Enzymes Small Group (Gochin)
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
Amino acids combine through what type of linkage?
peptide linkages
What determines the properties of proteins?
side chains of amino acids
Lysine, Arginine, Histidine are what type of amino acids?
a) basic
b) acidic
c) polar
d) non-polar
a)
Aspartic acid, glutamic acid are what type of amino acids?
a) basic
b) acidic
c) polar
d) non-polar
b)
Asparagine, glutamine, serine, threonine, tyrosine, and cysteine are what type of amino acids?
a) basic
b) acidic
c) polar
d) non-polar
c)
Glycine, tryptophan, tyrosine, phenylalanine, isoleucine, glycine, valine, alanine, leucine, and methionine are what type of amino acids
a) basic
b) acidic
c) polar
d) non-polar
d)
Which of the following amino acids contained in an enzyme active
site is likely to assist in binding of substrate if the enzyme is responsible
for cleaving an Asn – Lys bond?
a) Gly
b) Asp
c) Pro
d) Lys
e) Trp
b)
A prion is a protein that has misfolded from its regular globular structure to form
fibers of aggregated protein. To destroy a prion, which structure needs to be broken
down?
a) Primary
b) Secondary
c) Tertiary
d) Quaternary
e) Super-secondary
a)
What is a super-secondary protein structure?
fibrous protein
What are enzyme cofactors?
-Participate in an enzymatic reaction
-Bind in a separate site from the substrate
-Regenerated
What are the different types of enzyme cofactors
a) free ions
b) prosthetic groups (firmly linked)
c) coenzymes (loosely linked)
d) all of the above
d)
What is an enzyme containing its cofactor called?
a) holoenzyme
b) apoenzyme
a)
What is an enzyme requiring but devoid of its cofactors called?
a) holoenzyme
b) apoenzyme
b)
What group or groups do metal ions belong to (enzyme cofactors)?
a) free ions (essential minerals in diet)
b) prosthetic groups (firmly linked)
c) coenzymes (loosely linked)
d) A & B
e) B & C
d)
What group do small organic molecules belong to (enzyme cofactors)?
a) free ions (essential minerals in diet)
b) prosthetic groups (firmly linked)
c) coenzymes (loosely linked)
d) A & B
e) B & C
e)
What group do water soluble vitamins belong to (enzyme cofactors)?
a) free ions (essential minerals in diet)
b) prosthetic groups (firmly linked)
c) coenzymes (loosely linked)
d) A & B
e) B & C
c)
What coenzymes are derived from the water soluble vitamins?
a) vitamin B1
b) vitamin C
c) vitamin B6
d) vitamin B12
e) folate
f) all of the above
f)
An enzyme does which of the following?
a. It alters the balance of substrate and product
b. It changes the kinetics of the reaction
c. It maintains an aqueous environment around the substrate
d. It can perform the reaction on multiple different substrates
b)
What is a tissue specific form of an enzyme called?
a) holoenzyme
b) apoenzyme
c) isoenzyme
c)
What isoenzyme is found in the liver?
a) glucokinase
b) hexokinase
a)
What isoenzyme is found in the brain and muscles?
a) glucokinase
b) hexokinase
b)
Which enzyme is likely to have higher activity for its substrate glucose?
a) glucokinase
b) hexokinase
b)
When does the maximum velocity of an enzyme occur?
a) low saturation
b) mid saturation
c) fully saturated
c)
What is Km
substrate concentration at ½ Vmax
What isoenzyme has a lower Km?
a) glucokinase
b) hexokianse
b)
Hexokinase and glucokinase are isozymes which means that they
a) Are produced by the same gene with altered expression
b) Are produced by different genes
c) Share a common promoter
d) Have equivalent Michaelis Menten parameters.
