SEMMELWEIS UNIVERSITY Medical Biophysics II. Final - Theory topics
1/139
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
140 Terms
X-rays: Typical diagnostic wavelength and photon energy range.
100ev-200 keV min. wavelength
6pm-12nm
Structure and function of the x-ray tube.
A vacuum tube with an anode and a cathode. The cathode is heated and electrons are emitted, they are accelerated by a voltage difference in the tube. When the "brake" against the material of the anode, they release their high kinetic energy in the form of an x-ray. *x-ray tubes are of very low efficiency, most of the radiation is dissipated as heat.
The Duane-Hunt-law.
Gives the minimum wavelength of x-radiation produced by braking radiation (Bremsstrahlung) as a function of voltage by this formula: minimum wavelength = k/U, k is a constant
Spectrum of Bremsstrahlung.
A continuous spectrum illustrating the "breaking" radiation released by accelerating electrons. Increasing the voltage will result in electrons with higher kinetic energy, x-ray photons of higher frequencies will be emitted. Changing the heating of the cathode, will result in more electrons, thus a higher current.
The area under the curve represents the total emitted power.

Production of characteristic x-rays
Occurs when an electron with sufficient energy ejects an inner shell electron from the atoms of the anode. This state is unstable so an electron from an outer shell fills the vacancy and the excess energy is released in the form of x-ray radiation.
Due to the fact that it's caused by quantum transition (b/w defines energy states) its spectrum is linear and characterizes the matter of the anode.
Power and efficiency of the x-ray tube.
Efficiency is very low, 1%. Most of the energy from the accelerating electrons will be dissipated as heat. The anode must be cooled down and a material of high melting point should be used (tungsten). Can be calculated as the ratio between the power in (P=U I) and the power out. Power is proportional to the voltage, the current and the atomic number of the anode (tungsten).
Px=cxU^2IZ
Mechanisms and energy dependence of x-ray absorption.
two types of mechanism: photo-effect and compton-scattering. The photo-effect is the dominant process and it depends on the atomic number on the thirds power. formula : rho m = Cwavelength^3Z^3
thus, it is possible to differentiate btw. different materials in the body and produce structural image (by the
different absorption level)*for soft tissue imaging (have relatively low atomic number) we use soft radiation with bigger wavelength to compensate for their low atomic number.
X-ray contrast media.
To produce an image, the intensity of the radiation leaving the body is measured. Therefore, contrast in imaging depends on the difference in absorbance of the x-radiation, thus on the attenuation coefficient of the media (which is a product of the mass attenuation coefficient and the density). The two main absorption processes are photo effect and Compton scattering. Photo Effect has a high dependency on atomic number.
CAT-scan: Principles, generations.
A method based on xray which scans part of the body from different angles and provides a cross-sectional image of the body. x-ray alone can't give us information on the depth of the body, by summation of a lot of x-ray images that were taken from different angles we can
divide the area which we are investigating into voxels which are similar to pixels in an image.
The x-ray summation image.
when the elementary densities along the direction of the x-ray beam are summed. Produces the projection image.
X-ray image amplifier.
can be used in real time surgery bc the image is being screened to a monitor.
advantage: reduces the dosage needed for an x-ray image and produce digitizable optical image without using an x-ray absorption film
disadvantage: the amplified image is miniaturized with respect to the original x-ray image (spatial resolution decreases).
composed of a device containing 2 luminescence screens, electrode and photo cathode.
steps of the process:
a. body x-ray image appear on the first luminescence screen (the attenuated radiation from the body is proportional to the number of the optical photons)
b. luminescence light emitted to the photocathode produces photo-electrons which are proportional to the intensity of the radiation.Those electron being accelerated and focused by high voltage
c. the electron reaches the second luminescence screen and through the electron lens thus we get a real image, reversed, miniaturized and very luminous
DSA. (digital subtraction angiography)
Aim: creating an image which focus on the blood vessels steps of procedure:
a. x-ray image is take w/o contrast image (native)
b. contrast agent is injected→ additional image is taken (contrast)
c. by subtracting the native image from the contrast image we produce new images that concentrate on the vessels
Hounsfield unit, windowing in CAT scan.
Absorption coefficient and density values for CT imaging are measured in Hounsfield units.
N CT = 1000* (mu-mu W)/(mu W)
Windowing in CTis contrast manipulating technique in which a range of CT number values (which are density values) can be selected for the grayscale display of the image while others can be ignored.
For example; soft tissue window, lung window or brain window.
Production of high-energy x-rays.
Medical linear accelerators accelerate electrons using a tuned-cavity waveguide, in which the RF power creates a standing wave. Some linacs have short, vertically mounted waveguides, while higher energy machines tend to have a horizontal, longer waveguide and a bending magnet to turn the beam vertically towards the patient. Medical linacs use monoenergetic electron beams between 4 and 25 MeV, giving an X-ray output with a spectrum of energies up to and including the electron energy when the electrons are directed at a high-density (such as tungsten) target. The electrons or X-rays can be used to treat both benign and malignant disease. The LINAC produces a reliable, flexible and accurate radiation beam.
Volumetric flow rate, stationary flow.
Iv= ΔV/Δt
the change in volume per second (unit: m3 /s )
Stationary flow: a flow that is steady in time.
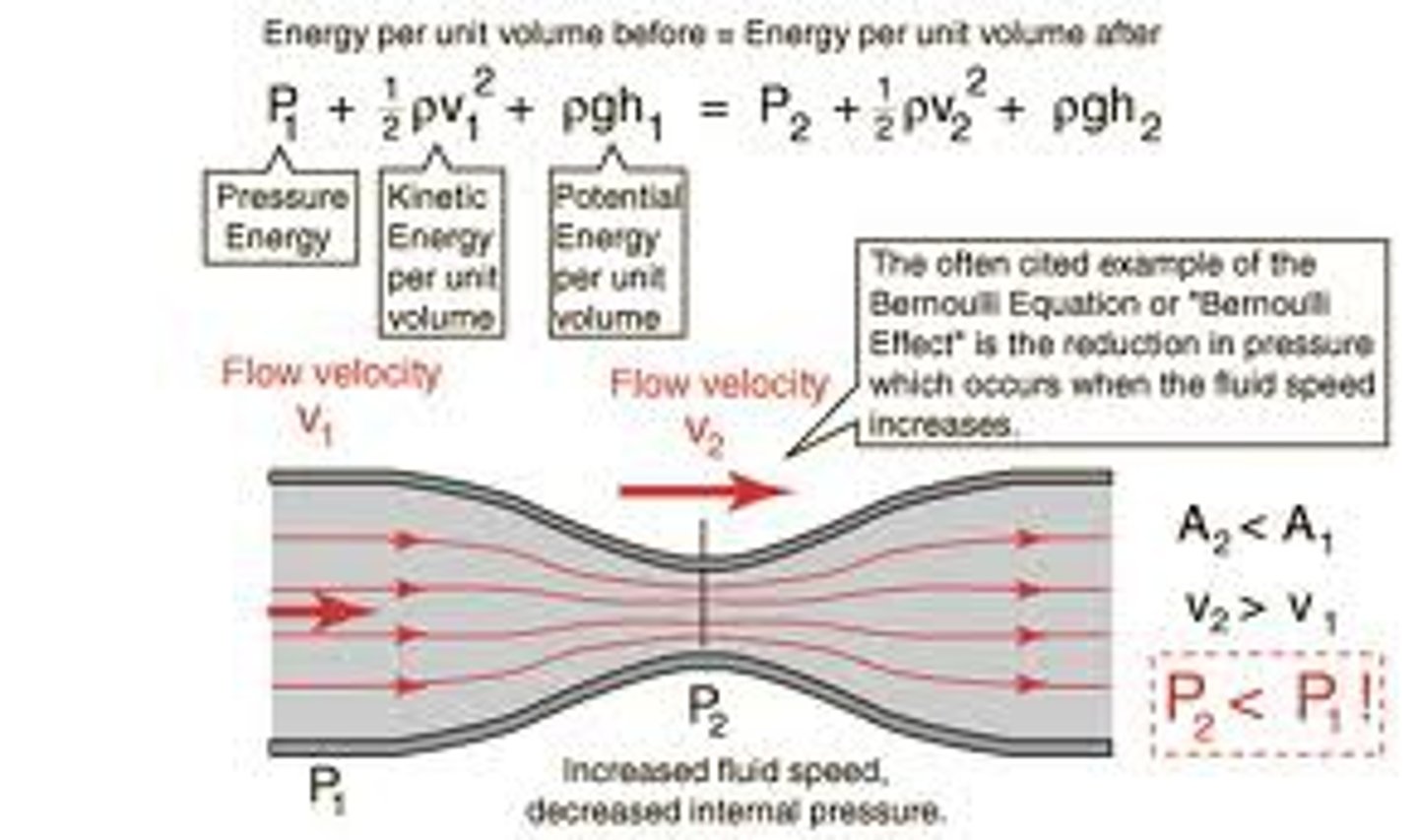
Bernoulli's law, plasma skimming.
