commercial cells
1/17
Earn XP
Description and Tags
overall eqns given for completeness but can be deduced
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
give the features of non rechargeable cells and explain why they are necessary:
porous separator - allows ions to move/flow/transfer
carbon rod - made from graphite to allow e- to flow
zinc - container for cell (gets used up)
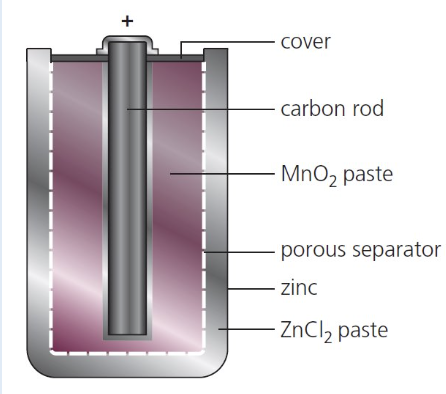
give an example of a non rechargeable cell:
either:
alkaline battery
dry cell
suggest why a non rechargeable cell often leaks after being used for a long time (1)
Zn oxidised

give an example of a rechargeable cell and give the 2 ½ eqns associated w/ it:
lithium ion:
Li+ + CoO2 + e- ⇌ LiCoO2 (+ve electrode)
Li+ + e- ⇌ Li (-ve electrode)
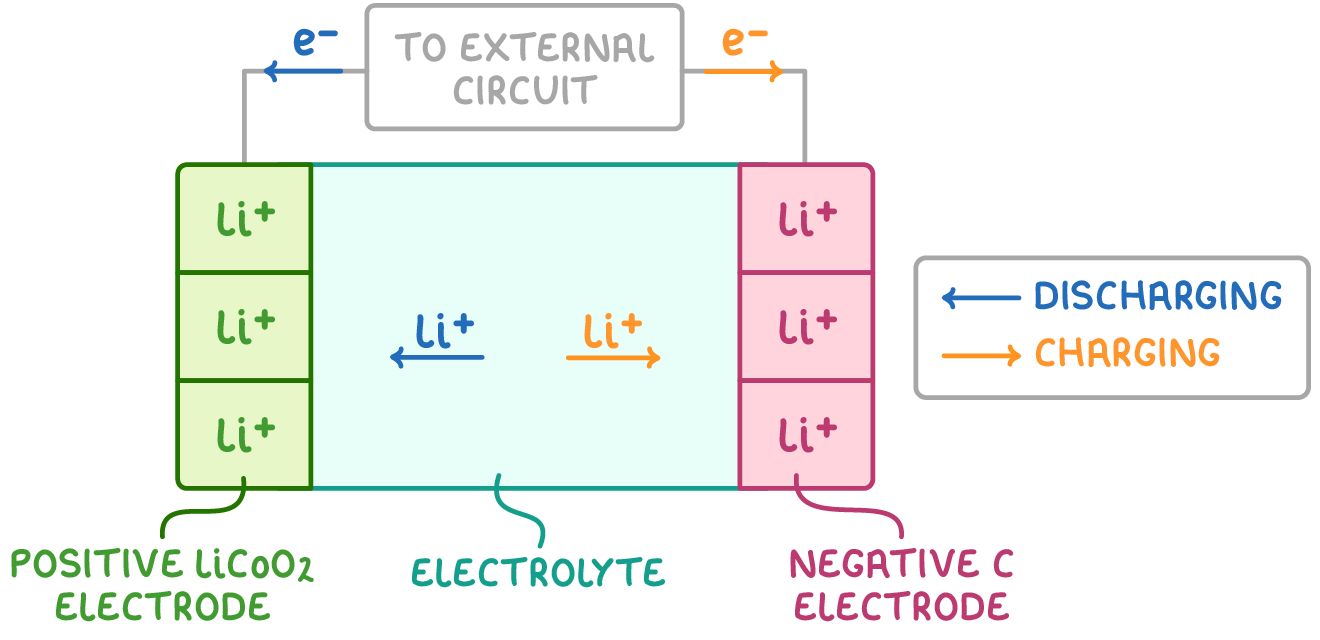
give the overall eqn for the reaction that occurs in a lithium-ion cell:
Li + CoO2 ⇌ LiCoO2
for a rechargeable reaction, what do the different directions of the reaction apply to?