b)
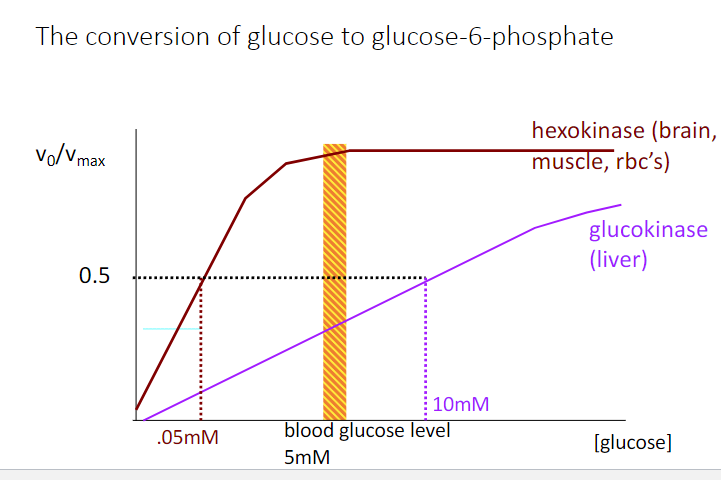
Indicate the glucokinase activity as a fraction of Vmax after a high carbohydrate
meal, in which blood glucose rises to 20 mM in the portal vein.
a) 0.45
b) 0.5
c) 0.6
d) 0.67
e) 1.0
d)
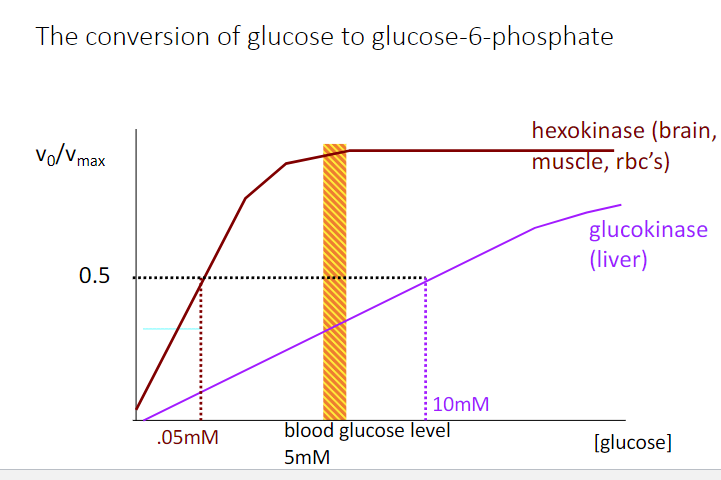
When blood sugar is low (4mM), what is the activity of glucokinase in the liver,
and what is the activity of hexokinase in the brain? (liver,brain)
a) 0.29, 0.99
b) 0.30, 1.00
c) 0.40, 0.99
d) 0.50, 0.50
e) 0.60, 0.40
a)
Blood tests on a newborn infant reveal hyperglycemia:
Fasting blood sugar: 10mM (normal is 5mM)
CBC: Normal
Electrolytes: Normal
BUN: Normal
Genetic test: mutation in liver enzyme glucokinase.
The mutant glucokinase is a known isoform having a Km = 30mM. k 2 , the catalytic rate constant, is unaffected. The liver cells of the newborn adapted within a few weeks by upregulating glucokinase production. How much more mutant glucokinase would be required in order to process 10mM glucose at the same
rate as the normal form of the enzyme?
a. 1.5 times as much mutant glucokinase
b. 2 times as much mutant glucokinase
c. 3 times as much mutant glucokinase
d. 4 times as much mutant glucokinase
e. 5 times as much mutant glucokinase
b)
The Michaelis-Menton constant KM is
a. The half-maximal velocity of an enzyme
b. The concentration of substrate at half maximal velocity of an enzyme
c. The enzyme concentration at half maximal velocity
c)
The catalytic rate constant k2 or kcat is
a. The half-maximal rate of the catalyzed reaction
b. The maximal rate of the catalyzed reaction
c. The substrate concentration at which maximal rate is achieved
d. The maximal velocity divided by the enzyme concentration
d)
An enzyme with higher affinity for its substrate has
a. A higher KM
b. A lower KM
c. A higher Vmax
d. A lower Vmax
b)
True or False
Allosteric enzymes do not obey Michaelis-Menten kinetics
cooperative S-Shaped binding curve
enzymes are typically multi-subunit
substate binding in one subunit induces a conformation change in a second subunit leading to a higher substrate affinity
a) true
b) false
a)
True or False
Glycogen phosphorylase is an allosteric enzyme. Cooperativity between subunits occurs with substrate binding as well as with allosteric effectors glucose-6-P, AMP and ATP, and with phosphorylation. There are three isozymes, liver, brain and muscle glycogen phosphorylase
a) true
b) false
a)
The TAUT conformer is:
a) low affinity substrate binding site
b) high affinity substrate binding site
a)
The relaxed conformer is:
a) low affinity substrate binding site
b) high affinity substate binding site
b)
True or false
Glycogen phosphorylase binds to glycogen in the R-state
• Allosteric inhibitors or activators
stabilize T- or R-state
• Phosphorylation increases activity
a) true
b) false
a)
What is the covalent mechanism for the activation of the enzyme that causes glycogen degradation.