Bernoulli's law: in a steady flow, the sum of all the energy in a fluid along a streamline is the same everywhere on the streamline. plasma skimming: The natural separation of red blood cells from plasma at bifurcations in the vascular tree, dividing the blood into relatively concentrated and relatively dilute streams.
Stokes drag law.
Describes the force drag acting on a particle being proportional to the radius of the particle.
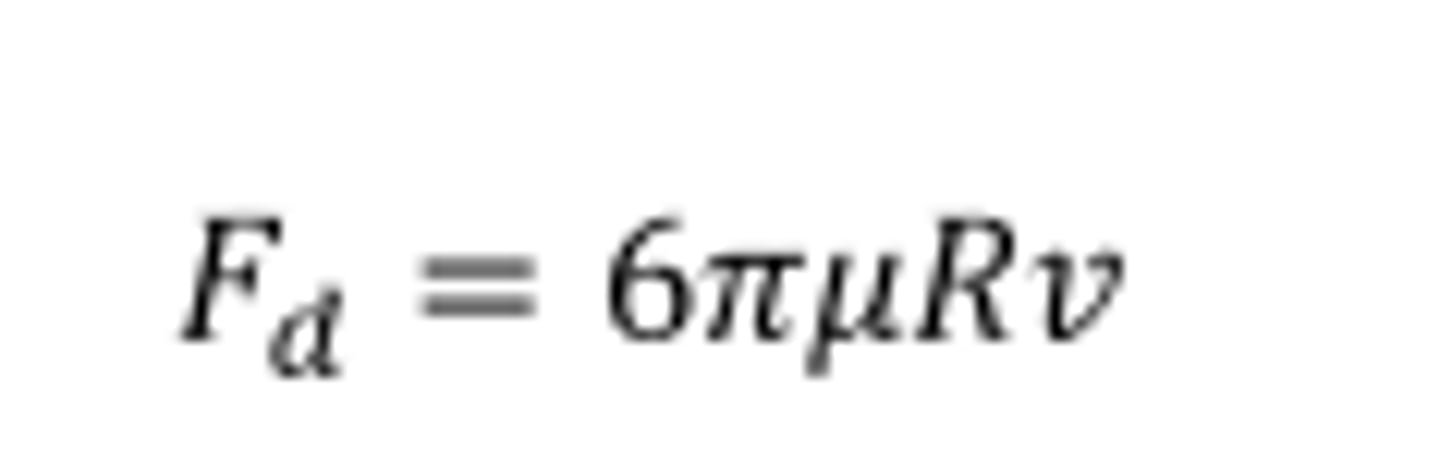
Laminar and turbulent flow.
laminar:
fluid layers do not mix, position of the flowing particles relative to each other is constant
turbulent: (above critical flow velocity). fluid layers mix, slower flow (compared to laminar flow)
Real fluids: Newton's law of friction.
Real fluids are those in which friction exists between its layers. Fluids which adhere to this law are "Newtonian fluids". The law states that the force needed to move one layer (of fluid) over the other, is proportional to the viscosity, the area and the velocity gradient.
Hagen-Poiseuille-law, flow resistance.
Hagen-Poiseuille-law- equation that describes stationary laminar flow of newtonian fluids in rigids tubes: (volumetric flow rate is dependent on the radius of the tube by the fourth power)
flow resistance: directly proportional to the length of the tube (l), viscosity of the fluid and inversely proportional to the square of cross section area!
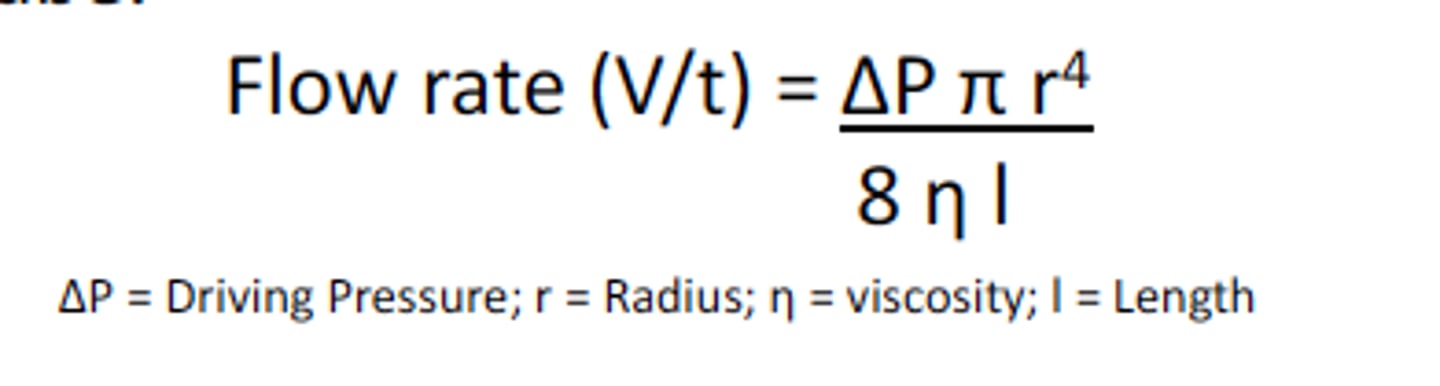
Continuity equation.
Along a tube (rigid tube) the volumetric flow rate is uniform along the entire length, which means that the same volume of fluid must pass through each cross section at a given moment. If the cross section becomes smaller → fluid velocity increases. For real fluids, which are not stationary, the average velocity is used in the formula.
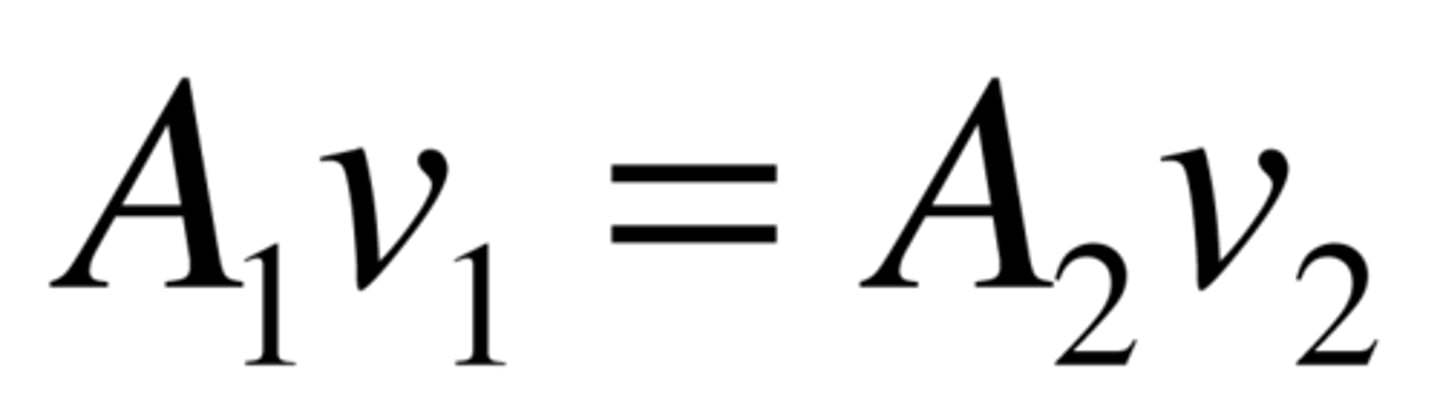
Reynolds-number, critical velocity.
