Alkanes
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
Alkane definition
Saturated hydrocarbons containing only single C–C bonds
General formula
CₙH₂ₙ₊₂
Bond type
Single covalent bonds only
Saturated meaning
No double or triple bonds; cannot undergo addition reactions
Homologous series
Alkanes form a homologous series with similar properties
Trend in boiling point
Increases as chain length increases
Trend in flammability
Decreases as chain length increases
Trend in viscosity
Increases as chain length increases
Solubility
Insoluble in water (non‑polar)
Reactivity
Generally unreactive due to strong C–C and C–H bonds
Main reactions
Combustion and substitution
Complete combustion
Alkane + oxygen → CO₂ + H₂O
Incomplete combustion
Alkane + oxygen → CO + C + H₂O
Substitution reaction
Alkane + halogen → haloalkane + hydrogen halide
Substitution condition
Requires UV light
Example substitution
Methane + chlorine → chloromethane + HCl
Uses of alkanes
Fuels (methane, propane, butane, petrol)
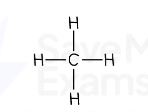
Methane formula
CH₄
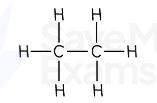
Ethane formula
C₂H₆
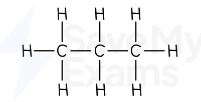
Propane formula
C₃H₈
Butane formula
C₄H₁₀
Key idea
Alkanes are saturated, fairly unreactive hydrocarbons used mainly as fuels
methane displayed formula
ethane displayed formula
propane displayed formula
butane displayed formula
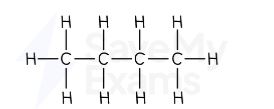
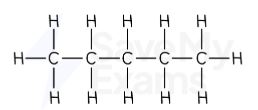
pentane displayed formula