LIFE 120 UNIT 2
1/123
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
124 Terms
Signaling pathways
cause chemical/abiotic factors to give the cell “orders”, telling the cell what chemical reactions it needs to do to survive
Metabolism
The totality of an organism’s chemical reactions, consisting of catabolic and anabolic pathways, which manage the material and energy resources of the organism.
metabolic pathway
A series of chemical reactions that either builds a complex molecule (anabolic pathway) or breaks down a complex molecule to simpler molecules (catabolic pathway).
catabolic pathways
A metabolic pathway that releases energy by breaking down complex molecules to simpler molecules.
Anabolic pathways
A metabolic pathway that consumes energy to synthesize a complex molecule from simpler molecules.
Energy
The capacity to cause change, especially to do work (to move matter against an opposing force).
Biosynthetic pathways
synthesis (creation) of new chemicals with a series of enzymes
Bioenergetics
1) The overall flow and transformation of energy in an organism. (2) The study of how energy flows through organisms.
Potential Energy
The energy that matter possesses as a result of its location or spatial arrangement (structure).
Chemical energy
potential energy stored in bonds available for release in a chemical reaction.
Kinetic energy
is energy associated with motion

Thermal energy
is kinetic energy associated with random movement of atoms or molecules. A type of kinetic energy
Thermodynamics
The study of energy transformations that occur in a collection of matter.
Includes: Closed system (thermos) and open system (organisms)
First law of thermodynamics
energy cannot be created or destroyed. Energy can be transferred and transformed into different energy types. (principle of conservation of energy)
Second law of thermodynamics
Every energy transfer or transformation increases the entropy of the universe
Entropy
A measure of molecular disorder, or randomness.
spontaneous process
A process that occurs without an overall input of energy; a process that is energetically favorable.
Basis of Biological Order and Disorder
Cells create ordered structures from less ordered materials
Organisms also replace ordered forms of matter and energy with less ordered forms
Free energy/Gibbs Free energy (G)
energy that can do work when temperature and pressure are ideal for the reaction, as in a living cell. The potential minimum energy a reaction requires to happen; the "free" energy usable for work.
If ΔG is lower than 0
it is spontaneous, Spontaneous processes can be harnessed to perform work
If ΔG is higher than 0
it is not spontaneous
The change in free energy (∆G) is
the difference between the free energy of the products and the free energy of the reactants. Predicts whether a reaction will happen on its own.
Free energy is a measure of
a system’s instability, its tendency to change to a more stable state
During a spontaneous change…
free energy decreases, and the stability of a system increases. Can perform work only when it is moving toward equilibrium
Equilibrium
forward and reverse reactions occur at the same rate; it is a state of maximum stability
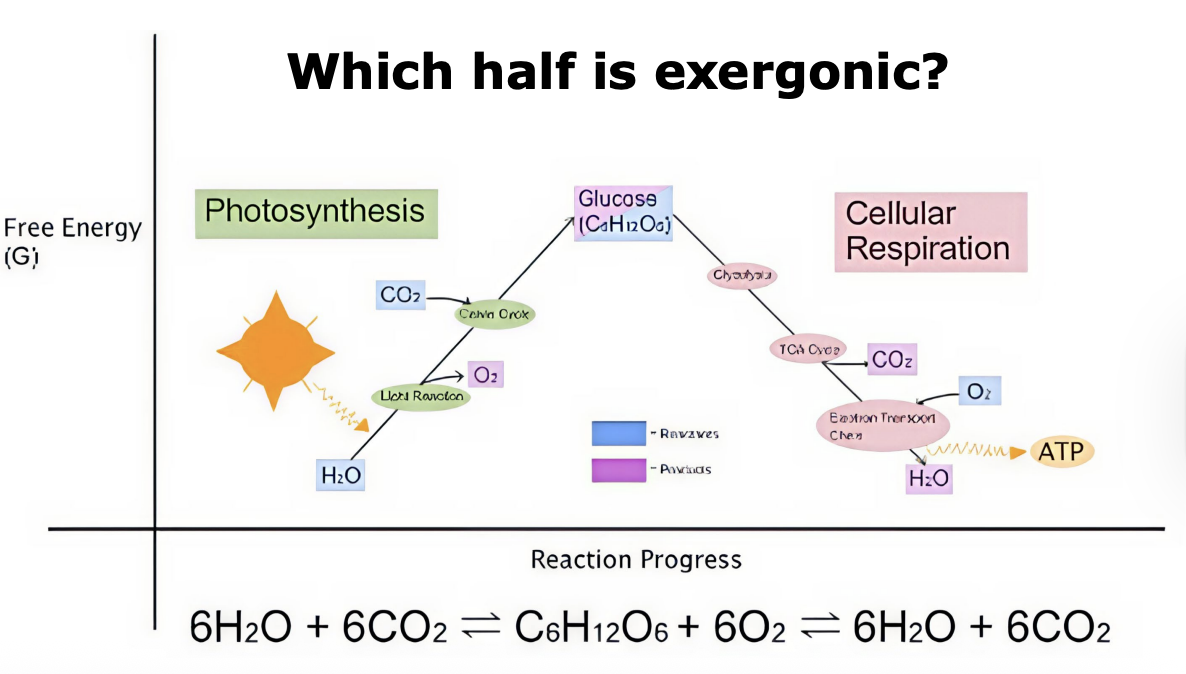
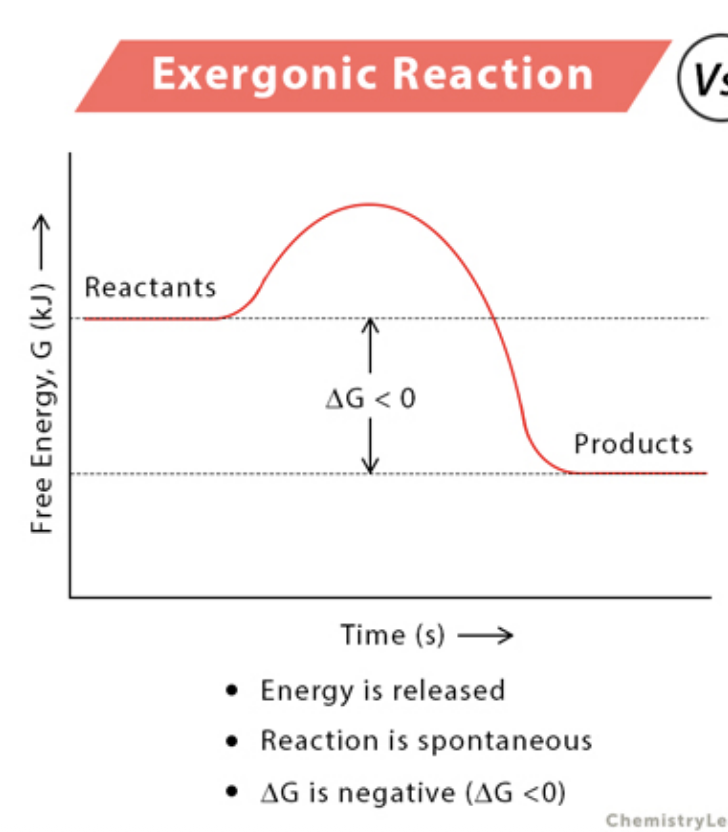
Exergonic reactions
a net release of free energy and is spontaneous; ∆G is negative. Reactants have more potential energy in their covalent bonds than the products. Ex: Cellular Respiration
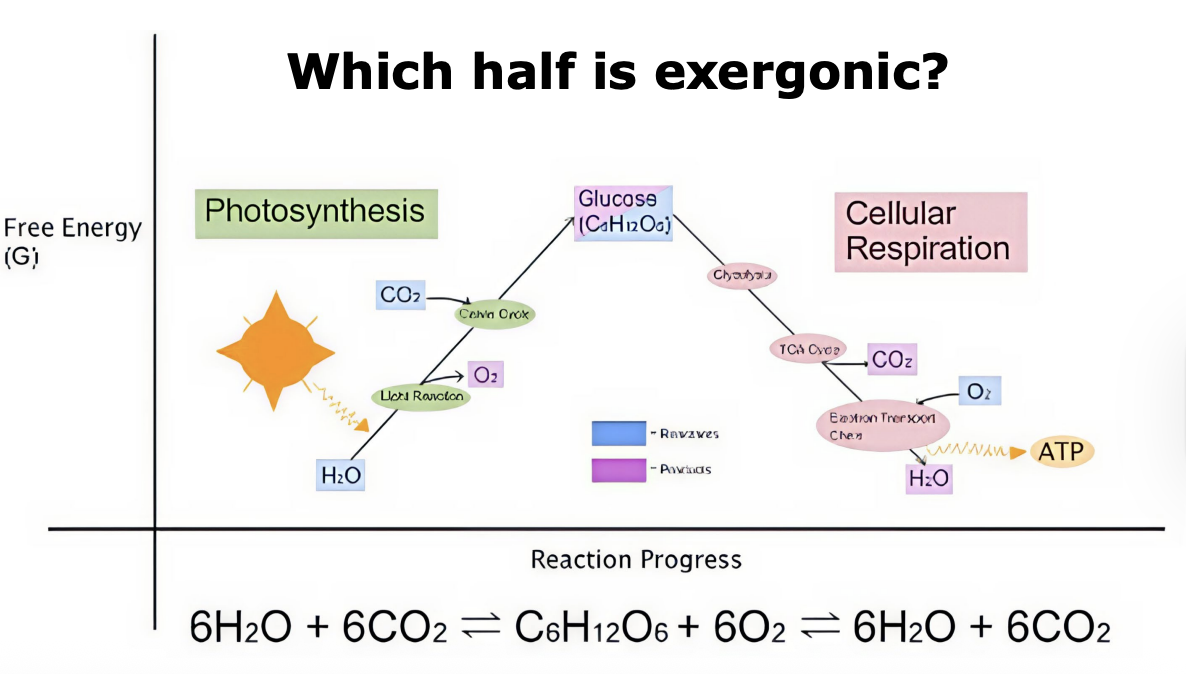
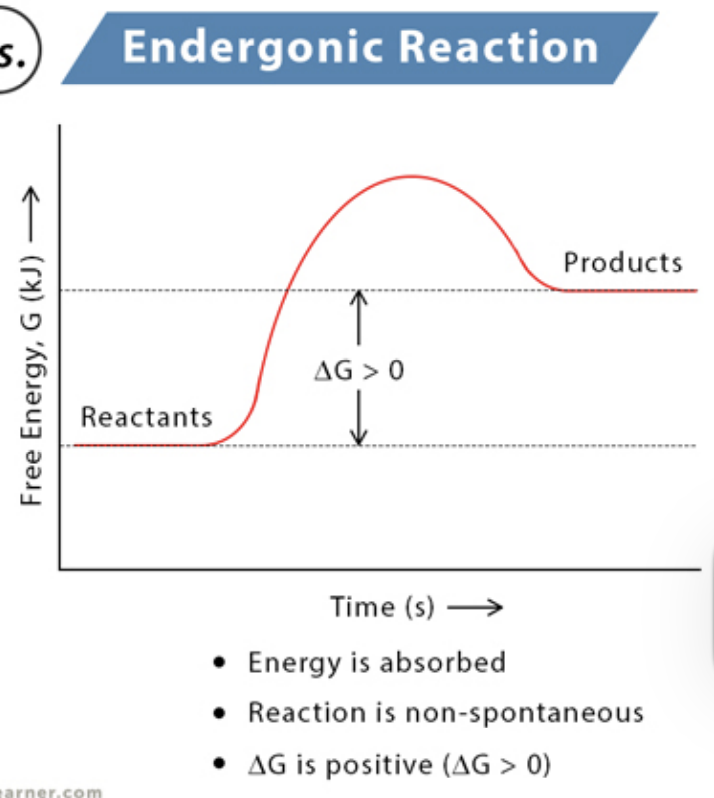
Endergonic Reactions
absorbs free energy from its surroundings and is nonspontaneous; ∆G is positive. Products have more potential energy than reactants. Ex: Photosynthesis
Reaction(energy) coupling
the use of energy released from an exergonic reaction to drive an endergonic reaction
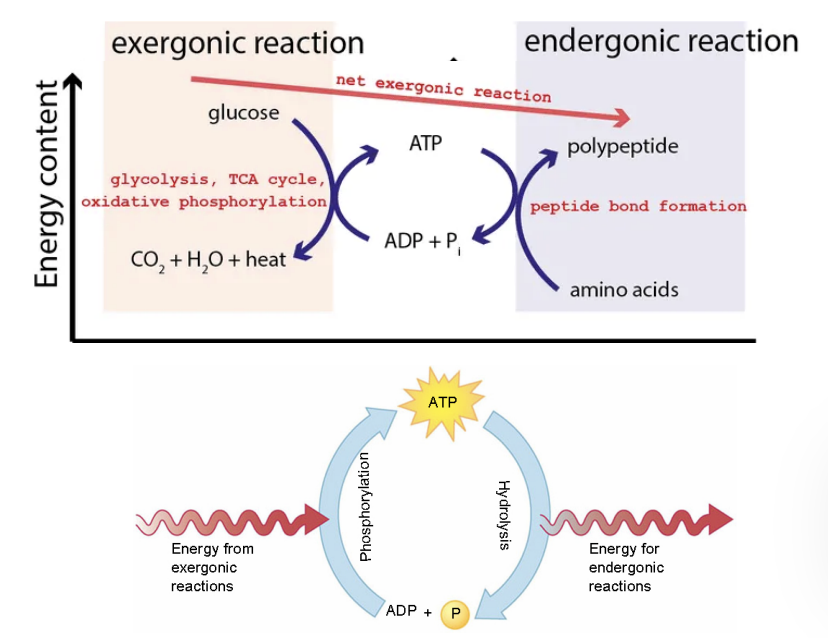
ATP drives endergonic reactions by…
phosphorylation of a reactant
phosphorylated intermediate