a) Dephosphorylation
b) Phosphorylation
c) Acetylation
d) Deacetylation
b)
What is a non-covalent effector of glycogen phosphorylase in muscles? (Two correct answers)
a) ATP
b) Glucose 6-phosphate
c) AMP
d) Phosphorylase kinase
b), c)
Which hormone results in the activation of glycogen phosphorylase in muscles?
a) Dopamine
b) Epinephrine
c) Glucagon
d) Insulin
b)
How does Ca2+ release during muscle contraction activate glycogen phosphorylase?
a) Ca2+ is an allosteric activator of glycogen phosphorylase.
b) Ca2+ is an allosteric activator of glycogen phosphorylase kinase.
c) Ca2+ is an allosteric activator of protein kinase A.
d) Ca2+ binds to calmodulin which binds to and activates glycogen phosphorylase.
e) Ca2+ binds to calmodulin which binds to and activates glycogen phosphorylase kinase.
f) Ca2+ release depletes ATP.
b)
What is the physiological advantage of having two mechanisms for activating muscle glycogen phosphorylase and the enzyme that phosphorylates it?
a) Hormonal regulation does not involve a phosphorylation cascade
b) Binding of a positive allosteric effector to muscle GP gives rise to a systemic hormonal response.
c) The two mechanisms can substitute for each other
d) Both mechanisms must be active for either to exert an effect
e) Cellular and organism level responses can occur independently
e)
A class of drugs called the chloroindole-carboxamides, used for the treatment of diabetes, inhibits glycogen phosphorylase by an allosteric mechanism. Which form do these drugs stabilize?
a) a, also known as taut
b) a, also known as relaxed
c) b, also known as taut
d) b, also known as relaxed
c)
![<p><span>Inhibition of an enzyme by a drug can take several forms. It can be reversible</span><br><span>(competitive or non-competitive) or irreversible (covalent). Reversible inhibition can best be evaluated using the Lineweaver Burke equation, a linear form of the Michaelis –Menten equation obtained by inverting both sides of the equation.</span></p><ul><li><p>M.M. <span>v0 = v max [S] / (K M + [S])</span></p></li><li><p><span>L.B. 1/v 0 = K M / (v max [S]) +1/v m</span></p></li><li><p><span>Note that the MM parameters can be read off</span><br><span>the linear L.B. plot. The Y-intercept = 1/Vmax and</span><br><span>the X intercept is -1/K M. The slope is KM/Vmax</span></p></li></ul><p>a) true</p><p>b) false</p>](https://knowt-user-attachments.s3.amazonaws.com/473a453d-3379-4779-8c73-cf985ede892a.png)
Inhibition of an enzyme by a drug can take several forms. It can be reversible
(competitive or non-competitive) or irreversible (covalent). Reversible inhibition can best be evaluated using the Lineweaver Burke equation, a linear form of the Michaelis –Menten equation obtained by inverting both sides of the equation.
