tubular reabsorption and secretion
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
tubular reabsorption
reclaiming those important solutes that were lost during filtration
virtually everything gets reabsorbed; glucose, amino acids, 99% water, etc.
transcellular route
molecules move through the apical surface into the cell and out through the basement membrane or lateral intercellular space to reach the interstitial fluid
requires facilitated diffusion (transport proteins and active transport)
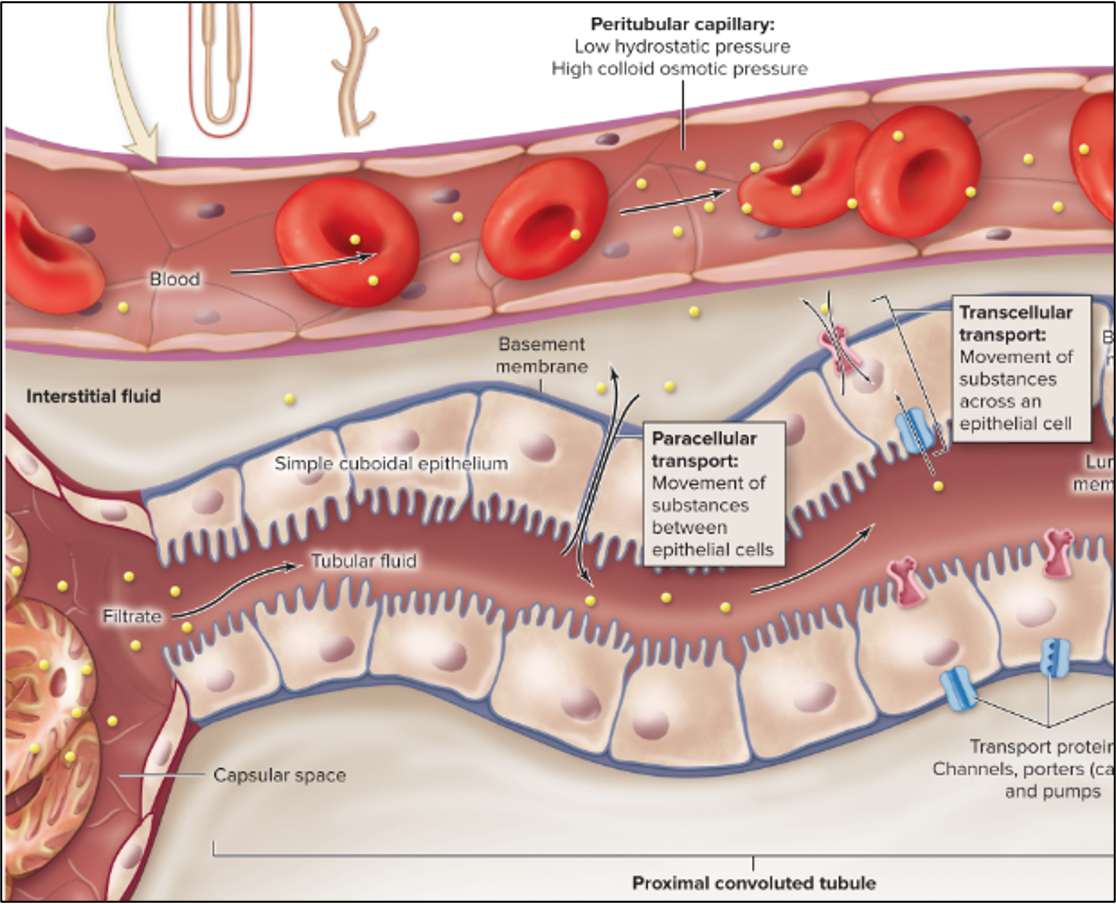
paracellular route
molecules move between cells into the interstitial fluid
tight junctions become leaky in PCT but tight enough to prevent pathogens
solvent drag moves solutes through the leaky junctions
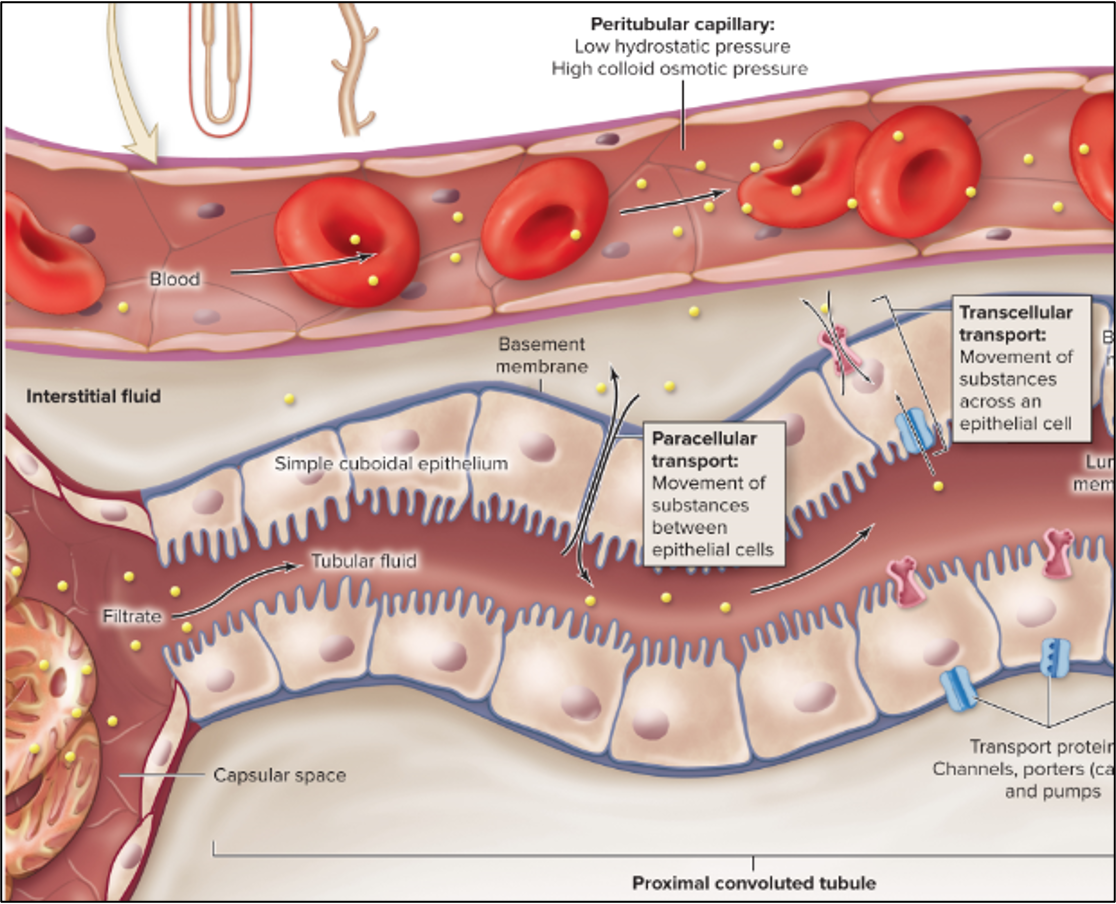
peritubular capillaries
uptake the molecules
low hydrostatic, high colloid osmotic
concepts of tubular reabsorption
Na+ is the most important ion in EC fluid. Reabsorption of Na+ closely linked to transport of many other ions and molecules. Na+ intake is equal to Na+ excreted
reabsorption of nutrients (glucose + AA) is directly linked to reabsorption of Na+
reabsorption of sodium sets up osmotic gradient in proximal convoluted tubule. Where Na+ goes, water flows
K+ intake is equal to K+ excreted and also influenced by Na+
water balance is regulated in distal convoluted tubule and collecting duct (under influence of ADH)
Na+ is the most important ion in EC fluid. Reabsorption of Na+ closely linked to transport of many other ions and molecules
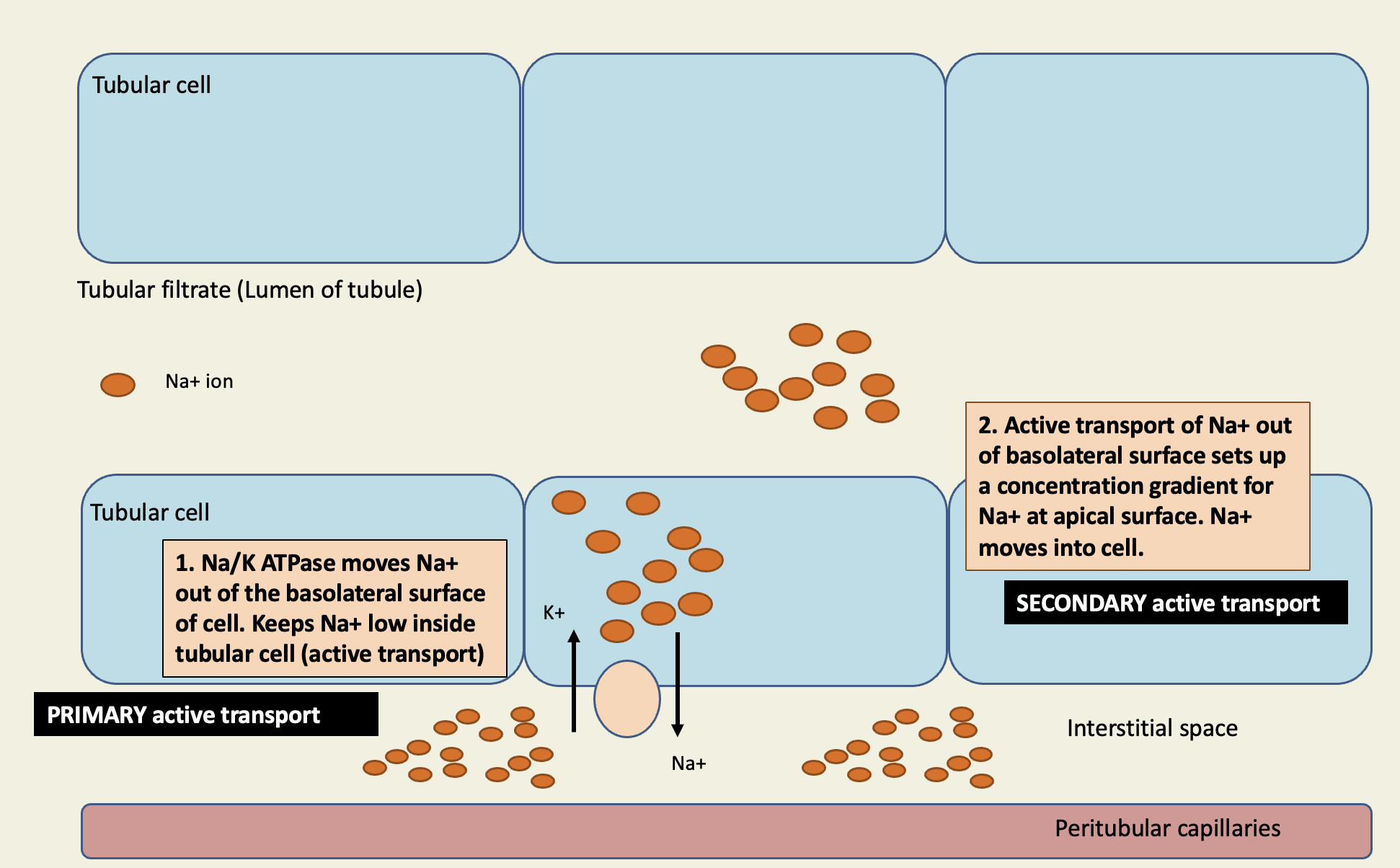
equal concentration of Na+ in filtrate and tubular cell
Na/K ATPase moves Na+ out of basolateral surface of cell (primary active transport)
makes Na+ low inside tubulate cell
active transport of Na+ out of basolateral surface sets up a concentration gradient for Na+ at apical surface. Na+ moves into cell via proteins (secondary active transport)
2/3 of Na+ reabsorbed in PCT through Na+/H+ exchanger
active transport of Na+ at basolateral surface allows exchange protein to function