A molecule that has been phosphorylated by ATP. It becomes less stable and gains energy.
Adenosine triphosphate is composed of…
1. ribose (a sugar)
2. adenine (a nitrogenous base)
3. and three phosphate groups
How is ADP and energy formed from ATP
A hydrolysis reaction removing a phosphate group
How does ATP drive endergonic reactions?
ATP drives endergonic reactions by coupling them to ATP hydrolysis.
It transfers a phosphate group to a reactant (phosphorylation), creating an unstable intermediate.
The breakdown of ATP (exergonic, ΔG < 0) makes the overall reaction spontaneous (ΔG total < 0).
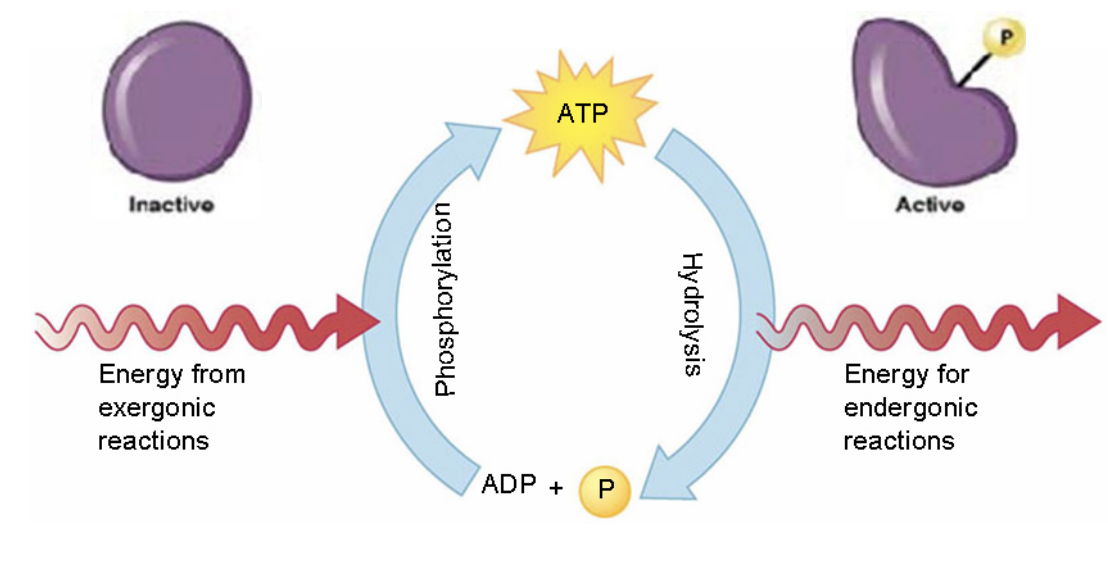
ATP is regenerated by..
The rephosphorylation of adenosine diphosphate (ADP). The energy to phosphorylate ADP comes from catabolic reactions in the cell
Activation energy
the energy barrier that prevents molecules from breaking down spontaneously
enzyme
A macromolecule serving as a catalyst, a chemical agent that increases the rate of a reaction without being consumed by the reaction. Most are proteins.
parts needed for a biochemical reaction to occur
1. Energetically favorable (-ΔG)
2. Overcome activation energy
3. Fast enough
Substrate
reactant that an enzyme acts on
The enzyme binds to its substrate, forming an enzyme-substrate complex
Active Site
the region on the enzyme where the substrate binds
Enzymes change shape due to…
chemical interactions with the substrate
This shape change allows the ACTIVE SITE to fit more snug around the substrate
How does induced fit help enzymes catalyze reactions?
When a substrate binds, the enzyme changes shape slightly to fit it better. Induced fit positions the active site’s chemical groups in the correct orientation to interact with the substrate. This enhances bond breaking/forming and lowers activation energy, increasing reaction rate.
How do temperature and pH affect enzyme activity?
Temperature and pH affect enzyme activity by influencing the enzyme’s shape. Changes in these environmental factors can alter the structure of the active site, which affects how well the enzyme binds to its substrate and catalyzes reactions.
What are optimal conditions for an enzyme?
Each enzyme has an optimal temperature and optimal pH at which it functions best. Under these conditions, the enzyme maintains its most active shape, allowing the highest reaction rate.
Why must enzyme activity be regulated in cells?
Chemical reactions in cells must be tightly regulated because uncontrolled enzyme activity could cause harmful or unnecessary reactions. Cells use regulatory mechanisms to activate or inactivate enzymes so reactions occur only when needed.
Activators
Compounds or enzymes that turn enzymes on and increase their activity (ex: transcription factors).
Inhibitors
Compounds or enzymes that turn enzymes off or reduce their activity (ex: intermediates in a metabolic pathway).
What are cofactors?
Cofactors help enzymes perform reactions and can sometimes regulate enzyme activity. They assist enzymes in carrying out reactions and may be inorganic (metal ions) or organic molecules. Examples include metal ions like iron (Fe) and molecules such as ATP that assist enzymes during reactions.
What is a coenzyme?
A coenzyme is an organic cofactor. Many coenzymes are derived from vitamins and help enzymes by carrying electrons or chemical groups. Examples include NADH, NADPH, and FADH.
Competitive inhibitors
bind to the active site of an enzyme, competing with the substrate
Noncompetitive inhibitors
bind to another part of an enzyme, causing the enzyme to change shape and making the active site less effective
What is allosteric regulation?
Allosteric regulation occurs when a molecule binds to a site on the enzyme other than the active site (the allosteric site), causing a shape change that can either stimulate or inhibit the enzyme’s activity.
What is cooperativity in enzymes?
Cooperativity occurs when the binding of one substrate molecule to an enzyme makes it easier for additional substrate molecules to bind, increasing the enzyme’s activity.
