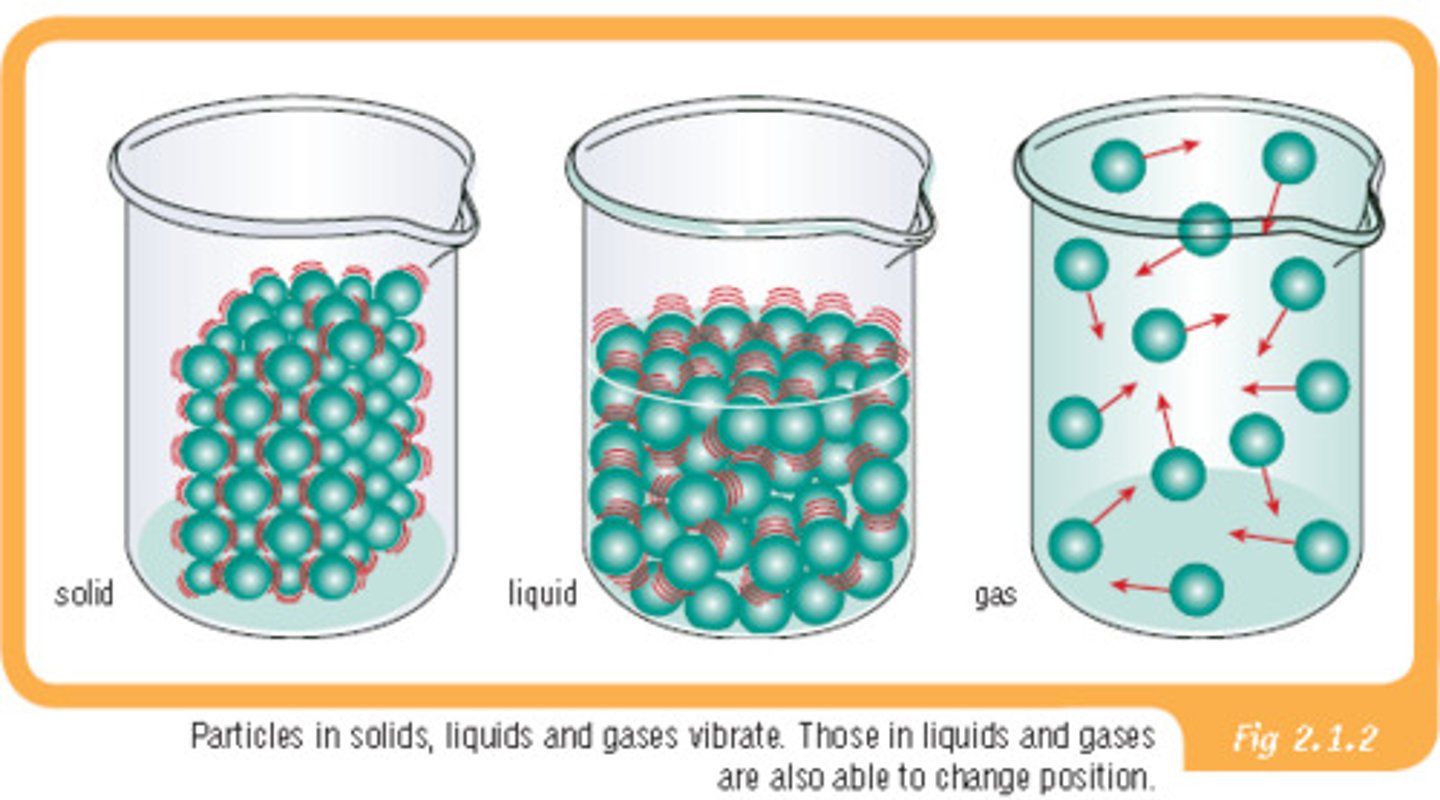
Bio111: Water and Carbon
1/29
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
30 Terms
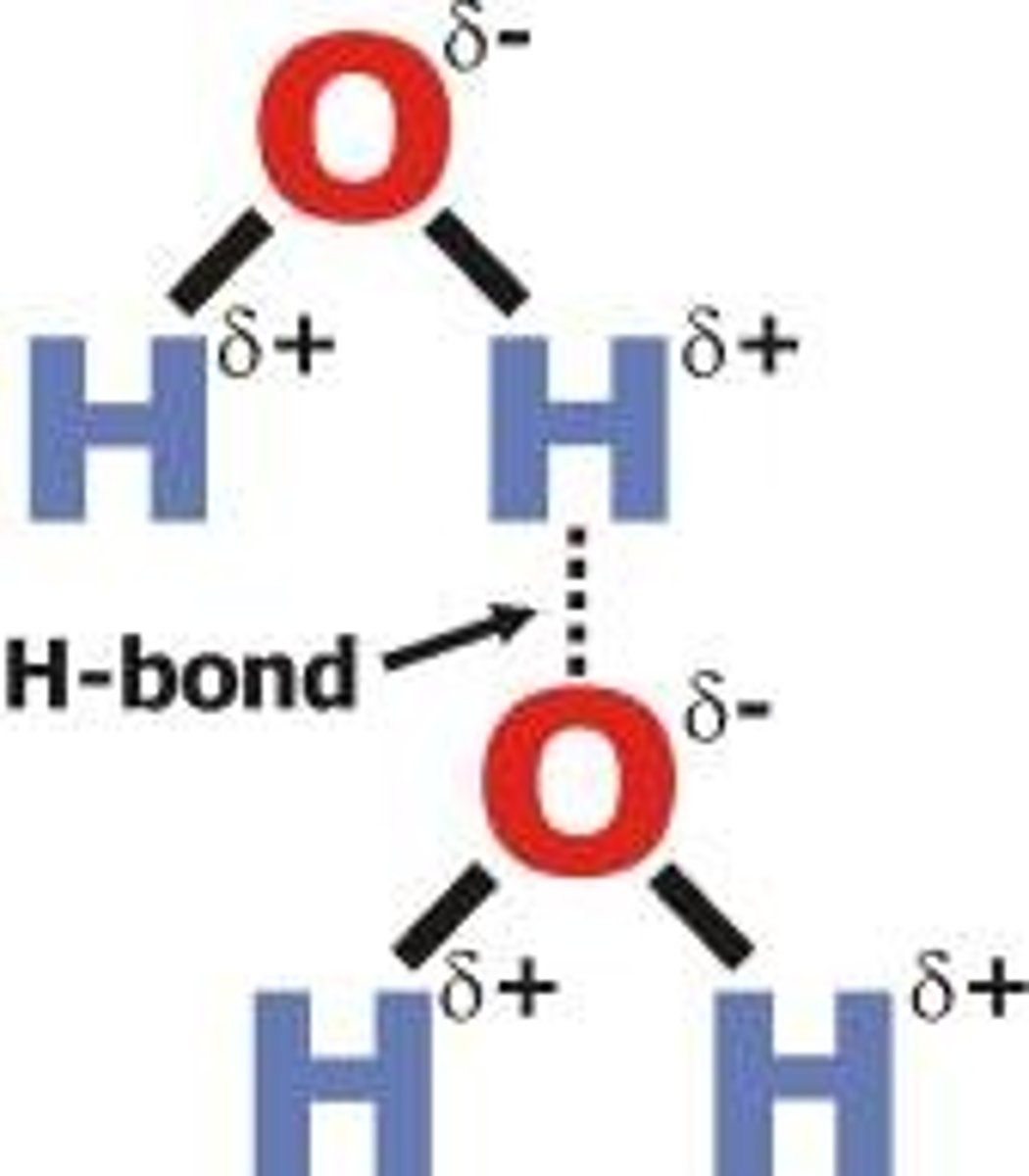
Hydrogen Bond
weak attraction between a hydrogen atom that is part of a polar covalent bond and another electronegative atom like oxygen or nitrogen