antiporter; H+ goes into lumen of tubule, Na+ goes into tubular cell
The reabsorption of nutrients (like glucose and AA) is directly linked to reabsorption of Na+
Sodium is pumped out at basolateral surface + concentration gradient developed
Na+ moves into cell through transport proteins
symporters move Na+ down gradient and glucose against gradient (secondary active transport)
2Na+ for 1 glucose
Tmax- max rate of reabsorption allowed
reached when molecules outnumber transporters
diabetes mellitus
in normal kidney, there’s always enough transport proteins for glucose
insulin not produced or not effective at stimulating glucose reabsorption
too much glucose in blood → too much glucose in filtrate → overwhelms glucose transporters
excreted in urine
Where are nutrients reabsorbed
67% in proximal convoluted tubule (phosphate, nutrients, Cl-)
25% in thick ascending limb of nephron loop (K+, Cl-, Na+) (reabsorption of Na+ is load dependent)
5% in distal convoluted tubule (Cl-, Na+)
3% in collecting duct (Na+ reabsorption is load dependent)
what happens when there’s an increase in Na+ intake
ECF volume and blood volume increase
sympathetic activity decreases
ANP increases
capillary osmotic pressure decrease
renin decrease
Na+ excretion increases
what happens when Na+ intake decreases
ECF volume and blood volume decrease
sympathetic activity increases
ANP decreases
capillary osmotic pressure increase
renin increase
Na+ excretion decrease
the reabsorption of sodium sets up an osmotic gradient in the proximal convoluted tubule. where Na+ goes, water flows: through transport proteins in apical membrane of the tubule cells
concentration gradient for Na+ at apical surface
water reabsorption in PCT is obligate water reabsorption
water MUST follow sodium (isosmotic)
65% water reabsorption occurs in PCT
passive transport of water
water moves via aquaporins and solvent drag
aquaporins
integral protein that moves water
solvent drag
paracellular transport; brings other molecules and ions with the water
