Topic 1 Atomic structure and the periodic table
1/56
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
57 Terms
Atoms
Atom have a radius of 0.1 nm (1 × 10⁻¹⁰ m)
Nucleus has a radius of 1 × 10⁻¹⁴ m
The nucleus is made up of protons and neutrons - overall positive charge
Electrons orbit the nucleus at different distances - energy levels or shells
Most of the mass of an atom is concentrated in the nucleus
Atoms have no overall charge because the positively charged nucleus and the negatively charged electrons cancel eachother out - number of protons = number of electrons
Particles
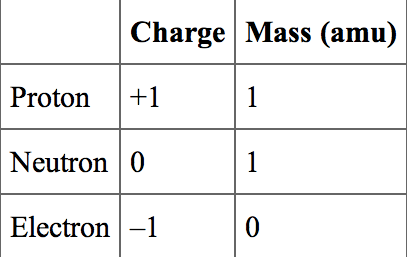
Mass number and atomic number
Mass number is the number of protons and neutrons
Atomic number is the number of protons (and electrons)
Protons determine the element of the atom
What is an atom?
An atom is the smallest part of an element that can exist
All substances are made of atoms
What is an element?
An element is a substance made up of atoms (only one tupe of atoms) that all have the same number protons in their nucleus
What are isotopes?
Isotopes are atoms of the same element that have the same number of protons but different number of neutrons
What is the formula for relative atomic mass of isotopes?
Ar = sum of (isotope abundance x isotope mass number) / sum of abundances of all isotopes
Cu -63 has an abundance of 69.2% and Cu -65 has an abundance of 30.8%.
Calculate the Ar of copper to 1.dp
Ar = (69.2 × 63) + (30.8 × 65) / 69.2 + 30.8
= 63.6 (1.dp)
The development of the model of the atom
Before the discovery of the electron, atoms were though to be tiny spheres that could not be divided (John Dalton)
Plum pudding model - the discovery of the electron led to the plum pudding model of the atom. The plum pudding model suggested that the atom is a ball of positive charge with negative electrons embedded in it (JJ Thompson)
Nuclear model - Rutherford fired positively charged alpha particles at a thin sheet of gold. From the plum pudding model, the particles were expected to pass straight through the sheet. Most of the particles did go through, but some were deflected more than expected and a small number were deflected backwards
→ Most of the alpha particles went straight through the foil. Therefore the atoms were mainly empty space
→ Some of the alpha particles were deflected. Therefore the centre of the atom must have a negative charge that repelled the alpha particles
→ Sometimes the alpha particles bounced straight back. Therefore the mass of the atom must be concentrated in the centre - called the nucleus
So Neils Bohr adapted the model by suggesting electrons orbit the nucleus at specific distances. Bohr’s work agreed with the results of experiments by other scientists. ‘Orbits’ are called shells or energy levels
Later experiments led to the idea that the positive charge in the nucleus in due to positive particles in the nucleus called protons
James Chadwick discovered that the nucleus contains neutrons - these have no charge and are neutral
What is a molecule?
2 or more atoms chemically bonded together
What is a compound?
A substance made up of 2 or more different elements chemically bonded together
What is a mixture?
2 or more elements or compounds that are not chemically bonded together
How can mixtures be separated?
Using physical processes, such as:
Filtration
Crystallisation
Simple distillation
Fractional distillation
Chromatography
(No new substances are formed)
What are the reactants and products in this reaction? methane + oxygen → carbon dioxide + water
Methane and oxygen are the reactants
Carbon dioxide and water are the products
Balance the equation: CH4 + O2 → CO2 + H2O
CH4 + 2O2 → CO2 + 2H2O
How can electronic structure be represented?
As numbers - 1st shell has 2 electrons, 2nd shell has 8 electrons, 3rd shell has 8 electrons (e.g neon - 2,8)
As a shell diagram showing electrons in each energy level
How are elements arranged in the periodic table?
Elements are arranged in order of increasing atomic (proton) number
Why is it called the periodic table?
Because similar chemical properties repeat at regular intervals across the table
What is a group in the periodic table?
A group is a vertical column of elements
Elements in the same group have similar chemical properties (react in a similar way)
Why do elements in the same group have similar properties?
They have the same number of electrons in their outer shell
How did early scientists arrange the periodic table?
They arranged elements by atomic weight, but this sometimes placed elements with different properties in the same group
How did Dmitri Mendeleev improve the periodic table?
Left gaps for undiscovered elements, when new elements were found, their properties fitted Mendeleev’s predictions, helping to confirm his ideas
Predicted their properties
Sometimes changed the order of atomic weights of specific elements so they fitted the patterns of other elements in the same group
Later discoveries and knowledge of isotopes explained why atomic weight order was not always correct
What is the difference between metals and non-metals in reactions?
Metals react to form positive ions (lose electrons)
Non-metals do not form positive ions (they usually gain or share electrons)
Where are metals and non-metals found in the periodic table?
Metals - left side and towards the bottom of the periodic table
Non-metals - right side and top of the periodic table
Properties of metals
Strong
Malleable
Conductors of heat and electricity
High melting and boiling points
Shiny
Properties of non-metals
Dull
Brittle
Do not conduct electricity
Low melting and boiling points
Low densities
What are transition metals?
Metals that can form more than one ion
They make very good catalysts
What are the elements in Group 0 called?
Noble gases
Why are noble gases unreactive?
They are unreactive because they have a full outer electron shell , giving them a stable electron arrangement
They do not have to lose or gain electrons to become stable
How many electrons do noble gases have in their outer shell?