Adhesion
attraction between molecules of different substances (like water and glass)
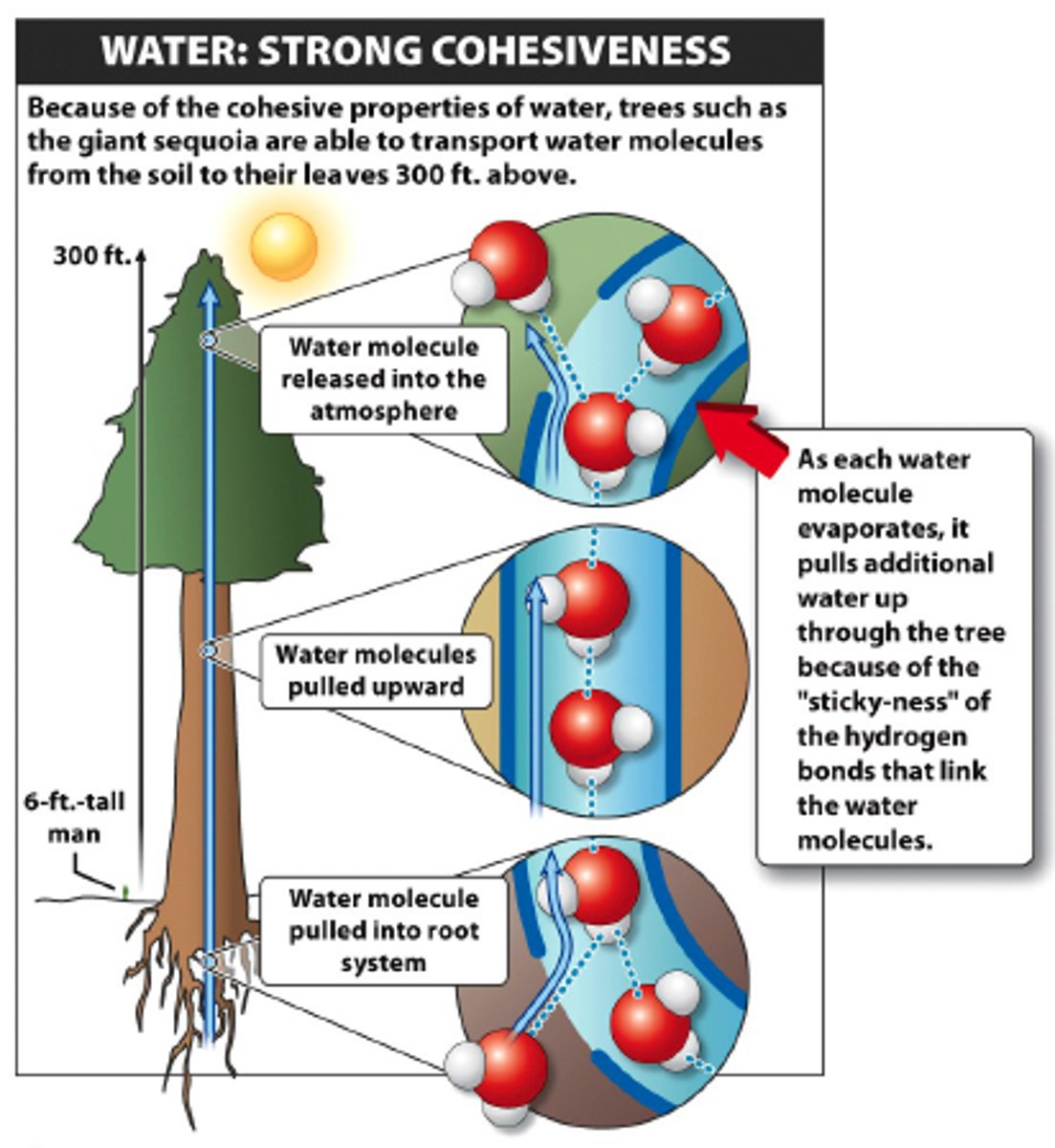
Cohesion
an attraction between molecules of the same substance (like water and water creating surface tension)
Polar
