Chapter 17.4: Titrations of Weak Acids and Weak Bases
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Weak Acid–Strong Base Titrations Differ from Strong Acid–Strong Base Titrations
Initial pH is higher.
• Weak acids do not release as much H+ as strong acids.
• Weak acids with lower Ka values have higher initial pH values.
…There is an initial rise in pH.
• Weak acids are poor at resisting pH changes until the pH is closer to their pKa values.
• Weak acids, with higher pKa values, have higher initial rises in pH.
…pH at the equivalence point is greater than 7:
• Weak acids do not release as much H+ as strong acids.
• The conjugate bases of weak acids are more basic than counterions of strong acids.
• Titrations of weaker acids have higher pH values at the equivalence point.
…The pH change near the equivalence point is less abrupt.
Weak acids with lower Ka values have more elongated (less and less abrupt) titration curves.
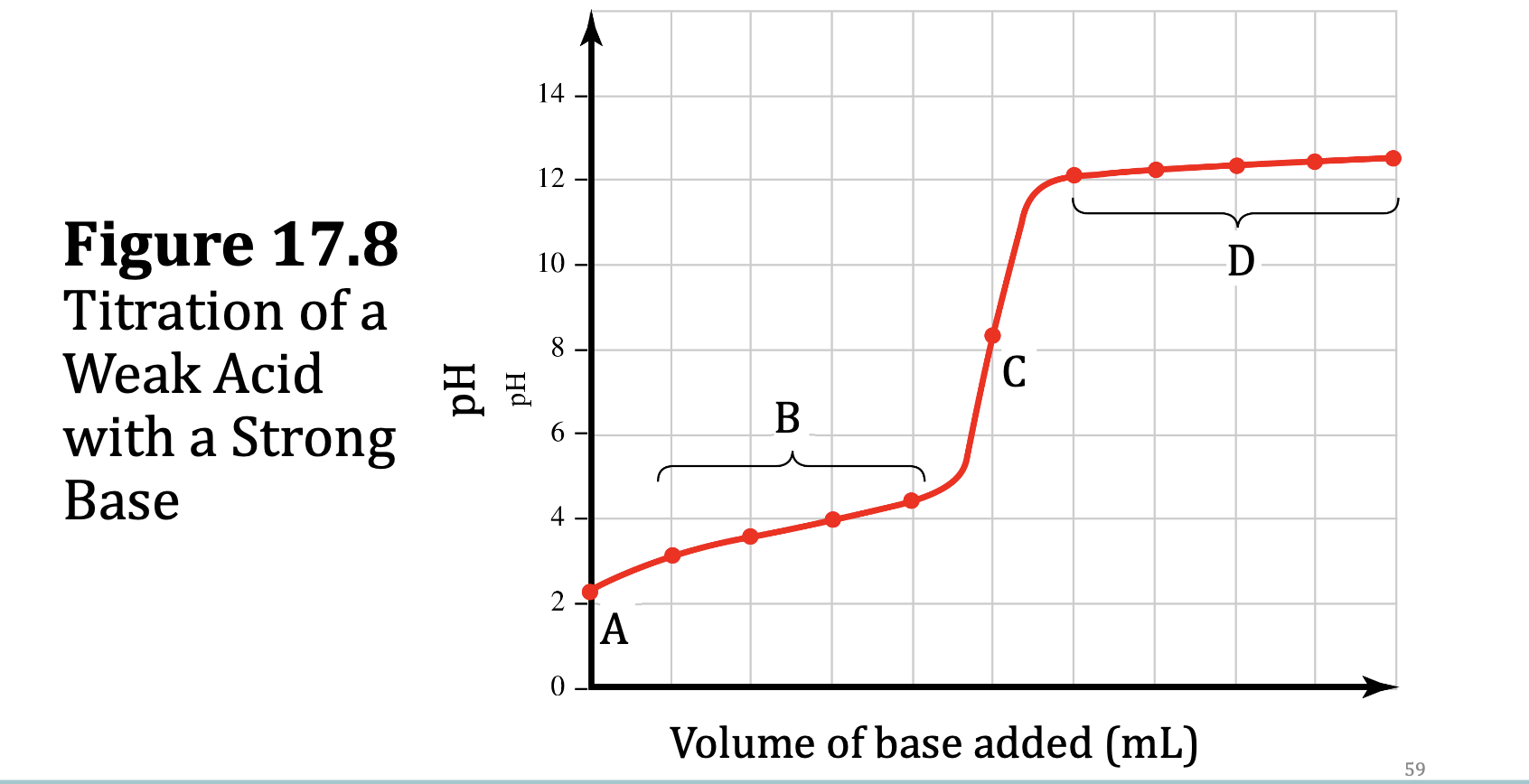
Calculating pH in Weak Acid–Strong Base Titrations Region A: Initial pH
The initial pH is the pH of a weak acid solution. As usual, the simplification is possible when <5% of the weak acid ionizes.