Reynolds number (Re=1160 in a rough walled tube), is a constant used to calculate the critical velocity at which flow becomes turbulent.
Determinants of blood viscosity.
Hematocrit (htc): the ratio between volume of cells and the total volume of the blood.
Normal range: 0.4-0.5.
Plasma viscosity depends on the plasma proteins.
Plasticity of RBC; their ability to change shape.
Aggregation of RBC Blood vessel diameter: with an increase above a certain point the blood's non newtonian behaviour it becomes more pronounced.
Brownian motion. Random walk.
Brownian motion is a phenomenon where particles have "random walk" (which itself exists in many other fields as well).
Brownian motion: the random motion of particles due to collisions with the surrounding molecules.
Basics of diffusion: Concepts, thermal motion.
The random motion of particles as a result of thermal motion, which is a result of the particle's thermal energy (which depends on its temperature).
Particles will interact with each other due to their own thermal motion, this interaction will cause the particles to change direction.
Fick's I. law.
the flow of a particle per unit time across unit area is proportional to the concentration drop.
Fick's II. law
Describes the spatial and temporal changes of the concentration gradient. Relevant when it is needed to consider the time dependence of diffusion (when studying metabolic processes, anesthesia, drug effects etc).
The diffusion coefficient. Einstein-Stokes-equation.
The diffusion coefficient gives the amount of material diffused across a unit area in a unit time driven by a concentration drop. unit m^2/s.
it depends on the size and the shape of the particle, on the interaction with the solvent and on the viscosity of the solvent.
Einstein-Stokes-equation: valid forspherical particlesin a viscous medium and used to calculate the diffusion coefficient.
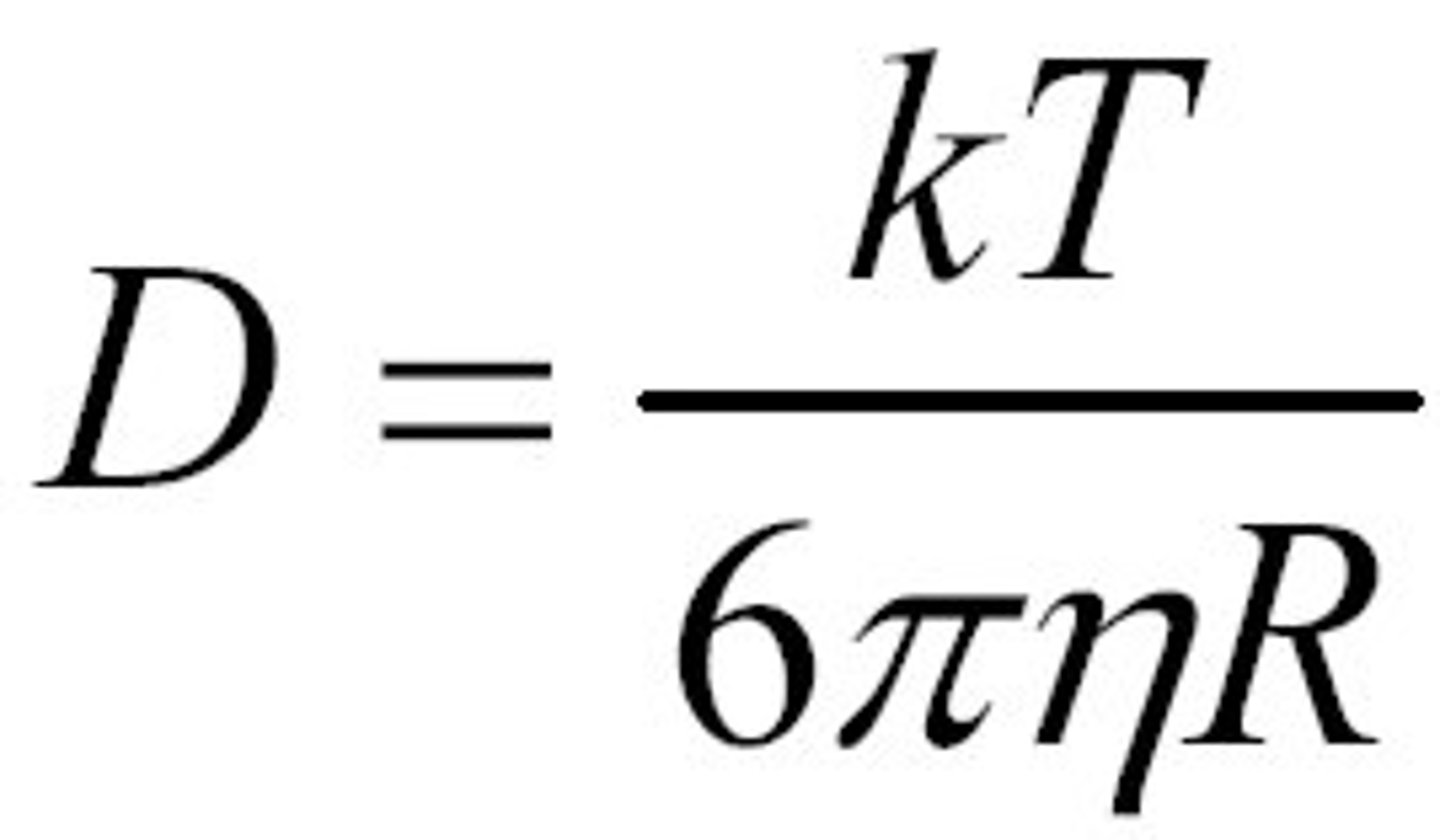
Thermodiffusion.
Diffusion that occurs due to a temperature gradient. Simple diffusion and thermodiffusion can compensate for one another in some situations (difference in temperature causes thermodiffusion which induces a concentration gradient which causes normal diffusion)
LT = matter diffused across a unit area at a unit temperature gradient
Heat transport, Fourier's law.
3 main mechanisms:
conduction: net energy flow (by contact) btw particles from high temp area with higher kinetic energy, and from cooler area particles by colliding and transfer part of their kinetic energy (w/o exchange of matter)
know fourier's law (conduction rate equation): where lambda= heat conductivity
we also have two other mechanisms:
convection: heat transfer by movement of fluid (e.g. hot ground→ the air next to it get hot lifted up, cold air sink down get hot by the ground floor and goes up)
thermal radiation: every matter radiate thermal radiation (does not require medium)
Physical quantities used for describing the transport of matter.
Particle flow rate: number of particles per unit time I = ΔN/Δt unit 1/s
Matter flow rate: moles per unit time I = Δv/Δt unit mole/s
Particle flow density/flux J = ΔI N/ΔA unit 1/m3s
Matter flow density/flux; J v = ΔI/ΔA unit mole/ m3 s
Gas exchange between blood and alveoli.
the amount of O2 and CO2 that diffuses across the membrane of an alveolus depends on:
- difference in partial pressure
- surface area of the membrane
- inversely proportional to the thickness of the membrane
by given diffusion coefficient for each of them, we can calculated the volumetric flow rate by using fick's first law
Osmosis, osmotic pressure, osmolarity.