Molecule with partial charges due to the presence of an electronegative atom like oxygen, nitrogen, phosphorous, or sulfur. Mixes with water.
pH
a measure of hydrogen ion (H+) concentration; as pH increases, H+ decreases
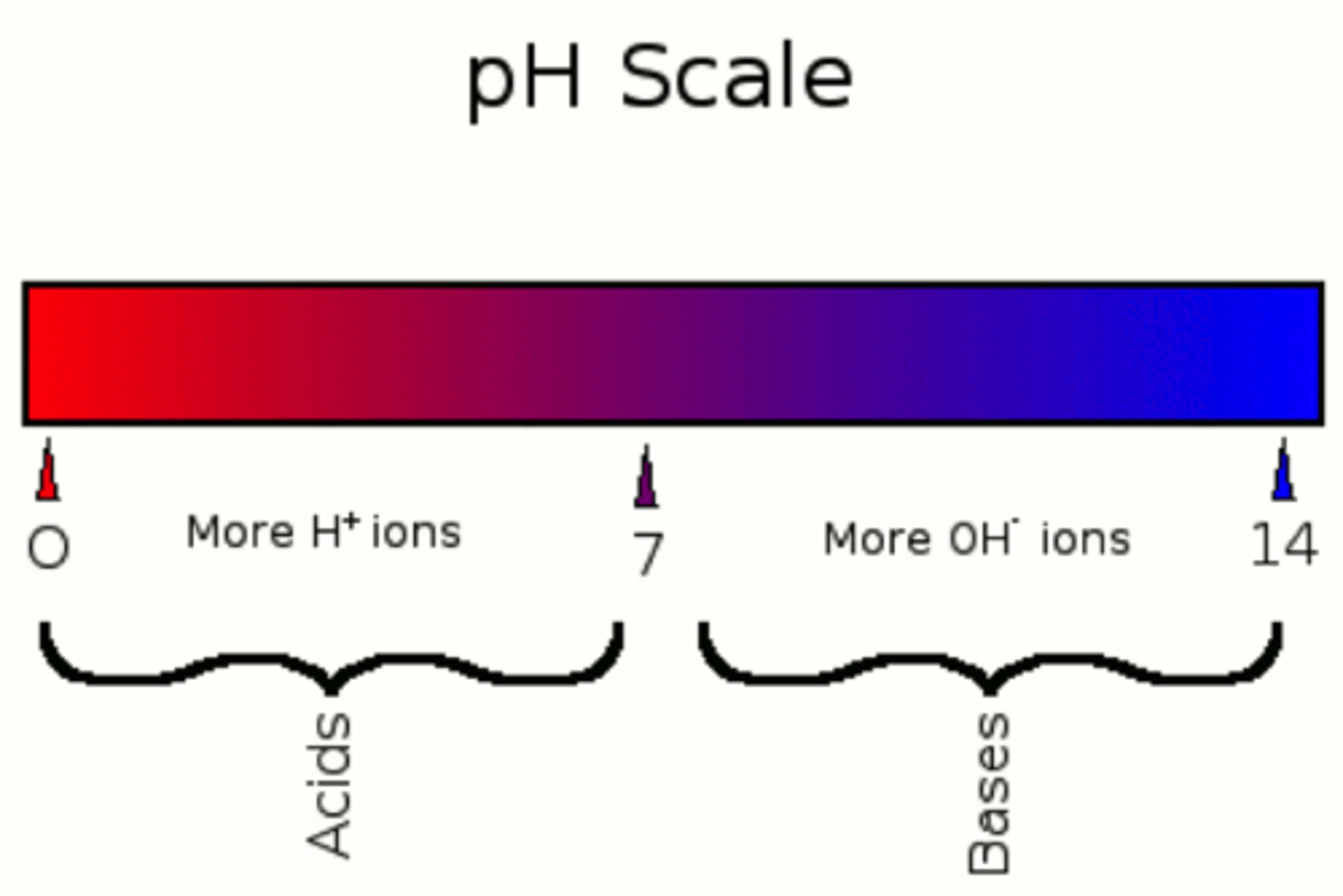
Acids
substances that release hydrogen ions (H+) when dissolved in water; cause pH to decrease
