Hypothalamic–Pituitary–Gonadal Axis
1/120
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
121 Terms
What is the hypothalamic–pituitary axis?
A regulatory system where the hypothalamus controls pituitary hormone secretion.
How does the hypothalamus communicate with the anterior pituitary?
Via the hypothalamic–hypophyseal portal system (blood vessels).
What structure connects the hypothalamus to the pituitary gland
infundibulum
Which hormones are transported to the posterior pituitary from the hypothalamus & are released from the posterior pituitary
ADH (vasopressin) and oxytocin
How are posterior pituitary hormones released
Produced in the hypothalamus —(axons from hypothalamic neurons)→(pituitary stalk)→ posterior pituitary → Released directly into the bloodstream
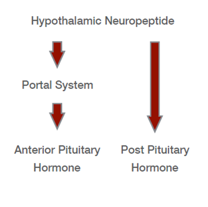
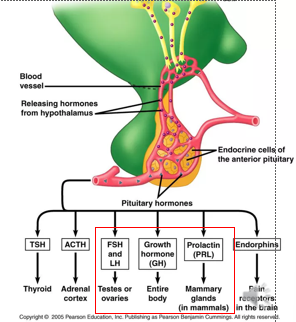
Which anterior pituitary hormones are released in response to hypothalamic signals
ACTH, TSH, LH/FSH, GH, Prolactin
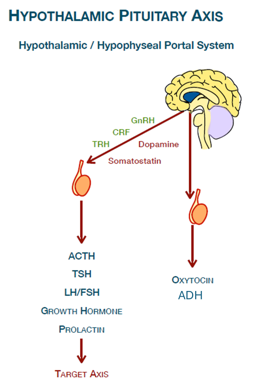
Name 3 hypothalamic hormones that stimulate anterior pituitary hormone release
GnRH (gonadotropin-releasing hormone)
TRH (thyrotropin-releasing hormone)
CRH (corticotropin-releasing hormone)
Which hormones do each of these stimulate the release of:
GnRH:
TRH:
CRH:
GnRH → Luteinizing Hormone (LH) and Follicle-Stimulating Hormone (FSH)
TRH → Thyroid Stimulating Hormone (TSH)
CRH → Adrenocorticotropic hormone (ACTH)
Name a hypothalamic hormone that inhibits anterior pituitary hormone release
Dopamine
What hormone does dopamine inhibit the release of
Prolactin
Which hypothalamic hormone inhibits growth hormone release
Somatostatin
Name an important clinical condition related to the posterior pituitary gland
Diabetes insipidus
What’s the problem in Diabetes insipidus
Deficiency of ADH or a reduced response to this hormone in the kidney.
Treatment for Diabetes insipidus
Desmopressin
Oxytocin is also released from the posterior pituitary. What is a clinical use of oxytocin?
Used for labour induction
4 key roles of anterior pituitary hormones
•Growth and metabolism
•Lactation
•Regulation of gonadal function
•Control of reproductive physiology
4 possible causes of Hyperprolactinemia
• Pituitary adenomas (prolactinomas)
• Hypothalamic or pituitary stalk lesions
• Drugs that inhibit dopamine signalling (e.g. antipsychotics)
• Pregnancy/lactation (physiological increase)
4 clinical features of Hyperprolactinemia
• Amenorrhoea
• Galactorrhoea
• Infertility
• Reduced libido
(body acts like it’s pregnant → no period, breast milk production, no need to get pregnant again)
What hormonal changes does Hyperprolactinemia cause that produces these clinical features
↓ Kisspeptin
↓ GnRH
↓ LH & FSH
3 types of treatment for Hyperprolactinemia
Dopamine agonists
Oral contraceptive pill / HRT
Surgical debulking (rarely)
Name 2 Dopamine agonists
Cabergoline
Bromocriptine
Which D receptor (no.) do the Dopamine agonists affect
D2
D2 receptor agonists inhibit prolactin secretion from what type of pituitary cells
