AS inorganic
1/83
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
84 Terms
describe how we can use dilute NaOH to identify group 2 cations:
add 6 drops of dilute NaOH solution to test tube containing metal ion solution (e.g. magnesium sulfate, Mg2+ etc.) and observe precipitate (if any) formed
keep adding dilute NaOH until it is in excess - record any changes you see
give 3 ways we can identify group 2 metal cations:
flame test (not in RP4)
by adding dilute NaOH
by adding H2SO4
what is seen for each of the group 2 metal cations when dilute NaOH is added?
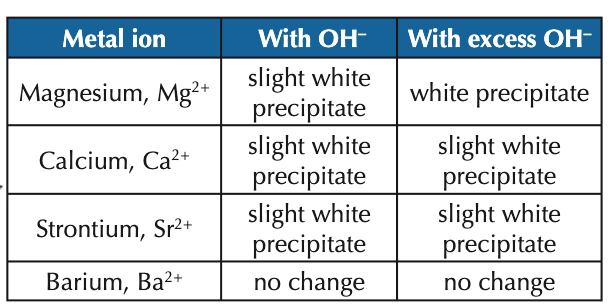
what would be the initial observation for a group 2 metal compound?
colourless solution
how can we test for ammonium (NH4+) ions?
add substance you are testing to test tube
add NaOH solution and shake
warm gently using a water bath
ammonia should be released - test fumes released by holding damp litmus paper at mouth of test tube
if ammonium ions present, damp litmus paper turns from red → blue
give the ionic eqn for the ammonium ion test:
NH4+ (aq) + OH- (aq) → NH3 (g) + H2O (l)
describe how we can use H2SO4 to identify group 2 metal cations:
add 10 drops of H2SO4 solution to test tube containing metal ion solution (e.g. magnesium sulfate, Mg2+ etc.) and observe precipitate (if any) formed
keep adding H2SO4 until it is in excess - record any changes you see
what is seen for each of the group 2 metal ions when H2SO4 is added?
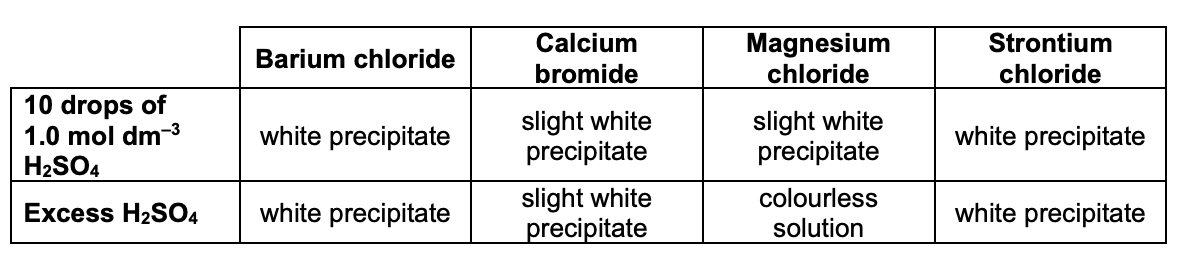
give 2 ways we can test for OH- ions:
using litmus paper
using ammonia solution and litmus paper
how can we test for OH- ions with just red litmus paper?
dip a piece of red litmus paper into the solution
if OH- ions are present, the paper will turn blue
how can we confirm a solution is ammonia based on the test for OH- ions?
take 5 drops of solution you are testing and place on filter paper
place this inside a petri dish w/ a lid
dampen a piece of red litmus paper w/ deionised water and place on other side of petri dish
replace lid and observe over a few minutes - if OH- ions present, litmus paper should turn blue as ammonia vapours produced
how does the test for OH- ions and litmus paper work?
we add deionised water as OH- ions form when NH3 comes into contact w/ water
this turns red litmus paper blue as ammonia solution vapours are alkaline
how can we test for CO32- ions in aqueous solution?
