Chapter 6: An Introduction to Energy, Enzymes, and Metabolism
1/67
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
68 Terms
2 types of chemical reaction
1. Catabolic reactions
2. Anabolic reactions
Anabolic reactions
Synthesis of molecules.
Energy is stores
Catabolic reactions
Broken down or degradation of molecules
Energy is released
Metabolism
total of chemical reaction or anabolic and catabolic reactions
First law of Thermodynamics = law of conservation of energy
Energy cannot be created or destroyed
Energy can be transfered from one place to another and can be transformed from one type to another
Second law of Thermodynamics
When Energy is transferred from one place to another, the level of disorder (entropy) is increase.
2 forms of Energy
Kinetic and Potential
Important types of Energy in Biology
light
heat
mechanical
chemical
electrical/ ion gradient
Entropy
measurement of disorder in the system
Why energy is unusable?
because of entropy, the degree of disorder cannot be used in an useful way
Spontaneous reaction
reaction that does not require energy.
ΔG = Δ H - T Δ S
ΔG is the free energy change
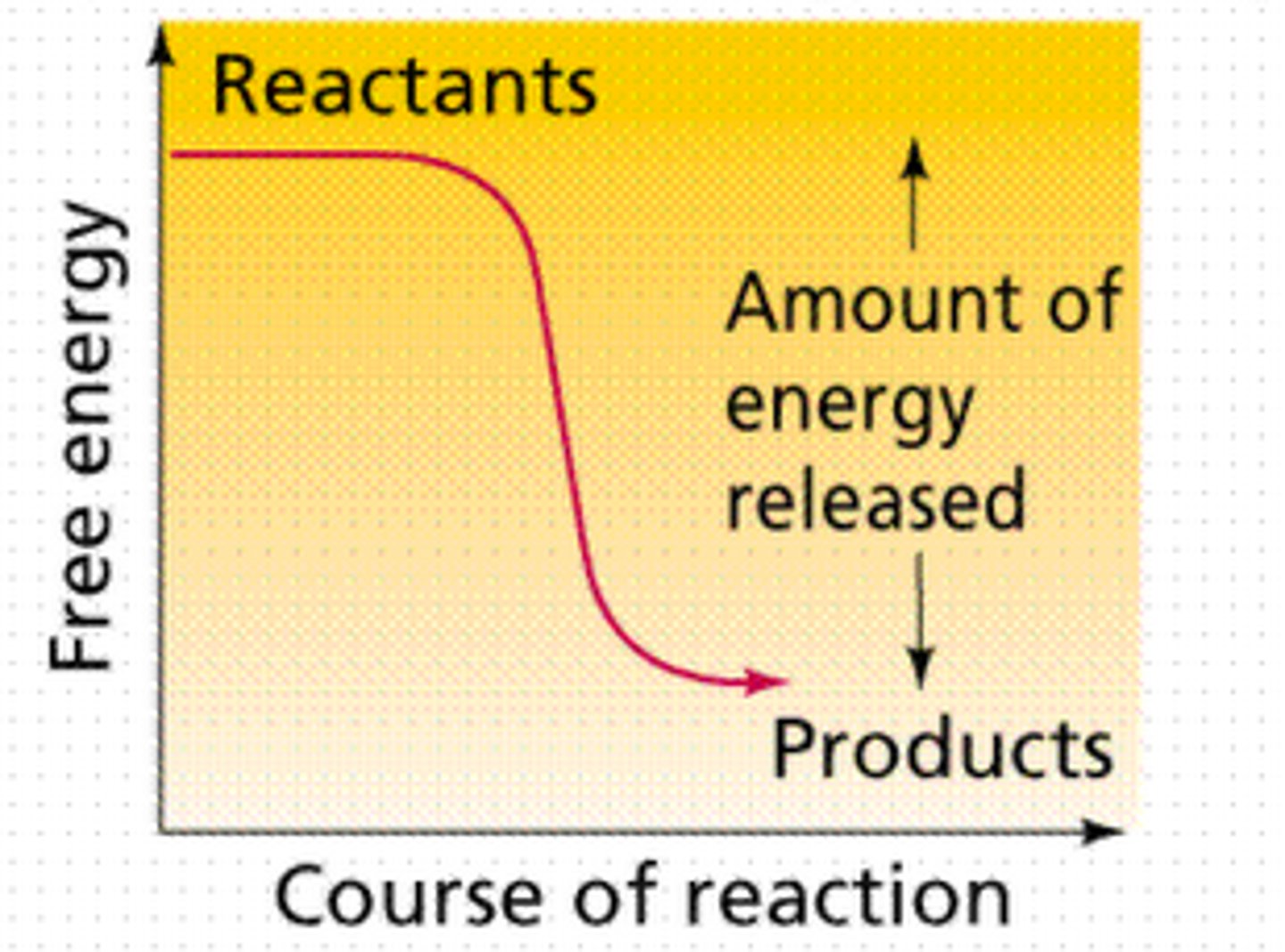
Exergonic reaction
spontaneous reaction
ΔG<0 or negative free energy change
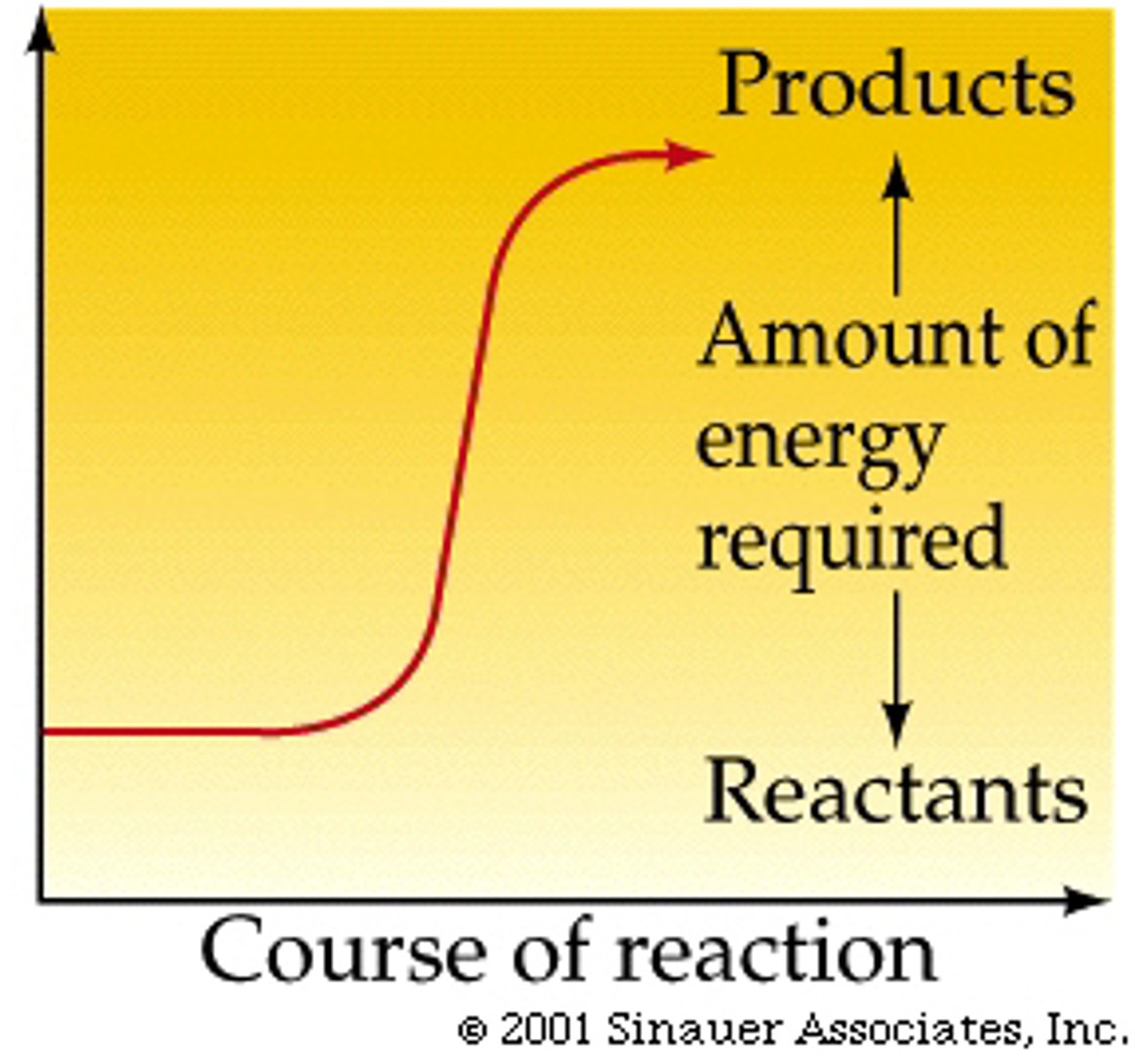
Endergonic reaction
it requires additional energy.