Pituitary lactotroph cells
Dopamine Agonists are derived from what
Ergot-derived (fungi)
Which Dopamine Agonist has a short/long half life
Bromocriptine
Cabergoline
Bromocriptine: Short (2–8 hour)
Cabergoline: Long (65 hours)
Which Dopamine Agonist has a higher D2 receptor affinity
Bromocriptine / Cabergoline
Cabergoline
How often is Bromocriptine administered
Multiple times daily
AEs of Bromocriptine
Nausea
Postural hypotension
CNS side effects
How often is Cabergoline administered
Once / Twice weekly
2 advantages of Cabergoline over Bromocriptine
Better tolerated
Improved clinical response
Growth hormone–releasing hormone (GHRH) secretion is stimulated/inhibited by what
also say where each stimulator/inhibitor comes from
Stimulated by Ghrelin (stomach hormone)
Inhibited by Somatostatin (hypothalamus) & IGF-1 (Insulin-like growth factor 1)(liver)
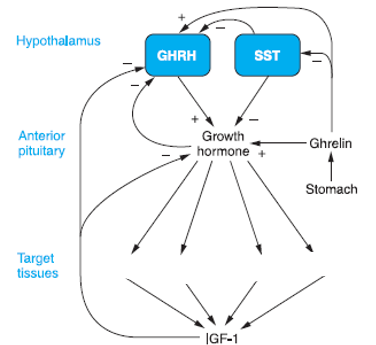
Name 4 target tissues of GH
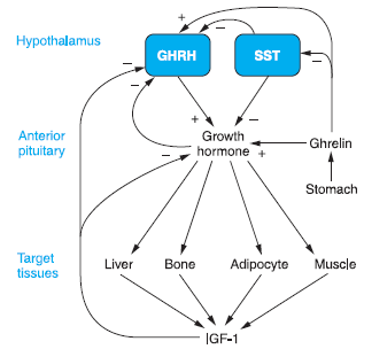
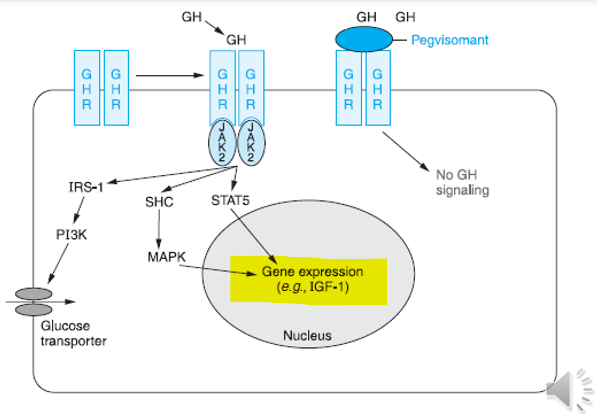
GH works through what 3 molecular signalling pathways
JAK2–STAT pathway
PI3K pathway
MAPK pathway
What causes the problem in Growth Hormone Deficiency
Caused by insufficient GH secretion from the anterior pituitary
4 Clinical features of Growth Hormone Deficiency
• Growth failure in children
• Increased fat mass
• Reduced muscle mass
• Reduced bone density
Growth Hormone Deficiency treatment (what type of med & name the drug)
Recombinant human growth hormone (Somatropin)
What is Growth Hormone Excess called in children vs adults
Gigantism (children)
Acromegaly (adults)
Compare growth pattern of Gigantism (children) & Acromegaly (adults)
Gigantism (children): excessive linear growth
Acromegaly (adults): enlargement of hands, feet and facial features, macroglossia, prognathism
What are 2 complications of acromegaly
Cardiomyopathy
Metabolic complications (insulin resistance, osteoporosis)
Cause of Growth Hormone Excess
GH-secreting pituitary adenoma
4 treatment options for Growth Hormone Excess
• Transsphenoidal surgery (first-line)
• Somatostatin analogues
• GH receptor antagonist
• Dopamine agonists
Name a GH receptor antagonist used in the treatment of Growth Hormone Excess
Pegvisomant
Somatostatin Analogues MOA
Bind somatostatin receptors (SSTR2, SSTR5) on pituitary somatotroph cells • Inhibit growth hormone (GH) secretion • Reduce IGF-1 production
Name the main 2 Somatostatin Analogue drugs
Octreotide
Lanreotide
You can get short/long acting formulations of Somatostatin Analogues. What is used for the short acting formulation & what is it’s duration of action
Octreotide
Duration of action: ~12 hours
What 2 drugs can be used for the long acting formulation & what is the duration of action
Octreotide LAR (Long-Acting Release)
Lanreotide
Intramuscular injection every 2–4 weeks
4 AEs of Somatostatin Analogues
• Gastrointestinal upset (~50%)
• Gallstones (~25%)
• Glucose metabolism alterations / diabetes (<5%)