add 2 cm³ Ca(OH)2 (limewater) to a test tube
to a different test tube, add 3 cm³ substance you are testing and an equal volume of dilute HCl
immediately place a delivery tube from this to the limewater test tube
if CO32- ions present, limewater goes cloudy
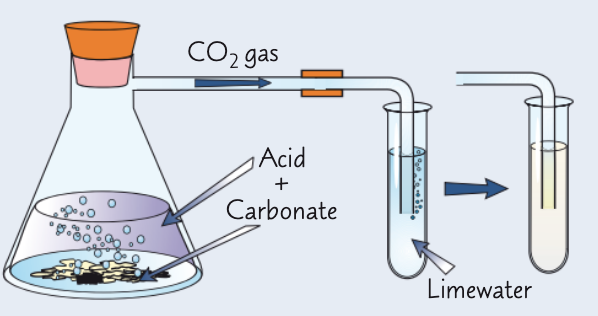
explain how HCl can help detect CO32- ions and give the eqn:
limewater turns cloudy if carbonate ions present as CO2 is produced in the reaction w/ dilute HCl (H+ ions)
CO32-(aq) + 2H+ (aq) → CO2 (g) + H2O (l)
how can we test for sulfate ions in aqueous solution?
add dilute HCl and acidified BaCl2
white precipitate of BaSO4 forms if SO42- ions present
(BaCl2 acidified by HCl but is in MS)
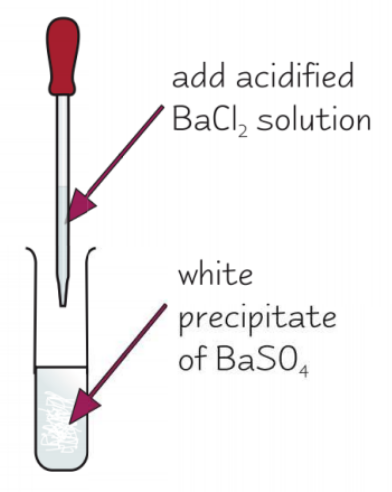
give the ionic eqn for the test for sulfate ions in aqueous solution:
Ba2+ (aq) + SO42- (aq) → BaSO4 (s)
how can we test for halide ions in aqueous solution?
add 10 drops of substance you are testing to a clean, dry test tube
add approx 5 drops of dilute nitric acid and shake
add 10 drops silver nitrate solution and record observations
add dilute ammonia (or ammonia solution if testing for I- ions and if so, work in a fume cupboard) and record further observations
why do we add nitric acid during the test for halide ions?
to remove CO32- and OH- ions, which would also form precipitates and interfere w/ the test
give the general eqn for the reaction of halide ions w/ silver nitrate:
Ag+ (aq) + X- (aq) → AgX (s)
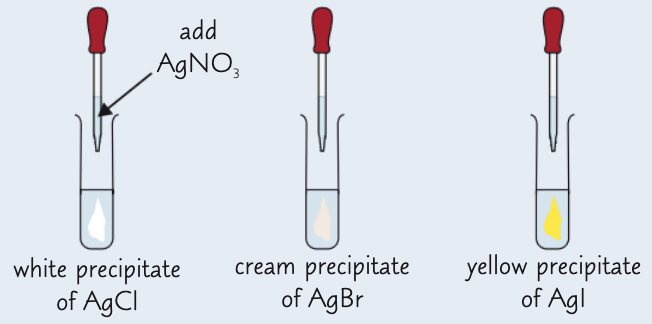
give the results of the silver nitrate test:
AgCl - white ppt
AgBr - cream ppt
AgI - yellow ppt
give the results of the silver nitrate halide ions test when aqueous ammonia/ammonia solution is added:
AgCl - redissolves
AgBr - redissolves slowly and needs a lot of ammonia
AgI - does not dissolve
how can we test for halide ions in solid salts?
