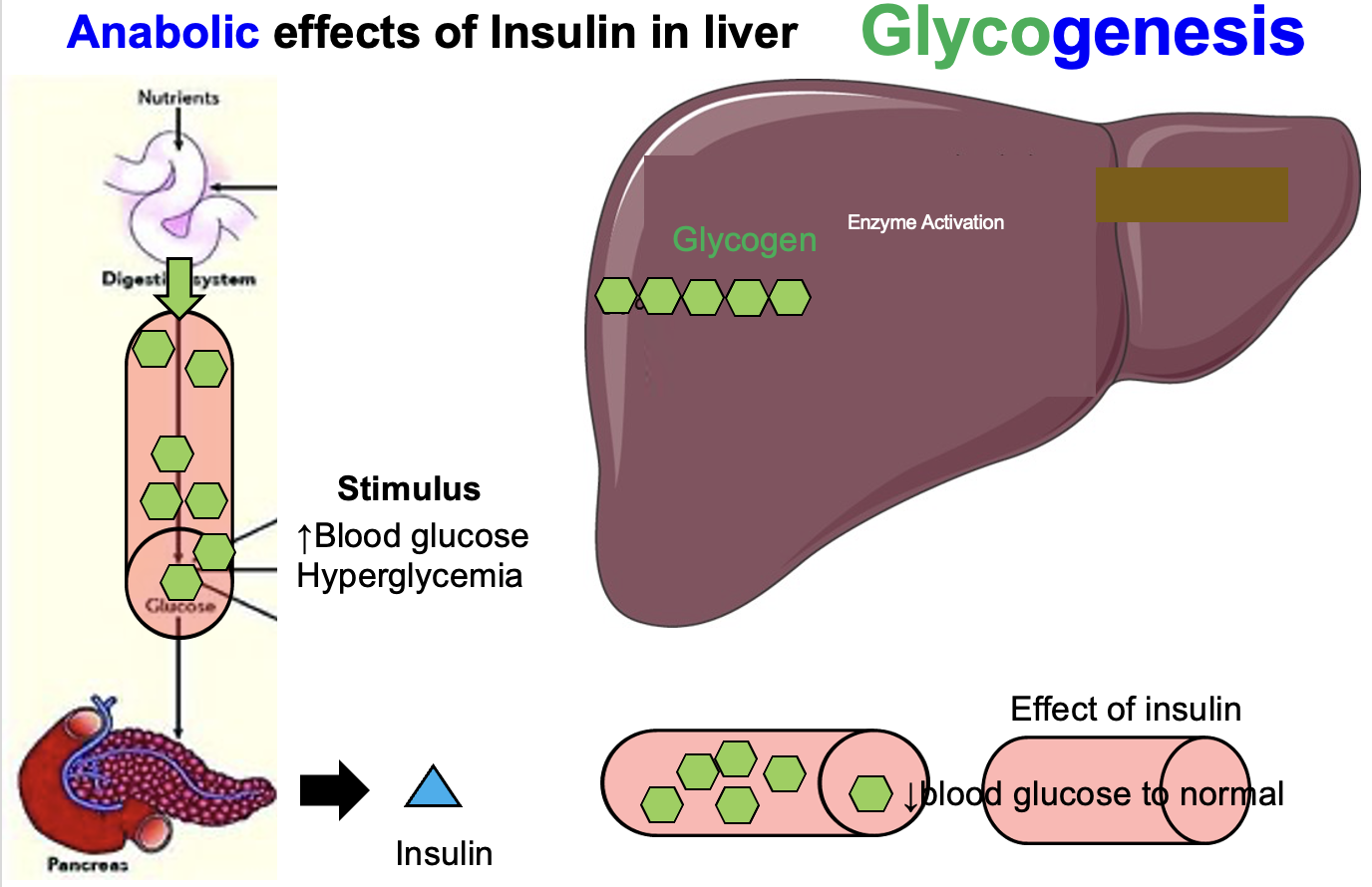
How does insulin regulate blood glucose in the liver after a meal?
After a meal, glucose absorbed from the small intestine (jejunum and ileum) enters the bloodstream and causes hyperglycemia (elevated blood glucose).
This increase in blood glucose stimulates pancreatic β-cells to secrete insulin.
Insulin acts on liver cells (hepatocytes) by stimulating the facilitated diffusion of glucose into the cells.
Inside hepatocytes:
• glucose is converted into glycogen
• this process is called glycogenesis
Result:
Excess glucose is stored in the liver as glycogen, which reduces blood glucose back to normal within about 30–60 minutes after a meal.
What effect does insulin have on muscle protein synthesis when amino acid levels are high?
When amino acid levels are high, insulin stimulates protein synthesis in skeletal muscle.
Insulin promotes:
• amino acid uptake into muscle cells
• activation of pathways that increase protein synthesis
Result:
Muscle proteins are built from amino acids, contributing to anabolic growth and tissue repair.
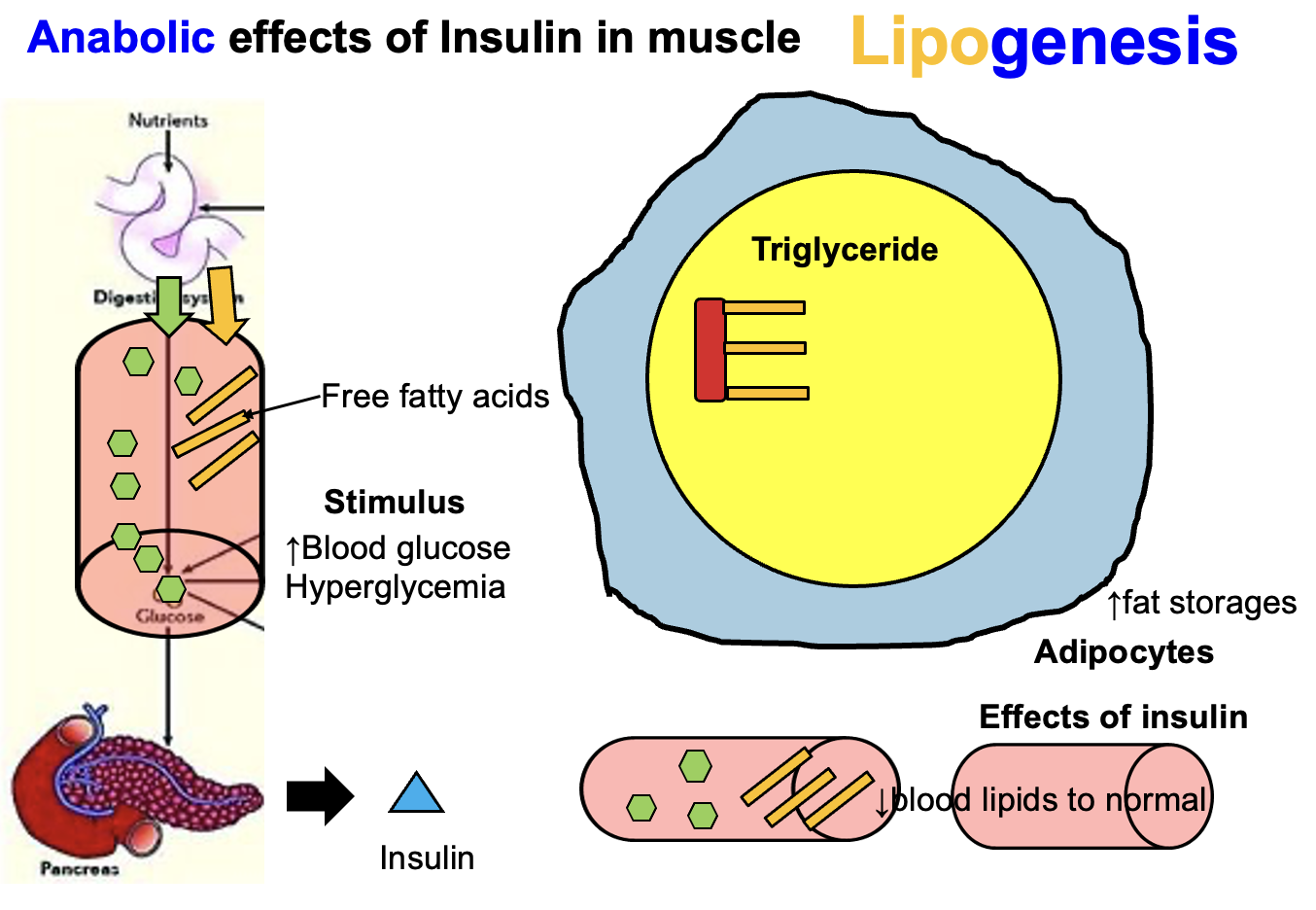
How does a carbohydrate-rich diet lead to triglyceride formation in adipose tissue?
