Ch. 2
0.0(0)
Studied by 1 personCard Sorting
1/60
There's no tags or description
Looks like no tags are added yet.
Last updated 8:58 PM on 8/22/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
61 Terms
1
New cards
What is matter? What states is it found in?
Matter is anything that can take up space and can be weighed
* **Solid** - particles tend to be densely packed and move very little
* **Liquid** - particles tend to be less densely packed and move more rapidly
* **Gas** - particles tend to be loosely packed and highly moveable
* **Solid** - particles tend to be densely packed and move very little
* **Liquid** - particles tend to be less densely packed and move more rapidly
* **Gas** - particles tend to be loosely packed and highly moveable

2
New cards
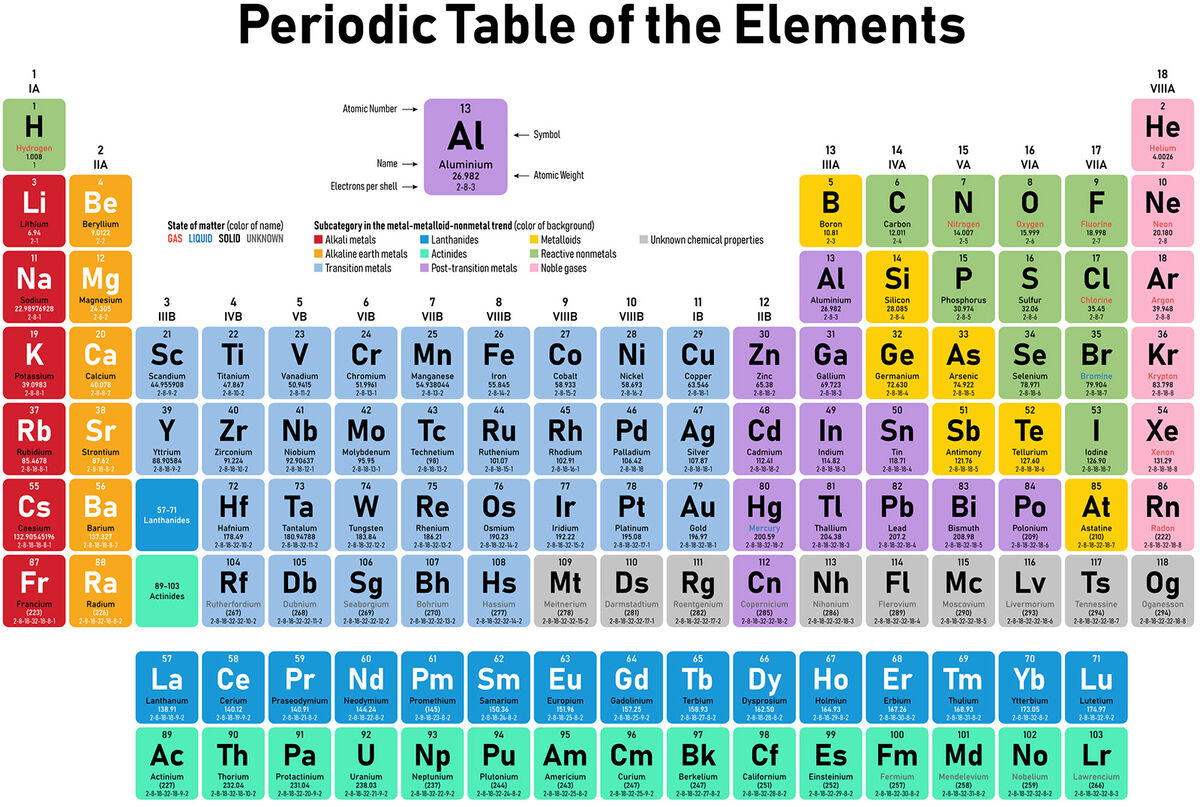
What are elements? How many are there on the periodic table?