Is allosteric because binding by a substrate to one active site affects catalysis in a different active site
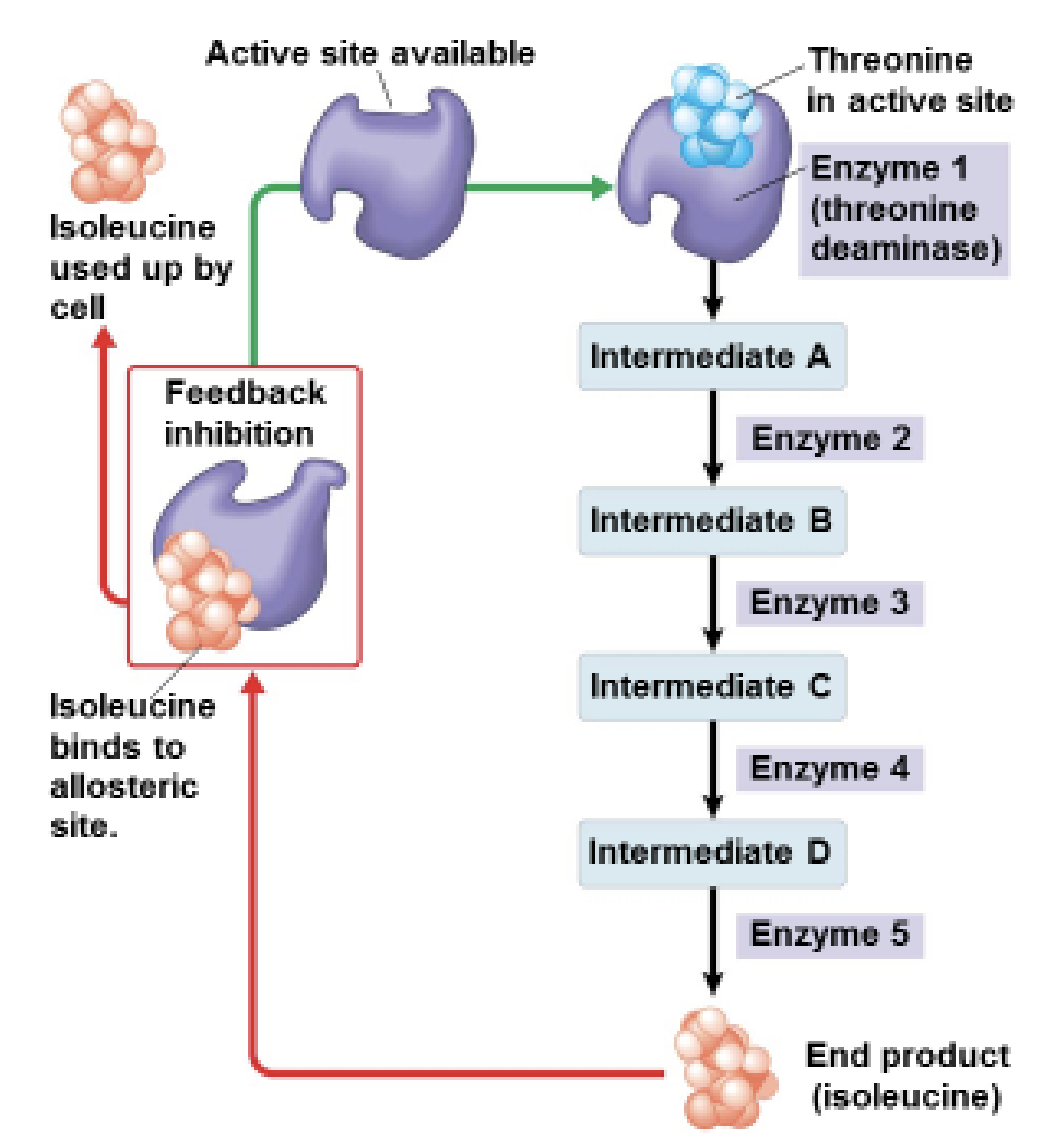
What is feedback inhibition?
A process where the final product of a reaction pathway shuts down an earlier enzyme.
This prevents the cell from making too much of that product.
central metabolism
The core, most important, chemical reactions.
Without these chemical reactions, the metabolism of specific chemicals, the cell would not live
cellular respiration
The catabolic pathways of aerobic and anaerobic respiration, which break down organic molecules and use an electron transport chain for the production of ATP. Exergonic and catabolic.
What is aerobic respiration?
Aerobic respiration is the process cells use to produce ATP by breaking down glucose using oxygen. It occurs mainly in the mitochondria and releases energy, carbon dioxide, and water.
What is the overall equation for aerobic respiration?
Glucose + Oxygen → Carbon dioxide + Water + ATP (energy)
fermentation
A catabolic process that makes a limited amount of ATP from glucose (or other organic molecules) without an electron transport chain and that produces a characteristic end product, such as ethyl alcohol or lactic acid.
What is anaerobic respiration?
Anaerobic respiration is the process of producing ATP without oxygen. It occurs in the cytoplasm and breaks down glucose through glycolysis followed by fermentation.
What are the products of anaerobic respiration?
Anaerobic respiration produces 2 ATP from glycolysis.
Can form lactic acid (in animals) or ethanol and CO₂ (in yeast) during fermentation.
How does ATP drive cellular reactions?
ATP drives reactions by phosphorylating a reactant, transferring a phosphate group to another molecule. This phosphorylation transfers energy to the reactant, making the reaction more likely to occur.
How does ADP gain energy to form ATP?
ADP gains energy by gaining electrons when a third phosphate group is added to form ATP. This is a reduction reaction, and gaining electrons means gaining energy.
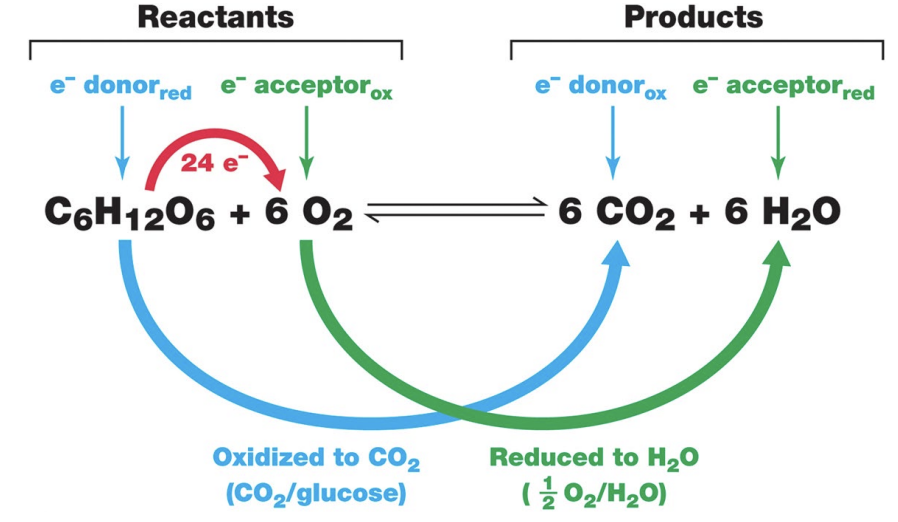
What is a redox reaction?
A redox reaction is a chemical reaction where electrons are transferred from one reactant to another. It includes two coupled processes: oxidation (loss of electrons) and reduction (gain of electrons).
What happens during oxidation and reduction?
Oxidation: a substance loses electrons and energy.
Reduction: a substance gains electrons and energy, reducing its positive charge.
How is energy obtained during cellular respiration? (Redox Terms)
Energy is obtained when glucose is oxidized to CO₂ during cellular respiration. As C–H bonds in glucose are broken and electrons are removed, energy is released and used to help form ATP.
What happens to electrons during cellular respiration?
During cellular respiration, electrons are transferred from glucose to oxygen. As electrons move through this process, energy is released and used to help produce ATP.
What role do NAD⁺ and FAD play in cellular respiration?
NAD⁺ and FAD are coenzymes that act as electron carriers in cellular respiration. They accept electrons removed from glucose and other organic molecules and transport them to later stages of respiration.
How does NAD⁺ become NADH?
A dehydrogenase enzyme removes two hydrogen atoms (2 electrons) from an organic molecule. NAD⁺ accepts the 2 electrons, becoming NADH, while one H⁺ ion is released.
Substrate-level Phosphorylation
Uses phosphorylation of substrates via kinase enzymes
Glycolysis – 2 ATP
Citric Acid Cycle – 2 ATP
Oxidative Phosphorylation
Relies on REDOX reactions
Electron Transport Chain: Helps make 28 ATP
Generates the most ATP
90% of all ATP in the cell is made via OxPhos
Define Glycolysis. Where does it occur?
Glycolysis is a 9-step pathway that breaks down glucose into two pyruvate molecules. It occurs in the cytoplasm and can happen with or without oxygen. The pyruvate produced can enter the citric acid cycle.
What are the main phases and products of glycolysis?
Glycolysis has two phases: energy investment and energy payoff. It produces a net gain of 2 ATP and 2 NADH, which carry electrons to the electron transport chain.