A diet rich in carbohydrates stimulates lipogenesis in adipose tissue.
Mechanism:
Glucose enters adipocytes and is used to produce glycerol.
At the same time:
• fatty acids diffuse into the adipocyte
Triglyceride formation:
• 1 glycerol molecule + 3 fatty acids → triglyceride
Result:
The triglycerides are stored in adipose tissue as fat for long-term energy storage.
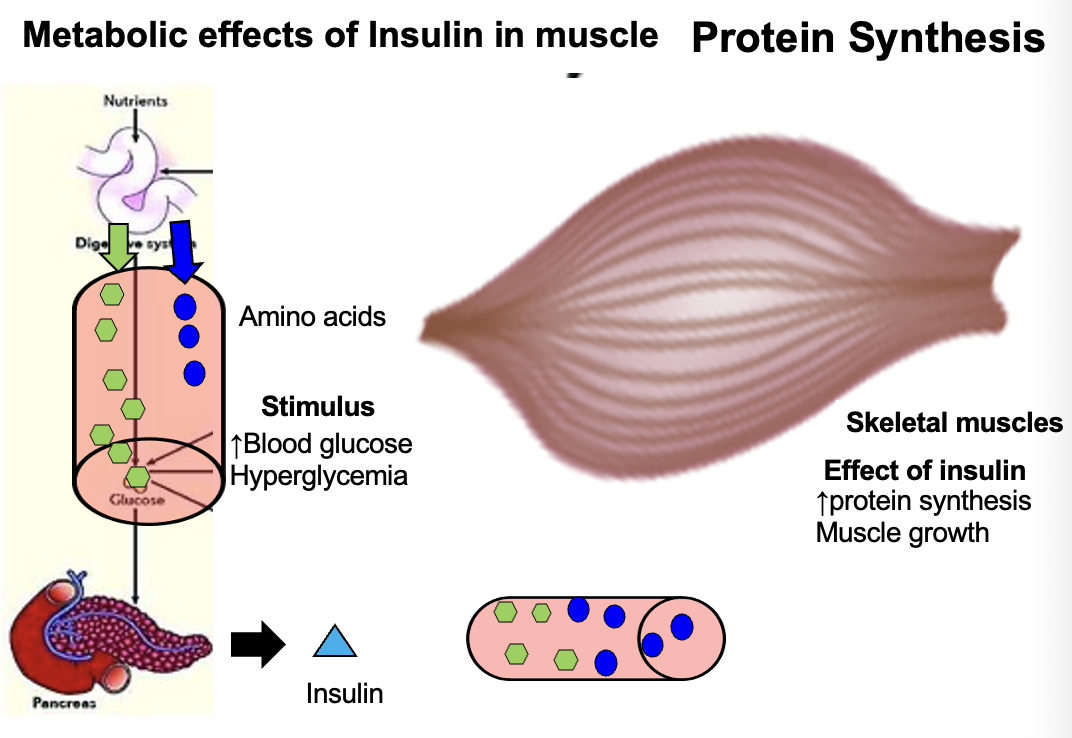
How does insulin affect muscle protein synthesis?
Insulin stimulates muscle protein synthesis WHEN amino acid levels are high.
Mechanism:
Insulin promotes amino acid uptake into muscle cells
Activates anabolic signaling pathways (e.g., mTOR)
Enhances protein synthesis
Key Point:
Insulin alone is not sufficient
Requires elevated amino acids (e.g., post-protein intake)
Example (Exercise Physiology):
After resistance training + protein meal →
↑ amino acids + ↑ insulin → maximal muscle protein synthesis
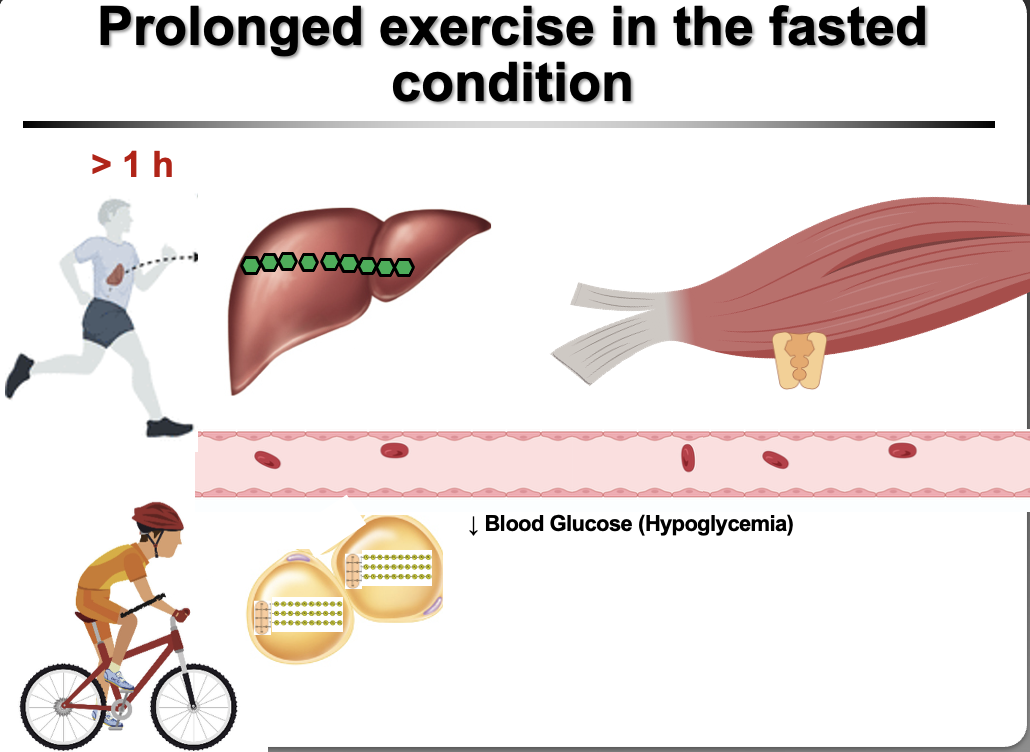
Why can prolonged exercise (>60–90 min) lead to hypoglycemia?
Prolonged exercise can cause hypoglycemia because muscle glucose uptake exceeds liver glucose production.
Mechanism:
Muscles increase glucose uptake during sustained exercise
Liver initially maintains blood glucose via:
Glycogenolysis
Gluconeogenesis
Over time, liver glycogen stores become depleted
Liver cannot keep up with muscle demand → blood glucose drops
Key Point:
Hypoglycemia risk increases after 60–90 minutes
Especially without carbohydrate intake
What are insulin counter-regulatory hormones, and how do they affect glucose metabolism during fasting or prolonged exercise?
Counter-regulatory hormones:
Glucagon
Epinephrine
Cortisol
Growth hormone (GH)
Overall Function:
👉 Oppose insulin → increase blood glucose levels
When are they released?