Osmosis:the phenomenon where solvent particles diffuse as a result of a concentration gradient. They will diffuse from an area of lower concentration to an area of higher concentration through a semipermeable membrane.
Osmotic pressure:example; a bag with sugar and water is placed inside of a vat of water. The bag is permeable only to water. The water is at a higher concentration outside the bag, therefore more water molecules from the outside will hit the bag barrier and enter it. This is diffusion. An increase in the water molecules inside the bag will cause an increase in pressure, this pressure difference between water on the inside and outside of the bag, is the osmotic pressure.
Osmolarity or "osmotic concentration" refers to the amount of solute particles per liter solvent (taking into consideration the number of dissociated ions if applicable. # dissolved ions x molarity = osmolarity). Blood is about 300mOsm/L/
Medical applications: isotonic solutions are necessary to keep RBCs alive.
Fundamentals of thermodynamics I.: types of systems, the human body as a thermodynamic system.
types of system:
a. open- exchange of matter and energy with the surrounding b. closed- exchange of energy only with the surroundingc. isolated- no exchange with the surrounding
the human body system function as an open system:exchange matter with the surrounding (e.g. gas exchange, water) exchange energy: energy is used to create mechanical work
Fundamentals of thermodynamics II.: change of internal energy.
The change in internal energy is the sum of the supplied heat and the work done on the system.
delta E = Q E + W
Q E = mc delta T
Fundamentals of thermodynamics III.: types of energies, internal energy and its components.
The internal energy is the sum of the individual energies of the particles that compose it. Electric, vibrational, rotational and interatomic interaction.
*the internal energy does not include the kinetic or potential energy of the system as a whole.
The 1st law of thermodynamics and its applications for biological systems.
Essentially a law of conservation of energy, which states that energy cannot be destroyed, rather converted from one form to another. Biological processes rely on the conversion of energy, for example; in cellular respiration where food is converted to energy in the form of ATP.
delta E = Q E + W
Q E = mc delta T
The 2nd law of thermodynamics, direction of spontaneous processes.
2nd law: heat will flow from higher temp area to lower temp area.
direction of spontaneous processes shifted to the process with which the highest entropy state is achieved.
the process is reversible as long as the combined entropy of the system and the surrounding remains constant (thermal or chemical equilibrium).
If the process is irreversible the combined entropy of the system and the surrounding is increased. (e.g. hot object is put in contact with cold object).
Entropy increases steadily in the universe (isolated system) and can not be decreased
The 3rd law of thermodynamics.
The entropy of a pure crystal at 0K is 0. This law can be explained from the definition of entropy; which is the number of microstates which characterize a given macrostate. Atoms in a crystal at temperatures higher than 0K would differ in their vibrational energies, thus at any given time there could be different positions (microstates) of atoms, which means the entropy is greater than 0. However at 0K (hypothetical temperature), thermal motion and thus vibration of atoms would not exist, therefore there would only be one microstate for the macrostate of the crystal.
Using Boltzmann's equation; S = k * ln(Ω) where Ω =1
ln(1)=0,
thus S=0.
Extensive and intensive quantities and their relations.
Extensive and intensive are quantities that serve characterization of thermodynamic systems.
Extensive: volume, mass, energy, charge, entropy, etc (quantities that can be change by splitting the system into 2 subsystems)
intensive: density, temperature, pressure (independent of the size of the system) the product of the intensive and extensive quantities give us the change in energy: Wi = yi * delta xi
Entropy and its connections with order, thermal and configurational entropy.
Entropy can be defined as a state of "disorder" in a molecular system. Entropy increases as the temperature of a system rises (it is the extensive variable of heat flux).
Configurational entropy is characterised as a flat part of the curve, where the temperature stays constant and molecular order changes. (which mean transition in state e.g ice-water)
Isobaric, isothermal, isothermal-isobaric systems.
Isobaric; at constant pressure,
Isochoric; at constant volume,
Isothermic; at constant temperature.
At constant pressure work done does not equal 0. Because
W = pΔV so W= change in volume, thus all the energy is converted to change in volume and change in temperature.
In Isochoric processes, ΔV = 0, so W = 0. Therefore ΔE = ΔQ and all energy is converted to heat. In isothermal processes all the energy will be invested in the work done.
Equilibrium conditions of different thermodynamic systems.
mechanical equilibrium: pressure throughout the system and in the surrounding is the same
chemical equilibrium: the rates of the forward and the backward reactions are the same
thermal equilibrium: two matter in contact with each other and there is no heat exchange
Thermodynamic potentials.
Gibbs free energy: gives the energy available in isothermal, isobaric,non isolated systems G = H − T S
● Himholtz free energy: gives the energy available in isothermal, isochoric,non isolated systems F = E − T S
● Enthalpy: total heat absorbed by a system at constant pressure H = E + pV
● Internal energy: ΔE = Q + W
Matter transport through the cell membrane.
3 types of membrane transport:
a. passive diffusion:fick's first law (+ extension): where the driving force is the chemical potential gradient as well as the electric potential gradient. Ficks first law is used with an extension to include this electrochemical gradient.
b. facilitated: require mediator molecules (channels or carrier): faster than simple diffusion, is selective (receptor protein must fit to the molecule), can be inhibited (as used in antibiotics). c. active: against electrochemical gradient- (e.g atp-driven, light
gated or coupled transporter)
The transport model and the Goldman-Hodgkin-Katz-equation.
Considers the continued diffusion of ions across the cell membrane while taking individual ion permeabilities into account P k . In a way it is a modified Nernst equation that gives the resting membrane potential more accurately.

Changes in the membrane potential as the function of time.
It's analog to charge or discharge of an RC circuit thus we can use those formulas for charging and discharge (look up said equations)
Resting transmembrane potential.
Negatively charged intracellular space and a more positively charged extracellular space. Higher concentration of K+ inside and a larger concentration of Na+ and Cl- outside.
Electric model of the membrane.
Analog of capacitor (two conductive layer and the membrane use as insulator material) the permeability of each substance can be modelled as conductivity (1/R)
Changes in the membrane potential as the function of space.
The space constant describes the propagation of action potential, in terms of the distance it travels before it is attenuated to 37% of its original value. Formula for the space constant:
Rm = transmembrane resistance. Ri = intracellular resistance Larger space constant provides a larger distance of propagation, can
be improved by increasing Rm as seen in the myelin sheath of axons

Diffusion of ions across the membrane, permeability.
depends on the electrochemical gradientand the number of the channels and carriers to a specific substance (due to the fact that that the cell membrane functions as an insulator). we can use the modulated onsager equation to calculate passive diffusion, which includes the diffusion coefficient D.
The Donnan-equilibrium.
Refers to theequilibrium state in which there is anuneven distributionof ions across a membrane, and the particles are not able to diffuse because of impermeability of the membrane. In cells this equilibrium is realized by the sodium potassium pump, which actively controls this uneven distribution.
Properties of the action potential.
1. unaltered ion concentration: only the permeability is changing (GHK equation is valid)
2. refractory period (dead time) 2 types:
- absolute: voltage gated of Na are inactive
- relative: action potential can occur but it requires much higher threshold stimuli to (prevent the backpropagation)
3. special AP: cardiac AP the duration is much longer (200-400 ms) thanks to additional channels, CA(2+) channels.
4. (''All or none''- stimuli have to exceed a threshold level to trigger action potential.
5. controlled by the opening and closing of voltage driven channels
6. action potential is very fast 1.5ms)
Propagation of the action potential, refractory period and its role.