Base
A substance that decreases the hydrogen ion concentration in a solution, while increasing the hydroxide ion (OH-) concentration.
Buffer
weak acid or base that can react with strong acids or bases to help prevent sharp, sudden changes in pH
Ion
An atom or group of atoms that has a positive or negative charge due to the gain or loss of electrons.
Ionic bond
A chemical bond resulting from the attraction between oppositely charged ions.
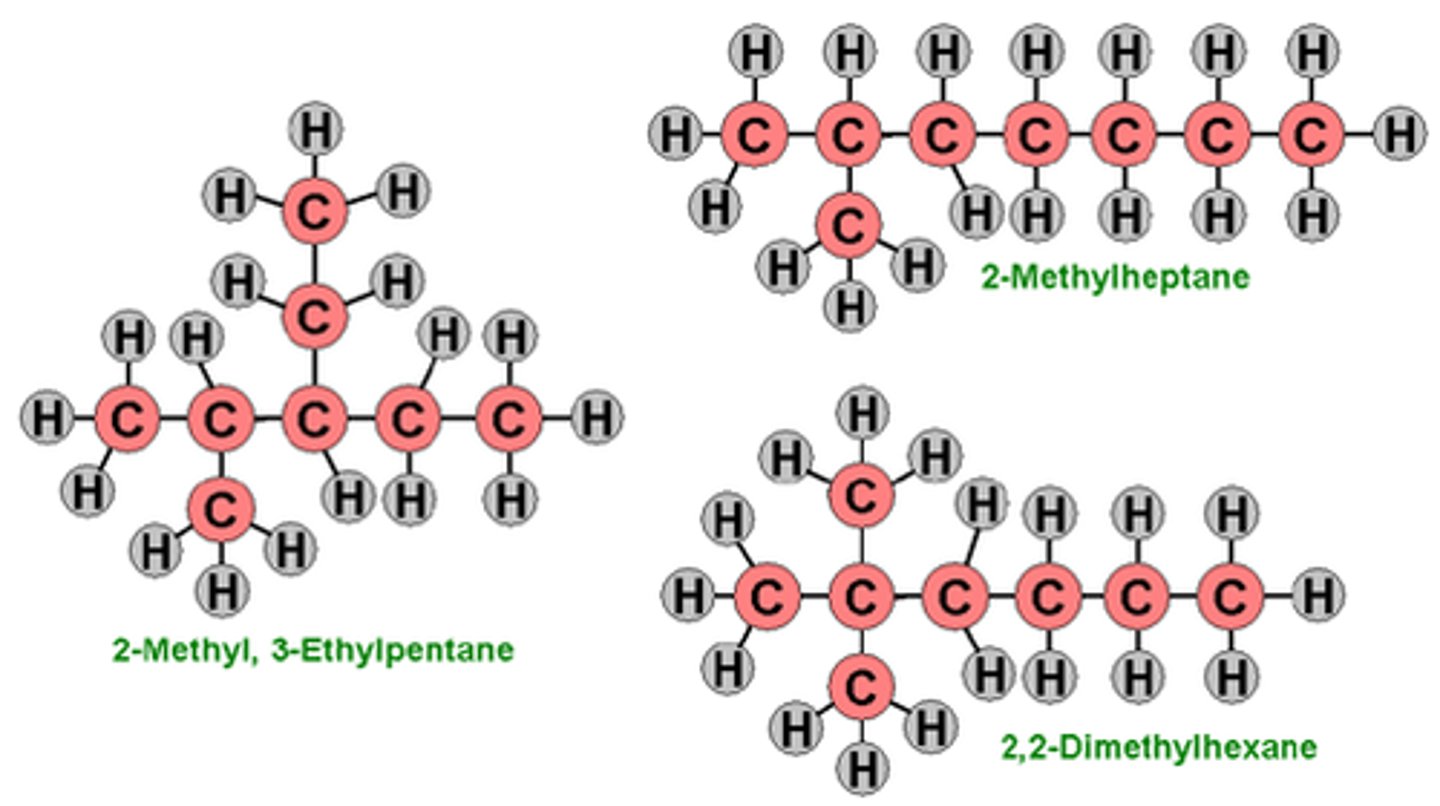
Isomers
Compounds with the same formula but different structures.
