Chapter 4 - Reactions in Aqueous Solutions | Definition
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
solution
a homogenous mixture of 2 or more substances
solute
the substance(s) present in the smaller amount(s)
solvent
the substance present in the larger amount
nonelectrolyte
a substance that, when dissolved, results in a solution that does not conduct electricity.
electrolyte
a substance that, when dissolved in water, results in a solution that can conduct electricity.

nonelectrolyte

weak electrolyte

strong electrolyte
A reversible reaction
The reaction can occur in both directions.
Hydration
the process in which an ion is surrounded by water molecules arranged in a specific manner.
Precipitate
insoluble solid that separates from solution
Solubility
the maximum amount of solute that will dissolve in a given quantity of solvent at a specific temperature.
Properties of Bases
bitter taste + slippery + soaps contain + Cause color changes in plant dyes + Aqueous base solutions conduct electricity.
Properties of Acids
sour taste + React with certain metals = h2 + React with carbonates and bicarbonates = co2
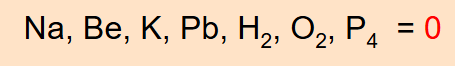
Free elements have an oxidation number of zero
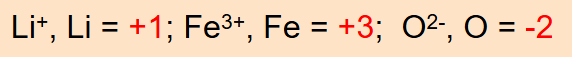
In monatomic ions, the oxidation number is equal to the charge on the ion.
Group IA metals
+1
IIA metals
+2
fluorine
-1
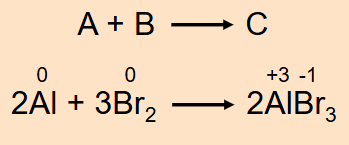
Combination Reaction
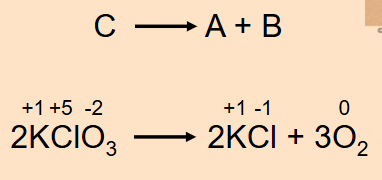
Decomposition Reaction
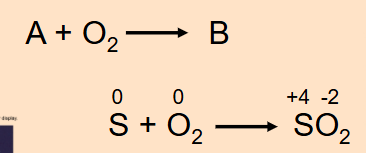
Combustion Reaction
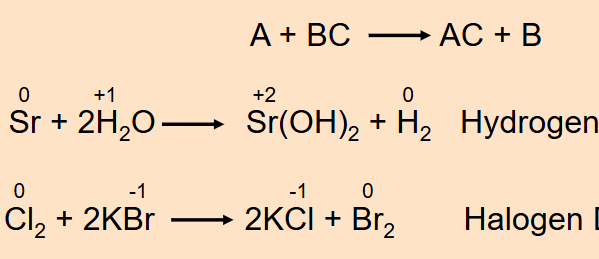
Displacement Reaction
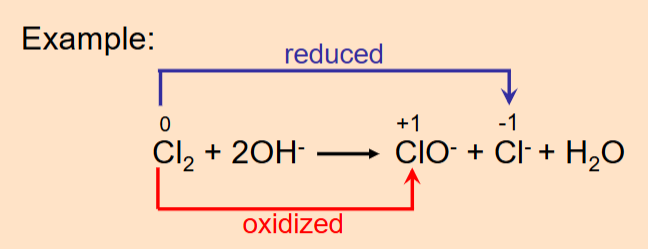
Disproportionation Reaction
The concentration of a solution
the amount of solute present in a given quantity of solvent or solution.
Formula of molarity
M=n/v
Dilution
the procedure for preparing a less concentrated solution from a more concentrated solution.
Formula of Dilution
n before dilution = n after dilution
titration
a solution of accurately known concentration is gradually added to another solution of unknown concentration until the chemical reaction between the two solutions is complete.
Equivalence point
the point at which the reaction is complete
Indicator
substance that changes color at (or near) the equivalence point

Acid-base reactions

Redox reactions