Chem Topic 7
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
In a hydrogen bond, what atoms could be bonded to the hydrogen
Nitrogen
Oxygen
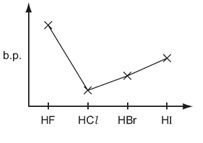
What is the trend in boiling temperatures of hydrogen halides up to Hydrogen iodide
HF has the highest, HCl the lowest, then they increase in BT
What is a disproportionation reaction
A reaction where an element in the same species is simultaneously reduced and oxidised
Oxidation number rules for flourine
Always -1
Oxidation number rules for hydrogen
Always +1 unless combined with a less electronegative element, where it becomes -1
Oxidation number rules for oxygen
-2 unless:
Combined with flourine which makes it +2
In a peroxide which makes it 1-
solubility of group 2 compounds
Nitrates: soluble
Chlorides: soluble
Hydroxides: increase solubility down the group
Sulfates: decrease solubility down the group
How does the CO2 test work chemically
Calcium hydroxide (lime water) reacts with CO2 to form calcium carbonate (insoluble) so the white precipitate turns the solution cloudy.
Test for sulfate ions
Dilute acid like nitric acid + barium nitrate
How can greater thermal decomposition of nitrate be distinguished by minor decomposition
Greater decomposition released brown fumes from the NO2
Thermal stability trends of group 1 & 2 nitrates
The stability increases down the groups, and group 2 is less stable (higher charge density → lower stability)
Formula for decomposition of metal nitrates
Metal nitrate → Metal nitrite + Oxygen
Metal nitrate → Metale oxide + Nitrogen dioxide + oxygen
Formula for decomposition of metal carbonates
Metal carbonate → Metal oxide + Carbon dioxide
Test for ammonium ions
Sodium hydroxide solution and damp red litmus
What is produced when hydrogen halides react with water
A colourless acid