Hypoglycemia
Prolonged fasting
Prolonged aerobic exercise
👉 ↓ insulin secretion
👉 ↑ counter-regulatory hormones
Specific Effects:
Glucagon & Epinephrine:
↑ glycogenolysis (liver) → ↑ glucose releaseCortisol:
↑ proteolysis (muscle) → ↑ amino acids for gluconeogenesisGrowth Hormone (GH):
↓ glucose uptake in peripheral tissues
Net Effect:
👉 ↑ blood glucose via catabolic pathways
Glycogenolysis
Gluconeogenesis
Lipolysis
Proteolysis
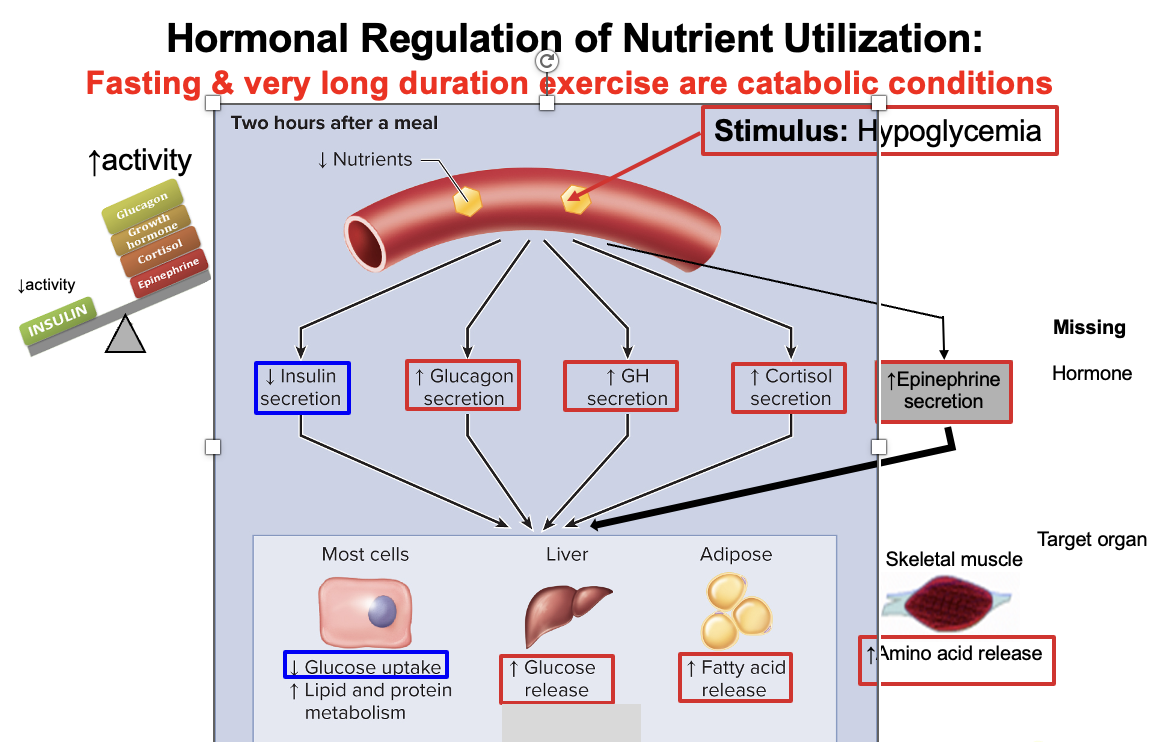
How is blood glucose maintained during exercise, and which hormones increase circulating glucose?
Maintaining blood glucose during exercise requires:
Liver glucose release
Glycogenolysis → breakdown of glycogen → glucose
Muscle glucose uptake
Muscles take up glucose for ATP production
Hormones that increase circulating glucose (counter-regulatory):
Glucagon → ↑ liver glycogenolysis
Epinephrine → ↑ glycogenolysis (liver + muscle)
Cortisol → ↑ proteolysis → ↑ gluconeogenesis
Growth Hormone (GH) → ↓ glucose uptake in tissues
Net Effect:
👉 Maintain blood glucose during exercise
👉 Prevent hypoglycemia
How do exercise intensity vs duration affect carbohydrate metabolism?
As EXERCISE INTENSITY ↑:
↑ Catecholamines (epinephrine, norepinephrine)
↑ Glycogenolysis (liver + muscle)
Muscle glycogen used FIRST
👉 Fast energy demand → use local (muscle) glycogen
As EXERCISE DURATION ↑:
↑ Liver glycogen use
↑ Muscle glucose uptake
↑ Liver glucose release
👉 Sustained exercise → rely more on blood glucose from liver
Key Distinction:
Intensity = muscle glycogen dominant
Duration = liver glucose becomes more important
How does insulin and glucose uptake change during exercise?
During exercise:
↓ Insulin levels
↑ Insulin sensitivity
↑ Glucose uptake into muscle cells
Key Idea:
👉 Muscles take up more glucose with LESS insulin
Mechanism:
Muscle contraction → GLUT4 translocation (insulin-independent)
Cells become more responsive to insulin
Why insulin decreases:
Prevents hypoglycemia
Allows counter-regulatory hormones (glucagon, epinephrine) to raise blood glucose
Net Effect:
Efficient glucose use
Maintained blood glucose levels during exercise
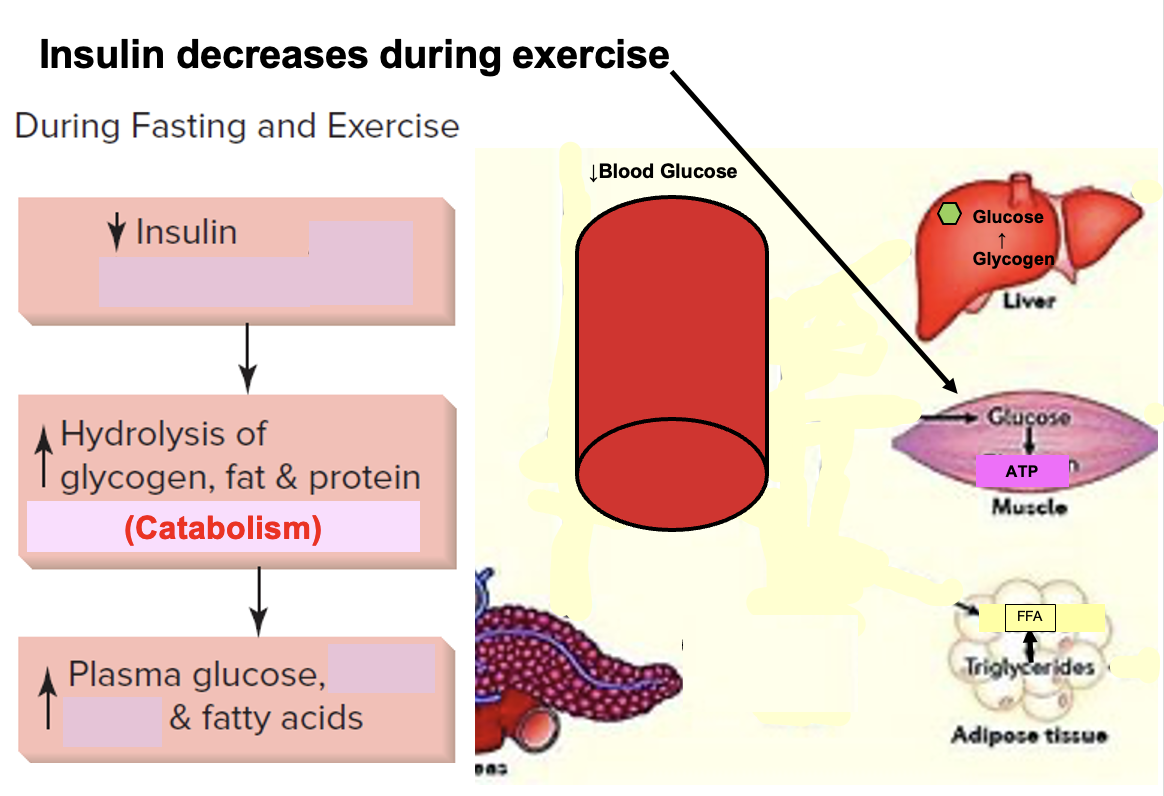
Why does insulin decrease during exercise, and what is the metabolic effect?