The propagation has a special exponential decay because of its RC circuit nature.
Refractory period is a time in which the cell is not able to respond to the stimulus by generating an action potential. It is characterised by an absolute refractory period and a relative refractory period (where a stimulus of much higher magnitude would be required to generate an action potential).
Electric signals measured on the body surface, for diagnostic purposes.
Based on dipole moment : d=Q*l
EKG:
a. a graph of the voltage vs. time of the electrical activity of the heart b. measured using electrodes placed on the skin
EEG: records the electrical activity of the brain
EMG: records the electrical activity of skeletal muscles by placing electrodes on diff. places on the skin
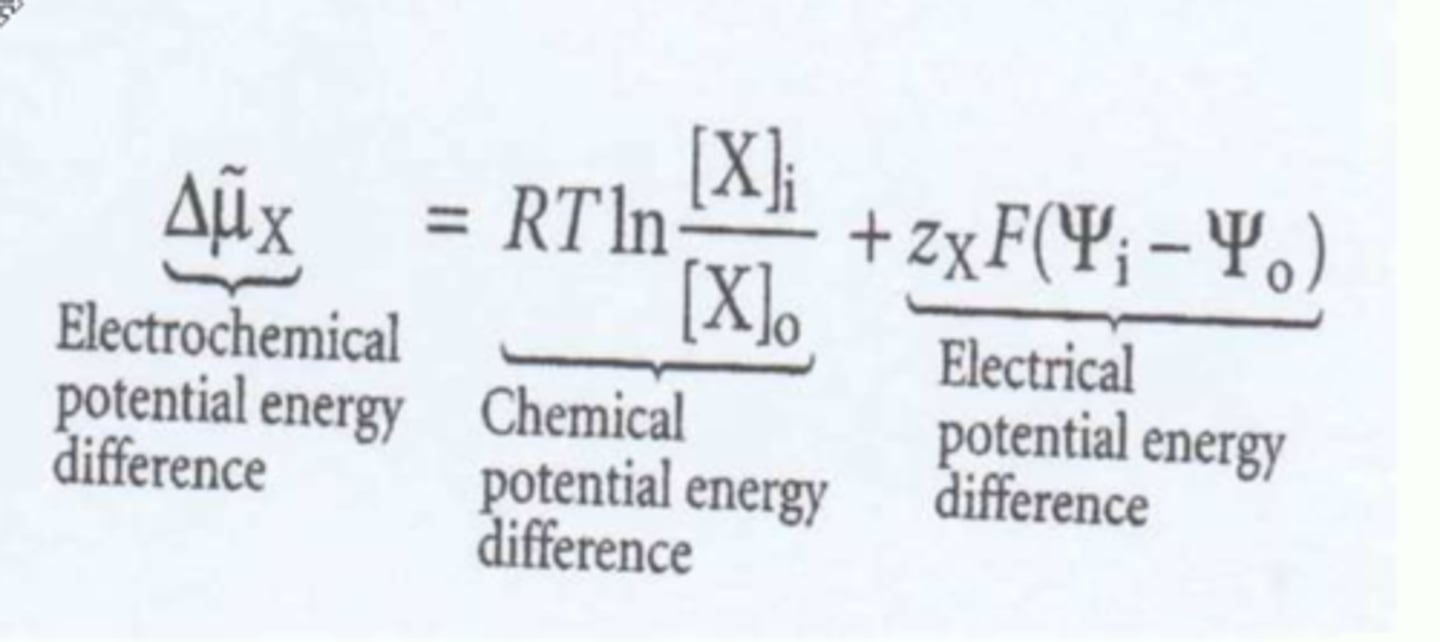
Electrochemical potential.
There are two components; charge difference/ potential across the membrane and chemical difference/ potential.
Ion currents during action potential.
Steps:
1. stimulus above threshold
2. voltage-gated channels of Na(+) open = Na+influx
3. voltage-gated channels of k(+) open = k+ outflux
4. voltage-gated channels of Na(+) inactive (partial)
5. voltage-gated channels of Na(+) close
6. voltage-gated channels of k(+) close
t is approx. 1-5 ms (muscle and neuron)
U is approx. 100 mv
Sound as a wave.
Sound is a mechanical wave - a "pressure wave" which are sinusoidal oscillations.
It is a longitudinal wave in liquid and in gases, which means that the oscillation is parallel to the direction of propagation.
In solids the waves can be either longitudinal or transverse.
Acoustic impedance, reflection, of sound, reflectivity.
Acoustic impedance:quantity that shows to what extent the medium resists when particles are brought into motion, practical equation:
Z = rho * c
(product of the density and the speed of sound in the specific medium)
(Z also can be measured by the ratio of the square root of density and compressibility)
reflection: At the boundary of 2 media part of the intensity of the sound reflect into the opposite direction and not penetrate the body the extent of this phenomenon is measured by the reflectivity equation.
*ultrasound diagnostic methods are based on that phenomenon.
reflectivity: measured by the ratio of the reflected intensity according
to this formula: R = JR/J0 = (Z1-Z2/Z1+Z2)^2
when R=1 → max reflection, when R=0 → no reflection
Imaging modes in sonography .
One dimensional A image (amplitude modulation): single transducer, time/distance corresponding to amplitude changes.
One dimensional B image (brightness modulation): single transducer where the amplitude is represented a brightness pixels on grayscale.
Two-dimensional B-image (2-D brightness modulations): an array of transducers, manipulated so that the wave direction can be controlled (allows back and forth scanning without needing to move the transducer).
M-image (motion):2D image where a specific direction is selected and is plotted as a function of time, to obtain positional information.
Reconstructed 3D image:tomography
Reconstructed 4D image:ultrasound movie (reconstructed 3D images as a function of time)
Generation and detection of ultrasound.
Generation: based on inverse piezoelectric effect in which voltage pulse is applied on the crystal which become deformed(and ''push'' the air particles next to it and by that generate ultrasound)detection: based on the piezoelectric effect: when the reflected signals from that examined body part compress and deform the transducer crystal, as a consequence voltage is formed (and being amplified by an amplifier)
The Doppler-effect, Doppler-shift.
Change in frequency and in wavelength of the wave as a result of relative motion of the source and the observer. In US imaging, the source can be considered to be the reflecting surface of the examined organ. The change in frequency can be attributed to the velocity of the moving source. Therefore by studying the change in frequency, the velocity of the surface can be measured as a function of time.
Effects of ultrasound, therapeutic applications.
1. the energy(?) that propagates in ultrasound is dissipated (dampes) in the medium in form of heat.
2. intensity of US proportional to pressure difference (high intensity can separates cells from each other in a tissue and form cavities.
3. we know that the higher the energy the higher the absorbance in the tissue (higher damping)
*We can use this knowledge and by focusing our ultrasound beam into the tumor for example we can create micro cavities in the tumor tissue and heat it and by that destroy it.
Absorption of ultrasound.
The absorption of US is important because some of the energy is dissipated in the form of heat.
General attenuation law is valid: Where μ = absorption coef f icient ~ F requency Absorption ability of tissues is characterised by the "damping" unit dB/(cm MHz)
The pulse-echo principle.
The distance b/w reflected boundary and the transducer can be calculated from the time elapsed b/w emission of the ultrasound pulse and its echo, if we know the velocity of sound in the given
medium by that formula: d= c* delta t /2
Propagation of ultrasound in air and in the body.
The velocity of sound depends on the media in which it propagates, specifically on the density and compressibility of the medium. Compressibility: K = -deltaV/V/deltap
Speed: c = 1/root rho K
How far it propagates depends on the attenuation along the depth it travels. μ = absorption coefficient ~ Frequency
Structure and properties of water.
structe: H2O tetrahedral shaped.in its liquid form its is rearrange as pentamer (H2O)5 or as octamer properties:
a. permanent dipole moment→ good solvent
anomalous density-temp. function→ the highest density is at 4 degrees celsius
large surface tension
anomalous phase diagram→ by exerting enough pressure ice
can be converted into water !