Calculating pH in Weak Acid–Strong Base Titrations Region B: Between Initial pH and Equivalence Point
• The addition of strong base has formed sufficient conjugate base to create a buffer solution in this region.
• Use the Henderson–Hasselbalch equation to determine pH here, using x= [OH]− .
Calculating pH in Weak Acid–Strong Base Titrations Region C: The Equivalence Point
At the equivalence point, only the conjugate base remains. The pH calculation is the same as that of a basic salt.
Calculating pH in Weak Acid–Strong Base Titrations Region D: After the Equivalence Point
Past the equivalence point, the pH depends on the concentration of excess OH− added.
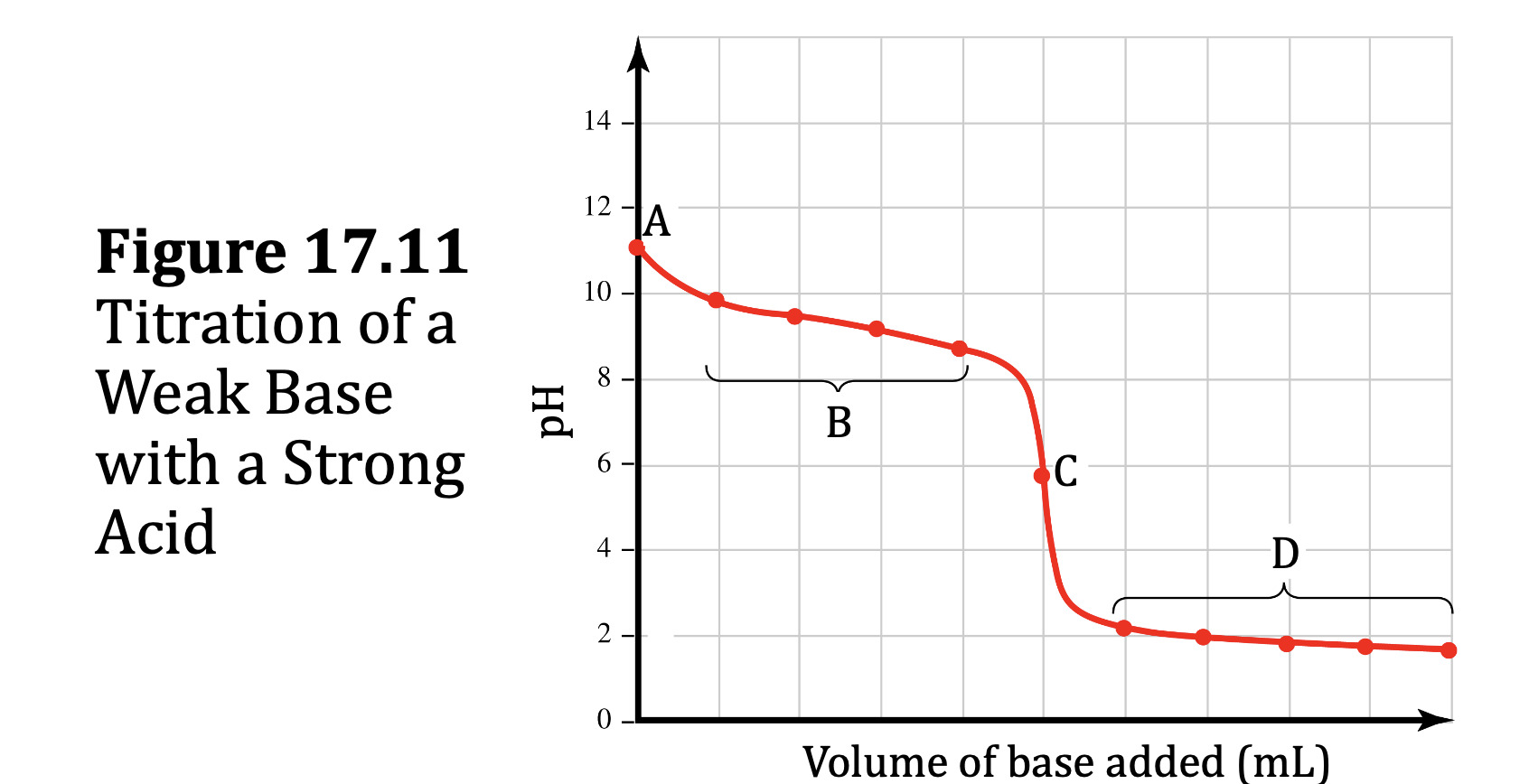
Weak Base–Strong Acid Titrations
• These curves are similar to weak acid–strong base titration curves, with these differences:
• The initial pH is basic, not acidic.
