BI108 Quiz 1
1/88
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
89 Terms
What does the null hypothesis imply? How do you reject it?
There is no relationship between variables and no significant difference between groups. To reject it, find the p-value, and if it's <0.05 H0 can be rejected
What does the alternate hypothesis imply?
There is a relationship between variables and there is a significant difference between groups
What is pH and how do you calculate it?
How acidic/basic a solution is, and it is calculated by -log[H+]
What is an acid?
A molecule that releases a proton when it dissolves
What is a base?
A molecule that acquires a proton/releases OH- when it dissolves
How do Van der Waals forces form?
As electrons move, their density may be temporarily asymmetrical, making fluctuating dipoles and allowing interactions (relatively weak)
What are macromolecules?
Polymers of smaller molecules (monomers) joined by covalent bonds
What kind of reaction is the formation of polymers?
Condensation reaction (releases water)
What elements form carbohydrates and in what ratio?
(CH20)n, where n is some factor to multiply by
What are the building blocks of carbohydrates?
Monosaccharides: 5-6 carbon sugars linked together
Carbohydrates: What is the structure of monosaccharides?
- a chain of carbons
- a carbonyl group
- several -OH hydroxyl groups
Carbohydrates: How do monosaccharides differ from each other?
- length of carbon chains
- orientation of carbonyl and hydroxyl groups
Carbohydrates: What are the different variations of monosaccharides?
disaccharides: 2 monosaccharides
trisaccharides: 3 monosaccharides
oligosaccharides: 3-20 monosaccharides
polysaccharides: 100s-1000s monosaccharides
Carbohydrates: What is a glycan?
many monosaccharides linked together
Carbohydrates: What is a glycosidic linkage?
a covalent bond between monosaccharides (specifically C-O-C)
Carbohydrates: What are the different orientations and examples of glycosidic linkages?
alpha: bent/branched networks, easier to break
- ex: starch (storage of glucose in plants), glycogen (storage of glucose in animals)
beta: linear fibers/sheets, harder to break because of hydrogen bonding
- ex: cellulose (structural components in plants), chitin (structural component in shells)
Carbohydrates: Forming glycosidic linkages occurs through which type of reaction?
Condensation
What are the main functions of carbohydrates?
1. energy storage and processing
2. structural support
3. identification of cells and cell parts
What makes lipids unique?
They are insoluble in water (hydrophobic) because they are comprised of C-C and C-H bonds that are nonpolar because they have similar electronegativity
Lipids: What are the 2 types of hydrocarbon chains?
Saturated: no double bonds, held together more tightly because they are straight, solid at room temperature (ex: animal fats)
Unsaturated: 1+ double bonds, bent shape, liquid at room temperature (ex: plant oils)
Lipids: What is the structure and function of fats?
Structure: 3 fatty acids with carboxyl groups interact with the hydroxyl group of one glycerol (3-carbon molecule) to form an ester bond.
Function: energy processing and storage
Lipids: What is the structure of steroids?
Carbon atoms arranged in 4 fused-rings, vary in R-groups attached to core structure
Lipids: What is the structure of phospholipids?
- 2 long hydrophobic tails
- a hydrophilic head with a phosphate group and a polar group (1+ charges or partial charges)
Lipids: How do phospholipids vary from each other?
Each has a different polar group and hydrocarbon tails that vary in length and/or saturation
What is an amphipathic lipid?
A lipid which has one region which is hydrophobic and one region which is hydrophilic.
Lipids: How do vesicles form?
They form spontaneously (without energy) because phospholipids are more stable that way based on:
- hydrophobic interactions of hydrocarbon tails
- hydrogen bonding between water and hydrophilic heads.