Explain what happens in the energy investment phase of glycolysis
Uses 2 ATP to phosphorylate glucose 2x into G3P
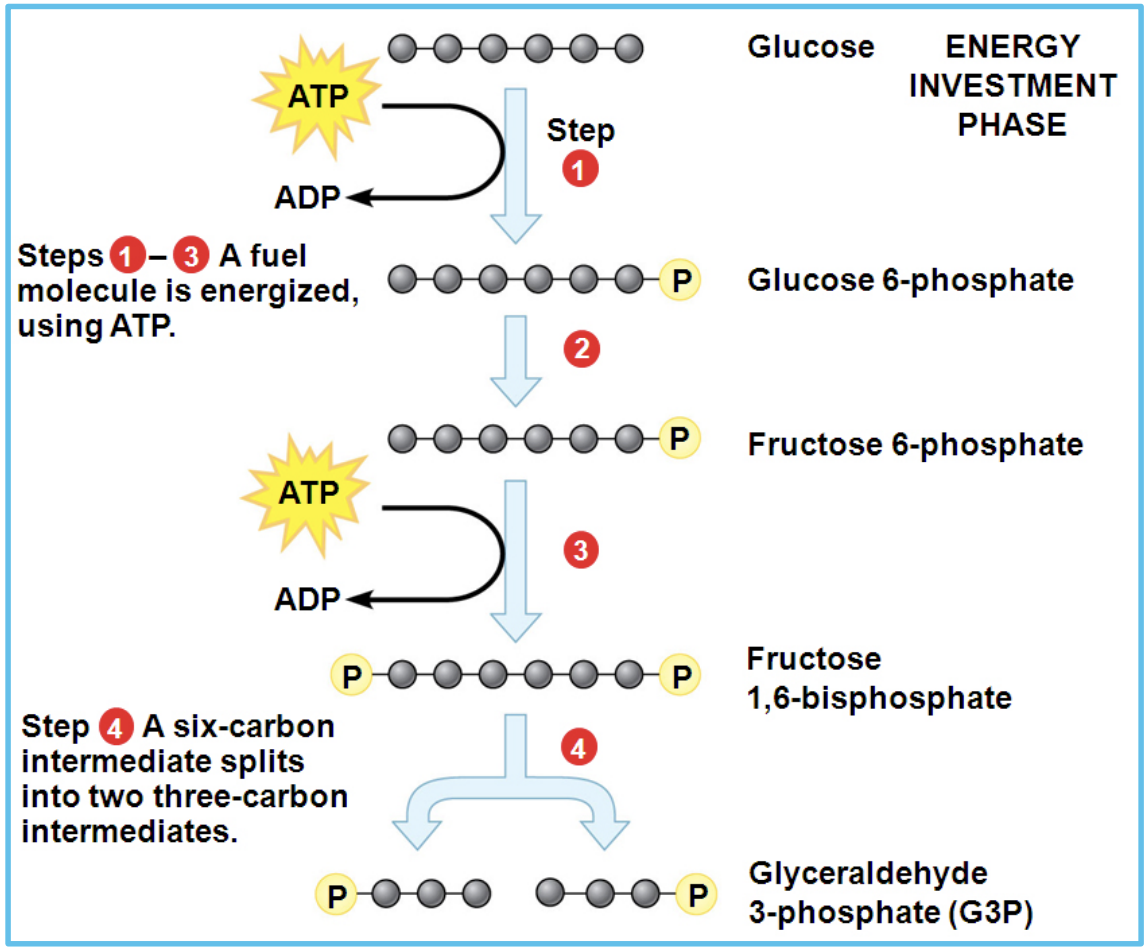
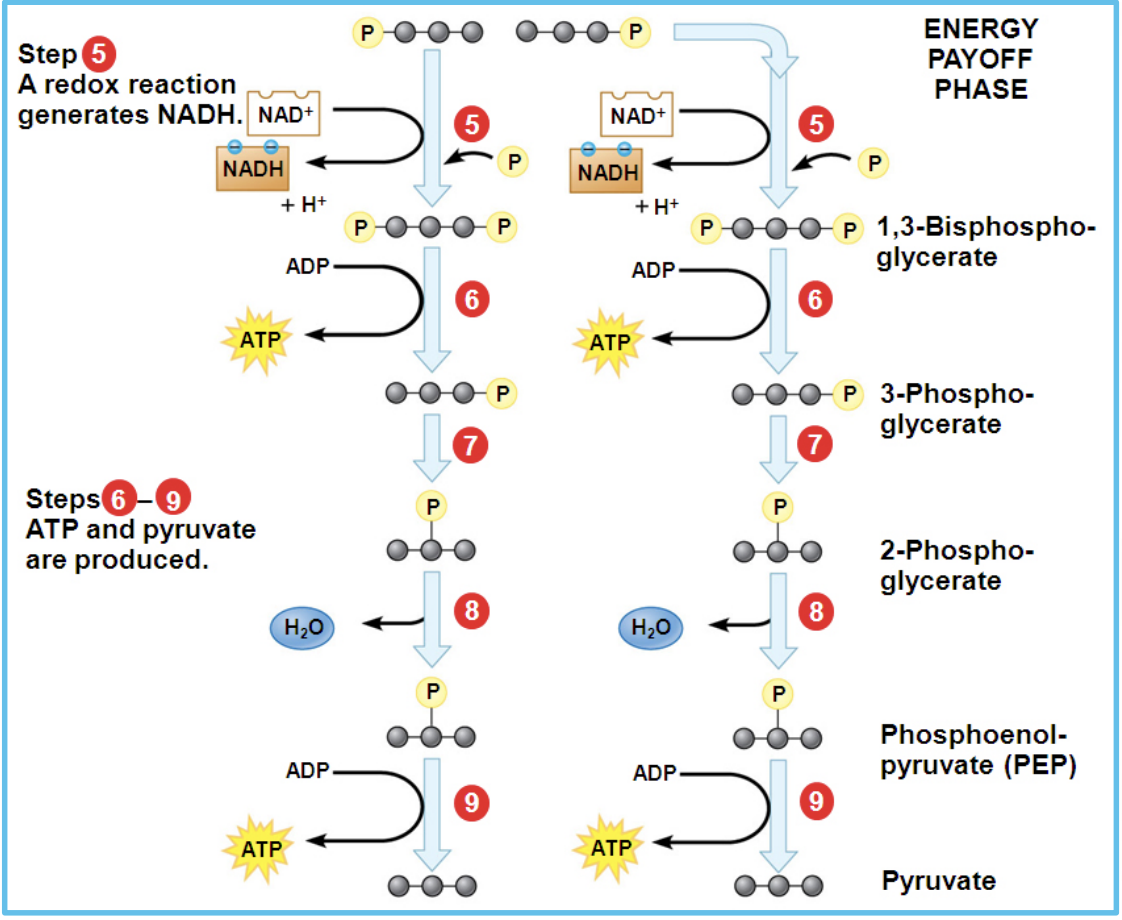
What happens during the energy payoff phase of glycolysis?
During the energy payoff phase, the two molecules of glyceraldehyde-3-phosphate are converted into two pyruvate molecules. In this process, 4 ATP are produced and 2 NAD⁺ are reduced to 2 NADH, resulting in a net gain of 2 ATP for glycolysis.
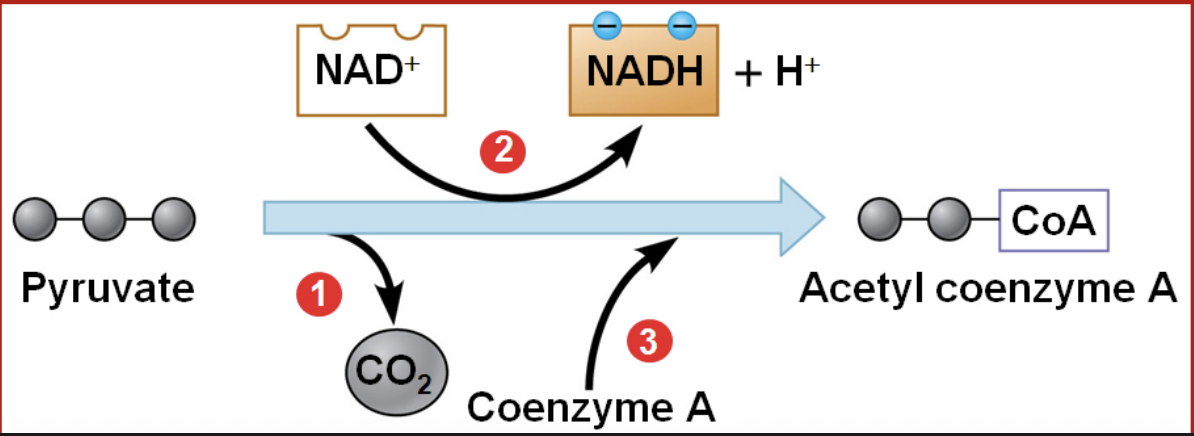
What happens to pyruvate in the presence of oxygen?
In the presence of O₂, pyruvate enters the mitochondrion and is converted into acetyl-CoA. During this conversion, NAD⁺ is reduced to NADH and CO₂ is released.
What energy molecules are produced in the citric acid cycle?
Each cycle produces 1 ATP, 3 NADH, and 1 FADH₂. Since one glucose forms two acetyl-CoA, the cycle runs twice per glucose molecule.