Pieces that we can’t break down and still keep their characteristics and there are 118 known elements on the PT
3
New cards
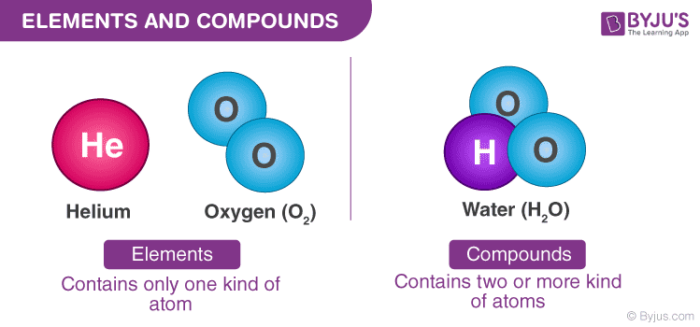
What are compounds?
Sticking together or bonding two different elements together which will have different characteristics from the element by itself – this gives us a lot of options to mix and match and make different functions and characteristics
4
New cards
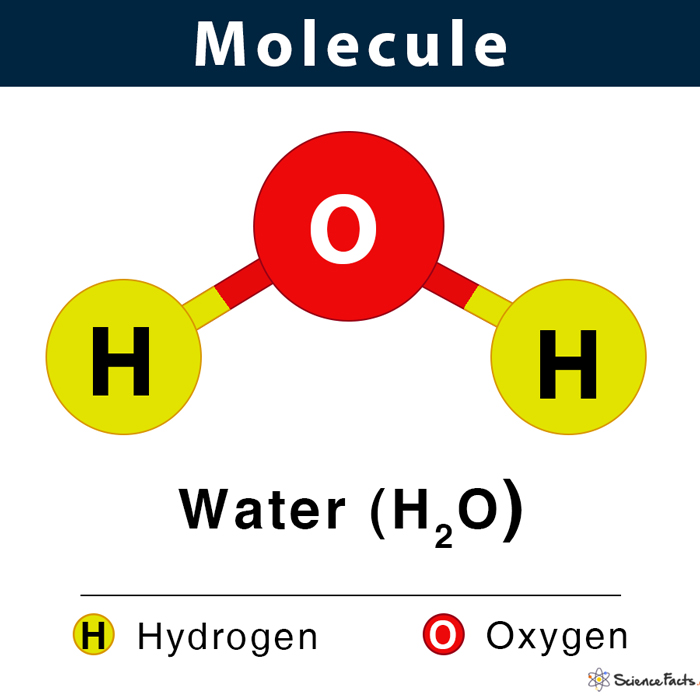
What are molecules?
Molecules are chemical substances consisting of atoms of one or more elements held together by covalent bonds
5
New cards
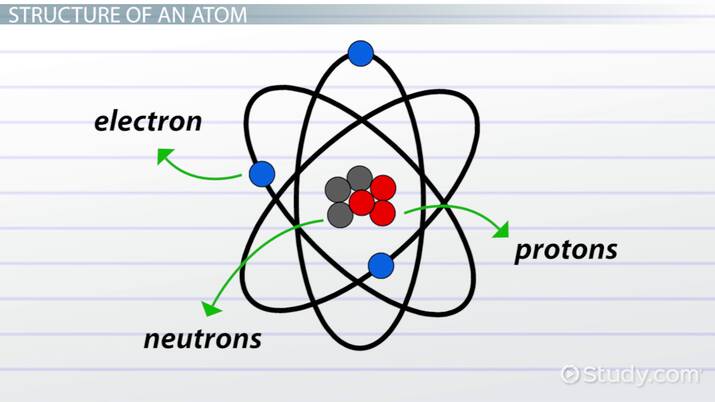
What are atoms?
