biochem 12
1/40
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
Protein separation methods
differential centrifugation, salting out
gel filtration chromatography, ion-exchange chromatography, affinity chromatography
Differential centrifugation
Separates soluble and particulate fractions of cell lysate
Salting out
Separates proteins based on solubility using high salt concentrations
Gel filtration chromatography
Separates proteins based on size and shape
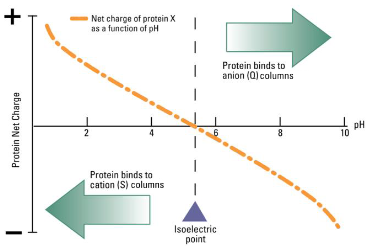
Ion-exchange chromatography
Separates proteins based on net charge at a given pH
Affinity chromatography
Separates proteins based on specific ligand binding
Protein characterization
Determination of physical and chemical properties of proteins
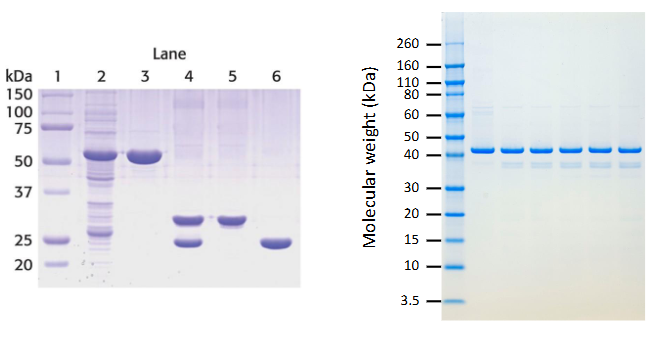
Molecular weight determination
Can be measured using SDS-PAGE or gel filtration
Isoelectric point (pI)
pH at which a protein has no net charge
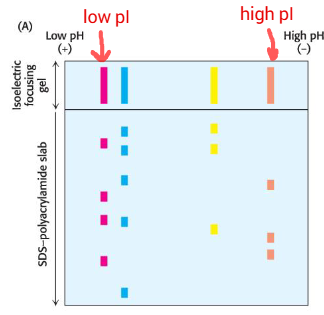
Isoelectric focusing
Technique to separate proteins based on pI in a pH gradient
UV-visible spectroscopy
Method to analyze protein absorbance properties
3D protein structure determination
Determining full structure using X-ray, NMR, cryo-electron microscopy
Polyacrylamide gel electrophoresis (PAGE)
Technique to separate proteins in a gel using an electric field
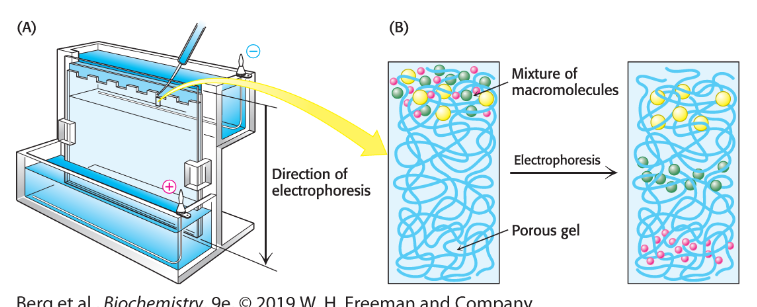
SDS-PAGE
Electrophoresis method that separates proteins based on size
SDS gives all proteins uniformly negative charge
small proteins move faster
Gel filtration vs SDS-PAGE similarity
Both separate proteins based on size
Gel filtration vs SDS-PAGE difference
Gel filtration uses native proteins while SDS-PAGE uses denatured proteins
Protein migration in SDS-PAGE
Proteins move toward positive electrode
Large vs small proteins in gel filtration
Large proteins elute first
Purpose of reducing conditions
Reveal subunit composition of proteins
Isoelectric focusing
Separation of proteins in a pH gradient based on pI
Proteins migrate until they reach pH where net charge is zero
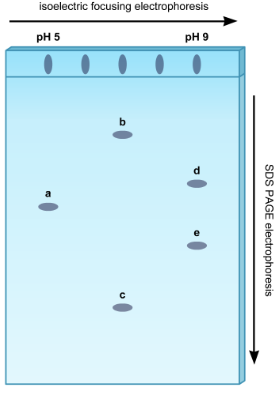
Two-dimensional electrophoresis
Combines isoelectric focusing (by pI) and SDS-PAGE (by size)
Spectroscopic detection of proteins
Measurement of protein concentration using light absorbance
Aromatic amino acids
Absorb UV light in proteins
Strongest chromophores in proteins
Tryptophan and tyrosine
how to determine highest pI value?
highest molecular weight?
highest pI value: farthest to right
highest molecular weight: highest up
Lambert-Beer equation
A = ε·c·l
Relationship between absorbance and concentration of a solution
Specific activity
Enzyme activity per amount of protein (U/mg)
Measure purity of protein during purification- increases as purification occurs
Visual evaluation of purification
SDS-PAGE shows fewer bands as purity increases
Protein structure determination methods
Techniques used to determine 3D protein structure
X-ray crystallography
Determines structure from diffraction of X-rays through protein crystals
Steps in X-ray crystallography
purify protein
crystallize protein (difficult)
collect diffraction data
calculate electron density
fit residues into density
pros:
no size limit
well established
cons:
difficult for membrane proteins
cant see hydrogen
Advantage of X-ray crystallography
No size limit and well-established method
Limitation of X-ray crystallography
Difficult crystallization and cannot see hydrogens
Nuclear magnetic resonance (NMR)
Determines protein structure in solution using magnetic fields
Steps in NMR
Steps needed
• Purify the protein
• Dissolve the protein
• Collect NMR data
• Assign NMR signals
• Calculate the structure
Advantage of NMR
No need for crystals and can observe hydrogens and dynamics
Limitation of NMR
pros:
Works best for small proteins
no need to crystallize protein
cons:
difficult for insoluble proteins
Cryo-electron microscopy (Cryo-EM)
Determines protein structure using frozen samples and electron microscopy
Steps in Cryo-EM
purify protein
place in em grid and freeze
collect images in microscope
calculate 3d structure
pros:
good for large proteins/complexes
requires little sample and no special treatment
cons:
new method, bad for small proteins
Advantage of Cryo-EM
Good for large proteins and complexes
Limitation of Cryo-EM
Not suitable for small proteins and equipment is expensive