Most noble gases have 8 electrons in their outer shell
Helium is an exception and has 2 electrons
What happens to the boiling points of noble gases down Group 0?
Boiling points increase going down the group along with relative atomic mass increasing
Properties of noble gases
Gases at room temperature
Non-flammable
Why can we predict the properties of Group 0 elements?
Because elements in the group have the same outer electron structure, their properties follow clear trends down the group
What are Group 1 elements?
Group 1 elements are called alkali metals
What do they have in common?
They all have one electron in their outer shell, which gives them similar chemical properties
How do the first three alkali metals react with water?
Lithium, sodium, and potassium react with water to produce:
a metal hydroxide
hydrogen gas
The reaction becomes more vigorous down the group (the more reactive the alkali metal is, the more violent the reaction)
How do alkali metals react with oxygen and chlorine?
With oxygen → form metal oxides
What are the colour flames when group 1 metals react with oxgen?
Lithium burns crimson
Sodium burns yellow
Potassium burns lilac
These reactions are why group 1 metals tarnish - they react with oxygen in the air to form a dull, metal oxide layer
What is the trend in reactivity in Group 1? (important)
As you go down group 1, the reactivity increases because the outer electron is further from the nucleus
These is increased shielding from the inner electron shells
So there is a weaker attraction between the electron and the nucleus, which makes the electron easier to lose, increasing reactivity
How do alkali metals react with chlorine?
With chlorine → form chloride salts
Group 1 metals react vigorously when heated in chlorine gas
As you go down the group, reactivity increases so the reaction with chlorine gets more vigorous
What are Group 7 elements?
Group 7 elements are called halogens
What do they have in common?
They are non-metals
Each halogen has seven electrons in its outer shell, giving them similar chemical properties
They exist as diatomic molecules (X₂)
How do halogens react with metals and non-metals?
With metals → form ionic halide salts
With non-metals → form covalent compounds
What is the trend in physical properties down Group 7?
Relative molecular mass, melting point, and boiling point increase going down the group
What is the trend in reactivity down Group 7?
As you go down group 7, the reactivity decreases because the outer electrons are further from the nucleus
There is increased shielding from inner electron shells
So there is a weaker attraction between the nucleus and the incoming electron, which makes it harder for the atom to gain an electron, decreasing reactivity
What is a halogen displacement reaction?
A more reactive halogen can displace a less reactive halogen from an aqueous solution of its salt
Example:
Cl₂ can displace Br⁻ from bromide solution
What is filtration used for?
Used to separate an insoluble solid from a liquid
Filtration practical
1) Set up filter paper in a funnel over a container (e.g a conical flask)
2) Pour the mixture into filter paper
3) The liquid passes through the tiny holes in the filter paper
4) The solid material is left in the filter paper
5) At the end, the liquid is separated from the solid
What is crystallisaiton used for?
Used to separate a soluble solid from a liquid
Crystallisation practical
1) Pour the solution into an evaporating dish
2) Gently heat the solution using a water bath or Bunsen burner until some of the solvent evaporates
3) Allow the solution to cool so it becomes saturated
4) Crystals of the solute form as the solution cools
5) Remove the crystals by filtration and dry them between filter paper
What is simple distillation used for?
Used to separate a liquid from a solution
Example:
Used to produce drinking water from sea water
Simple distillation practical
1) Pour the solution containing the liquid and dissolved solid into a distillation flask
2) Connect the flask to a condenser; ensure cold water runs continuously through the condenser to keep the internal glass cold
3) Heat the solution using a Bunsen burner
4) As we heat the solution, the liquid start to evaporate turning into vapour, which rises through the glass tube
5) The vapour passes over the thermometer, and then enters the condenser, where it cools and condenses back into a liquid
5) The vapour passes into the condenser turning back into a liquid
6) Collect the distilled liquid in a beaker. At the end, the solid remains in the flask, while the pure liquid is collected separately
What is fractional distillation used for?
Used to separate a mixture of different liquids with different boiling points
Example:
Crude oil
Fractional distillation apparatus
In the flask we have a mixture of two different liquids
The flask is attached to a long column containing glass beads - this is called the fractionating column. They give a larger surface area, so the vapour can condense and evaporate many times, improving the separation of liquids with different boiling points.
The rest of the apparatus is the same as simple distillation
Fractional distillation practical
1) Gently heat the mixture. Both liquids begin to evaporate, but the liquid with the lower boiling point evaporates more easily. The vapours enter the fractionating column
2) In the fractionating column, the vapours condense and drip back into the flask, then evaporate again. This repeated evaporation and condensation increases the concentration of the lower boiling point liquid in the column
3) The warm vapours pass the thermometer, and the temperature reading rises
4) Vapours pass into the condenser and cool, turning back into a liquid. When the thermometer reaches the boiling point of the first liquid, the first liquid is collected
5) When the thermometer reaches the boiling point of the second liquid, the second liquid is collected
Liquids with very similar boiling points are harder to separate effectively
What is chromatography used for?
Used to separate and identify the different substances in a mixture
Chromatography practical
1) Draw a pencil line near the bottom of a sheet of filter paper. Use a pencil as it is insoluble
2) Place a small dot of each substance (e.g ink or dye) on the pencil line
3) Pour a small amount of solvent (e.g water or ethanol) into a beaker and place the bottom of the paper in the solvent without submerging the dots
4) As the solvent moves up the paper, it dissolves the substances, carrying them at different rates depending on their solubility and attraction to the paper
5) Separate spots appear on the paper, allowing you to identify or compare substances