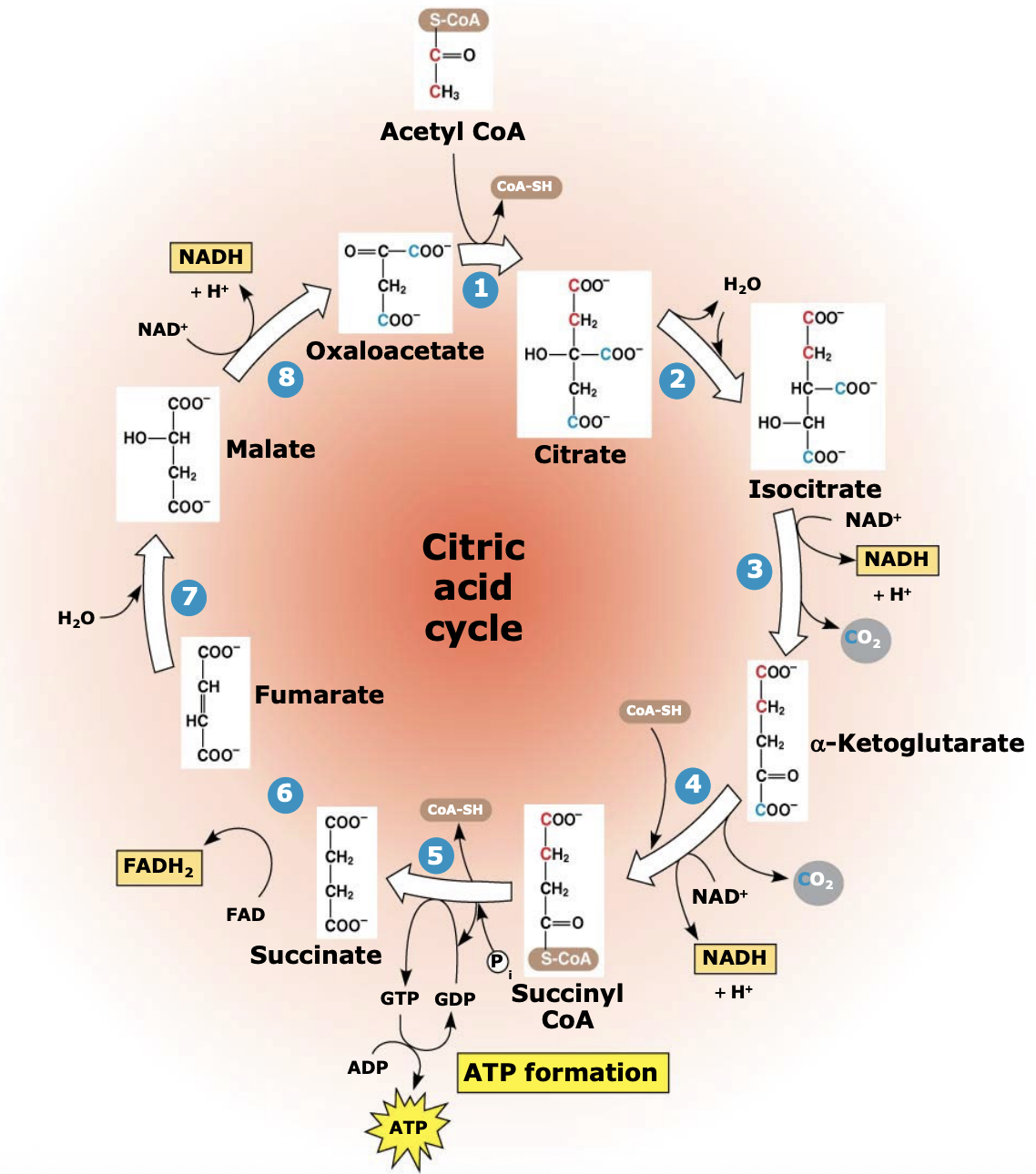
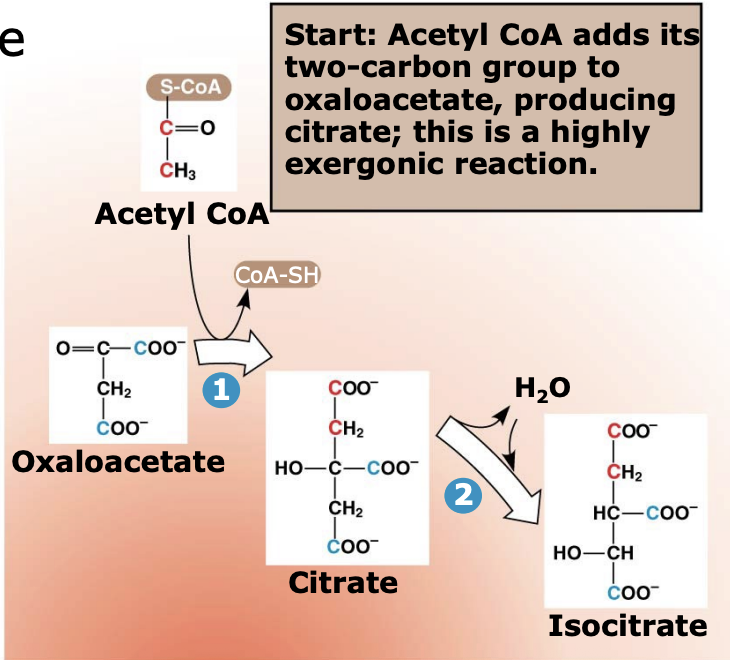
What happens in Step 1 of the citric acid (TCA) cycle?
Acetyl-CoA (2C) combines with oxaloacetate (4C) to form citrate (6C). Coenzyme A is released and reused. This reaction forms the first compound of the citric acid cycle.
Why is Step 1 of the citric acid cycle important?
This step is highly exergonic (ΔG ≈ −7.7) and essentially irreversible, helping drive the cycle forward. It is also the only step in the TCA cycle that forms a carbon-carbon (C-C) bond.
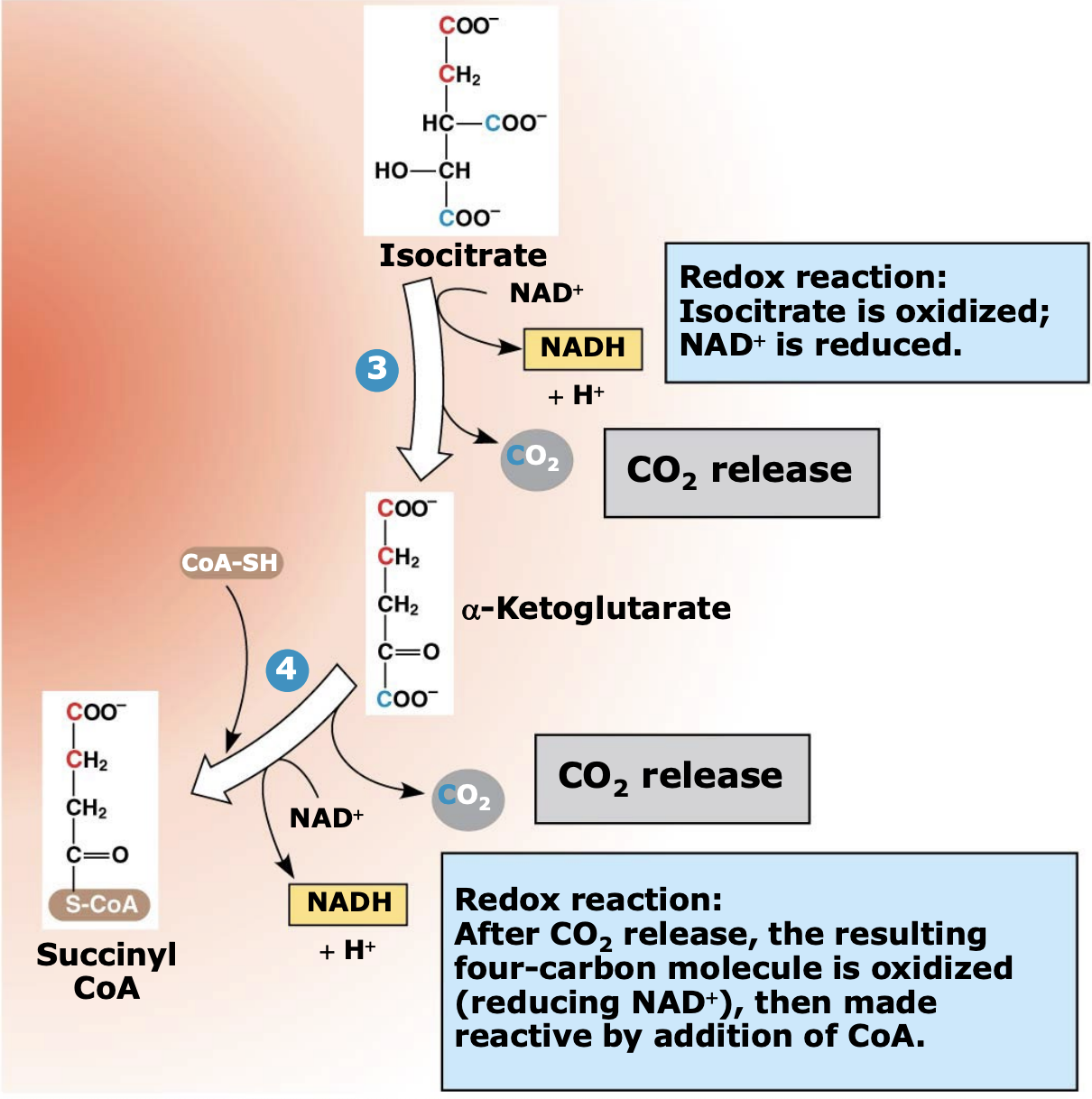
What happens during Steps 2–4 of the citric acid cycle?
Isocitrate is oxidized and loses carbon atoms as CO₂. During these reactions, two NAD⁺ are reduced to two NADH, and a 4-carbon molecule (α-ketoglutarate) is formed and then converted to succinyl-CoA.