ΔG>0 or positive free energy change
Hydrolysis of ATP
Reaction favors formation of products because of
ΔG = -7.3 kcal/mole
Energy liberated can drive a variety of cellular processes
Cells use ATP hydrolysis
to drive endergonic reactions
An endergonic reaction and an exergonic reaction are coupled together to proceed spontaneously
Endergonic reaction will be spontaneous if net free energy change for both processes is negative
Phosphrylation
a phosphate is directly TRANSFERRED from ATP to glucose
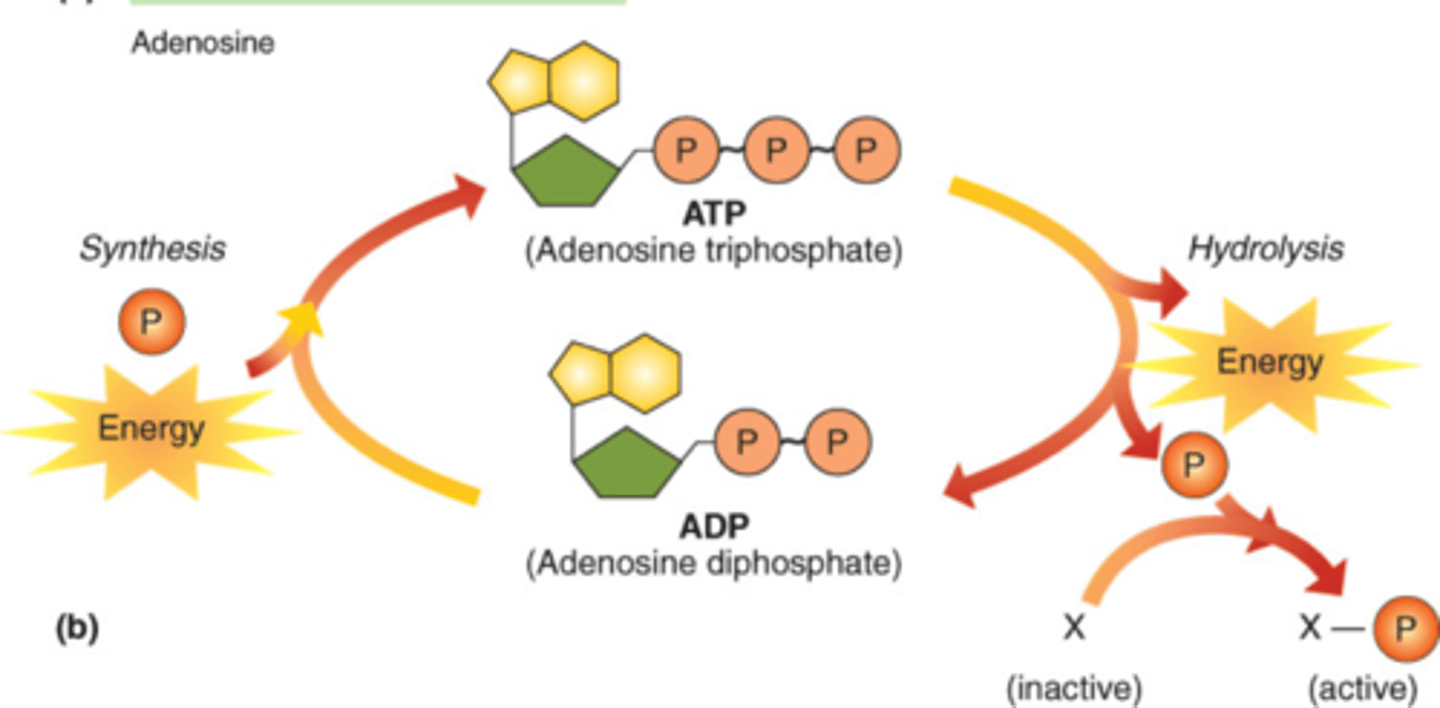
ATP cycle
To use ATP the bond breaks by hydrolysis between the 2nd and 3rd phosphate releasing energy (exergonic) to synthesize ADP. To go back to ATP a free phosphate must attach to ADP. This requires energy input.
Proteomes
all the proteins that cells make
Enzymes
protein catalyst in living cells.
May change the local environment of reactants.