Atoms are the smallest unit of matter that still retains the properties of an element
6
New cards
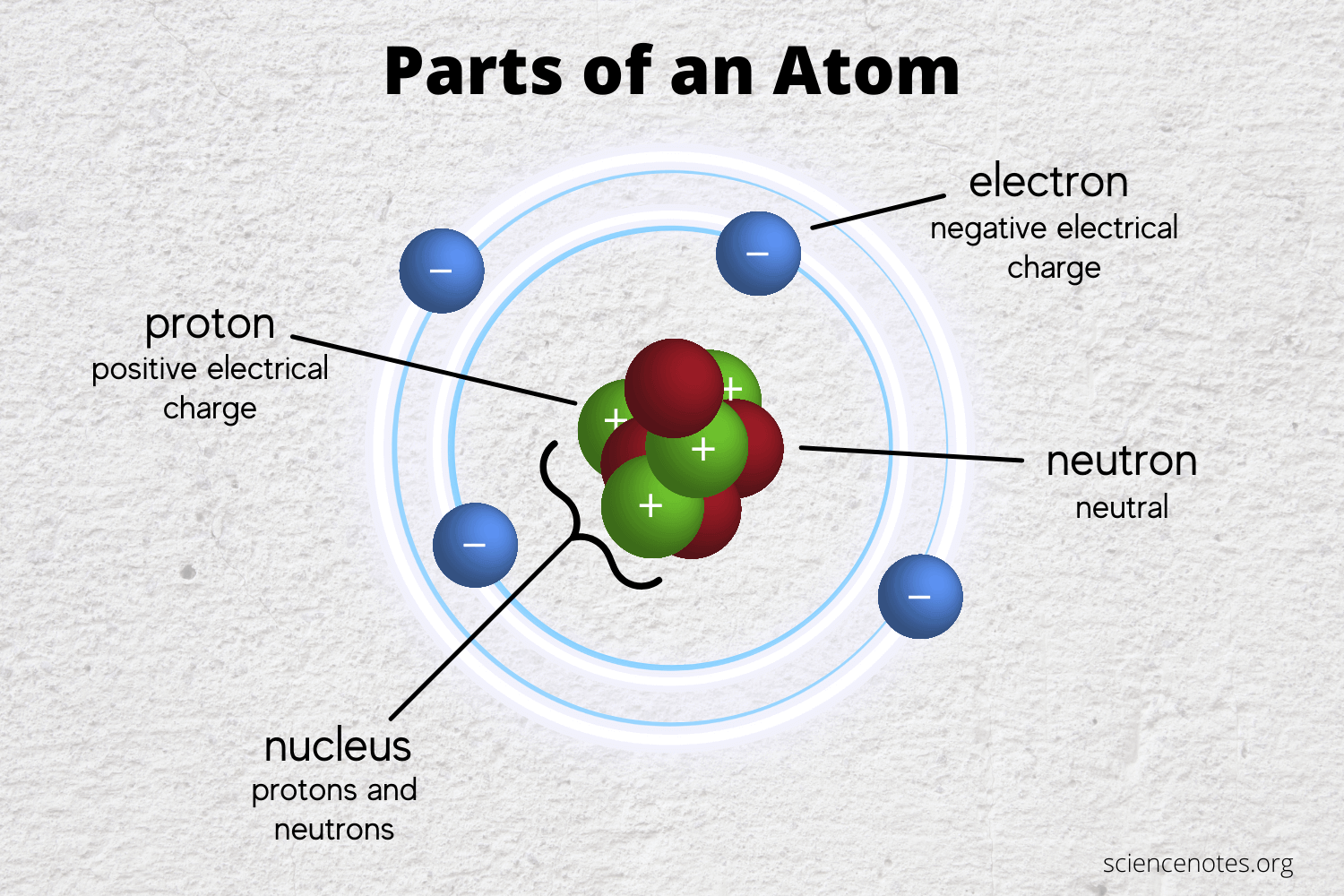
Atoms can be broken down into subatomic particles, however if the atom is broken down further it would lose the properties that make up that element. What are those subatomic particles?
**Protons (+)** have a **positive charge** and located in the nucleus or core of the atom
**Neutrons** have a **neutral charge** and also located in the nucleus or core of the atom
**Electrons (-)** have a **negative charge** and are located outside the nucleus creating an electron cloud made up of levels called electron shells
**Neutrons** have a **neutral charge** and also located in the nucleus or core of the atom
**Electrons (-)** have a **negative charge** and are located outside the nucleus creating an electron cloud made up of levels called electron shells
7
New cards
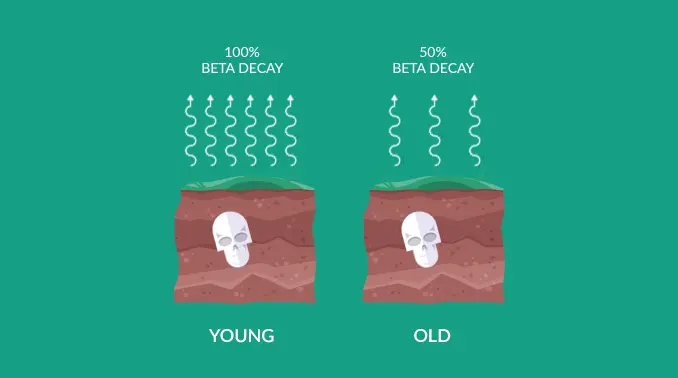
What is carbon dating?
