Chemistry: Energy Unit
1/25
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
26 Terms
Energy
Capacity for doing work or supplying heat
Potential Energy
Energy that is stored (converted to work or heat)
Kinetic Energy
Energy of motion (related to temperature and heat due to motion of molecules)
Heat
Energy that is transferred from one object or substance to another because of a difference in temperature between them
Thermochemistry
The study of energy changes that occur during chemical reactions and during changes in state
Specific Heat (heat capacity)
Amount of heat required to raise the temperature of 1 gram of a substance by 1°C
Specific Heat
q = (m) (c)( ΔT)
Calorimetry
The measurement of the transfer of heat into or out of a system during a chemical reaction or physical process.
Calorimeter
An insulated container that is used to measure heat change
Energy and Change of State
The stronger the attractions between the molecules in a substance the greater the energy input for phase changes.
System
The specific part of the universe in which we focus our attention (Chemicals)
Surroundings
Everything else besides the system
Exothermic
Heat is released by the system to surroundings (feels warm)
Endothermic
Heat is absorbed by the system (feels cold)
Bond Energy
Is released when bonds form. The same bond energy must be added to break the bonds and separate the atoms again.
Thermochemical equation
A chemical equation that includes the enthalpy change of the reaction.
ΔH
Change in energy during a reaction
If ΔH is positive
The reaction is endothermic
If ΔH is negative
The reaction is exothermic
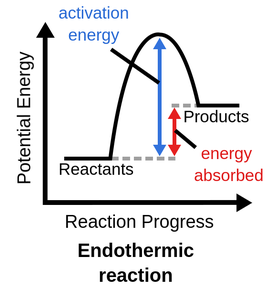
Endothermic Diagram
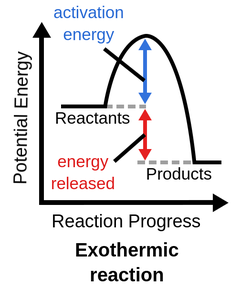
Exothermic Diagram
Hess’s Law
Two or more thermochemical equations can be added together to give a final equation.
Hess’s Law: Rule #1
Reverse a chemical → change the sign of ΔH (+ or -)
Hess’s Law Rule #2
If you multiply the equation you multiply the ΔH, too.
Hess’s Law Rule #3
Add up ΔH to determine the ΔH for the overall rxn.
ΔHrxn°
The standard enthalpy of reaction: the change in enthlapy for a reaction carried out under standard conditions.