Sometimes they participate in the chemical reaction
Importance of enzymes
lowers the activation energy to overcome the bonding and achieve the transition state
2 ways of enzyme have to lower activation energy
Straining the reactants
Bringing them close together
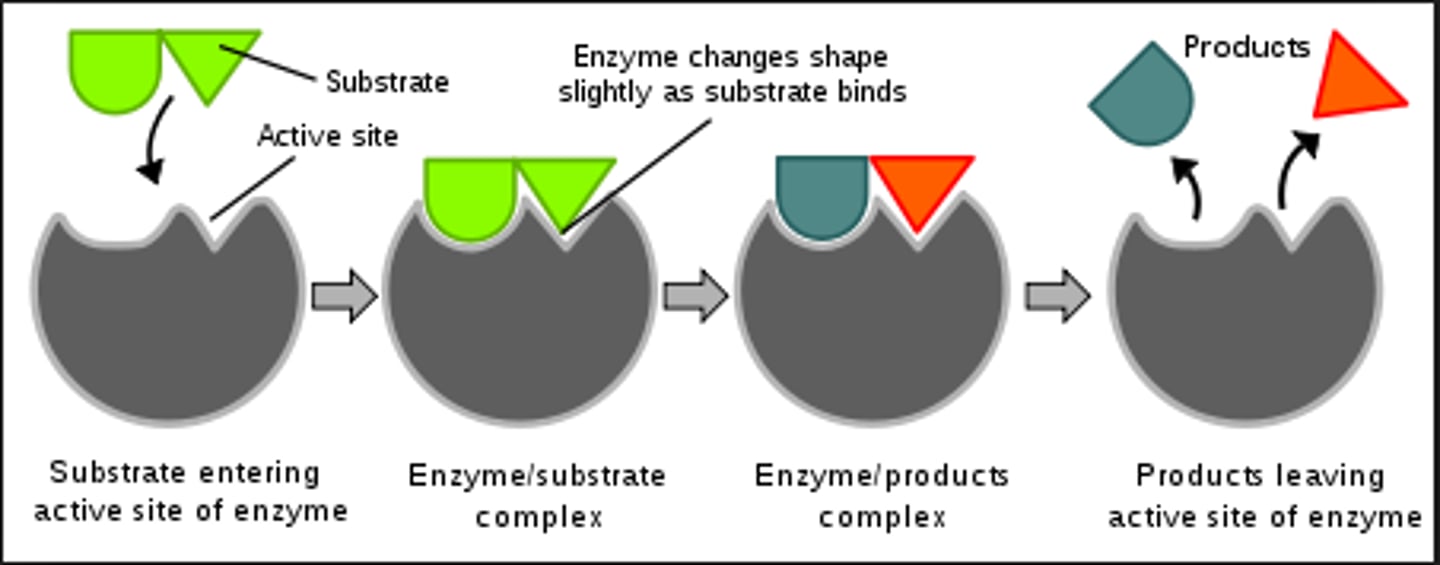
Active site
location in an enzyme where the reaction takes place
Substrate
reactant that binds to enzyme at the active site, and are part of chemical reaction
Enzyme-substrate complex
binding between an enzyme and substrate
Key features of enzymes
Ability to bind the substrates with a high degree of specificity.
Induced fit.
Enzymes are reusables.
Induced fit
interaction between enzymes and substrates involve conformational changes that causes the substrate to bind tightly to enzyme. It lowers the activation energy
Conformational change
alteration in the shape usually the tertiary structure of a protein as a result of alteration in the environment ph, temperature, ionic strength) or the binding of a ligand (to a receptor) or binding of substrate (to an enzyme)
Affinity
degree of attraction between an enzyme and its substrate
Enzyme-catalyzed reaction
Step 1:
substrate binds to enzyme
Step 2:
enzyme undergoes conformational change that binds the substrates more tightly. This induced fit strains
chemical bonds within the substrates and/or brings closer together
Step 3:
substrates are converted to products.
Step 4:
Products are releases. Enzyme is ready to be reused.