carry out in a fume cupboard and wear gloves:
place a small spatula of the solid you are testing into a clean, dry test tube
slowly add a few drops of concentrated H2SO4 and record observations
test gas w/ moist blue litmus paper and record observations
(repeat w/ acidified K2Cr2O7 / lead (II) nitrate solution)
give the observations for the tests for halide ions in solid salts:

(not part of the practical but useful!) how can we test for all acids? what denotes a +ve result?
add Na2CO3
+ve result = effervescence - as CO2 produced
(not in RP4 but useful to know) identify a reagent/test that could be used to distinguish between aqueous solns of SO2 and SO3 w/ the same concs:
use universal indicator:
SO2: orange-red (higher pH)
SO3: red (lower pH)
how does the reactivity of the group 2 elements w/ water change going down the group?
increases

does Be react w/ water? if so, how? + give the eqn
no :(
does Mg react w/ water? if so, how? + give the eqn
reacts very slowly w/ water but readily w/ steam
water: Mg + 2H2O (l) → Mg(OH)2 + H2
steam: Mg + H2O (g) → MgO + H2
state two observations you would make when magnesium reacts w/ steam:
bright white light
white solid (NOT ppt)
does Ca react w/ water? if so, how? + give the eqn
reacts steadily w/ water
Ca + 2H2O → Ca(OH)2 + H2
does Sr react w/ water? if so, how? + give the eqn
reacts fairly quickly w/ water
Sr + 2H2O → Sr(OH)2 + H2
does Ba react w/ water? if so, how? + give the eqn
reacts rapidly w/ water
Ba + 2H2O → Ba(OH)2 + H2
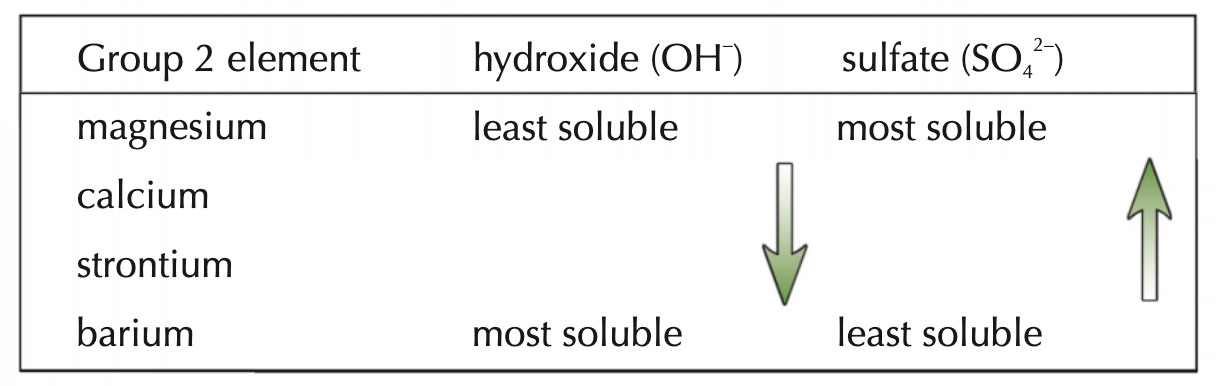
how does solubility of group 2 compounds change down the group?
compounds of group 2 w/ singly charged anions - OH- - increase in solubility down the group
compounds of group 2 w/ doubly charged anions - SO42- - decrease in solubility down the group
is BaSO4 soluble in water? why is this significant?
not soluble in water
precipitates out of solution so forms a white precipitate when BaCl2 and dilute HCl are added if SO42- ions are present
summarise the test for sulfate ions:
add dilute HCl and BaCl2
white precipitate of BaSO4 forms if SO42- ions present
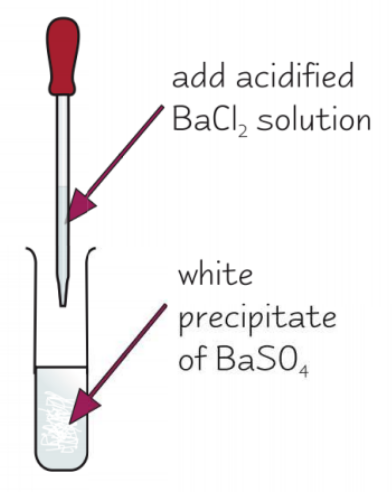
why is HCl added in the test for sulfate ions?