• Local injection-site reactions
3 other clinical uses of Somatostatin Analogues (other than GH excess)
• Neuroendocrine tumours
• Acute variceal bleeding (via splanchnic vasoconstriction)
• TSH-secreting pituitary adenomas (TSHoma)
Pegvisomant MoA
GH receptor antagonist
4 effects of Pegvisomant
Reduces IGF-1
Increases growth hormone
May increase tumour size
Symptom control
How is Pegvisomant administered
Subcutaneous administration - daily injection
How is the action of Pegvisomant monitored
Measuring IGF-1 levels
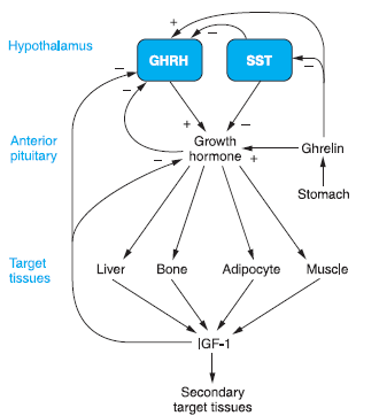
In the Hypothalamic–Pituitary–Gonadal (HPG) Axis, what stimulates the release of GnRH
Kisspeptin
In the HPG axis, what is the action of GnRH
GnRH is released in pulses and acts on the anterior pituitary, stimulating the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH).
Effects of FSH in females
FSH stimulates granulosa cells, promoting follicle development and oestrogen production
Effects of LH in females
LH acts on theca cells to produce androgens and later triggers ovulation and progesterone production
Effects of FSH in males
FSH acts on Sertoli cells to support spermatogenesis
Effects of LH in males
LH stimulates Leydig cells to produce testosterone
How does feedback happen in the HPG axis
Sex steroids such as oestrogens and testosterone provide negative feedback to the hypothalamus and pituitary, helping regulate the system
What’s the problem in Hypogonadotropic Hypogonadism
Reduced gonadal function due to insufficient stimulation by LH and FSH
3 causes of Hypogonadotropic Hypogonadism
• Hypothalamic dysfunction
• Pituitary disorders
• Chronic illness or medications
What (if any) changes do you see in what hormones in Hypogonadotropic Hypogonadism
• Low testosterone / oestrogen
• Low or inappropriately normal LH and FSH
3 options for treatment of Hypogonadotropic Hypogonadism
Hormone replacement therapy (HRT)
Pulsatile GnRH therapy
Gonadotropin therapy
For what goal would Hormone replacement therapy (HRT) be used over the other 2 options
Goal: Restoration of sex hormone levels
Effective on: libido, bone density, muscle mass, sexual development
For what goal would Pulsatile GnRH therapy be used
Goal: Fertility
(when the defect is hypothalamic)
For what goal would Gonadotropin therapy be used
Goal: Fertility
What does Gonadotropin therapy consist of
hCG (LH-like activity)
FSH
In the menstrual cycle, when are LH & FSH peaking
Day 14
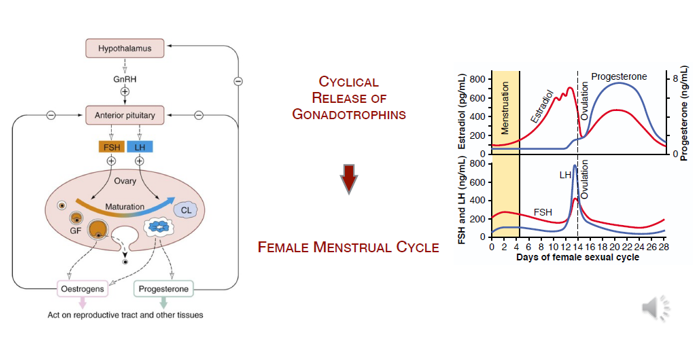
Does LH/FSH promote steroidogenesis
LH —(thecal cells)→ Steroidogenesis
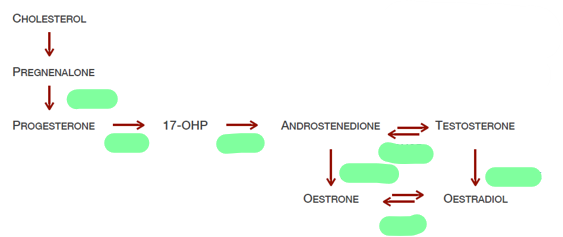
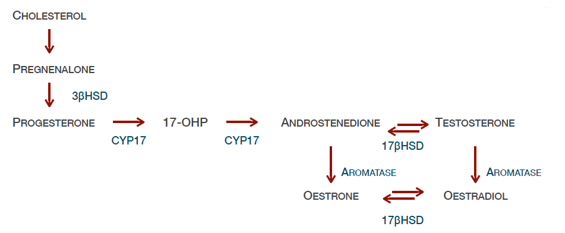
Name 5 drug types that modulate the HPG axis
• GnRH agonists
• GnRH antagonists
• Gonadotropins
• Estrogens and progestins
• Androgen therapy
GnRH agonists MoA
• Continuous stimulation of GnRH receptors
• Initial flare effect
• Subsequent suppression of LH and FSH
4 uses of GnRH agonists
• Endometriosis
• Prostate cancer
• Precocious puberty
• Assisted reproduction