V max
maximal rate or velocity of a chemical reaction
Concentration reaches a plateau
It means most of the active site are occupied with substrate. Further the line the effect is negligible
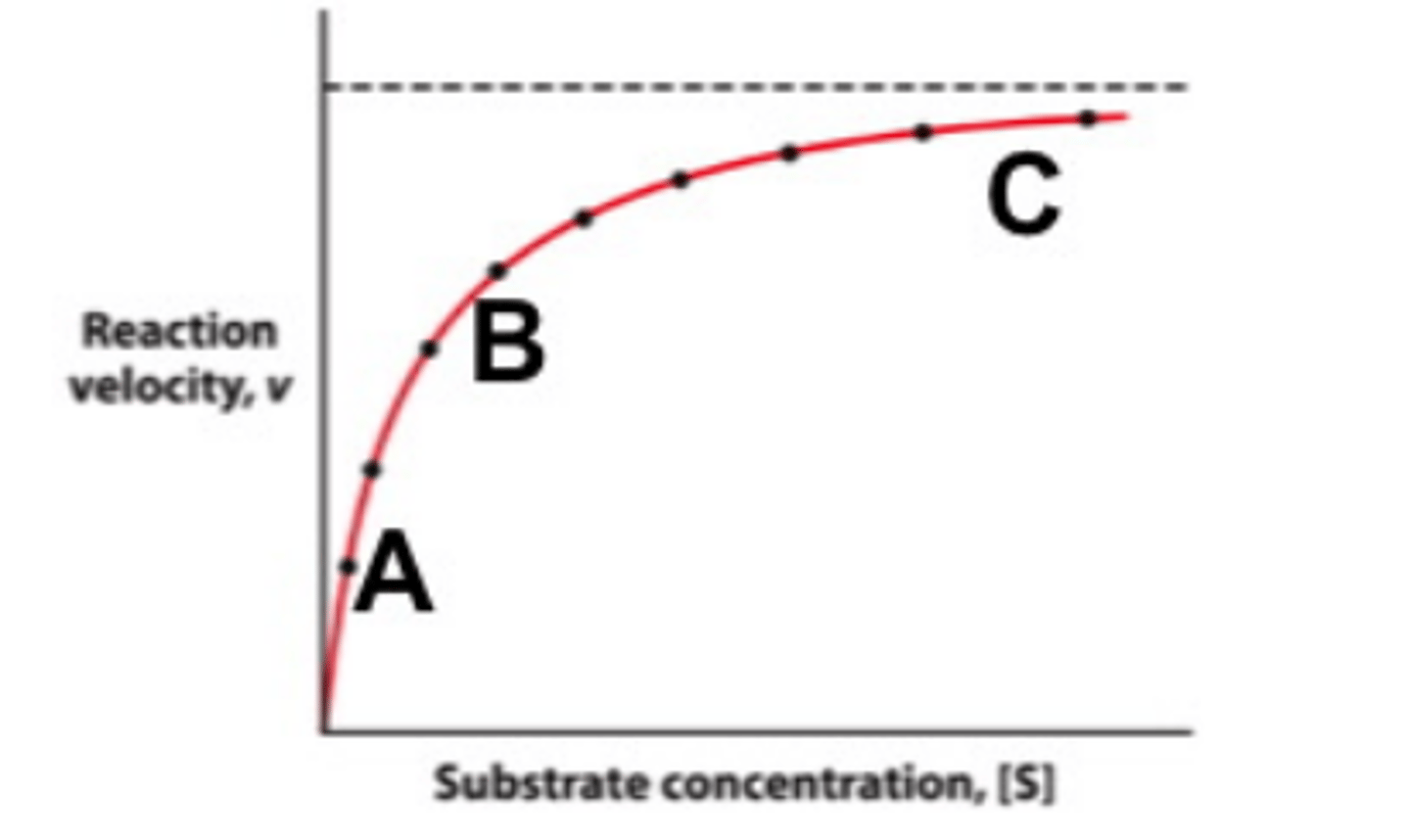
K m
substrate concentration at which velocity is half its maximal value
Reversible inhibitors
cells use them to modulate the enzyme function.
It allows enzyme to be reusables when inhibitor concentration is lowered
Competitive inhibitors
compete with substrate for the ability to bind to enzyme
Characteristics of competitive inhibitors
Non covalently bond
Its structure is like the enzyme's substrate.
It increases K m
It requires higher K m to achieve the same rate of chemical reaction.
Adding more substrate, increases the chances to create real substrate and overcome inhibitor.
Characteristics of noncompetitive inhibitor
Non covalently bond
It lowers the V max, so K m is the same.
It binds outside of the enzyme, allosteric site.
It never reaches the velocity of original reaction.
At the beginning the slope are the same because the system is still open.
V max is different because the substrate will never accommodate well.
Allosteric site
The place on an enzyme where a molecule that is not a substrate may bind, thus changing the shape of the enzyme and influencing its ability to be active
Irreversible inhibitors
Permanently inactive an enzyme to further use.
Covalently bond
Factors that influence Enzyme function
Reversible and irreversible Inhibitors.
Prosthetic groups.
Cofactors.
Coenzymes.
They are affected by environment.
Only function in a narrow range of pH
Why enzyme function changes when pH is changed?
because:
- Enzyme cannot maintain 3 dimensional shape
- The ezyme becomes unstable because the proton amount wil be altered by the change in pH
Prosthetic groups
small molecules that are permanently attached to the surface of an enzyme and aid enzyme function
Cofactors
inorganic ions that temporarily bind to the surface of an enzyme and promote chemical reaction
Coenzyme
Organic molecule that is a necessary participant in some enzymatic reactions; helps catalysis by donating or accepting electrons or functional groups; e.g., a vitamin, ATP, NAD+.
