Hydrogels: Degradable & Resorbable Materials
1/35
Earn XP
Description and Tags
Lecture 2
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
What is a hydrogel?
A water-swollen polymer network held together by crosslinks that allow the material to absorb large amounts of water while maintaining structural integrity.
Why are hydrogels useful for biomedical applications?
Their high water content mimics biological tissues and allows diffusion of nutrients, metabolites, and drugs.
What mechanisms can hold hydrogel networks together?
Covalent crosslinks (PRIMARY)
Ionic interactions
Hydrogen bonds
Bio-recognition interactions (affinity)
Hydrophobic interactions
Polymer crystallites
Physical entanglements
What makes hydrogels water-swellable?
At least one polymer in the hydrogel must be hydrophilic.
Types of hydrogels: complexation hydrogels
held together by specific forces, including hydrogen bonds, hydrophobic group associations, and affinity complexes.
Types of hydrogels: ionic hydrogels
Hydrogels with charged polymer backbones that may be:
Neutral
Anionic
Cationic
Ampholytic
Types of hydrogels: interpenetrating network (IPN) hydrogel
Two or more polymer networks interlaced but not covalently bonded. Both properties are expressed.
What are the two main types of hydrogel crosslinks?
Chemical (covalent)
unreacted functional groups
Physical (non-covalent)
multifunctional junctions
physical entanglements
chain loops
What is Mc in hydrogel networks?
Molecular weight between crosslinks
density determines mesh size, porosity, and mechanical properties.
What pore sizes allow cell and tissue penetration?
Macropores (>50 nm, <300 µm)
What pore sizes control drug and solute diffusion?
Mesopores (2–50 nm) and micropores (<2 nm)
What hydrogel property affects diffusion coefficient?
Pore volume fraction
What causes hydrogel swelling?
Water binding to hydrophilic polymer chains and solutes diffuse through free water channels.
What occurs at hydrogel swelling equilibrium?
Thermodynamic swelling force = polymer chain retractive force.
What properties are affected by swelling extent?
Solute diffusion
Surface properties
Molecular mobility
Optical properties
Mechanical properties
What happens when a crosslink in dried state swells?
network expands
What happens when a crosslink in a swelled state swells?
less network expansion
What is PHEMA and where is it commonly used?
Hydrogel (Poly(2-hydroxyethyl methacrylate)
used in contact lenses because it is biocompatible and swells when hydrated, also inert, and permeable
What happens when methacrylic acid is added to PHEMA?
Swelling decreases due to hydrophobic interactions.
What is poly(vinyl alcohol) (PVA) and where is it used?
A non-toxic hydrophilic polymer that forms physically crosslinked hydrogels via freeze–thaw cycles.
(forms quasi-permanent crystallites)
used in blood-contacting hydrogels
Why is PEG or PEO widely used in biomaterials?
Biologically inert and resistant to protein adsorption
Used in tissue engineering as a crosslinking constituent and PEGylation of drugs.
because of H in back, absorbs most water of any material
Why are degradable biomaterials useful in medicine?
They provide short-term function and degrade after healing
eliminating the need for surgical removal (no secondary intervention)
Examples:
Sutures
Fixation devices
Drug delivery matrix
Tissue engineering scaffolds
What is degradation and biodegradation?
Chemical cleavage of covalent polymer bonds (hydrolysis, oxidation, enzymes).
Bio = breakdown caused by biological agent
What is erosion and bioerodible?
Physical loss of material mass or shape. Doesn’t have to be degradation.
i.e. a sugar cube
bio = water-insoluble polymer converted to water-soluble by physical (dissolution) or chemical (cleavage) processes
What is bioresorption or bioadsorption?
Removal of polymer degradation products by the body.
Surface erosion vs. bulk erosion
Surface = Water penetration is slower than polymer degradation, so erosion occurs only at the surface. Maintains bulk properties.
Bulk = Water penetrates the entire polymer faster than degradation occurs. Water uptake followed by erosion.
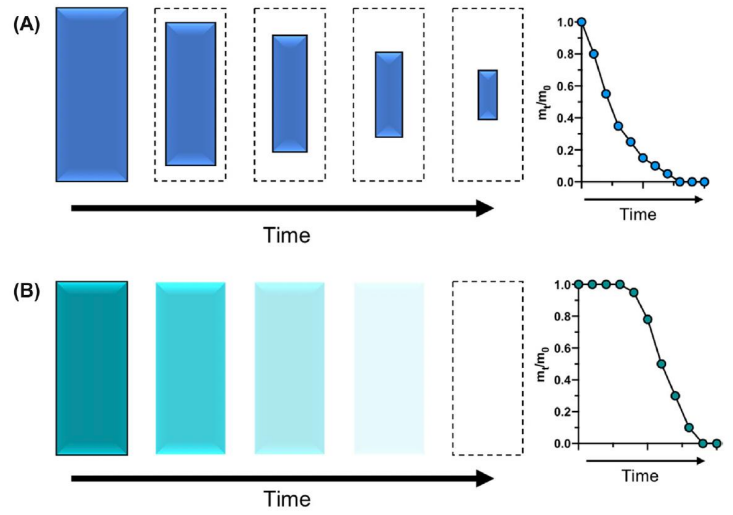
What polymer bonds enable degradation?
Hydrolytic groups incorporated into the polymer backbone.
Why are polyanhydrides useful for drug delivery? (degradable polymer)
They limit water penetration and undergo surface erosion, producing near zero-order drug release.
What are polyesters (PLA and PGA) place in degradable polymers?
they erode rapidly and rely on crystallinity and hydrophobicity to tune degradation rate
What polymer characteristics influence degradation rate?
Morphology
Crystallinity
Stereochemistry (tacticity)
Hydrophobicity (more hydrophobic = less water in = less degradation)
Degradation routes: What causes oxidative degradation?
Reactive peroxide radicals generated during inflammatory responses.
Degradation routes: What chemical processes cause degradation?
Hydrolysis
Enzymatic degradation
pH changes
Photodegradation
What state do materials go from and to when degraded?
water insoluble —> soluble
What are smart or stimuli-responsive biomaterials and why are they useful for medicine?
Materials that change properties in response to environmental stimuli.
i.e. Ionic strength
Electric fields
Light
Enzymes
They can trigger controlled drug release in response to physiological conditions.
How do temperature-responsive hydrogels work?
They undergo reversible phase separation due to changes in hydrophobic/hydrophilic balance.
Ex: Poly(N-isopropylacrylamide) (PNIPAM)
How do pH-responsive hydrogels work?
Swelling behavior changed by polymer balance of ionization.