• The pH at the equivalence point is acidic, not basic.
• The pH at the half-equivalence point is equal to the pKa of the conjugate acid of the weak base being titrated.
• The final pH is acidic, not basic.
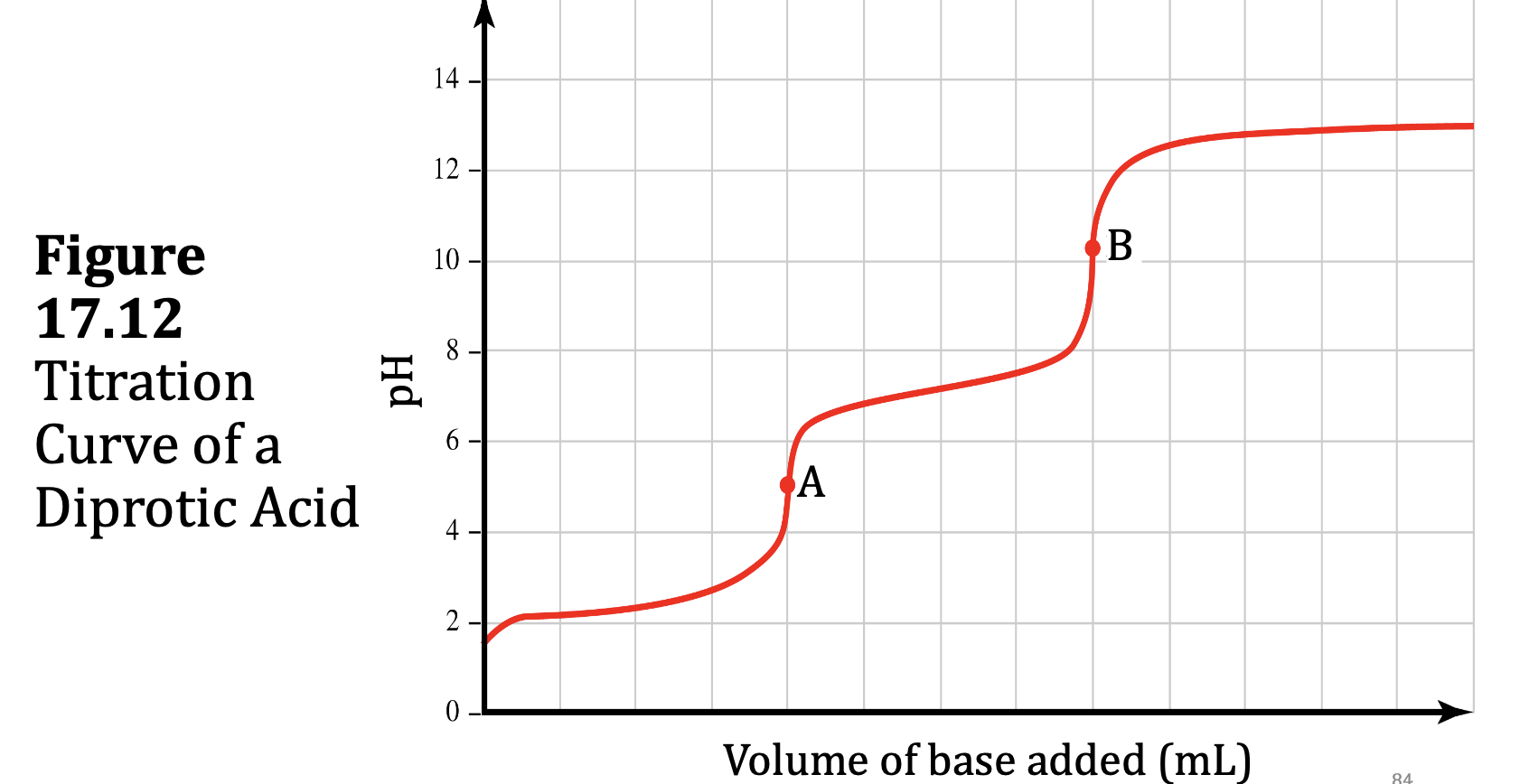
The diprotic acid malonic acid ionizes as follows:
H2CH3H2O4(aq) + H2O(l) ⇌ HCH3H2O4−(aq) + H3O+(aq)
Ka1 = 1.42 × 10–3
HCH3H2O4−(aq) + H2O(l) ⇌ CH3H2O42–(aq) + H3O+(aq)
Ka2 = 2.01 × 10–6