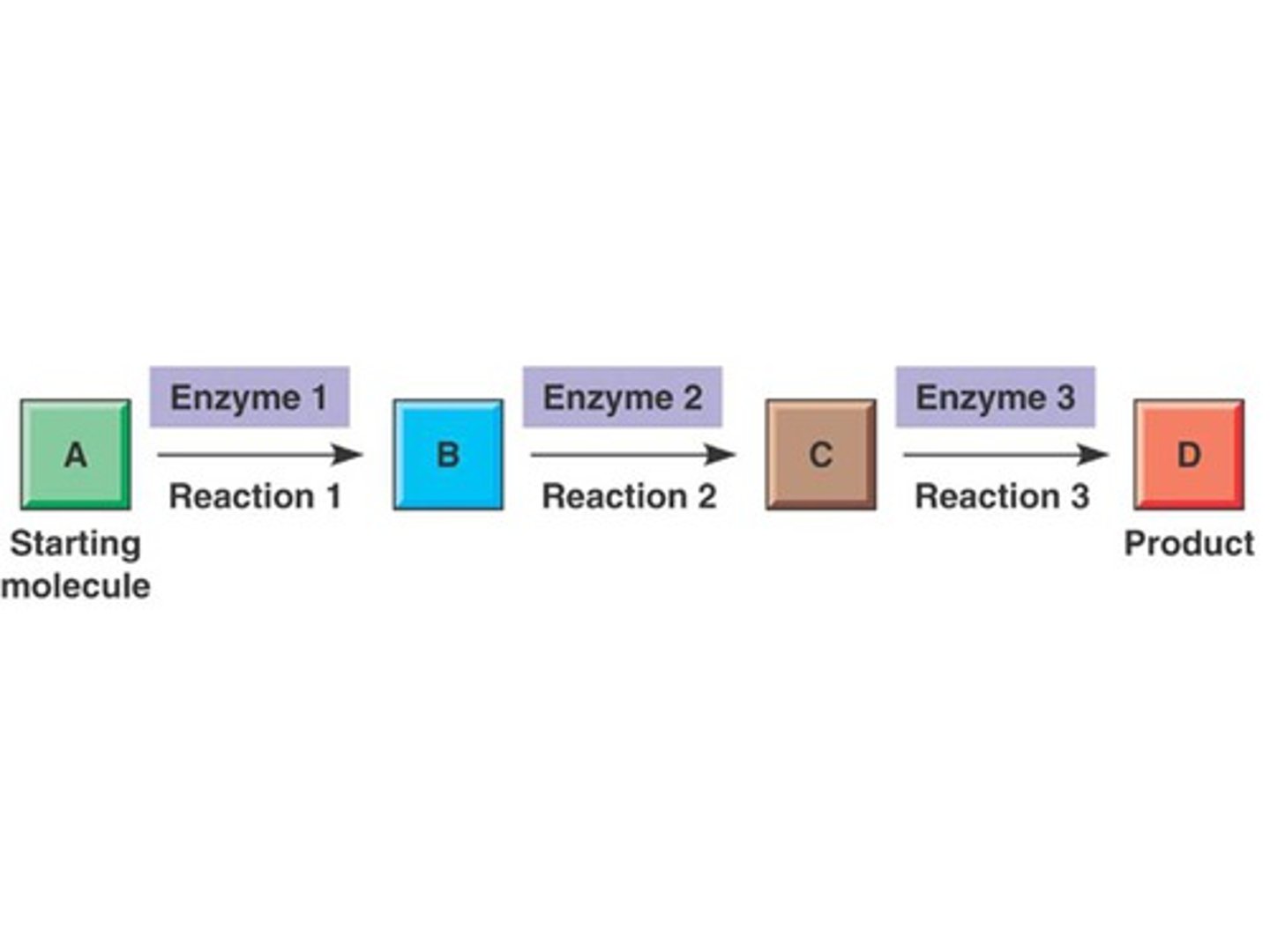
Metabolic pathways
A series of chemical reactions that either builds a complex molecule or breaks down a complex molecule into simpler compounds when it's needed
Energy intermediates
released energy of the broken covalent bonds is stored in them, which are directly used to drive endergonic reactions
E.g: ATP, NADH
Substrate-level phosphorylation
enzyme transfers a phosphate from organic molecule to ADP, thereby making ATP
Chemiosmosis
energy is stored in an ion electrochemical gradient is used to make ATP from ADP and Pi
2 ways to make ATP
Substrate-level phosphorylation
Chemiosmosis
Redox reaction
are important in the metabolism of small organic molecules
Oxidation
loose electron (less energy)
Reducing agent
Reduction
Gain electron (has more energy)
Oxidizing agent
NAD+
nicotinamide adenine dinucleotide.