Phase diagram of water.
Pressure over temperature; same chemical, different phases.one of the special properties that makes it different = "anomaly" of water is that it has a negative melting curve. When temperature is constant and pressure is increased, ice will melt.
Anomalous behavior of water.
Density-temp function → highest density at 4 celcius
phase state → at some temp, at high pressure ice becomes
water
high surface tension , on hydrophilic surface water molecules
''climbing''.
(floating water bridge)
Structure of biopolymers.
Protein - AA - covalent
Nucleic acid - nucleotide - covalent
Polysaccharide - sugar - covalent
Protein polymer - protein - secondary
Structural hierarchy of proteins.
3 main structural hierarchy:primary→define the sequence of a.a→ peptide bone secondary→alpha-helix, B-sheet→ H-bonds, secondary bonds, tertiary→define the 3D str. of the polypeptidehierarchy of the bonds:weak bonds: H-bonds,van der waals, hydrophobic-hydrophobic interaction, dipole dipole, dipole-dipole bondscovalent bonds: disulfide bridge→ b/w to cystin aa side chains connects diff part of the polypeptide
Protein-stabilizing interactions.
Secondary:
● Hydrogen bonds
● Electrostatic interactions
● Van Der Waals interactions
● Hydrophobic/Hydrophilic interactions
Covalent
● Disulfide Bridges (Between Cysteine side chains). Hydrogen bonds can orient in two ways:Parallel coupling:which requires a very high amount of force to breakSerial coupling:which in comparison requires much less force to break.
Protein folding.
The protein funnel hypothesis assumes that protein's native state corresponds to its free energy minimum under the solution in physiological condition.
Biopolymer elasticity.
Determined by two parameters:
Contour length (L):the length of the biopolymer when it is stretched to its maximum. Depends on the number of monomers that compose it and it's length.
Persistence length:a measure of the stiffness of a biopolymer.
If L>>>I: the chain will be flexible (DNA)
If L<<
Structure and elasticity of DNA.
Primary structure:
- esters of nucleotides forms with phosphoric acid
- each nucleotide contains heterocyclic base (contain N) and
pentose (deoxyribose)
- bases= pyrimidine/purine derivatives
- pyrimidine base: thymine, cytosine
- purine: guanine, adenine
*in the polynucleotide chain, deoxyribose connects with phosphodiester bonds. --> sugars backbone of the chains
*sugar= most flexible component in the nucleic acid.
2. 2 independent DNA chains form a double helix with antiparallel orientation by H-bonds b/w the bases of each dna strand.
There are 3 different conformations (A-DNA, B-DNAis the familiar one, Z-DNA)
tertiary structure: nucleosome (dna-histone protein)
Steps of sensory signal transduction.
Stimulus → Receptor potential (on sensory receptor) → Action potential (on nerve cell) → CNC
Information coding by the receptor potential.
Amplitude coding, where the intensity of the stimulus corresponds to the receptor potential.When the stimulus exceeds a given threshold, an action potential will be generated.
Information coding by the action potential.
Frequency coding, where the increase in the voltage of the receptor potential will lead to higher frequency of the AP of nerve. gives information about:•modality (type) (depends of which nerve does the AP) • intensity
(strength) frequency coding • duration • localization
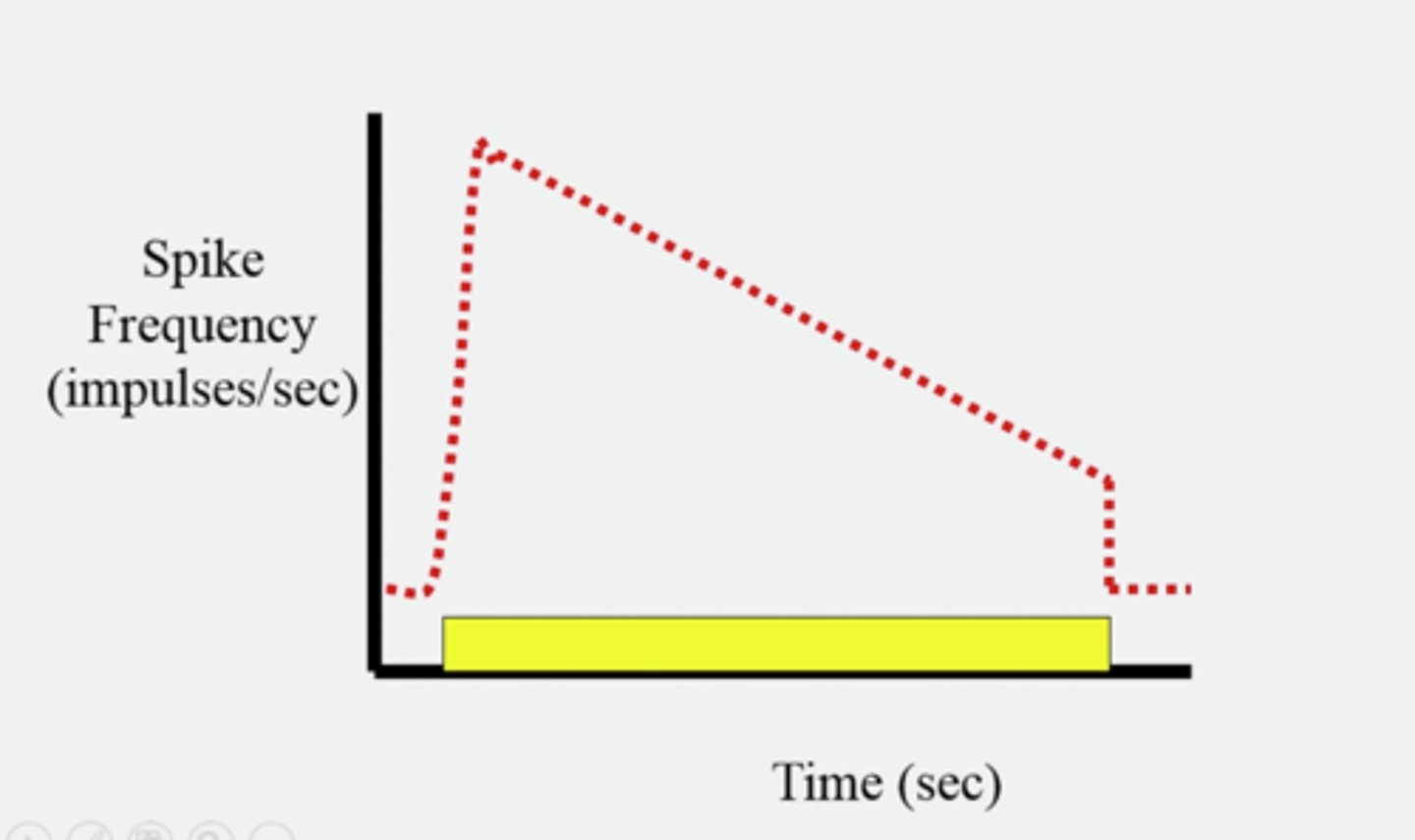
Sensory adaptation.
A process by which a stimulus causes a decreasing frequency of action potentials (receptors get used to the stimulus)
Slow adaptation, tonic receptors: pain and cold receptors:
Rapid adapting, phasic receptors: pressure, smell, heat.
Stevens' Law.
sensation is a power function of the relative stimulus, where the exponent n is characteristic for the modality of sensation.
Sensation = constant * (x/x0)^n
Weber-Fechner law.
Describes the relationship between the intensity of the stimulus and the perceived sensation. Relative change of the stimulus is proportional to the change in sensation intensity. This law is only valid for sensations between 100-1000 Hz but fails at lower or higher frequencies.