potassium balance is continually modified based on dietary intake. K+ intake is equal to K+ secreted/excreted
most K+ in ICF, so if any changes occur where K+ leaves cell and enters ECF, small changes have large effects on K+ levels
internal K+ balance modified by:
insulin- stimulates K+ uptake into cells via Na+/K+ ATPase after a meal or when K+ levels in ECF rise
epinephrine- exercise causes tremendous release of K+ ions into ECF
K+ is intrinsic vasodilator in skeletal muscle
exercises also results in epinephrine secretion which stimulates K+ uptake into cells
H+ ion concentration- decrease in H+ ion concentration in ECF results in K+ movement into cells (Na+/H+ pump)
how is K+ regulated in the nephron (K+ in = K+ out)
most K+ reabsorbed in PCT and nephron loop (thick ascending limb)
K+ dragged by water in PCT
actively transported into cells in TAL (Na+K+2 Cl- pump)
DCT and collecting duct fine tune K+
reabsorption by tubule cells or intercalated cells
secretion by principal cells
levels of Na+ in filtrate
more Na+ to DCT + collecting duct, more Na+ that is reabsorbed
the more Na+ reabsorbed into tubular cells = more K+ secreted into tubular fluid
aldosterone
stimulates Na+ uptake in principal cells
in response to increase in Na+ uptake, principal cells secrete K+
hyperkalemia
increase in blood K+ concentration; repolarization of ventricles impacted; can lead to v-fib
hypokalemia
decrease in blood K+ concentration; atrial and ventricular arrhythmias
water balance is regulated in the distal convoluted tubule and collecting duct
deprived of H2O, increased plasma osmolarity
osmoreceptors stimulated, ADH secretion increased, thirst increased, increased aquaporins, increased H2O reabsorption, increased urine osmolarity and decreased urine volume, plasma osmolarity down
increased H2O, decreased plasma osmolarity
inhibits osmoreceptors, ADH secretion decreased, thirst decreased, decreased aquaporins, decreased urine osmolarity and increased urine volume, increased plasma osmolarity