Dehydration Synthesis (Condensation)
A chemical reaction in which two molecules covalently bond to each other with the removal of a water molecule.
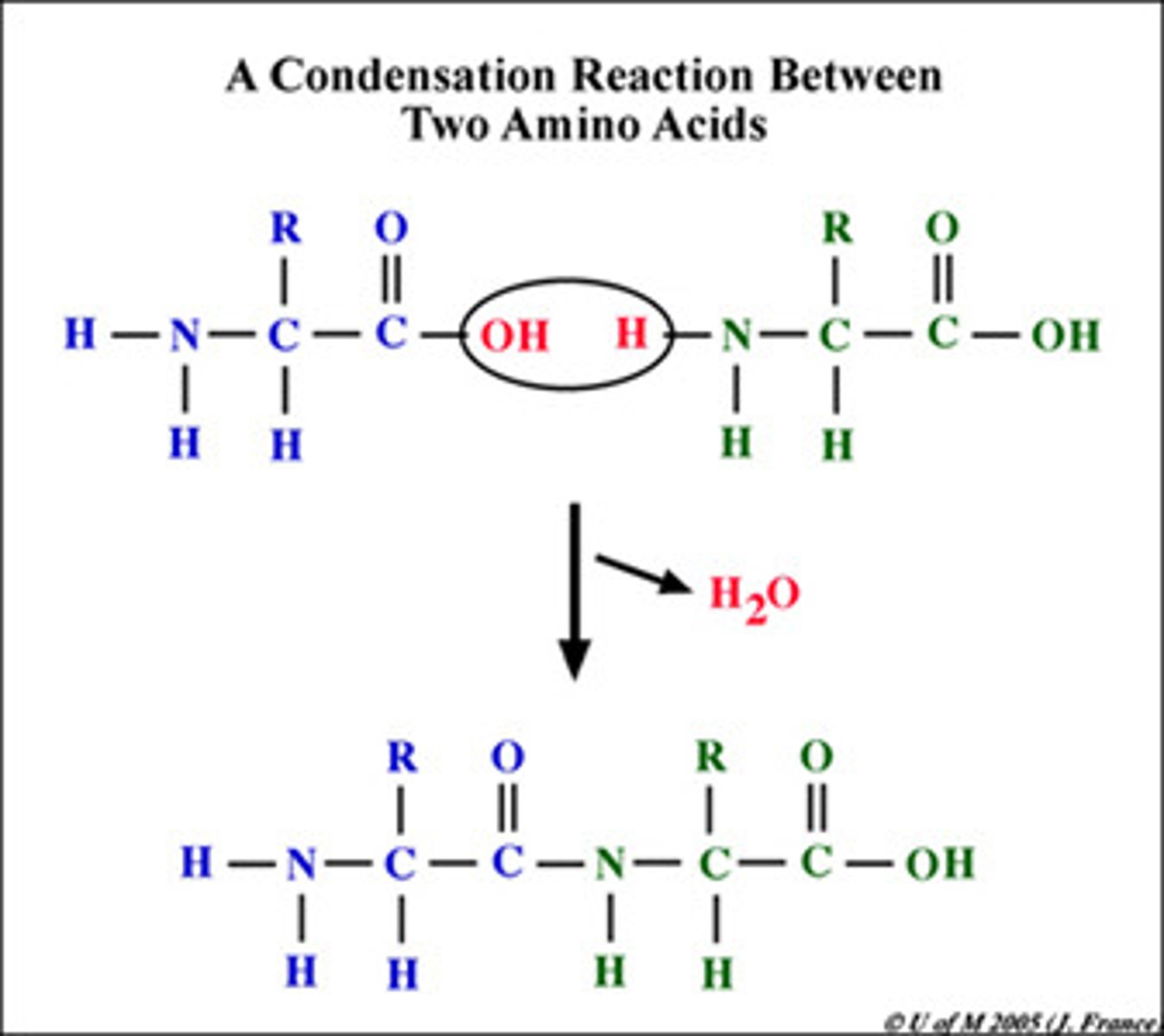
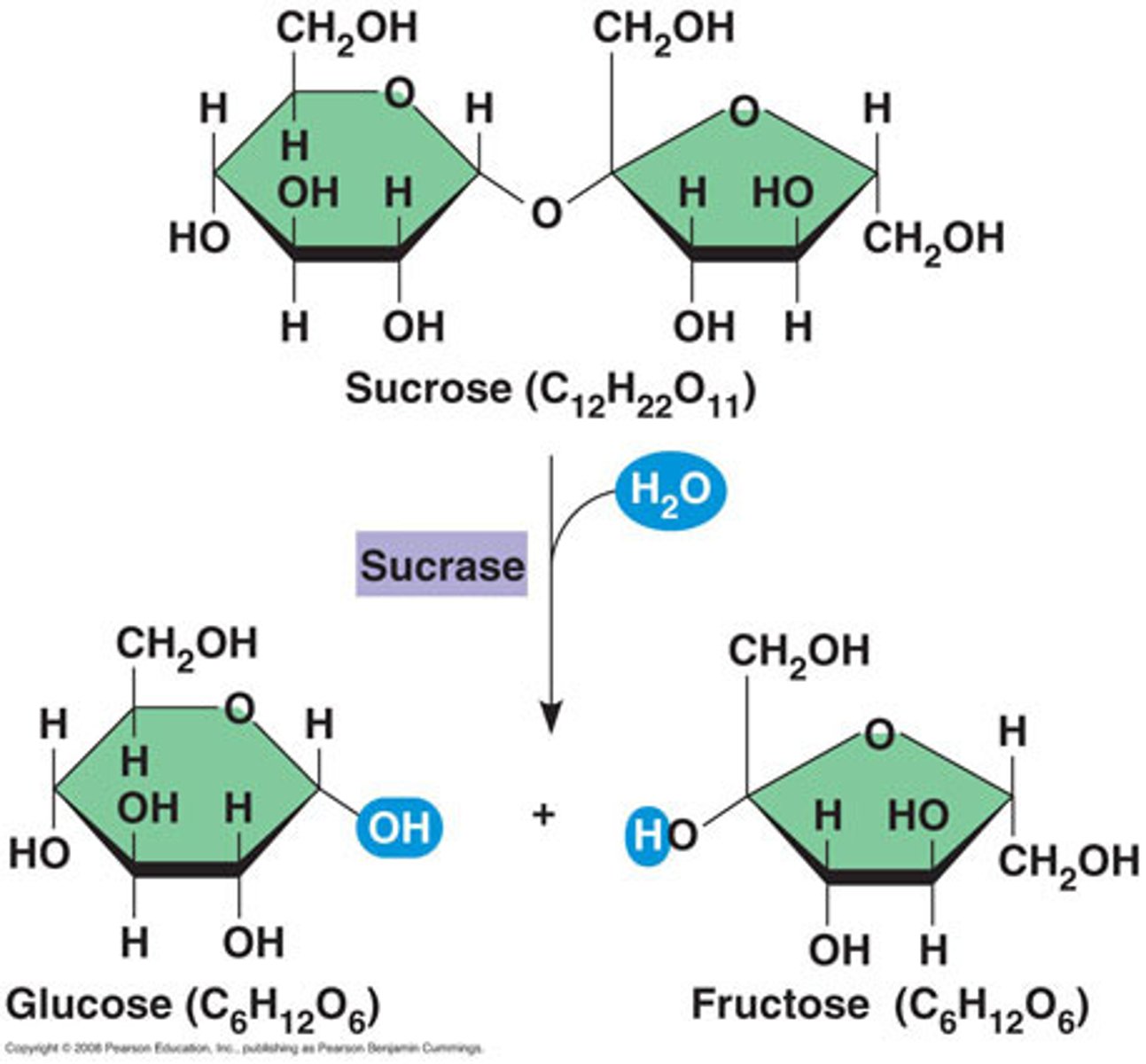
Hydrolysis
A chemical process that splits a molecule by adding water.