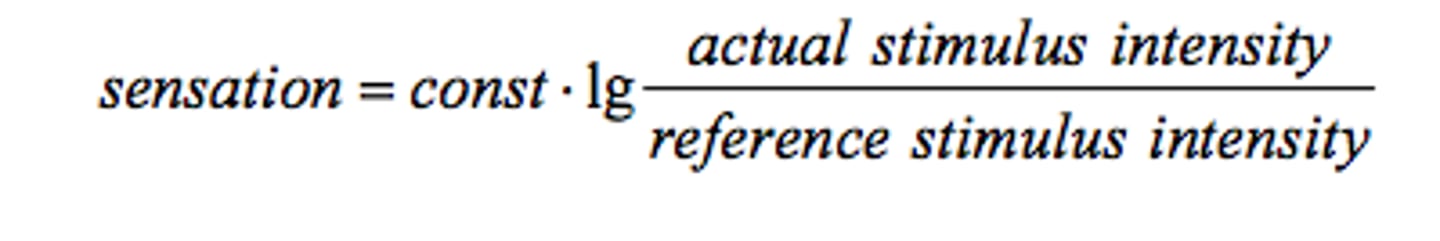
Biophysics of hearing I.: the outer ear.
Made of the auricula, external auditory canal and eardrum
The auricula steers the sound waves towards the external auditory canal, which then transmits the sound towards the eardrum.
The eardrum is brought into resonance by the sound wave and will transmit the vibrations towards the middle ear.
Biophysics of hearing II.: the middle ear.
contain the auditory ossicles: malleus,incus, stapes and eustachian tube.role of middle ear: pressure amplification due to cross sectional area different b/w eardrum and the oval window and auditory ossicles→converting the pressure from air medium into liquid medium (without it, it would be complete reflection and no signal will occur)
eustachian tube: pathway of atmospheric pressure equilibrium
Biophysics of hearing III.: Békésy's hearing model.
Mechanism in the inner ear, by which there is a spatial frequency coding. The basal membrane stretches from the oval window to the cochlea. The incoming sound vibrations will cause "surface waves" on the membrane. Depending on the relative location of the wave, different inner hair cells are stimulated, and this contributes to the frequency resolution.
Biophysics of hearing IV.: signal transduction in hair cells.
the basal membrane distorted due to the ''surf wave'' '(depends on frequency of the sound waves) which cause the hair cells to tilted→cation gates are open (k+ & Ca+2) → depolarization occur → neurotransmitters released into afferent nerve cells→ depolarization occurs→ frequency of AP increase.
*outer hair cells also amplifying narrow range of frequency of the sound by an active work (by contraction tilted the stereocilia and transduce AP) → positive feedback
Signal amplification by hair cells
Outer hair cells have a positive feedback mechanism by which they contract, as a result of sound. When they contract (due to a motor protein "prestin") they move the tectorial membrane. This occurs at a narrow frequency range and causes a large amplification within that region.
The phon scale.
represent the loudness level according to weber-fechner lawwhich describe the sensation as a log function of the relative stimuli, expressed in decibel. (not accurate as sone scale)

The sone scale.
A subjective perception of loudness level. Based on the observation that every 10 phon increase in loudness level is perceived as a doubling of the sound sensation. 1 son = 40 phon, 2 son= 50 phon, etc.
Reaction steps of light sensation.
light travels from the cornea→ to the anterior chamber→ to the lens→ to the vitreous humor→ to the optic nerve→ to the photoreceptor cells.
light photon is being absorbed, rhodopsin → conformational change occur→ enzymes are being activated→ big number of Na+ channel is inhibited→ hyperpolarization→ the release of inhibitor transmitter is reduced.
Photoreceptors of the retina.
Cones
6.5 Million
Daylight vision
Colour sensitive
located at center/fovea of retina
Small convergence = high spatial resolution
Lower frequency sensitivity
Rods:
120 million
twilight vision
Not colour sensitive
Located at retinal periphery
Many rods per ganglion - higher convergence-lower resolution
Higher frequency sensitivity
Basis of color sensing.
Colours are not a physical property b.c not all the colours can be defined by wavelength (e.g. brown).
The human eye has 3 different color sensitive receptors (each is sensitive in a range of wavelengths (blue region,red-yellow region and green region). By mixing them in various weighting factors it is possible for us to sense any color.

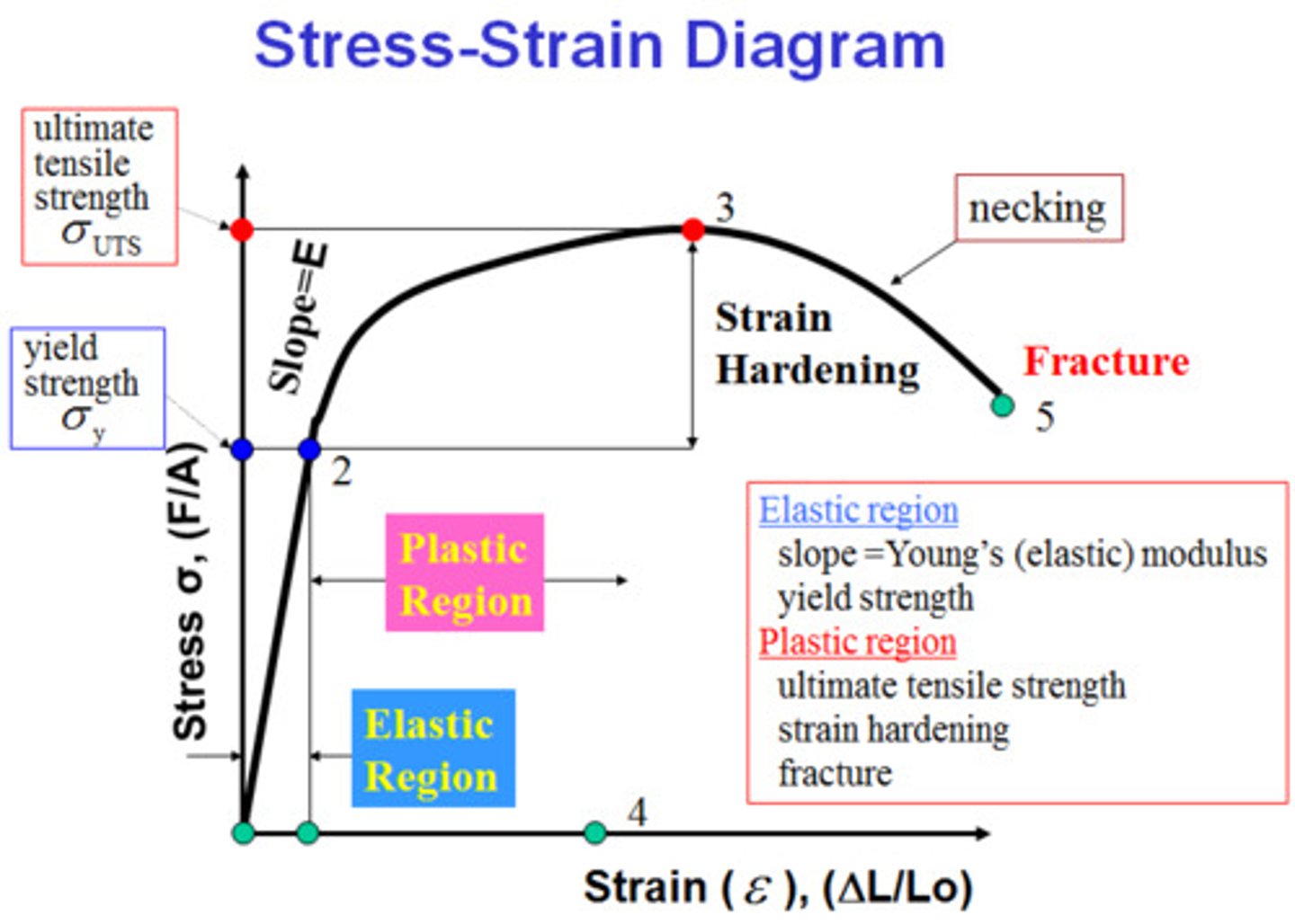
Biomechanics I.: Stress-strain diagram and its ranges.