During exercise → ↓ insulin
Why insulin decreases:
👉 Allows mobilization of stored fuels
Effects of ↓ insulin:
↑ Glycogenolysis (liver) → ↑ blood glucose
↑ Lipolysis (adipose tissue) → ↑ free fatty acids
Fuel usage:
Glucose → used by muscles for ATP
Fatty acids → oxidized for ATP (especially during prolonged exercise)
Key Concept:
👉 Low insulin = catabolic state → fuel release
Big Picture:
Liver → releases glucose
Adipose → releases fatty acids
Muscle → uses both for energy
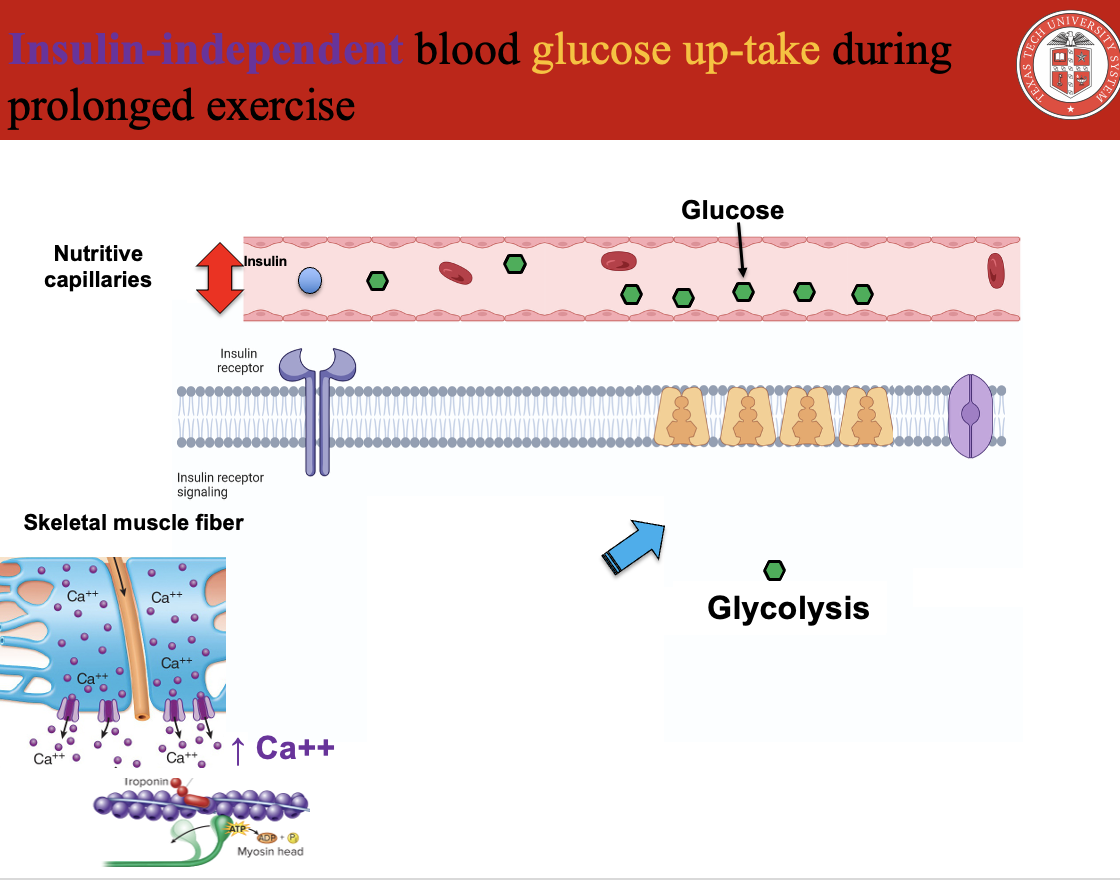
How does exercising muscle take up glucose without insulin?
Glucose uptake during exercise is INSULIN-INDEPENDENT
Key Mechanism:
Muscle contraction →
↑ Ca²⁺ release from sarcoplasmic reticulum↑ Intracellular Ca²⁺ triggers:
Muscle contraction
GLUT-4 translocation to sarcolemma
GLUT-4 in membrane →
Facilitated diffusion of glucose into muscle cells
Important Distinction:
At rest: insulin → GLUT-4 translocation
During exercise: contraction (Ca²⁺) → GLUT-4 translocation
Key Concept:
👉 Glucose uptake does NOT require insulin during exercise
GH, Cortisol, Epinephrine = glucagon. insulin does the opposite
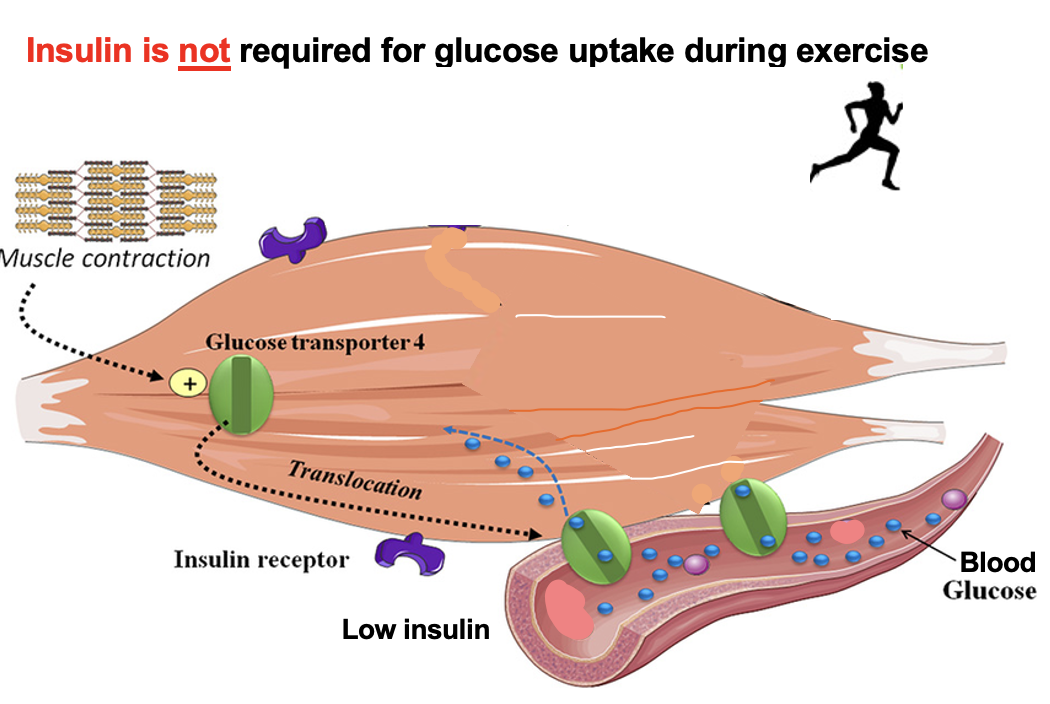
How does Ca²⁺ movement enable insulin-independent glucose uptake during muscle contraction?
Muscle contraction → Ca²⁺ released from the sarcoplasmic reticulum (SR) into the sarcoplasm → ↑ intracellular Ca²⁺ → activates Ca²⁺-dependent pathways (CaMK, AMPK) → GLUT4 translocates to the membrane → ↑ glucose uptake independent of insulin
During prolonged aerobic exercise (e.g., 3 hours at ~65% VO₂ max), how do key hormones change to maintain blood glucose?