It's composed by two nucleotide; one with adenine base and other one with nicotinamide base.
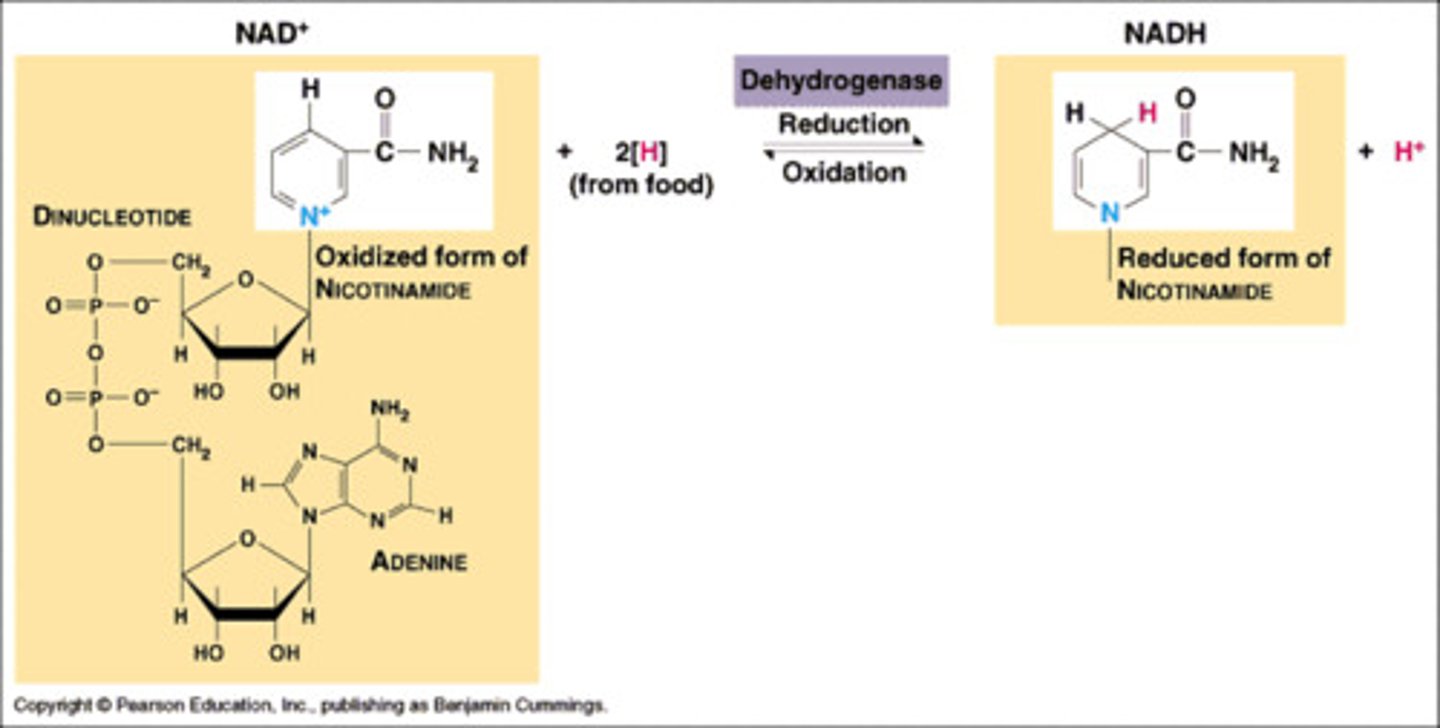
Function of NADH
it's use to make ATP.
It can donate electrons to other organic molecules and thereby energize them to form covalent bonds
NAD+ bonding to NADH
redox reaction
two electrons and H+ are incorporated to ring
Regulation of metabolic pathways
Gene regulation
Cellular regulation
Biochemical regulation
Characteristics of metabolic pathways
Regulations ensure that cell synthesizes molecules when they're needed.