to remove carbonates and sulfites, as they would also produce a white precipitate
how is Ti extracted from its ore? + give the eqn
Mg is used to extract Ti from rutile
TiO2 is first converted to TiCl4, which is then reduced by Mg in a furnace at 1000oC
TiCl4 + 2Mg → Ti + 2MgCl2
name and describe a use of BaSO4:
barium meals:
BaSO4 is opaque to X-rays
patients swallow a barium meal (suspension of BaSO4) which coats the tissues so they show up on an X ray
BaSO4 is insoluble so passes through your body after
how is sulfur removed from flue gases? + give the eqns
flue gas desulfurisation:
powdered CaO or CaCO3 mixed w/ water to make an alkaline slurry
when flue gases mix w/ alkaline slurry, acidic SO2 gas reacts w/ calcium compounds to form CaSO3, which is harmless
CaO + 2H2O + SO2 → CaSO3 + 2H2O
CaCO3 + 2H2O + SO2 → CaSO3 + 2H2O + CO2
give 3 general uses of group 2 compounds:
to neutralise acids
antacids (indigestion remedies)
to neutralise acidic soils
give the general ionic eqn for the reaction of a group 2 metal w/ water:
X2+ (s) + 2H2O (l) → Ca2+ (aq) + 2OH- (aq) + H2 (g)
state the role of water when reacting w/ group 2 metals:
oxidising agent
explain why different observations are made when aq BaCl2 is added separately to aq MgSO4 and aq Mg(NO3)2:
BaSO4 is insoluble but Ba(NO3)2 is soluble
solutions of barium hydroxide are used in the titration of weak acids.
state why magnesium hydroxide solution could not be used for this purpose (1)
Mg(OH)2 is insoluble
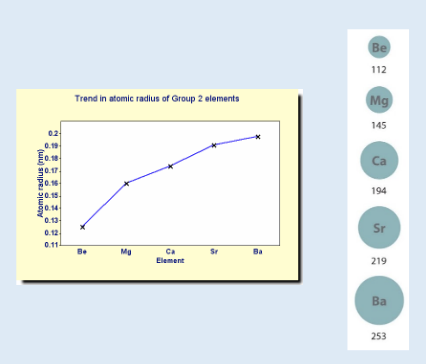
how does atomic radius change down group 2?
increases
as more e- shells are added
so size increases
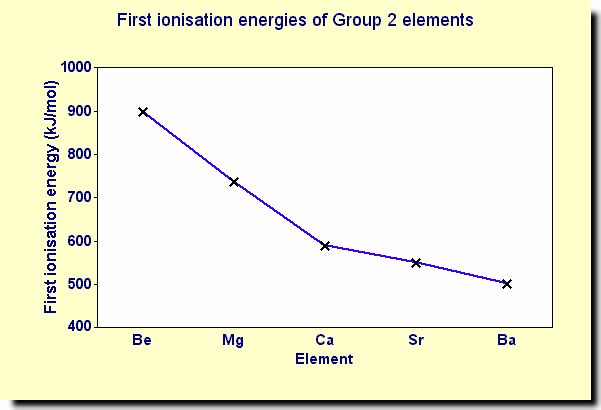
how does 1st IE change down group 2? why?
decreases
outer e- in higher E level / increase in shielding / atoms larger / more shells
∴ weaker attraction between ion and lost e-
(there is more +ve charge in the nucleus but this is overridden by the extra shells)
how does reactivity change down group 2? why?
increases
as 1st and 2nd IE decrease down the group - it is easier to lose e- and form 2+ ions as you go down
what is the general trend of mpt down group 2?