M.M. v0 = v max [S] / (K M + [S])
L.B. 1/v 0 = K M / (v max [S]) +1/v m
Note that the MM parameters can be read off
the linear L.B. plot. The Y-intercept = 1/Vmax and
the X intercept is -1/K M. The slope is KM/Vmax
a) true
b) false
a)
What type of inhibition when Km increases, Vmax unchanged
a) competitive inhibition
b) non-competitive inhibition
a)
What type of inhibition when Km unchanged, Vmax decreases
a) competitive inhibition
b) non-competitive inhibition
b)
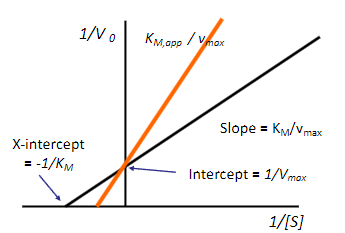
Based on the figure what type of inhibition occurs? (look at orange line)
a) competitive inhibition
b) non-competitive inhibition
a)
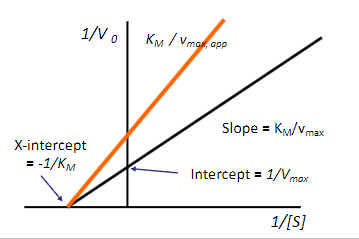
Based on the figure what type of inhibition occurs? (look at orange line)
a) competitive inhibition
b) non-competitive inhibition
b)
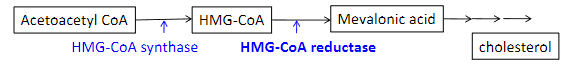
Statins are a class of drugs which lower hepatic cholesterol production by inhibiting an enzyme called HMG-CoA reductase. The result is an increase in LDL receptors and a lowering of LDL particles in the bloodstream
a) true
b) false
a)
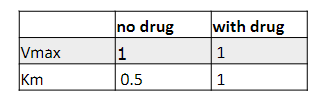
Is TUCOMCHOL a competitive inhibitor?
a) Yes
b) No
a)
We have invented a new statin, called Tucomchol, and we are investigating its potency and mechanism of action. We
measure enzymatic activity of HMG-CoA reductase as a function of HMG-CoA concentration and plotted it in a linear
form. Based on the mechanism of action, what would you expect about the molecular structure of Tucomchol?
analog of substrate
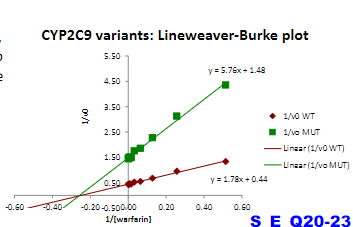
Warfarin is an anticoagulant used to prevent blood clots. It is metabolized from its active form by a CYP450 enzyme CYP2C9.
A major challenge in warfarin therapy is its narrow therapeutic range,
with even a mild degree of over-anticoagulation potentially leading to
hemorrhagic complications. Genetic variations in the CYP2C9 enzyme
occur in the population. One CYP450 variant, CYP2C9*2, exhibits
particularly low activity compared to the “wild-type” enzyme.
CYP2C9 and CY2C9*2 enzyme activity was measured for substrate
warfarin, and plotted in a linear form
a) true
b) false
a)
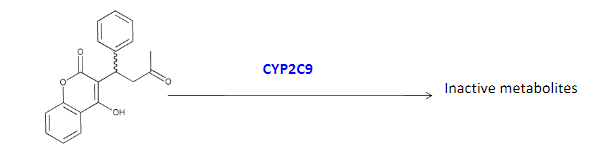
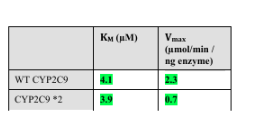
How does the mutation in CYP2C9*2 affect metabolism of warfarin compared to wild-type CYP2C9?
A. reduced binding affinity for warfarin,
B. reduced turnover rate (k2 ),
C. causes non-competitive inhibition
b)
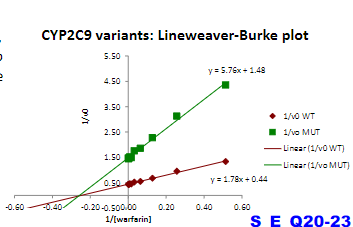
Which individual who would be more susceptible to the effects of warfarin
a) Homozygous WT
b) Heterozygous one CYP2C9 WT allele, one CYP2C9 2 allele
c) Homozygous for CYP2C92
c)
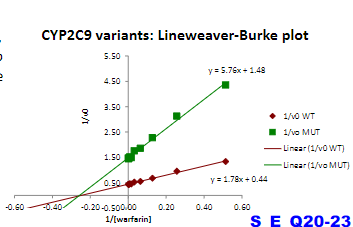
Which individual who should get the highest dose of warfarin:
a) Homozygous WT
b) Heterozygous one CYP2C9 WT allele, one CYP2C9 *2 allele
c) Homozygous for CYP2C9*2
a)