Radiocarbon dating, or carbon-14 dating, is a scientific method that can accurately determine the age of organic materials as old as approximately 60,000 years
8
New cards
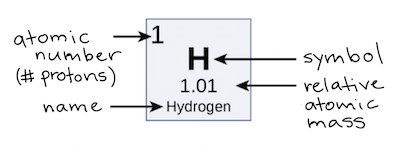
Atomic number vs. mass number
the number of **protons** (number placement on the periodic table like Nitrogen is number 7), however in all neutral atoms, the number of electrons is the same as the number of protons whereas the mass number is protons plus neutrons
9
New cards
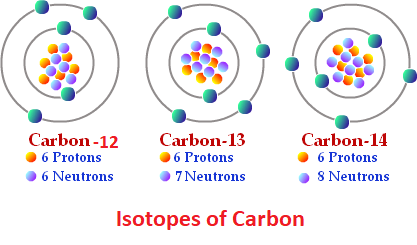
What are isotopes?
Atoms with the same number of protons but different number of neutrons (can vary)
10
New cards
What energy molecule do we primarily use?
ATP

11
New cards
What is an exothermic energy reaction?
A chemical reaction that releases energy by light or heat

12
New cards
What is an endothermic energy reaction?
Any chemical reaction that absorbs heat from its environment

13
New cards
Energy levels
These energy levels are designated by a number and the symbol “n” with n1, n2, etc. Since n1 is closest to the nucleus its energy will be the lowest of the shells. The outermost shell contains the most energy
14
New cards
What are valence electrons?
The electrons that we have on the outermost energy level of an atom
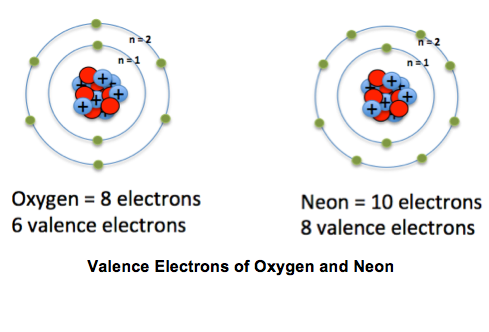
15
New cards
What is an ion?
An ion is a charged atom or molecule. It is charged because the number of electrons do not equal the number of protons in the atom or molecule
16
New cards
Cations vs. anions
**Cations** are positively charged atoms created by the loss of one or more electrons (Na+, Ca2+, or Mg2+)
**Anions** are negatively charged atoms created by the gaining of one or more electrons (Cl-, SO42-, or PO42-)
**Anions** are negatively charged atoms created by the gaining of one or more electrons (Cl-, SO42-, or PO42-)
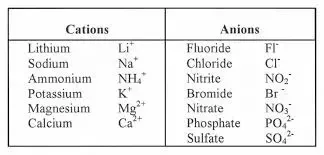
17
New cards
What are chemical bonds?