forward reaction = discharge (being used)
backward reaction = recharge (being charged)
give the 3 types of cell:
non rechargeable (irreversible)
rechargeable (reversible)
fuel cells
describe how an alkaline hydrogen-oxygen fuel cell works:
H2 and O2 gases are supplied to separate platinum-containing electrodes
an anion-exchange membrane separates the electrodes, allowing passage of anions (OH-) and water but not the gaseous reactants
the electrolyte is an aq KOH soln
e- flow from the -ve electrode through an external circuit to the +ve electrode, while OH- ions migrate through the anion-exchange membrane towards the -ve electrode
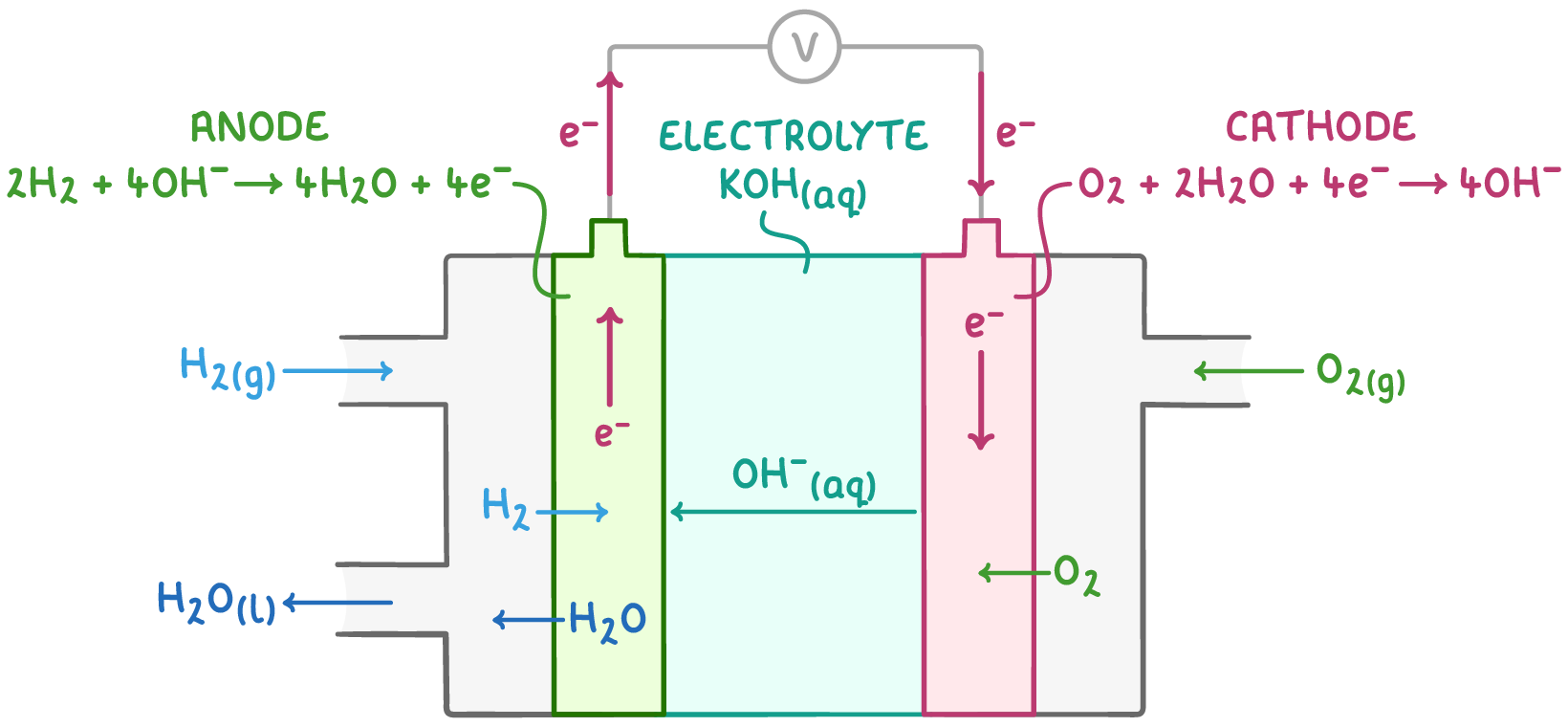
give the eqn for the reaction at the -ve electrode of an alkaline hydrogen-oxygen fuel cell:
H2 (g) + 2OH- (aq) → 2H2O (l) + 2e-
give the eqn for the reaction at the +ve electrode of an alkaline hydrogen-oxygen fuel cell:
O2 (g) + 2H2O (l) + 4e- → 4OH- (aq)
give the overall eqn for the reaction that occurs in an alkaline hydrogen-oxygen fuel cell:
2H2 + O2 → H2O
give a benefit of using non-rechargeable cells:
cheap
give a drawback of using non-rechargeable cells:
difficult to dispose of/waste issues
give 3 benefits of using rechargeable cells:
less waste as metal compounds reused
cheaper in the long run
lower environmental impact
give 2 drawbacks of using rechargeable cells:
need E source to recharge
some waste at end of useful life
give 3 benefits of using hydrogen fuel cells:
converts more of the available E from combustion of H2 into KE of car
no pollutants (CO2, SO2 etc.) as only waste product is H2O
efficient
no need to recharge
give 4 drawbacks of using hydrogen fuel cells:
need constant fuel supply
H2 is flammable and explosive
high cost
difficult to source H2 (usually made using fossil fuels)
suggest why the recharging of a lithium cell may lead to the release of CO2 into the atmosphere (1)
electricity from recharging the cell may come from power stations burning fossil fuel