lecture #1 Chem rocks
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Terrigenous Sediments
form from weathering/erosion of preexisting rocks, everything we have studied up to now. usually transported into the basin of deposition
Chemical Sediments
direct chemical precipitation of minerals in the depositional basin
Biogenic Sediments
precipitated within the depositional basin by a biogenic agent, including bacteria
rock with most carbonates
biogenic rocks
clastic
refers to any grains that are transported before final deposition
chemical and biogenic sediments
are commonly non-clastic
Siliciclastic influence
High clastic input (Bangladesh) kills reefs; minimal input (Great Barrier Reef) allows carbonates to grow.
Hydrocarbon source rock
Upper Cretaceous- Turonian
Lower Cretaceous- Aptian
Upper Jurassic- Kimmeridgian
Upper Jurassic- Oxfordian
how are ions introduced into rivers and ground water by
chemical and biological degradation of primary mineral grains creates a dissolved load
How do you concrete the salt into the basin? (Why seawater has dissolved salts)
Evaporation removes water and leaves salts behind
To be freshwater, the salt amount must be
less than one gram per liter
% of the weight that is dissolved salts in salinity
3.5
What mineral do u get when mixing sodium and chloride?
Salt
Tops Ions?(most abundant)
Chloride and Sodium
Ocean salinity is stable cuz?
input rate= removal rate
how many elements got a measurable value in sea water? ( but they make up only 3.5 of the weight
85
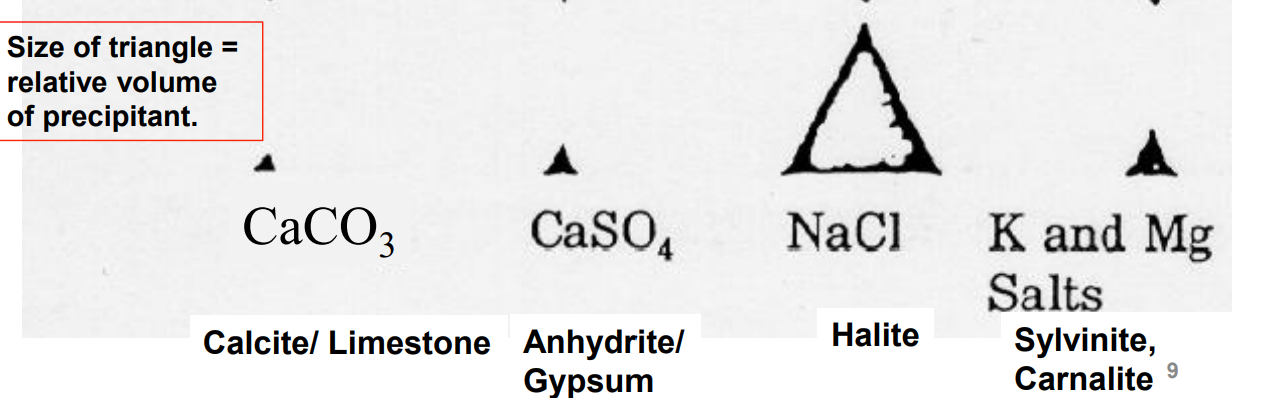
what’s the order of evaporation for sea water?
-calcium carbonate
-gypsum/anhydrite( 19% of water remaining)
-halite(9.5% left)
-bitter salts(4% left)
volumes of precipitants for sea water evaporation
most common rock type for carbonate
limestone(chalk)
most common chemical composition
(CaCo3)
most abundant modern and ancient chemical sediment
limestone
major host rock for hydrocarbons
limestone
Gypsum facts
precipitated in restricted marine bodies/saline lakes
CaSO4*H2O
Anhydrite facts
Gypsum converts into this by dehydration ( depths of over 50-60C)
Selenite
A giant, bladed crystal variety of Gypsum
what are the most common bitter salts with evaporation of sea water
sylvinite( KCl)and carnallite (MgCl)
carbonate equilibrium

pH control with carbonate equilibrium
increase in CO2 → lowers pH and dissolves CaCO3
decrease in CO2 → raises pH and precipitates CaCO3
what causes the loss of CO2 ( control of CO2)
increase in temp and decreases in water pressure
Why reefs/carbonates are found in low latitudes?
warm water holds less CO2/ promotes CaCO3 precipitation
Equator ( middle section of the continents)
warmer waters area shows in a greater increase in reefs ( Southwest Asia and South America)
What is the correlation between ocean temperature, latitude, and calcium carbonate saturation?
Low latitudes, higher temps, more saturated= more probability of reefs
what makes a carbonate sediment?
inorganic precipitation of CaCO3 ( examples are called Ooids and whitings)
what are the 3 ways that biogenic carbonate is made
direct extraction- skeletons/shells
photosynthesis- plants/bacteria remove CO2( precipitation)
fecal pellets- repackaging of sediments
vadose zone
Unsaturated zone above the water table where dissolution occurs
karst
Landscape formed by this dissolution
Speleothems
Precipitation features inside caves
when water is getting out of the ground( gw precipitation) it’s called
discharge
water coming in from the ground within the cave ( gw dissolution)
recharge
Phreatic Zone
Saturated zone below the water table
Geodes
Form slowly in the phreatic zone via mineral replacement
Travertine
Carbonate precipitates from springs, often in thin layers.
Spring Controls
Precipitation is controlled by changes in temperature, chemistry, or flow rate.
chert
rock made of Amorphous Silica-SiO2
Formed abiotically or from Diatoms(biotic hard parts)