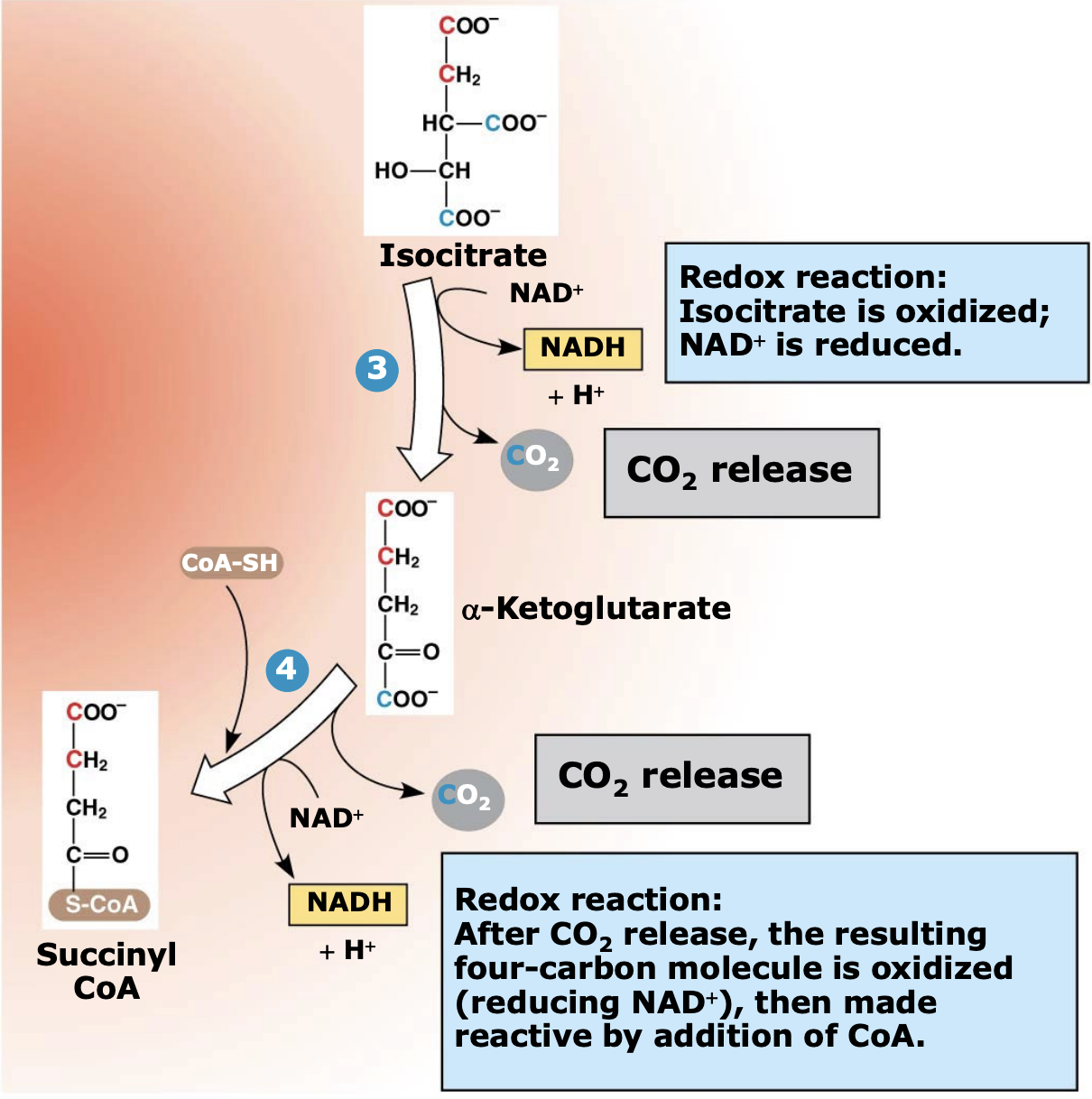
Why is succinyl-CoA important in the citric acid cycle?
Succinyl-CoA is a high-energy intermediate formed when α-ketoglutarate combines with CoA-SH. The stored energy in this compound is later used to help produce ATP in the next step of the cycle.
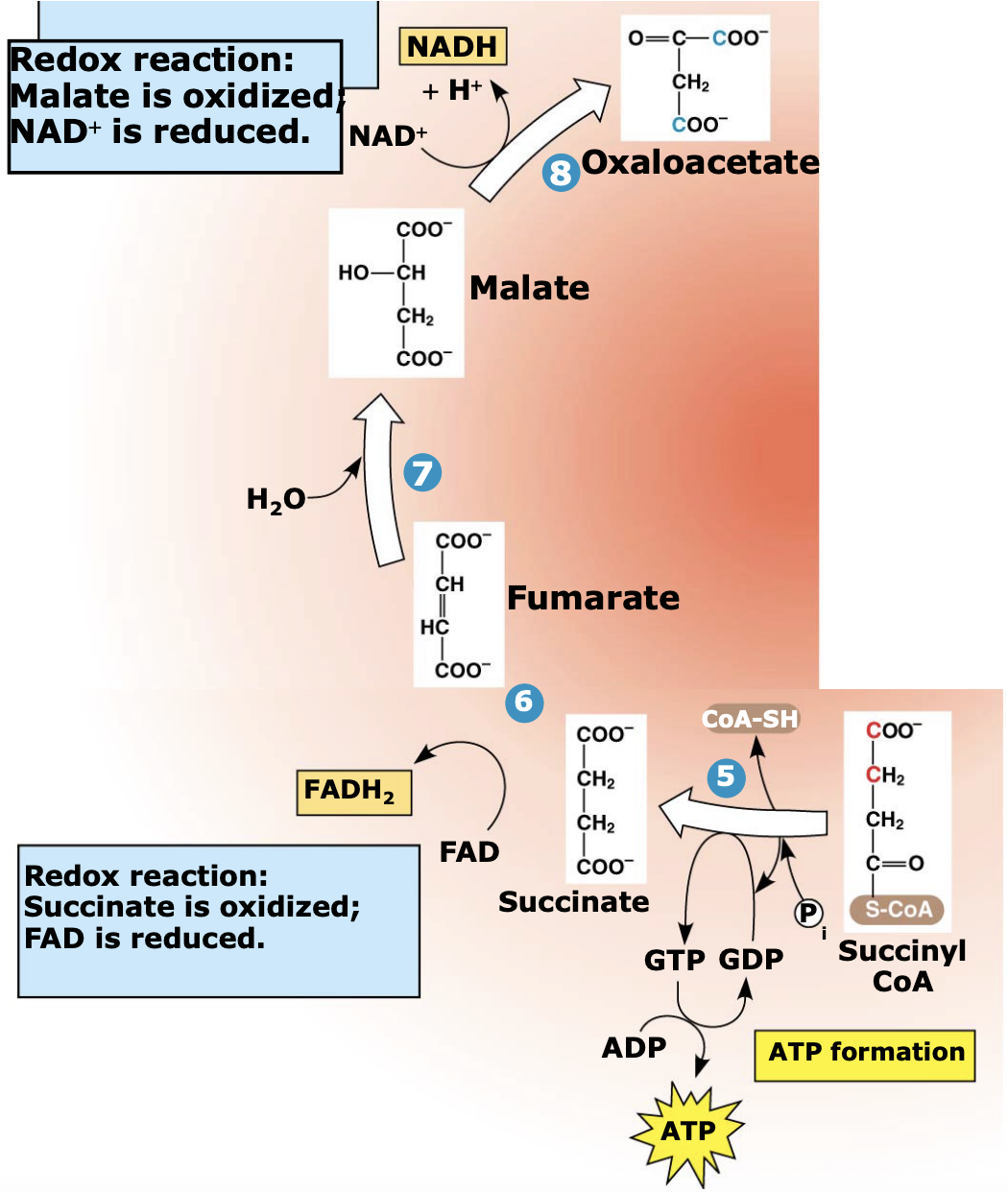
What happens in Steps 5–8 of the citric acid cycle?
Succinyl-CoA loses its CoA, which drives the formation of GTP (later converted to ATP). The remaining 4-carbon molecules are then oxidized to regenerate oxaloacetate, allowing the cycle to continue.
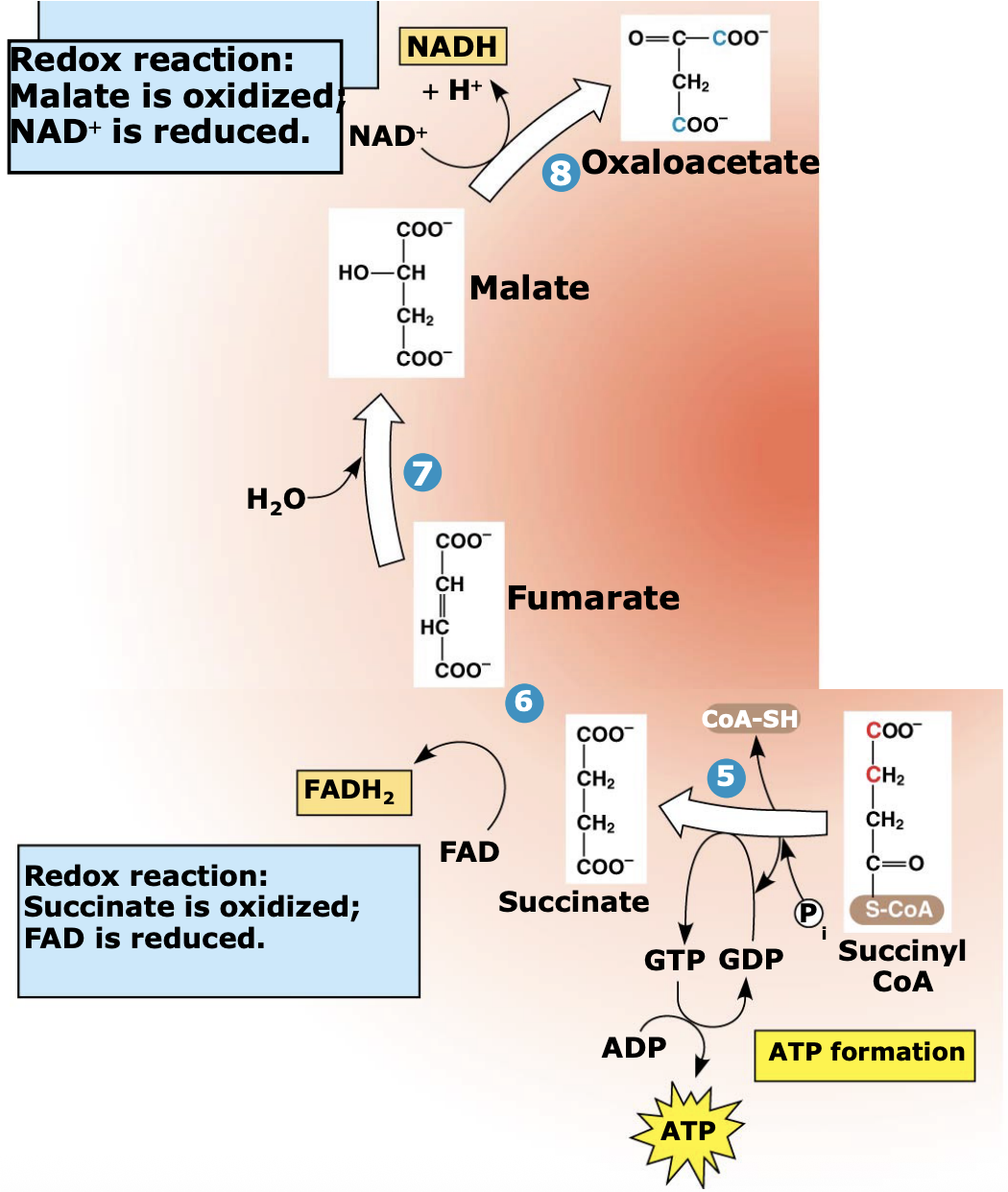
What energy molecules are produced in Steps 5–8 of the citric acid cycle?