Chemical bonds are interactions that stabilize the outer energy levels of atoms
In a chemical reaction, new chemical bonds form between atoms, or existing bonds between atoms are broken
In a chemical reaction, new chemical bonds form between atoms, or existing bonds between atoms are broken
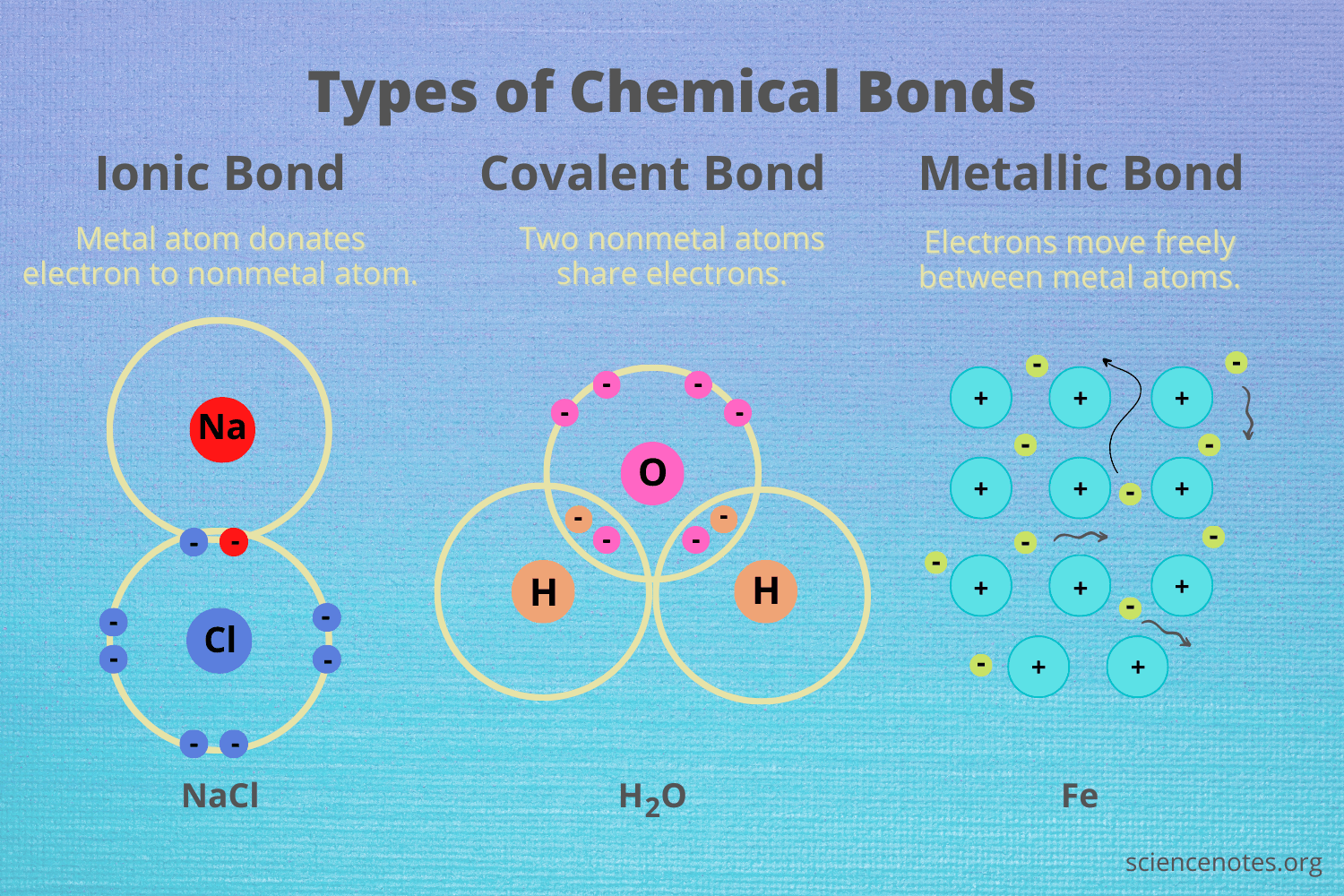
18
New cards
Reactive atoms
If the outer shell is **not full**, the atom will form chemical bonds with other atoms until it satisfies its outermost shell requirements. The atom is said to be **reactive**. **Hydrogen, Lithium, and Sodium** are all reactive because their valence shells are not full
19
New cards
Inert atoms
If the outer shell **is full**, the atom will not react with other atoms and is said to be **inert. Helium and neon,** for example, are called **inert gases or noble gases**
20
New cards
What are ionic bonds?