generally decreases
as the atoms (and ions) get bigger as you go down the group, delocalised e- are further from the +ve nuclei
so it takes less energy to break metallic bonds
Mg does not follow this trend (because of how its ions are arranged)
how reactive are the halogens in general?
fairly reactive
how does reactivity change down the group? why?
decreases down the group
as halogens want to gain e- to become -ve ions so reactivity decreases down the group as shielding increases down the group
how does the bpt of the halogens change down the group? why?
increases down the group
as molecules get larger
so VDWs between molecules get stronger
how does electronegativity change down group 7? why?
decreases down group
as nucleus is more shielded
so weaker attraction between nucleus and bonding pair
what is a disproportionation reaction?
reaction where a single element is both oxidised and reduced
give the full and ionic eqn for making bleach. what type of reaction is this? what observations are made?
2NaOH + Cl2 → NaClO + NaCl + H2O
ionic: 2OH− + Cl2 → Cl− + ClO− + H2O
disproportionation, yellow green gas Cl → colourless solution
give the full and ionic eqn for the reaction of chlorine w/ water. what type of reaction is this?
ionic: Cl2 + H2O ⇌ 2H+ + Cl- + ClO-
Cl2 + H2O ⇌ HCl + HClO
disproportionation
give the eqns for the reaction of chlorine w/ water IN SUNLIGHT:
2Cl2 + 2H2O ⇌ 4HCl + O2
2HClO → 2HCl + O2
why is Cl added to water, despite its limitations?
ClO- ions kill bacteria → safe to swim in/drink
only a small amount is added/benefits outweigh risks
give the strengths of adding Cl to water:
kills disease causing microorganisms
prevents algae growth
removes discolouration
give the limitations of adding Cl to water:
Cl gas is harmful
liquid Cl is corrosive
Cl can react w/ organic compounds in water to make chlorinated compounds (which can be cancerous)
describe a halogen displacement reaction and give the eqn for the displacement of Br by Cl2 in KBr:
a solution of a more reactive halogen can displace a less reactive halide from its salt
2KBr + Cl2 → 2KCl + Br2
how does oxidising ability of halide ions change down the group?
attraction between outer e- and nucleus decreases down group as ions get bigger
so oxidising ability decreases
give the eqns for halides reacting w/ concentrated sulfuric acid:
NaX + H2SO4 → NaHSO4 + HX
give the further reaction for some halides reducing sulfuric acid:
2HX + H2SO4 → X2 + SO2 + 2H2O
give the overall eqn for halides reacting w/ concentrated sulfuric acid and reducing it further:
2NaX + 3H2SO4 → 2NaHSO4 + X2 + SO2 + 2H2O
give the half eqns for some halides reducing sulfuric acid:
2X- → X2 + 2e-
H2SO4 + 2H+ +2e- → SO2 + 2H2O
name and explain the observations for the reactions of NaF and NaCl w/ H2SO4:
HF and HCl are not strong enough reducing agents to reduce the H2SO4 further
not a redox reaction - oxidation states of halogens and sulfur remain the same
observation: misty white fumes of HF / HCl
name and explain the observations for the reaction of NaBr w/ H2SO4:
HBr is a strong enough reducing agent to reduce H2SO4 further
second eqn = redox: Br oxidised (from -1 to 0) and S reduced from (+6 to +4)
observations: misty fumes of HBr, red-brown vapour Br2, choking fumes of SO2
what are the overall and ionic eqns for the overall reaction of sodium bromide w/ sulfuric acid, w/ the sulfuric acid being further reduced?