Prolonged exercise → risk of hypoglycemia → ↑ counterregulatory hormones:
↑ epinephrine
↑ glucagon
↑ cortisol
These hormones → ↑ hepatic glucose output (glycogenolysis + gluconeogenesis) → maintain/↑ blood glucose during exercise
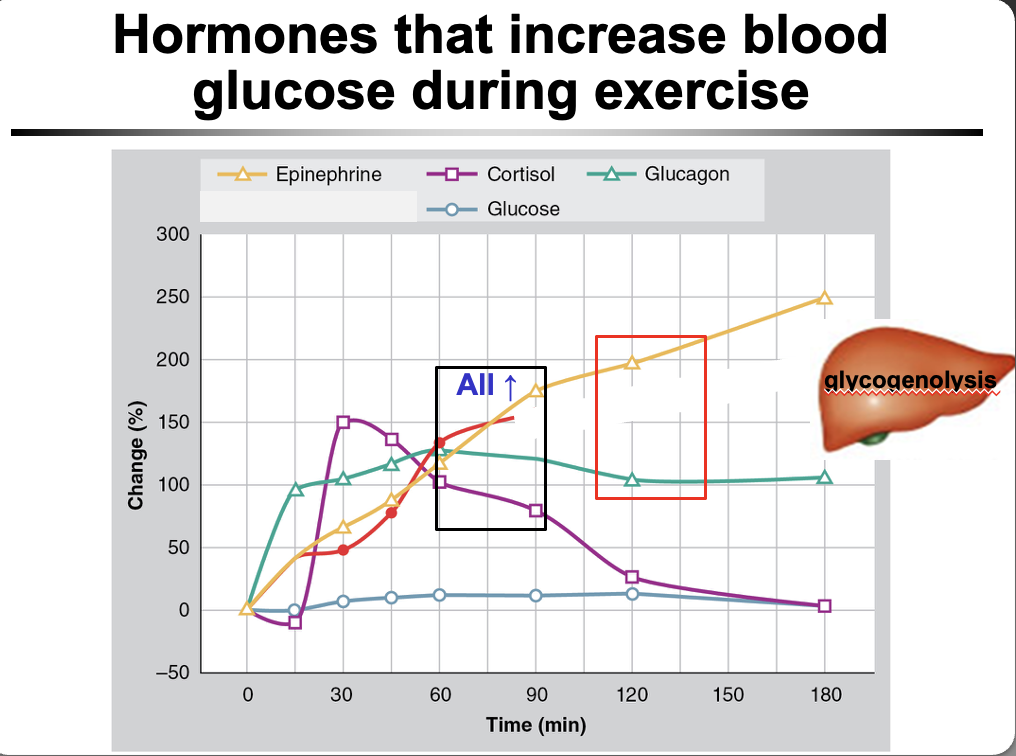
What stimulates glucagon secretion during prolonged exercise, and what is its purpose?
Prolonged exercise → ↓ blood glucose (hypoglycemia) → stimulates pancreatic α-cells → ↑ glucagon secretion
Glucagon → ↑ glycogenolysis + ↑ gluconeogenesis in liver → ↑ blood glucose to prevent hypoglycemia
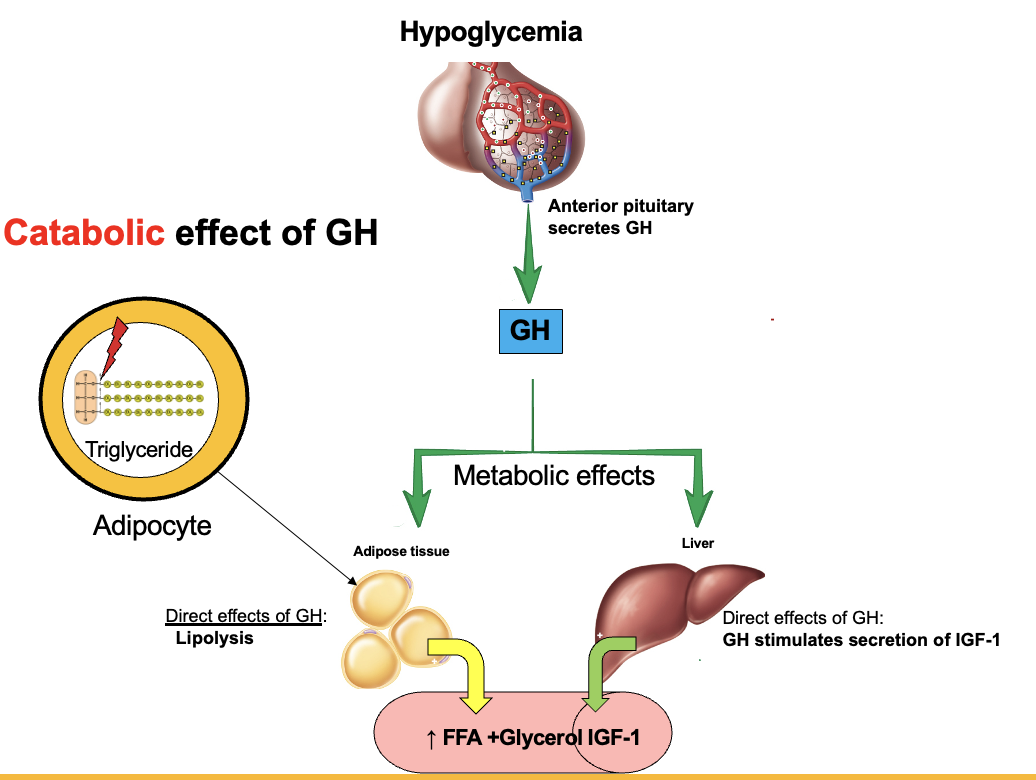
What are the main metabolic effects of growth hormone (GH)?
GH → anabolic for proteins: ↑ amino acid uptake → ↑ protein synthesis
GH → catabolic for lipids: ↑ lipolysis → fats used for energy → spares protein for growth
What are tropic hormones, and how do ACTH and growth hormone differ in their targets?
Tropic hormones → act on other endocrine glands → stimulate hormone release
ACTH (adrenocorticotropic hormone) → targets adrenal cortex → stimulates cortisol production
Growth hormone (somatotropic hormone) → non-tropic/systemic → acts on many tissues (liver, bone, muscle) → widespread growth and metabolic effects
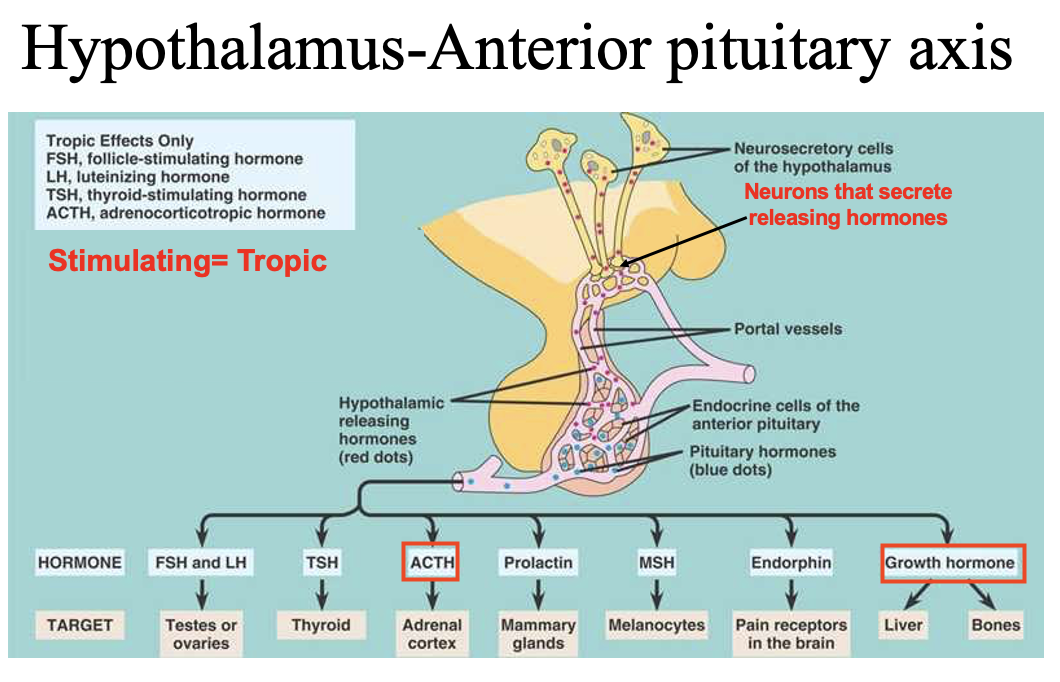
What is the key structural and functional difference between the neurohypophysis and adenohypophysis?