During these steps, oxidation reactions produce 1 FADH₂ and 1 NADH, which carry high-energy electrons to the electron transport chain.
The electron transport chain is located
in the inner membrane (cristae) of the mitochondrion
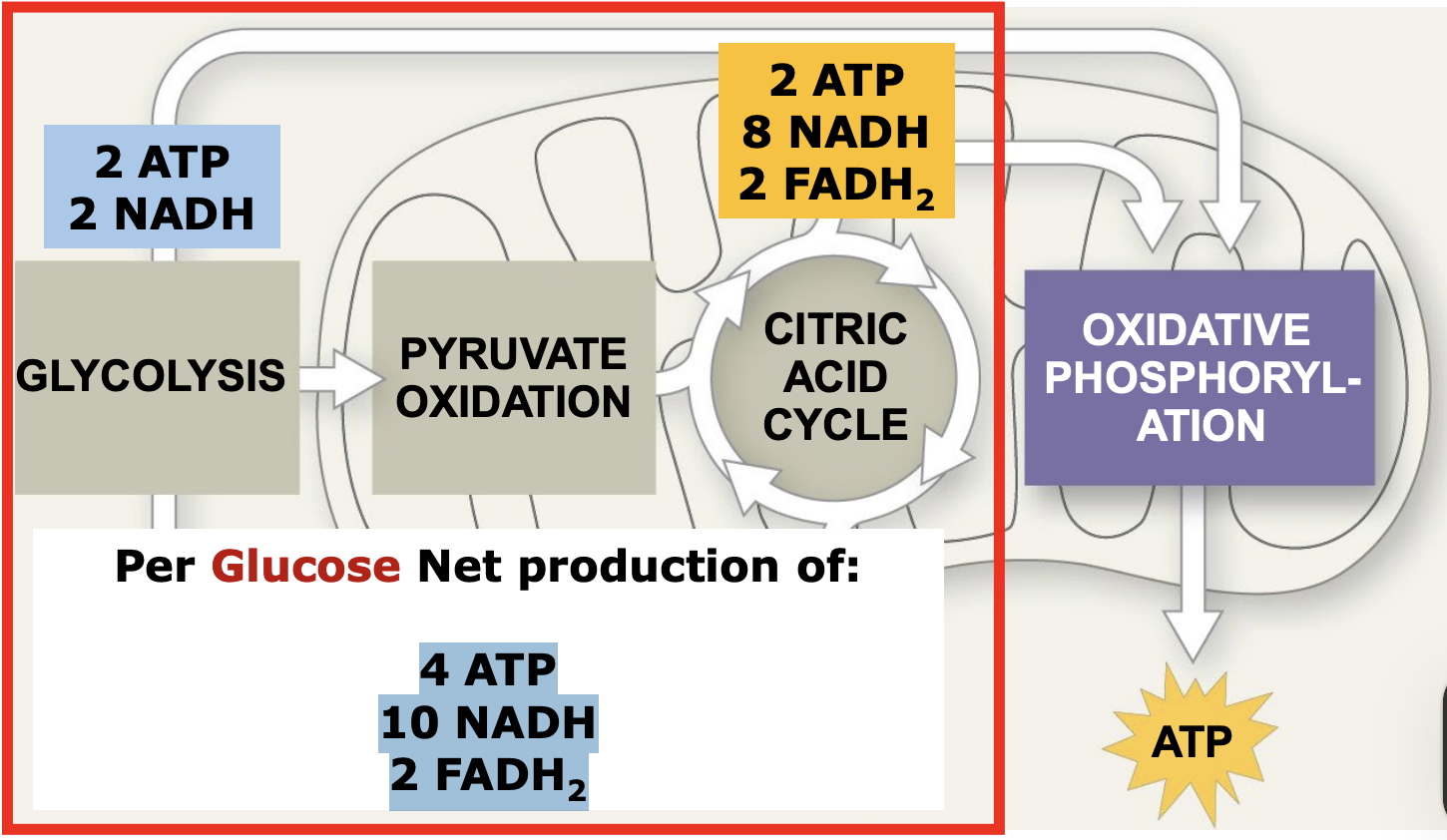
What is the net production of essential molecules by the time ETC starts?
4 ATP
10 NADH
2 FADH2
What is the electron transport chain made of?
The electron transport chain is made of proteins organized into multiprotein complexes in the membrane. These proteins transfer electrons through a series of redox reactions.
How do electron carriers work in the electron transport chain?
Electron carriers alternate between reduced and oxidized states as they accept and donate electrons, passing them down the chain.
What happens to electrons at the end of the electron transport chain?
Electrons lose free energy as they move down the chain and are finally transferred to oxygen (O₂), forming water (H₂O). The released energy helps power ATP synthesis by ATP synthase.
What happens to electrons as they move through the electron transport chain?
Electrons are passed through Complexes I, III, and IV by mobile electron carriers. At Complex IV, electrons are transferred to oxygen (the final electron acceptor), which combines with H⁺ ions to form water (H₂O).
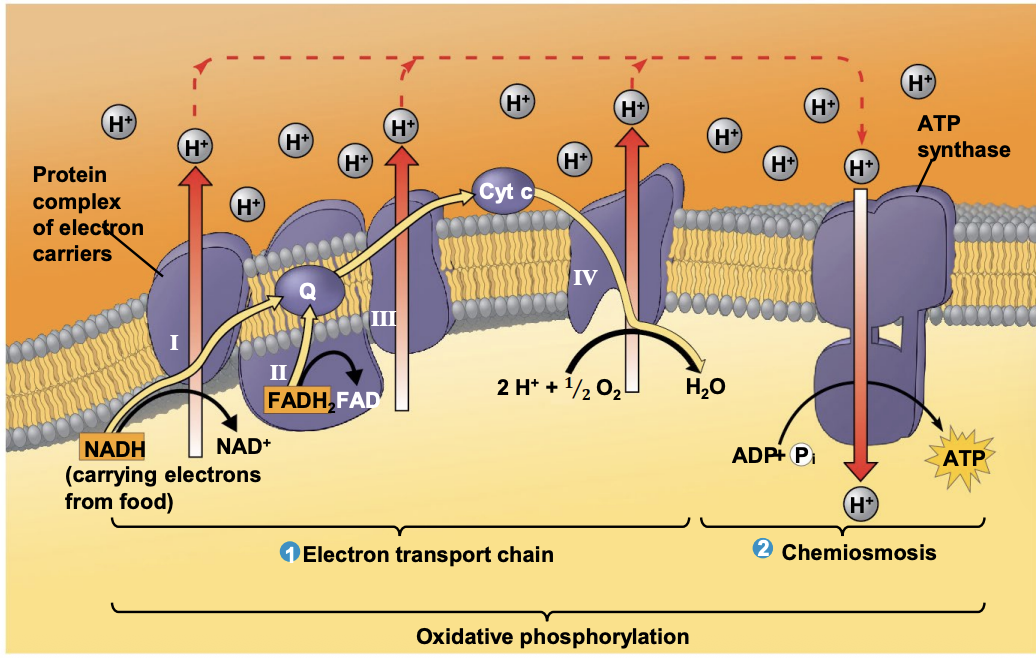
What is chemiosmosis in cellular respiration?
Chemiosmosis is the process where H⁺ ions diffuse down their concentration gradient across the inner mitochondrial membrane, releasing energy that is used to produce ATP.
How do H⁺ ions move across the inner mitochondrial membrane during chemiosmosis?
The inner mitochondrial membrane is not permeable to H⁺ ions, so they can only move back into the matrix through ATP synthase, a protein complex.
How does ATP synthase produce ATP?
As H⁺ ions pass through ATP synthase, the released energy is used to phosphorylate ADP, forming ATP. 1 ATP per H+
What are the types and purpose of fermentation?
Fermentation regenerates NAD⁺ from NADH so glycolysis can continue. There are two main types:
Lactic acid fermentation → produces lactate
Alcohol fermentation → produces ethanol and CO₂
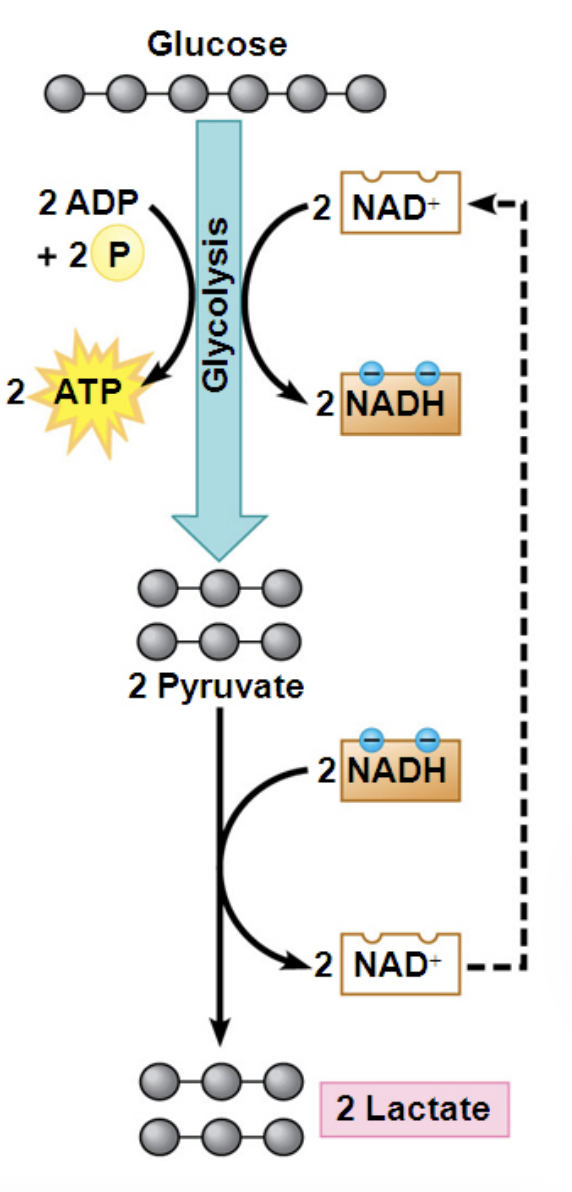
What happens during lactic acid fermentation?