ionic: 2Br− + 3SO42- + 6H+ → 2HSO4- + Br2 + SO2 + 2H2O
overall: 2NaBr + 3H2SO4 → 2NaHSO4 + Br2 + SO2 + 2H2O
give and explain the eqns for NaI reacting w/ sulfuric acid:
NaI(s) + H2SO4 (l) → NaHSO4 (s) + HI(g)
HI can reduce H2SO4 further: 2HI(aq) + H2SO4 (l) → I2 (s) + SO2 (g) + 2H2O(l)
HI is a very strong reducing agent and can reduce the SO2 again to S and H2S: 6HI(g) + H2SO4(l) → 3I2(s) + S(s) + 4H2O(l)
6HI(g) + SO2(g)→ H2S(g) + 3I2(s) + 2H2O (l)
give and explain the observations for NaI reacting w/ sulfuric acid:
misty white fumes (HI)
purple vapour (I2)
yellow solid formed (S)
rotten egg smell (H2S)
black solid formed (I2)
choking fumes (SO2)
give the overall and overall ionic eqns for NaI reacting w/ H2SO4 and the observations:
overall: 8NaI + 9H2SO4 → 4I2 + 8NaHSO4 + H2S + 4H2O
ionic: 8I− + SO42- + 10H+ → 4I2 + H2S + 4H2O
observations: purple vapour released, black solid formed and a rotten egg smell
how can we test for halide ions in aqueous solution?
add 10 drops of substance you are testing to a clean, dry test tube
add approx 5 drops of dilute nitric acid and shake
add 10 drops silver nitrate solution and record observations
add dilute then concentrated ammonia and record further observations
give the general eqn for the reaction of halide ions w/ silver nitrate:
Ag+ (aq) + X- (aq) → AgX (s)
give the results of the silver nitrate test:
AgCl - white ppt
AgBr - cream ppt
AgI - yellow ppt
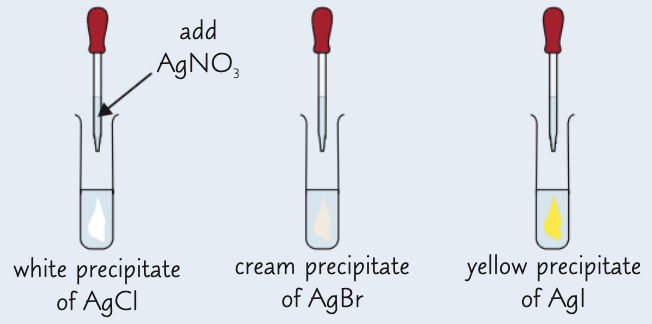
give the results for the silver halide ppts dissolving in ammonia:
AgCl - dissolves in dilute ammonia to form a colourless solution
AgBr - dissolves in concentrated ammonia to form a colourless solution
AgI - does not dissolve
how do we classify elements? how is this determined?
according to their block - s, p, d or f
block is determined by proton no. (or last orbital in e- configuration)
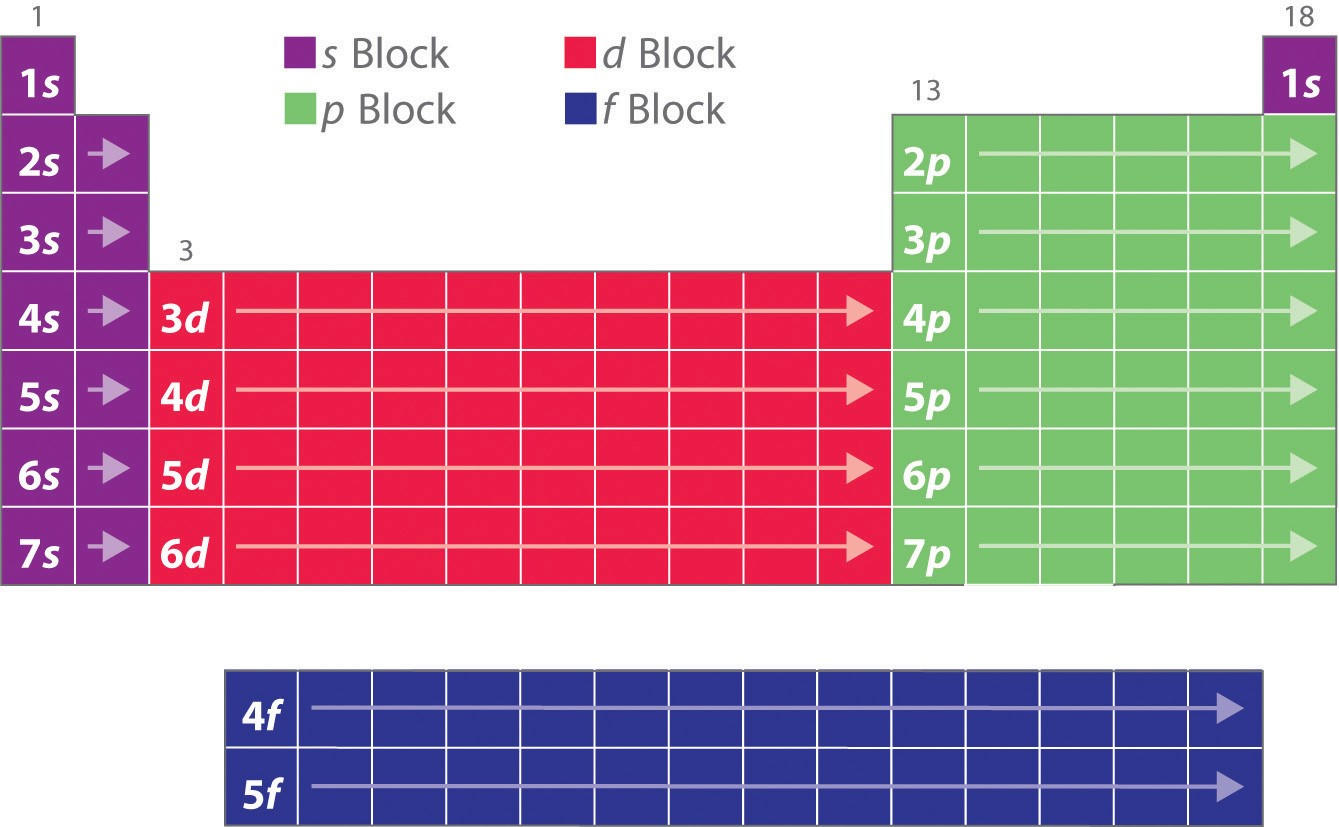