Neurohypophysis (posterior pituitary) → no endocrine cells → contains axons/axon terminals of hypothalamic neurons → releases hormones made in hypothalamus (ADH, oxytocin)
Adenohypophysis (anterior pituitary) → has endocrine cells → stimulated by hypothalamic releasing hormones → synthesizes and secretes its own hormones
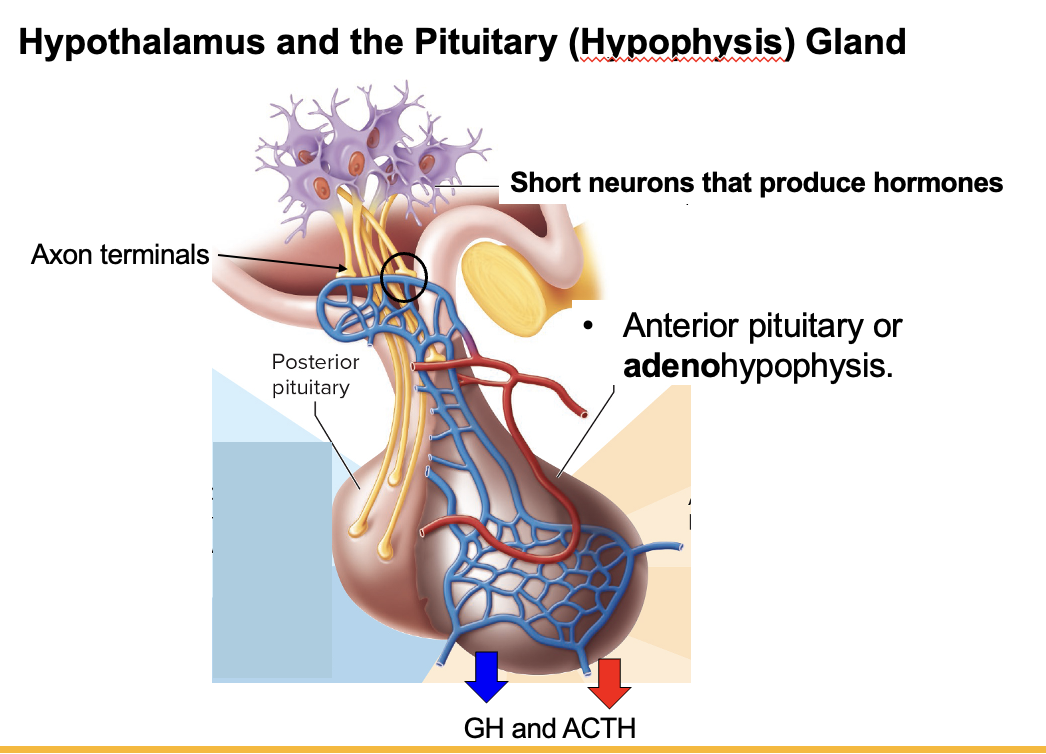
How does the hypothalamus → anterior pituitary (adenohypophysis) signaling pathway work?
Hypothalamic neurons (cell bodies in hypothalamus) → secrete releasing hormones (RH) into capillary bed (hypophyseal portal system) → RH travel to anterior pituitary → bind receptors on endocrine cells → anterior pituitary releases hormones (e.g., GH, ACTH) into blood → act on target tissues/organs
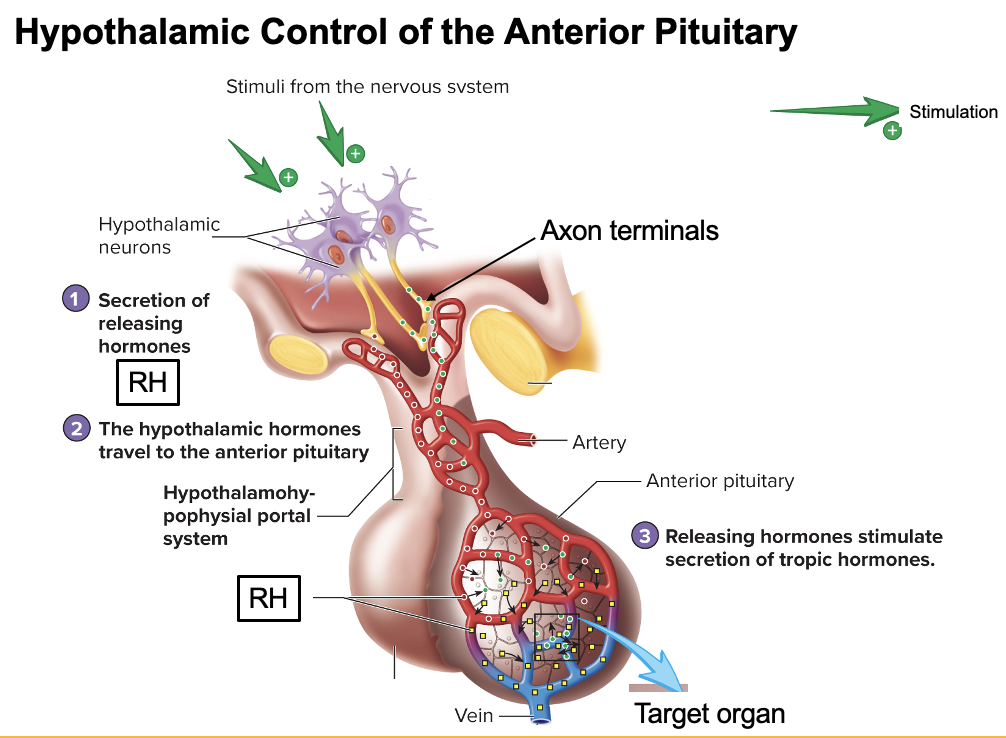
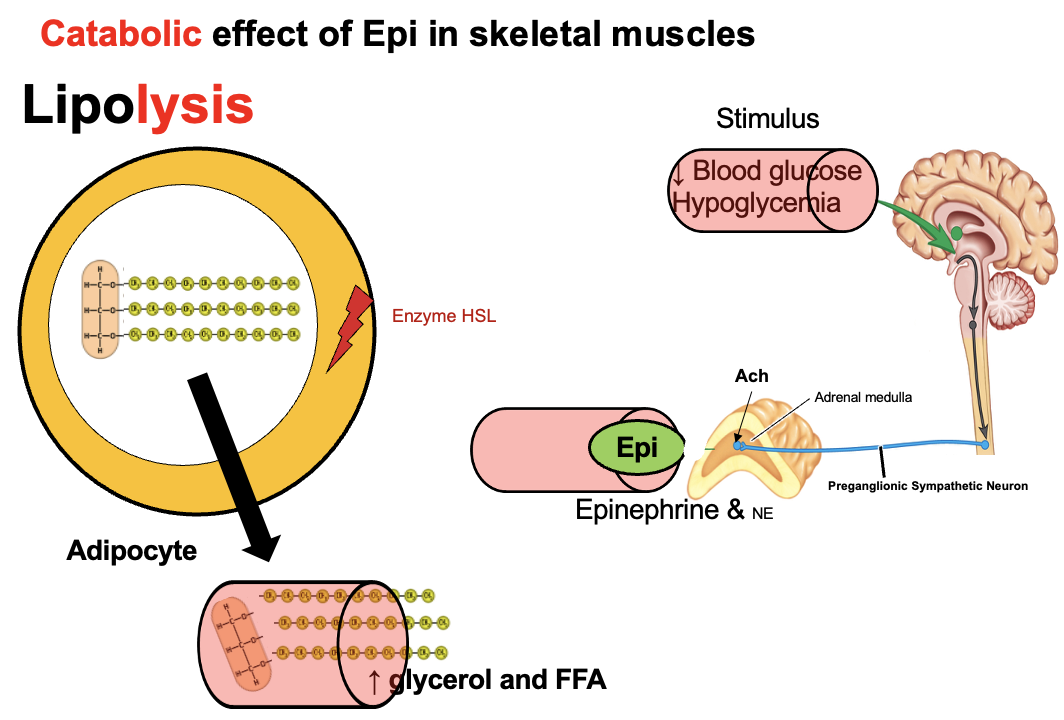
How does growth hormone (GH) promote lipolysis in adipose tissue?