Each step is catalyzed by a specific enzyme
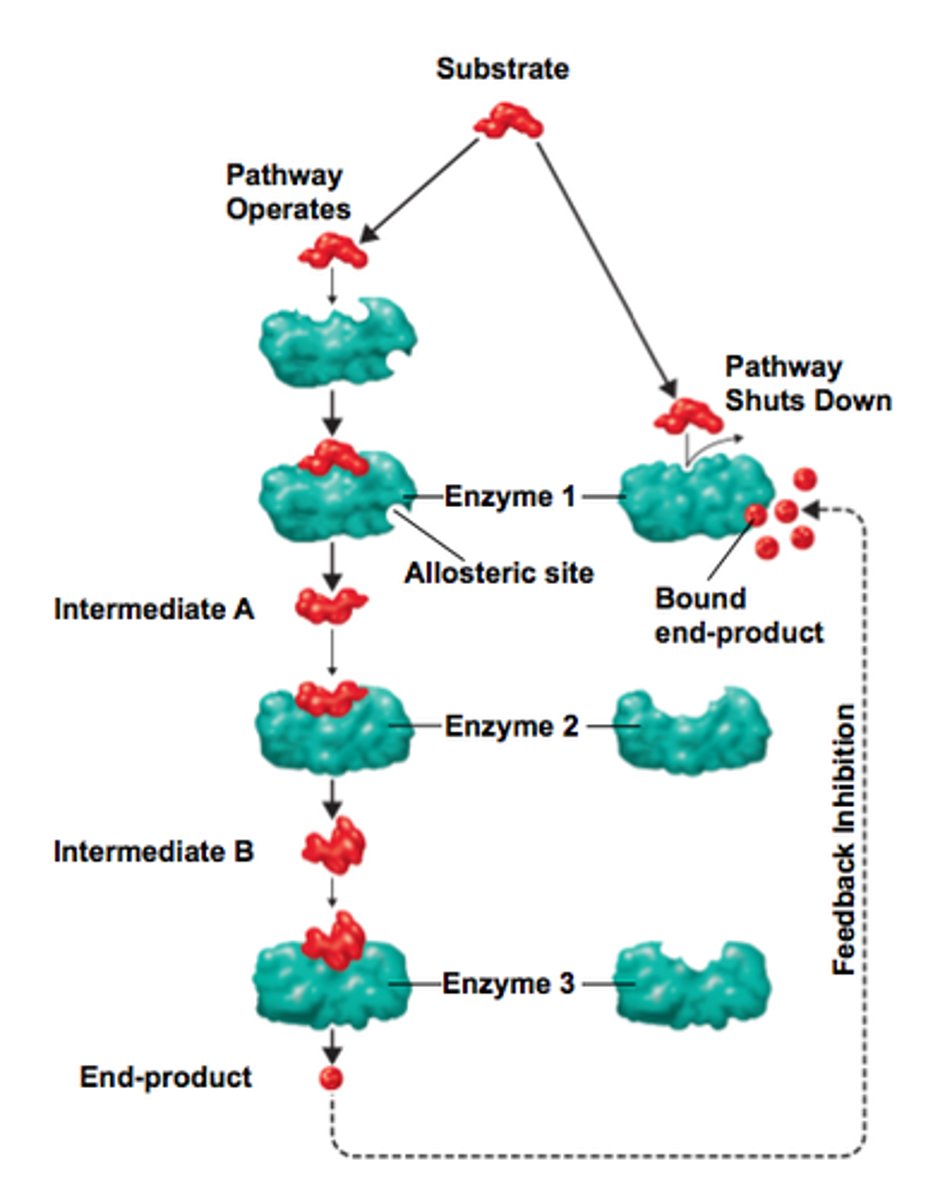
Feedback inhibitors
ability of the cell to use the end product of metabolic pathway to react/inhibit with the initial step through the allosteric site , making a conformation change in the enzyme that shuts down pathway.
In order to prevents the over accumulation of the product in the cell
Recycling of organic molecules
saves a great deal of energy for living organisms
mRNA degradation
they're are degraded by 5' to 3' exonucleases or by exosomes
Exonuclease
enzyme that cuts a nucleic acid chain at the end
Exosome
multiprotein complex that degrades mRNA
Proteasome
degrades proteins in ekaryotes and archea
Autophagy
lysosomes digest intracellular material
Difference between competitive and noncompetitive inhibitor on effects of enzyme function
- competitive inhibitor bind to active site, while noncompetitive inhibitor binds to allosteric site
- only the effects of competitive inhibitor can be overcome by increasing substrate concentration
Function of catabolic pathways
Two functions:
- recycle macromolecules
- produce energy intermediate e.g: ATP, which is use to drive endergonic reactions
Direction and rate of chemical reaction
Thermodynamics govern the direction but not he rate of ____
The change in free energy determines the direction of the ____
Catalyst influence in the rate of ____
Chemical reactions depend on
rate and direction