how does atomic radius change across a period? why?
decreases
as no. of protons increases, +ve charge of nucleus increases
this means outer e- are pulled closer, making radius smaller
w/ same shielding (as even though we are adding extra e-, they are added to the same shell, so they don’t increase the size or shield the +ve charge of the nucleus)
what is first ionisation energy?
minimum energy needed to remove 1 mole of e- from 1 mole of atoms in the gaseous state
what is the general trend of ionisation energy across a period? why?
generally increases - takes more energy to remove an e-
as no. of protons increases, so +ve charge of nucleus increases
so pulling force on outer e- increases
why may first IE sometimes decrease across a period?
an e- may be being removed from a different subshell, so requires less energy to be removed
how does the mpt change across period 3 from Na → Al? why?
there is a general increase from Na → Al as metallic bonding gets stronger, so more energy is required to break the metallic bonds
this is because we go from Na+ to Mg2+ to Al3+ so there is a smaller radius and more delocalised e-
so there is a stronger attraction between the +ve ions and the delocalised e-
how does the mpt change across period 3 when we get to Si? why?
increase - has highest mpt
as Si is a macromolecular compound w/ very strong covalent bonds
which require a large amount of energy to break
how does the mpt change across period 3 from P → Ar? why?
P4, S8 and Cl2 are all molecular substances, so their mpt depends on the strength of the IMF - in this case VDW - between the molecules
the more atoms, the stronger the IMF, so S8 > P4 > Cl2
Ar is monatomic so has a very low melting point
which ion has the largest radius and why? F-, Mg2+, Na+, O2-
O2- - most -ve, fewest protons so weakest attraction between outer e- and nucleus