GH → binds receptors on adipocytes → ↑ activity of hormone-sensitive lipase (HSL) → breaks triglycerides into free fatty acids (FFA) + glycerol → released into blood → used for energy
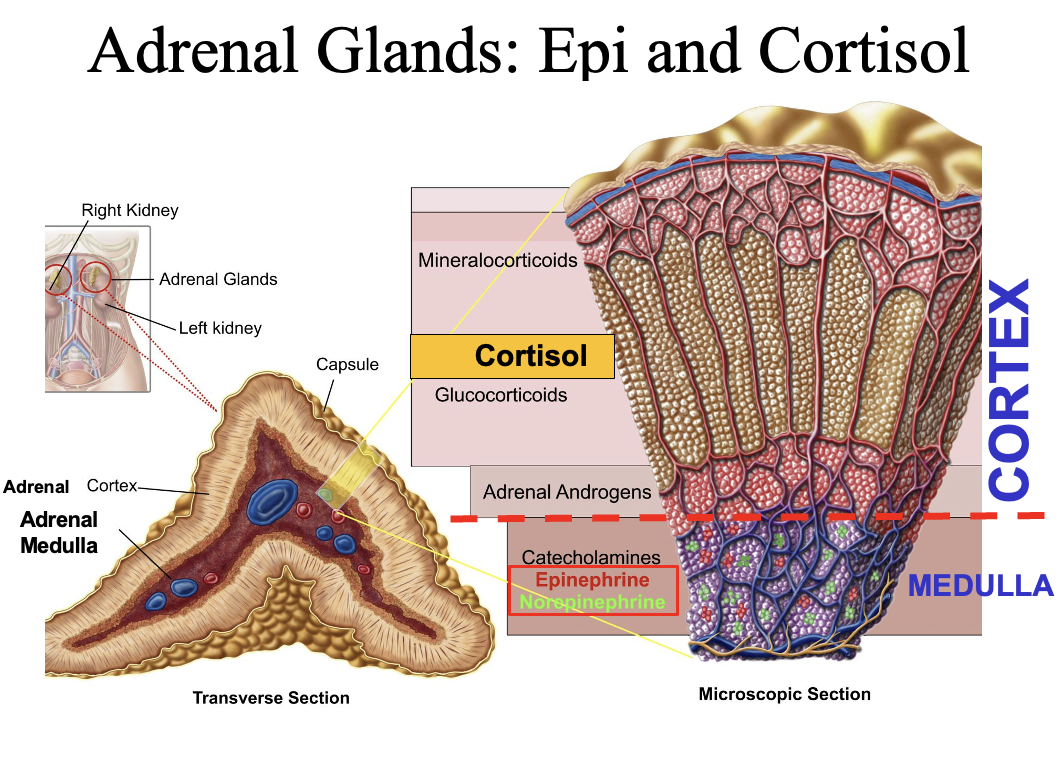
What hormones are produced by the adrenal cortex vs. adrenal medulla?
Adrenal cortex (3 layers):
Mineralocorticoids → aldosterone
Glucocorticoids → cortisol
Sex hormones → androgens/estrogens
Adrenal medulla:
Part of sympathetic nervous system
Secretes catecholamines (epinephrine, norepinephrine) into blood
Hypothalamus and adrenal medulla = ONLY TWO NEUROENDOCRINE ORGANS
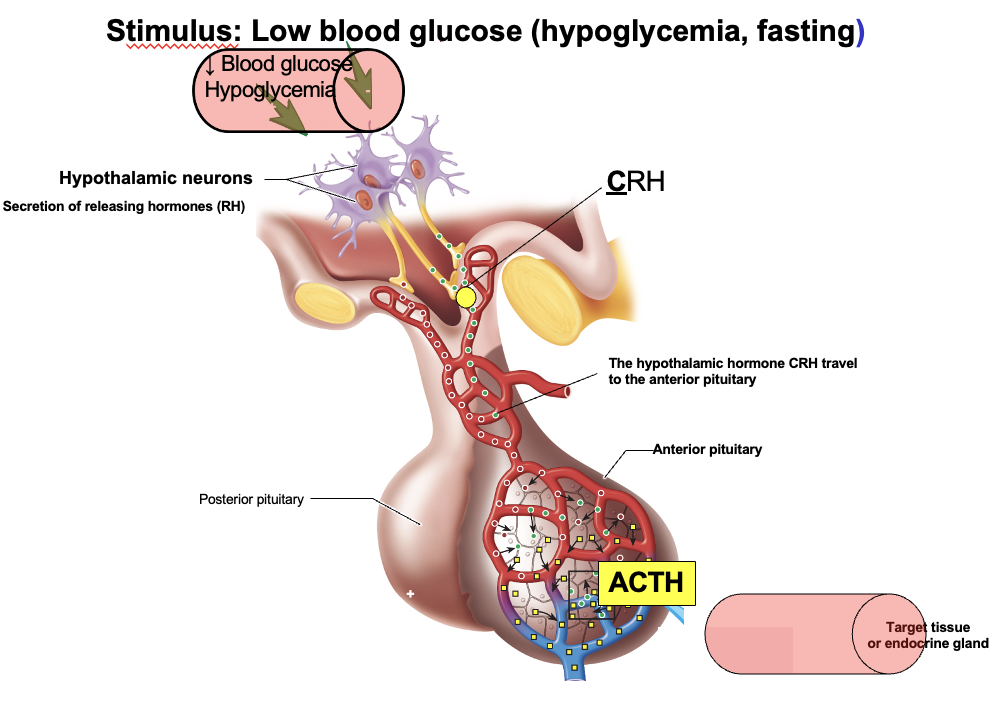
How does hypoglycemia activate the hypothalamus to restore blood glucose?
Hypoglycemia → stimulates two hypothalamic pathways:
Sympathetic pathway → hypothalamus → medulla oblongata → activates adrenal medulla → ↑ epinephrine
Neuroendocrine pathway → hypothalamus releases RH into capillary (portal system) → anterior pituitary → ↑ hormones (e.g., ACTH → cortisol)
→ Both pathways work to ↑ blood glucose
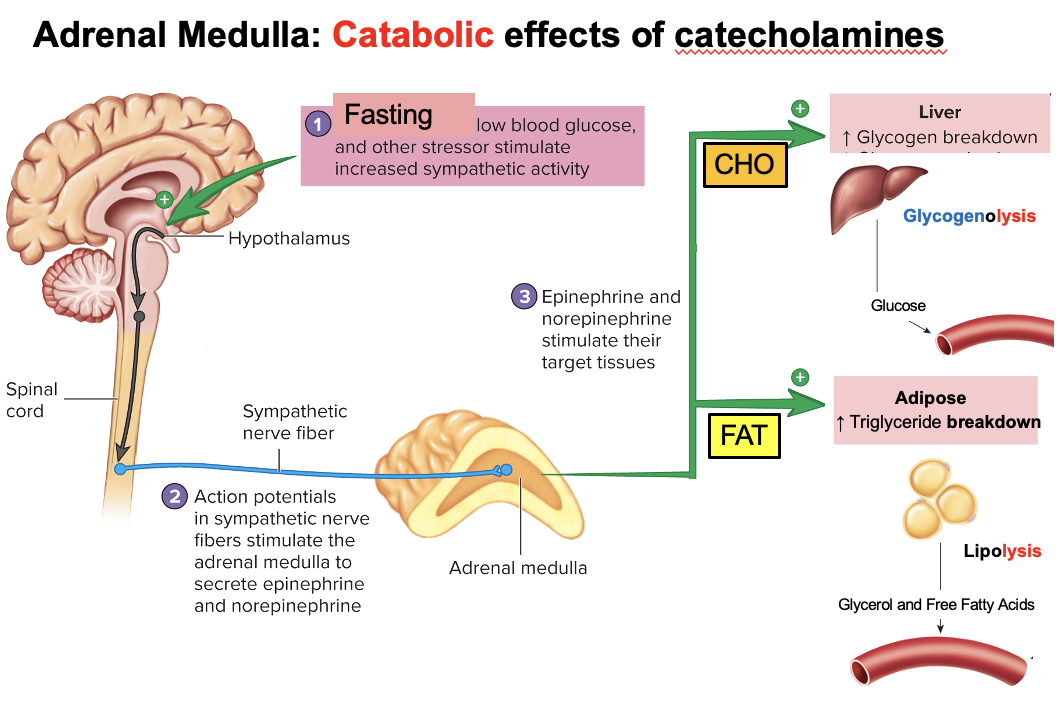
What is the role of the adrenal medulla during exercise or stress?