countercurrent multiplier
generates salt concentration gradient within interstitial fluid (nephron loop)
more concentrated deeper into medulla
occurs in descending and ascending limb
H2O balance in collecting duct is dependent on salt concentration
descending limb permeable to water but not salt
ascending limb permeable to salt but not water
combination of two allows water to be reabsorbed into interstitial fluid
countercurrent exchange
maintains salt concentration gradient within interstitial fluid (vasa recta)
blood vessels exchange water and salts as blood passes by nephron loop
water balance is regulated by several hormones in the collecting duct
aldosterone: stimulates Na+ reabsorption and water follows (increases Na+ channels)
ADH: stimulates aquaporin synthesis
reabsorption
the movement of molecules from the tubular filtrate through the cells of the tubule and into the peritubular capillaries
proximal convoluted tubule- ions, all nutrients, lipid soluble solutes, water, wastes
nephron loop
thin segment- water by osmosis
thick segment- Na+, Cl-, K+ ions by secondary active transport
distal convoluted tubule- Na+, Cl- (primary active) via aldosterone, Ca+ via PTH
collecting duct- Na+ (primary active) via aldosterone, water, osmosis via ADH
tubular secretion fine tunes the filtrate composition
movement of substances from capillaries to the filtrate
reverse of reabsorption
need to correct some of the reabsorption that has happened
remove drugs
remove wastes (40-50% urea)
remove excess K+ (aldosterone driven secretion)
maintaining optimal blood pH
collecting duct is critical in maintain acid/base balance of blood
increased H+
type A cells secrete H+ into filtrate
Type A cells release HCO3- into blood to bind to H+ to increase pH
decreased H+
type B cells release H+ into blood
type B cells secrete HCO3- into filtrate
diuretics
medications that promote water and salt removal from body in urine
carbonic anhydrase inhibitors (acetazolamide)
diuretic that decreases Na+ reabsorption in renal tubule (PCT)