Monomer
small chemical unit that makes up a polymer
Polymer
molecules composed of many monomers; makes up macromolecules
Hydrophilic
water loving, polar molecule
Hydrophobic
water hating, nonpolar molecule
Covalent Bond
A chemical bond formed when two atoms share electrons
Carbon
atom with 4 valence electrons that forms the backbone of all organic molecules
Nonpolar
Molecules like oil in which all atoms have similar electronegativity and the electron distribution is equal between atoms in a covalent bond (i.e. C-H, C-C, O-O)
Transpiration
The process where plants absorb water through the roots and then give off water vapor through pores in their leaves
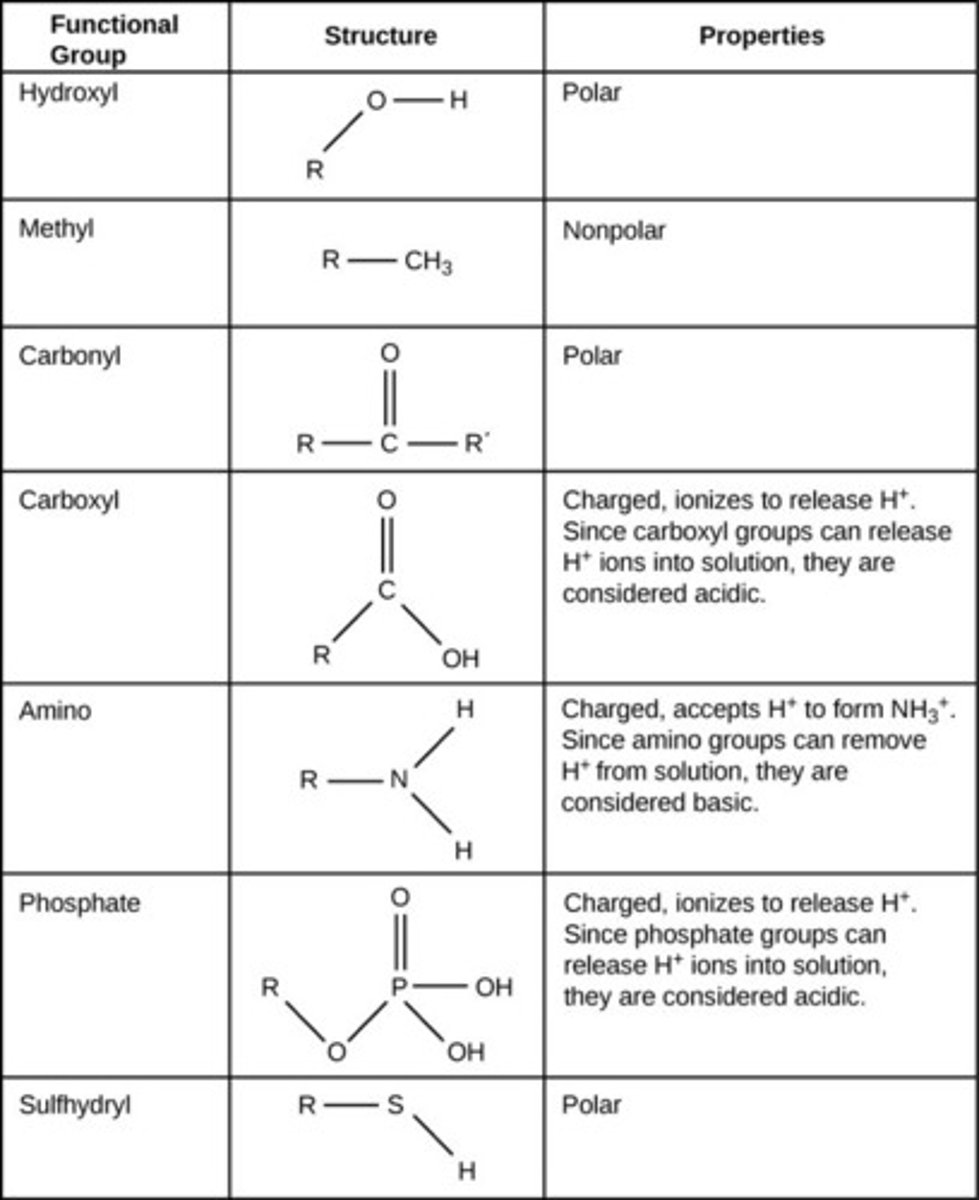
Functional Groups
the components of organic molecules that are most commonly involved in chemical reactions
Spontaneous Reaction
occurs naturally and favors the formation of products at the specified conditions without a constant input of outside energy