Adrenal medulla → releases catecholamines (80% epinephrine, 20% norepinephrine)
Exercise/stress → ↑ sympathetic activity → ↑ catecholamine release
Effects:
↑ glycogenolysis
↑ lipolysis
→ ↑ blood glucose and energy availability
What is the full neural pathway that stimulates the adrenal medulla during hypoglycemia/stress?
Hypoglycemia/stress → hypothalamus → synapses in medulla oblongata → descends to lumbar spinal cord → activates preganglionic sympathetic neurons
Preganglionic neurons release acetylcholine (ACh) onto adrenal medulla
→ adrenal medulla secretes epinephrine (± norepinephrine)
What are the main target tissues and metabolic effects of epinephrine from the adrenal medulla?
Epinephrine targets:
Liver (hepatocytes) → ↑ glycogenolysis → ↑ glucose release (CHO)
Adipose tissue (adipocytes) → ↑ lipolysis → ↑ free fatty acids + glycerol (FAT)
→ Overall: catabolic effects to increase energy availability during stress/exercise
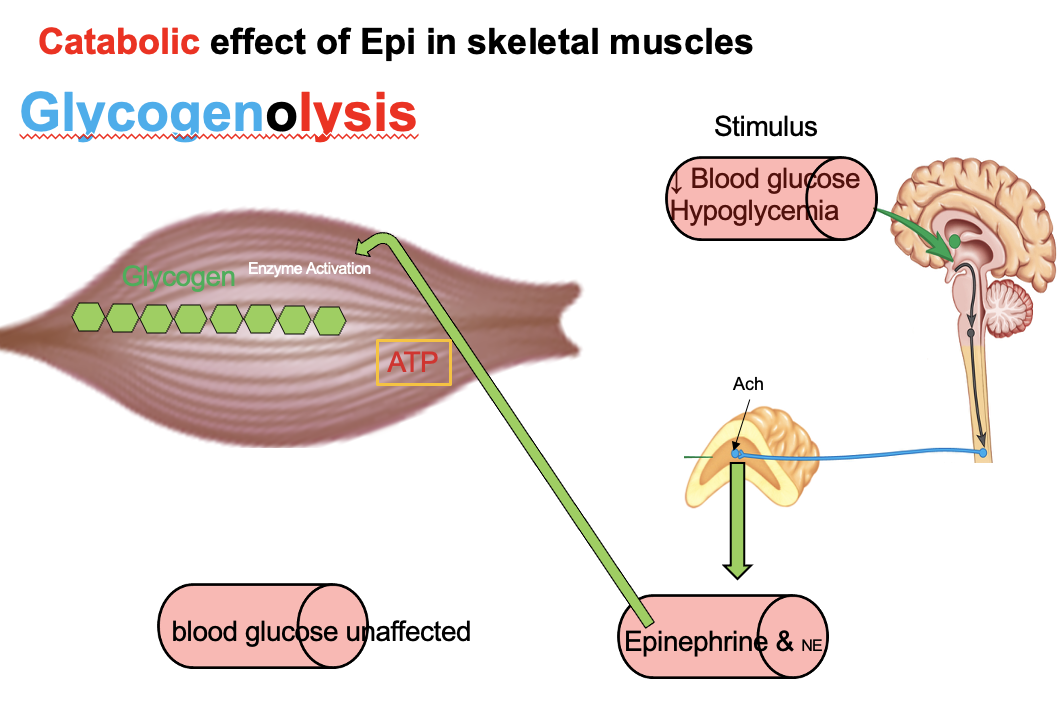
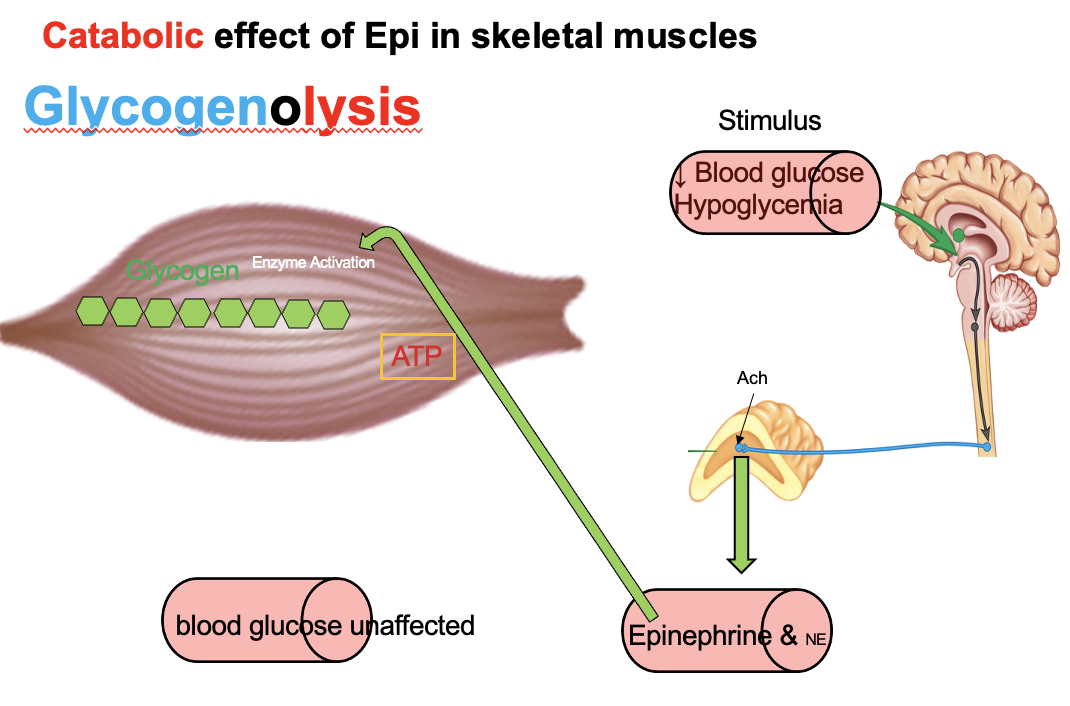
How does epinephrine affect glycogenolysis in liver vs. skeletal muscle, and why can only the liver raise blood glucose?
Epinephrine → ↑ glycogenolysis in liver + skeletal muscle
Liver:
Has glucose-6-phosphatase → converts G6P → free glucose
→ releases glucose into blood → ↑ blood glucose
Skeletal muscle:
Lacks glucose-6-phosphatase
G6P stays in cell → enters glycolysis → used for local energy
→ Only the liver can increase blood glucose levels
Glucagon and Epinephrine have the liver as the target organ
Why can’t skeletal muscle release glucose into the blood during epinephrine-stimulated glycogenolysis?
Epinephrine → ↑ glycogenolysis in liver and muscle
Skeletal muscle:
Lacks glucose-6-phosphatase
Cannot convert G6P → free glucose
G6P stays in cell → enters glycolysis → produces ATP
→ Muscle uses glucose locally, cannot release it into blood
How does epinephrine support energy production during fasting?
Fasting → epinephrine → ↑ lipolysis in adipose tissue
Triglycerides → free fatty acids (FFA) + glycerol
FFA → used for energy (β-oxidation)
Glycerol → used for gluconeogenesis
→ provides fuel and helps maintain blood glucose
How does CRH regulate ACTH and the adrenal cortex during stress or hypoglycemia?
Stress/hypoglycemia → hypothalamus releases CRH → travels via portal system to anterior pituitary
CRH → stimulates anterior pituitary → ↑ ACTH
ACTH → tropic hormone → stimulates adrenal cortex → ↑ cortisol secretion