In lactic acid fermentation, pyruvate is reduced by NADH to form lactate. This process regenerates NAD⁺ and does not release CO₂.
Where does lactic acid fermentation occur and why is it important?
Lactic acid fermentation occurs in some bacteria and fungi (used to make yogurt and cheese) and in human muscle cells when oxygen is scarce, allowing glycolysis to continue producing ATP.
What happens during alcohol fermentation? Where does it occur?
In alcohol fermentation, pyruvate is converted into ethanol and CO₂, and NADH is oxidized to NAD⁺, allowing glycolysis to continue. It occurs in yeast and some bacteria and is used to make bread, beer, and wine.
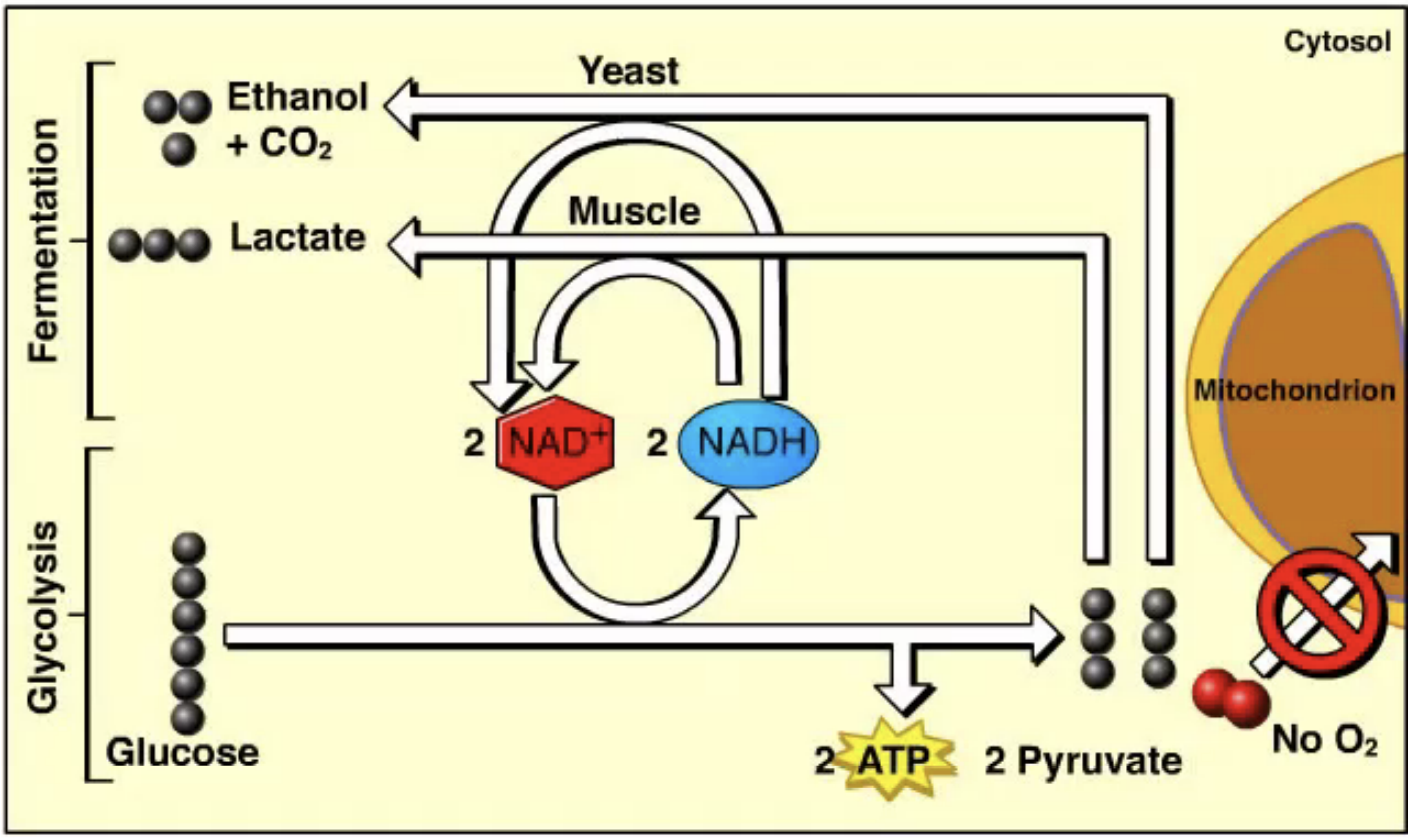
What are obligate anaerobes vs. facultative anaerobes?
Obligate anaerobes can only live without oxygen and are poisoned by it (ex: some bacteria in stagnant ponds or deep soil).
Facultative anaerobes can live with oxygen (cellular respiration) or without oxygen (fermentation) (ex: yeast and many bacteria).
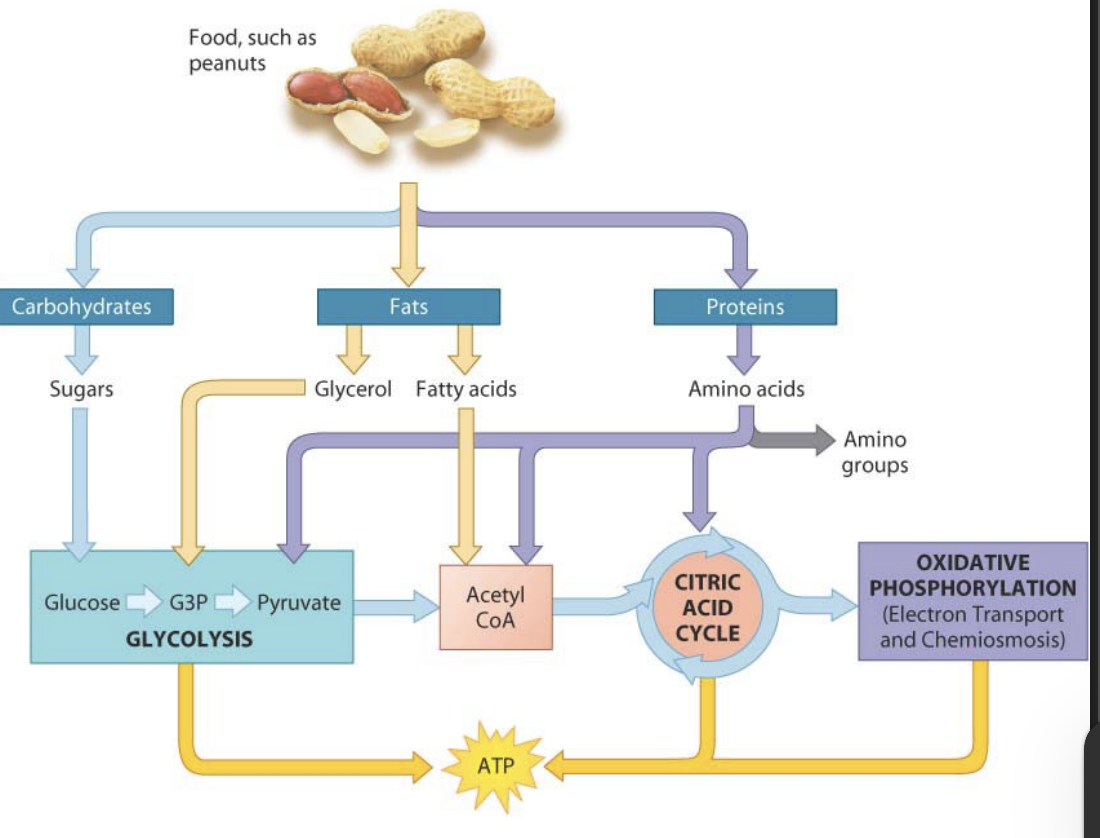
How do carbohydrates, proteins, and fats enter cellular respiration?
Carbohydrates are broken down into glucose, which enters glycolysis.
Proteins are broken into amino acids, which can lose their amino group and enter glycolysis or the citric acid cycle.
Fats are broken into fatty acids and glycerol, which are converted to enter glycolysis or the citric acid cycle.
What is photosynthesis and why is it important?
Photosynthesis is the process that converts solar energy into chemical energy stored in organic molecules. It supports almost all life by providing energy either directly or indirectly.
Who performs photosynthesis and what are autotrophs?
Photosynthesis is carried out by plants, algae, and cyanobacteria. These organisms are autotrophs, meaning they produce organic molecules from CO₂ and other inorganic substances, acting as producers in the biosphere.
What are autotrophs, heterotrophs, and photoautotrophs?
Autotrophs make their own organic molecules from inorganic sources (like CO₂).
Heterotrophs obtain organic molecules by consuming other organisms.
Photoautotrophs use sunlight to produce organic molecules and are the primary producers of the biosphere.