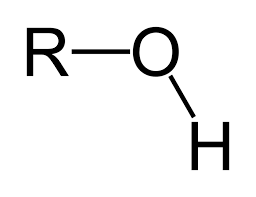
Functional groups: Hydroxyl
Polar
Functional groups: Methyl
Nonpolar
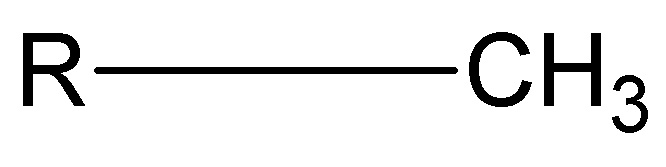
Functional groups: Carbonyl
Polar

Functional groups: Sulfhydryl
Polar
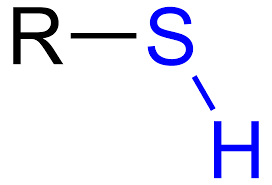
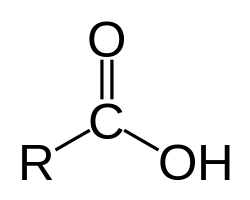
Functional groups: Carboxyl
Charged, ionizes to release H+ (acidic)
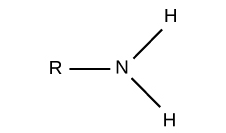
Functional groups: Amino
Charged, accepts H+ to form NH3 (basic)
Functional groups: Phosphate
Charged, ionizes to release H+ (acidic)

What are the building blocks of nucleic acids?
Nucleotides
Nucleic Acids: what are nucleotides made of?
A phosphate group, a 5-carbon sugar, and a nitrogenous base
Nucleic Acids: How do nucleotide connect?
They join through covalent bonds between the phosphate group of one and the sugar of another, called phosphodiester bonds
Nucleic Acids: How is the polarity of nucleotides descirbed?
Using 5’ to 3’ polarity, additional ones are added to the 3’ end.
Nucleic Acids: Describe the primary structure of DNA
It is the series of nucleotides consisting of the 4 bases (A, C, T, G) written in the 5’ to 3’ direction. They are connected along the sugar-phosphate backbone so the nitrogenous bases stick out
Nucleic Acids: Describe the RNA primary structure
It is similar to DNA in 5’ to 3’ polarity and the sugar-phosphate backbone, but Thymine is replaced by Uracil and it is single-stranded
Nucleic Acids: Describe the DNA secondary structure
Forms a double helix, but only if strands line up antiparellel, each G has an opposing C, and each A has an opposing T
Nucleic Acids: What is a purine?
A nitrogenous base that has 2 rings (A and G)
Nucleic Acids: What is a prymidine?
A nitrogenous base that has one ring (T and C)
Nucleic Acids: Describe the RNA secondary structure
Generally single stranded, but some form double helices when A-U and G-C hydrogen bond, forming a stem and loop structure
Nucleic Acids: Describe RNA teritiary structure
Multiple stem and loop structures twist and fold into a 3D shape
Nucleic Acids: What is a gene?
Instructions of nucleotides to make proteins
Proteins: Name the 6 functions of proteins
Catalysis: making reactions happen
Transporting materials
Movement
Cell structure
Defense
Signaling and communication
Proteins: Describe the structure of amino acids
amino group: NH2
Carboxyl group: COOH
R-group: what makes each one unique
Proteins: How to R-groups in amino acids vary?
they can be fully charged, partially charged, or not charged
They can be hydrophobic (clumping together) or hydrophilic (faces surrounding aqueous environment)
Proteins: Describe the primary structure of proteins
Amino acids linked with peptide bonds creating a polar peptide-bonded backbone with relatively flexible bonds
Proteins: Describe the secondary structure of a protein
Either fold into:
Alpha helix where R-groups stick out and are free to interact
Beta-pleated sheet wheee N-H and C=O groups stick out free to interact
Proteins: Describe the tertiary structure of a protein
Proteins fold again including secondary structures with 4 possible interactions:
Hydrogen bonds between partial charges of N-H and C=O groups in backbone and/or R-groups
Ionic bonds between full charges on R-groups
Covalent bonds (disulfide bridges) between S atoms in R-groups
Hydrophobic interactions between hydrophobic regions of R-groups (more stable that way)
Proteins: How is function ensured when the tertiary structure is forming?
Chaperone proteins ensure folding lines up for interactions
Proteins: What is denaturation?
The unfolding of proteins
Proteins: Describe the quaternary structure of proteins
Folded subunits stick together with similar interactions in tertiary
What are peripheral proteins?
Membrane proteins that are on the exterior or interior of the bilayer. They interact with water so they are polar and charged.
What are transmembrane proteins?
Membrane proteins that span the width of the bilayer, making the two ends that interact with water polar/charged and the middle that doesn’t nonpolar (amphipathic)
What determines the permeability of a membrane?
Length of hydrocarbon tails → longer tails = more hydrophobic
Saturation of hydrocarbon tails → saturated = more hydrophobic
Cholesterol presence → bulky rings cause packing, so makes it more hydrophobic
What determines if a molecule can pass through the membrane vs bump into it?