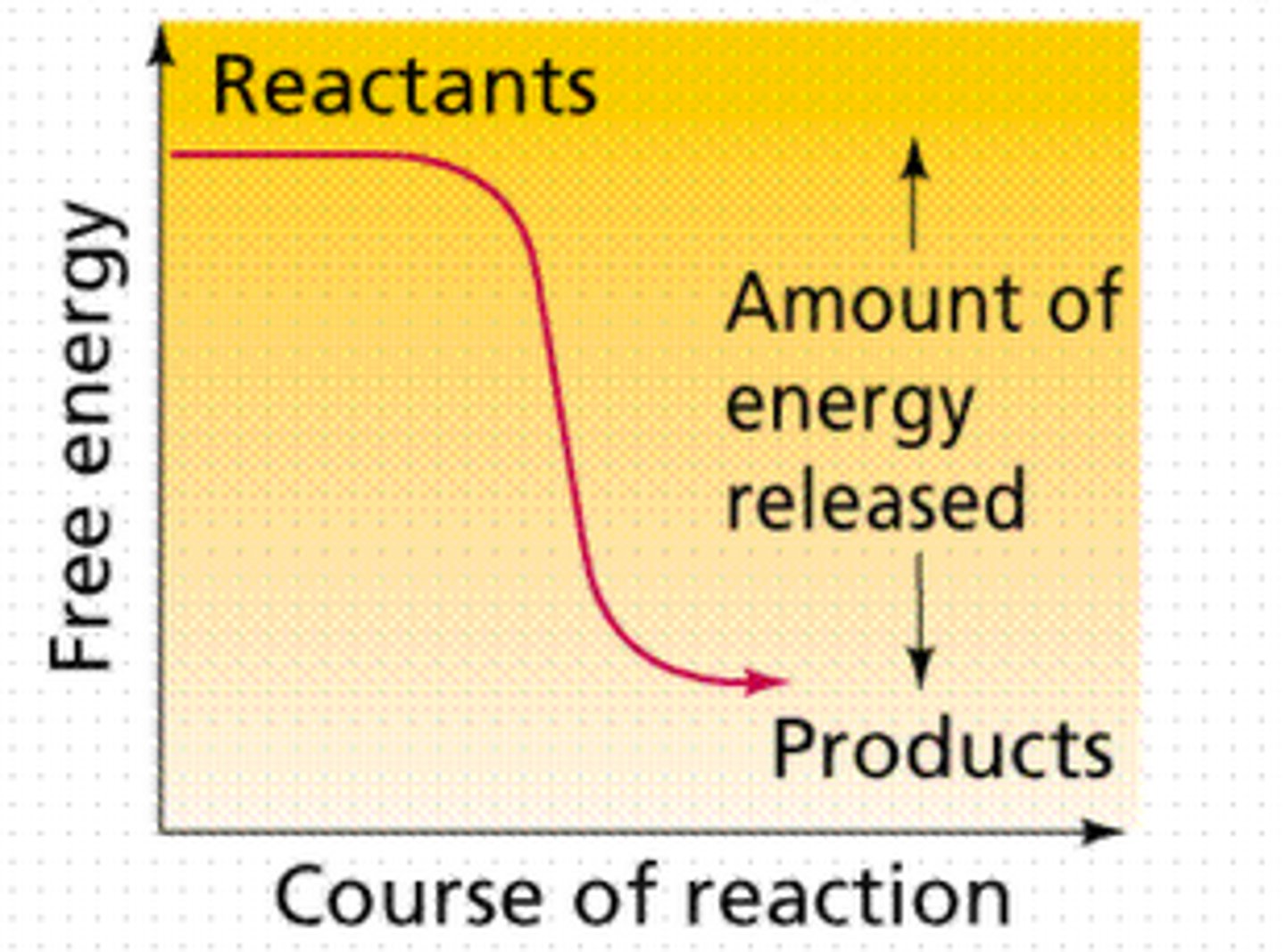
Second Law of Thermodynamics
When energy is changed from one form to another, some useful energy is always degraded into lower quality energy (usually heat), which increases the entropy of the universe
Surface Tension
A measure of how difficult it is to stretch or break the surface of a liquid

Specific Heat
The amount of energy required to raise the temperature of 1 gram of a substance by 1 degree celcius; water has a high specific heat (it requires a lot of energy to change its temperature)
Solvent
A liquid substance capable of dissolving other substances; water is often called the "universal solvent" but only dissolves other POLAR substances
First Law of Thermodynamics
Energy can be transferred and transformed, but it cannot be created or destroyed.
Potential Energy
Energy that is stored and held in readiness (i.e. in a chemical bond)
Kinetic Energy
energy of motion (i.e. thermal energy, the movement of atoms due to heat)