Stress: amount of force per area: F/A (N/m^2), Pascal. Strain:measure of length deformation from original length: m/m (dimensionless).
Stress is proportional to strain
Proportional region: where the stress is proportional to the strain, and there is no hysteresis (part of the elastic region).
Elastic region:reversing the stress will result in recovery of the length, no permanent deformation occurs. Hysteresis is possible.
Plastic region: after this critical threshold, irreversible deformation occurs.
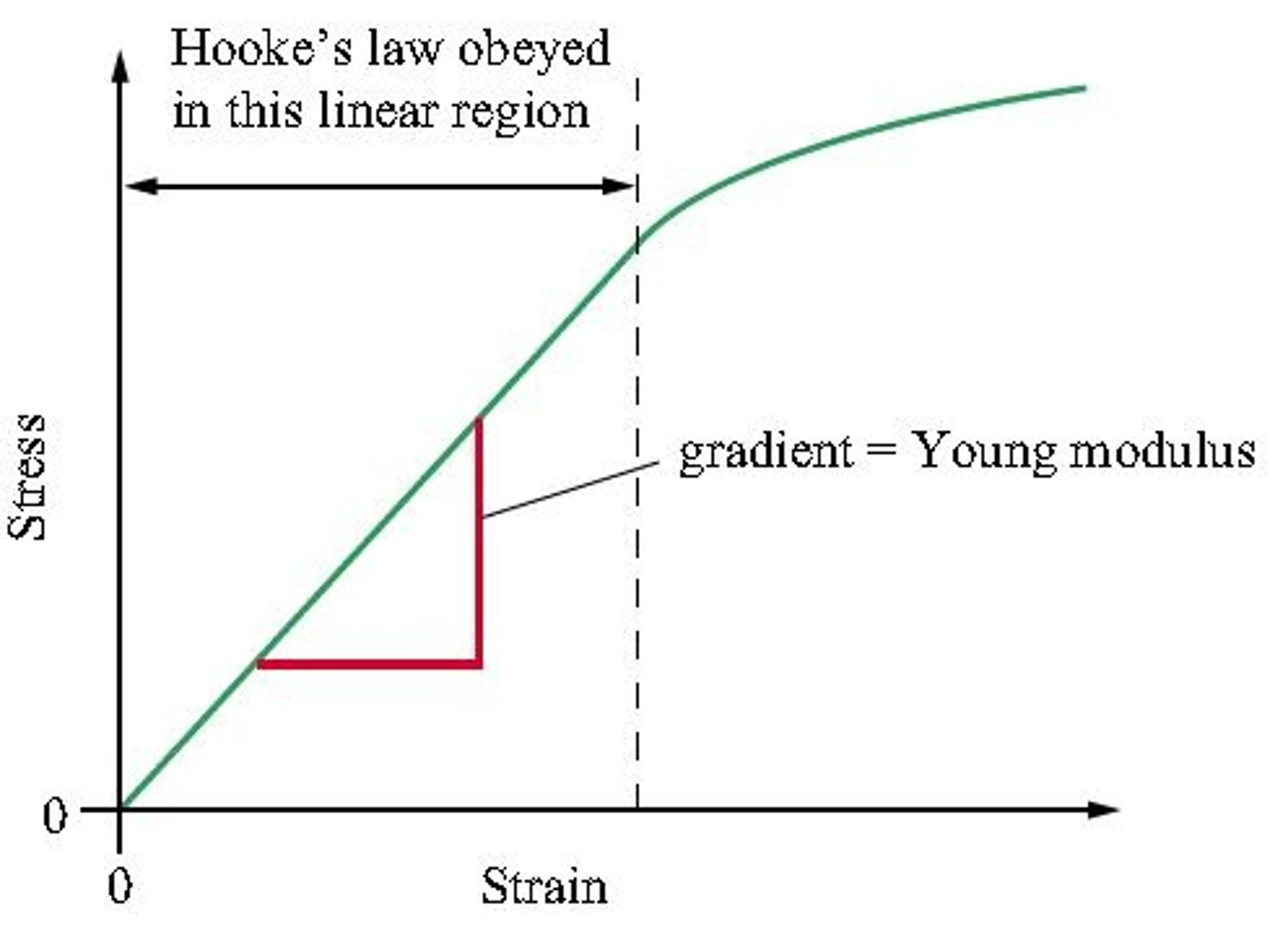
Biomechanics II.: Hooke's law, Young-modulus.
Young modulus v.s. hooke's law (look at formulas)
hooke's law:spring constant depends on the dimension of the body (cross-section and length) andon the material (E young modulus constant).
Can be calculated by the external force acting on the body and the deformation.
young's modulus-measure the stiffness of the materials (stress/strain) and the constant depends only on the type of the material.
Biomechanics III.: Laplace-Frank-equation.
The tangential stress is proportional to the intramural pressure and the ratio between the inner radius and the wall thickness. Relevant when considering hypertension, as the increased intramural pressure will cause an increase in the tangential stress, which will cause an increase in the inner radius and a decrease in the wall thickness, thus Aneurysm is formed. This dangerous cycle continues and may cause rupturing of the wall.
Viscoelasticity I.: mechanical model
viscous matter: resist shear flow and strain linearly with time when stress is applied
elastic matter strain when stretched and immediately return to their original state once the stress is removed
visco-elastic body→ characteristic of biopolymers:viscous and elastic behavior appears simultaneously
Model: parallel connection of spring and dashboard

Viscoelasticity II.: stress-relaxation, energy dissipation.
Stress-relaxation is a property of visco-elastic materials (such as muscles), where if the strain is held constant, there will be a decrease in stress as a function of time. (example: when stretching a muscle, the stress will decrease when the strain is held constant).
This decrease in stress can be expressed by this equation: phi = phi0*e^-t/Rho, rho = time constant.
Stress dissipation/hysteresis:
occurs when the path to recovery after deformation is different than the path to deformation. The energy difference between those two paths, is the energy loss or dissipation.
Biomechanical characteristics of bone and enamel.
Bones made of:a. organic substance: mainly collagen b. inorganic:hydroxyapatite
Enamel composed of : only inorganic molecules: mainly hydroxyapatite
*The inorganic compositionimproves the strength of the bone/enamel.
*enamel is the hardest substance in the body
Biomechanics of elastic arteries, distensibility.
Distensibility is defined as the change in vessel volume under pressure. When considering relative expansion over transmural pressure, the slope of the graph is the distensibility. At the beginning of the expansion, the elastic properties of the artery play a role. After
a certain point, the collagenic properties take over and prevent overstretch.
Structure and types of motor proteins.
The globular head at the N-terminus is the motor domain(ATPase) and provides a specific binding-site for the respective cytoskeletal filament.
The C-terminus binds to the surface that is being moved
classification based on the substrate they bind to:
1. actin cased: myosin
2. microtubule based: kinesin, dyneins,dymatize
3. DNA & RNA polymerase
4. rotatory motors: found in the mitochondria,flagellar motor
5. mechano-enzyme: ribosome
Processivity, typical force range and working distance of motor proteins.
Processive motor proteins are characterised by duty cycles which are larger than 1. Meaning that they remain attached most of the time. Ex: Kinesin, DNA and RNA polymerase.force range: piconewton scaledistance:nanometer scale