mild diuretics- thick ascending loop is load dependent so a decrease in Na+ reabsorption in PCT results in increased Na+ reabsorption in nephron loop
loop diuretics (Furosemide, Lasix)
affect K+, 2 Cl-, Na+ pump (ascending limbs)
affect H2O balance because countercurrent is affected and changes osmotic gradient for H2O balance
thiazide diuretics (indapamide)
affects Cl-, Na+ pump in DCT
kidney stones
hardened or crystallized deposits formed in kidney
renal pelvis through bladder
most passed but >5 mm can block ureter
pain from peristaltic contractions against stone
dehydration, high salt/protein diets, obesity
symptoms:
pain in back to groin
peristaltic generation of pain
frequent urination
classification
calcium
struvite
uric acid
glomerulonephritis
inflammation contributes to a variety of disorders within the kidneys
degradation of basement membrane
hypertrophy and proliferation of cells
complement mediated cell lysis
structural damage to glomerular filtration membrane reduces surface area
end-stage renal disease
characterized by extreme inhibition of normal kidney function; result of progressive CKD
fluid imbalances
metabolic acidosis
anemia
urea retention in blood
untreated patients can become comatose and die
diagnosed by:
complete physical history
blood tests
urinalysis
treated with dialysis or kidney transplant
filtering blood across artificial glomerular filtration membrane
hemodialysis
blood drained from arm and replaced back into arm
dialysate and blood flow in opposite directions for better exchange
peritoneal dialysis
dialysate fills abdomen behind peritoneum and blood is filtered across directly cross peritoneum
waste fluid removed every 4-6 hrs and replaced with fresh dialysate
not as efficient as hemodialysis, higher risk of peritoneal infection
allows for activity during dialysis dwell time
kidney transplant
new kidney transplanted in anterior portion of abdominopelvic region
damaged or diseased kidneys usually left in place
ABO blood typing, HLA matching