Nature of the molecule (smaller and uncharged molecules go through)
Structure of the membrane (less hydrophobic = more permeable)
What is entropy?
The tendency for systems to tend toward a state of randomness or disorder
What is osmosis and when does it occur?
The diffusion of water across membranes, occurs when:
2 solutions are separated by a lipid bilayer
solutions have different concentrations of dissolved ions/molecules
dissolved substances cannot move across the membrane, but water can
How is potential energy determined?
If a molecule has a high chance of moving (i.e. glucose at a high concentration wants to move to a lower concentration).
What is passive transport?
spontaneous movement of ions/molecules without the input of energy due to movement down the concentration gradient
What are the two proteins possible in facilitated diffusion?
Channels: transmembrane proteins that acts as tunnels to allow ions and other small molecules to pass
Carriers: proteins that allow molecules to bind and change shape (conformation) to move molcule
What are the properties of channel proteins?
Specificity: specific R-groups only admit 1 ion
Regulation: gated channels can open or close based on the presence/attachment of another molecules causing it to conform (i.e. ATP to chloride channel) or an electrical charge difference
Structure: interior of channel is hydrophilic due to polarity or charged R-groups
What are the properties of carrier proteins?
Specificity: specific R-groups only admit 1 ion
Regulation: controlled by the presence/attachment of another molecules causing it to conform
How do molecules bind to carrier proteins?
Molecules have kinetic energy where each one has an equal chance of hitting a binding site
What is exocytosis?
Active transport that allows larger molecules (i.e. waste) to leave the cell via vesicles
What is endocytosis?
Active transport that allows larger molecules to enter the cell via vesicles
What is active transport?
Movement of molecules up/against a concentration gradient (requires energy)
What are characteristics of Archaea?
live in extreme habitats (i.e. hot springs, deep sea) that are frozen, lack oxygen, very acidic, or very basic
unique hydrocarbon chains
unique carbohydrates in their cell walls
What are characteristics of Bacteria?
have cell walls and flagella
mostly single-celled, bit sometimes multicellular
most have a single circular chromosome (double helix)
What are characteristics of Eukaryotes?
multicellular or single-celled
nucleus is studded with pores that regulate what enters/exits the nucleus
Organelles: Cell membrane
Composed of lipid bilayer and membrane proteins
Function: regulates molecules in and out of the cell
Organelles: Ribosomes
Complex of RNA and protein molecules
Function: amino acids are linked and proteins are created using mRNA instructions
Organelles: Cell wall
a stiff wall made of cellulose fibers
Function: prevents plant cells from moving and helps them survive in harsh conditions
Organelles: flagella
long-thin projections
Function: propel the cell. through water
Organelles: nucleus
contains genetic material (usually chromosomes)
Function: DNA replication and gene expression
Organelles: nuclear envelope
2 membranes around the nucleus
Function: regulates trafficking of molecules (RNAs, proteins) via nuclear pore complexes
Organelles: mitochondria
surrounded by 2 membranes, inner one forms tubes and sacs
Function: site of cellular respiration and ATP production
Organelles: Vacuole
more prominent in plant cells
Function: stores key molecules, waste, and provides/regulates turgor pressure
Organelles: Choloroplasts
2 membranes, has stacks (vesicles)
Function: capture sunlight and convert to bonds in photosynthesis
Organelles: Endoplasmic Reticulum
forms a network of tubules and sacs near the nuclear envelope
Rough ER: areas with ribosomes for proteins to enter and be modified, folded, and transported
Smooth ER: no ribosomes, chemically modifies small molecules
Organelles: Golgi Apparatus
located near ER in a sequence of stacks close together
Function: receives proteins from the ER to package and store them
Organelles: Cytokeleton
dynamic network of long fibers (actin filaments, intermediate filaments, microtubules)
Function: movement and support
Organelles: Extracellular matrix
proteins and carbs
Function: protection and support in animal cells
What organelles are specific to plant cells?
cell wall
vacuoles are more prominent in plant cells
chloroplasts
What organelles are specific to animal cells?
Extracellular matrix
What is the endomembrane system?
The process of creating and transporting lipids and proteins:
ER → Golgi → vesicles → membrane
Why do organelles have a high surface-area-to-volume ratio?
Small volume = more likely for molecules to collide to interact
High surface area = more space for processes