Chemical bonds created by the **electron transfer** from one atom to another atom
21
New cards
Ionic bonds
The transfer of electrons creates **two ionized atoms**: one a **cation** and the other an **anion**
Being of opposite charges, the two atoms attract one another. This attraction is an **ionic bond** EX: Na+ + Cl- yields NaCl (table salt)
Being of opposite charges, the two atoms attract one another. This attraction is an **ionic bond** EX: Na+ + Cl- yields NaCl (table salt)
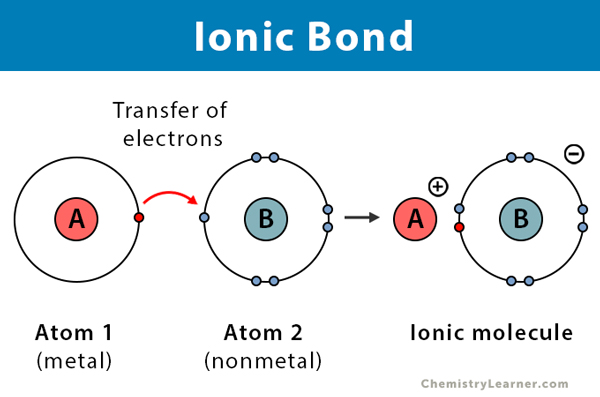
22
New cards
What electron transfer happens?
This is where trading of the electrons will happen (**loss or gain**) – one is going to give up electrons and **become positive** because it **gave a negative charge** and the other is **going to gain that negative charge** and will become negative and usually occurs when one is a **metal** and the **other is a nonmetal**
23
New cards
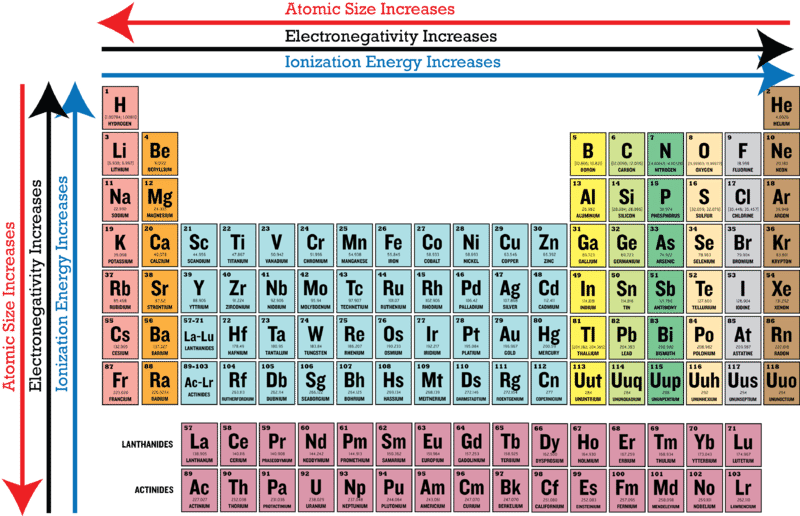
What is electronegativity?
A measure of the attraction of an atom for bonding electrons in molecules compared to that of other atoms
24
New cards
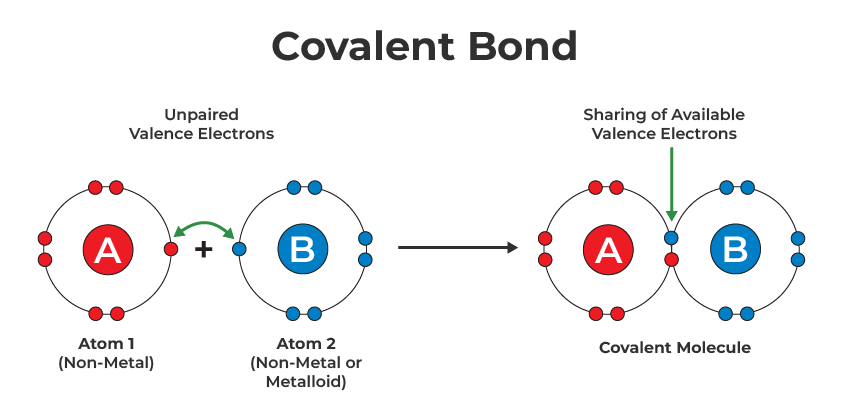
What are covalent chemical bonds?
Chemical bonds that **form between neutral atoms** rather than ionized atoms
25
New cards
What do covalent bonds do?