6 AEs of GnRH agonists
• hot flashes
• decreased libido
• vaginal dryness
• erectile dysfunction
• mood changes
• bone mineral density loss (long-term
Name 2 GnRH antagonists
Cetrorelix
Ganirelix
GnRH antagonists MoA
Competitive blockade of GnRH receptor
Immediate suppression of LH and FSH
GnRH antagonists use
Assisted reproduction (IVF)
Name 3 Gonadotropins
• FSH
• LH
• hCG (LH-like activity)
3 Clinical uses of Gonadotropins
• Ovulation induction
• Infertility treatment
• Hypogonadotropic hypogonadism
Oestrogens actions
• Development of female secondary sexual characteristics
• Endometrial proliferation
• Regulation of the menstrual cycle
• Feedback on the hypothalamic–pituitary axis
3 uses of oestrogens
• Contraception
• Post-Menopausal hormone replacement therapy
• Endocrine disorders (e.g. hypogonadism
Synthetic oestrogens have what added benefit over natural
Improved oral bioavailability
Route of admin for Oestrogens
Transdermally/Topically
Progesterone/Progestins actions (4)
• Preparation of the endometrium for implantation
• Maintenance of pregnancy
• Regulation of the menstrual cycle
• Negative feedback on LH and GnRH secretion
3 clinical uses of Progesterone/Progestins
• Hormonal contraception
• Hormone replacement therapy
• Treatment of gynecological disorders (e.g. endometriosis)
True/False progesterone has high oral bioavailability
False - Extensive hepatic metabolism
Route of admin for progesterone
Orally (synthetic progestins developed with improved stability)
Intramuscularly
Intrauterine devices (levonorgestrel)
What improved with each generation of progestin preparations
Reduced androgen activity
Name 4 1st gen progestin preparations
First generation (estrane): norethindrone, norethynodrel, norethindrone acetate, ethynodiol diacetate
Name 3 2nd gen progestin preparations
Second (gonane): levonorgestrel, norethisterone, norgestrel
Name 4 3rd gen progestin preparations
Third (gonane): desogestrel, gestodene, norgestimate, drospirenone
Name 5 4th gen progestin preparations
Fourth: dienogest, drospirenone, nestorone, nomegestrol acetate and trimegestone


Name 2 Emergency Contraception drugs
Levonorgestrel
Ulipristal acetate
Which of the 2 Emergency Contraception drugs is a progesterone receptor modulator / a progestin
Levonorgestrel (progestin)
Ulipristal acetate (progesterone receptor modulator)
MoA of Emergency Contraception drugs
• Delay or inhibition of ovulation
• Prevent LH surge
• Reduce probability of fertilisation
(Does not terminate an established pregnancy!!)
Levonorgestrel must be taken within how many hours
72 hrs
Ulipristal must be taken within how many hours
120 hours
What do Selective Oestrogen Receptor Modulators (SERMs) do
They act as oestrogen receptor agonists or antagonists depending on the tissue
Selective Oestrogen Receptor Modulators (SERMs) MoA
• Bind to estrogen receptors (ERα / ERβ)
• Tissue-selective activity due to recruitment of co-activators or co-repressors
• Can produce estrogenic or anti-estrogenic effects