Covalent bonds **share** their electrons with each atom – they’re not trying to steal electrons because there isn’t enough of a difference in electronegativity to allow them to take the electrons away from the other so instead they line themselves up so the orbitals overlap and each get the electron that they need
26
New cards
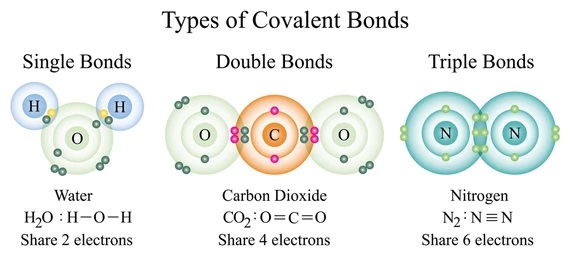
Single covalent bonds vs, double covalent bonds
**Single covalent bonds** exist when one pair of electrons are shared between two atoms while **double covalent bonds** exist when two pairs of electrons are shared between two atoms
27
New cards
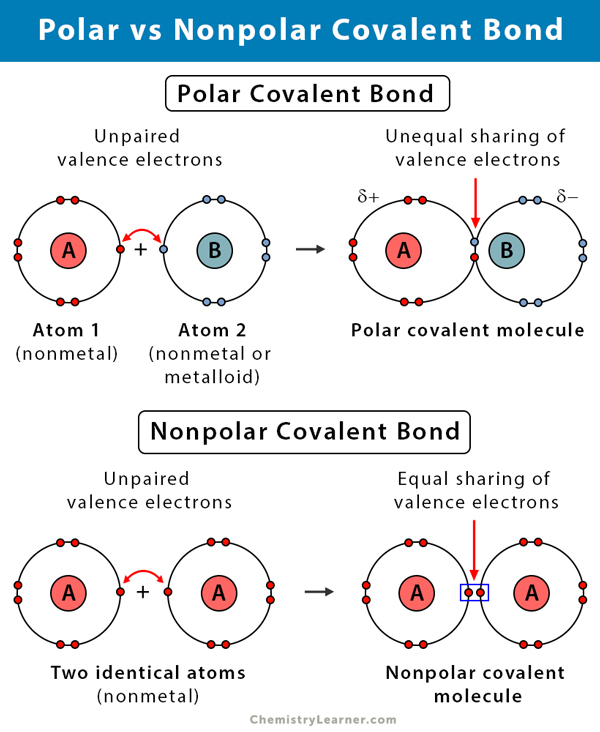
What is polarity?
This is where there is an imbalance in electronegativity – one is more electronegative so it’s able to pull things a little closer to it so it becomes **partially negative** and the other will be **partially positive** because the electrons are further away from it
This can be important because they can interact with stuff like ions that are fully charged because they have this partial charge, so it’s a weaker interaction but it’s still there
This can be important because they can interact with stuff like ions that are fully charged because they have this partial charge, so it’s a weaker interaction but it’s still there
28
New cards
Nonpolar covalent bonds vs. polar covalent bonds
**Nonpolar covalent bonds** are those where the atoms **share** the electrons **equally** (would be oil or fat)
**Polar covalent bonds** are created by **unequal** sharing of the electrons due to the electronegativity of the atoms (water is formed by these bonds). Polar things don’t like to mix with nonpolar things (water and oil)
**Polar covalent bonds** are created by **unequal** sharing of the electrons due to the electronegativity of the atoms (water is formed by these bonds). Polar things don’t like to mix with nonpolar things (water and oil)
29
New cards
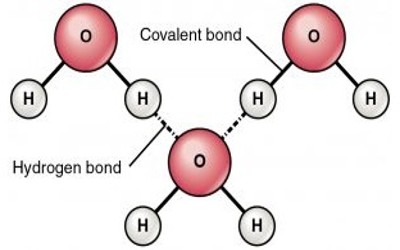
What is a hydrogen bond?
A **hydrogen bond** is the weak attractive force occurring when the small positive charges on the hydrogen atoms of one polar molecule can be attracted to the negative charges on another polar molecule, and this can change the shapes of the molecules or pull adjacent molecules together
30
New cards
What are attractive forces?
Forces between separate molecules that are not bonds, not as strong as bonds (very weak compared to covalent bonds). They are important because it keeps things in specific arrangements to try to allow separate objects/molecules to still stick closer together than they would elsewise
31
New cards
What are Van der Waals forces?
A molecule has its electrons constantly moving and sometimes those electrons happen to be on one side of the molecule so that side of the molecule becomes slightly negative for a moment and the other side becomes slightly positive and so they can get these temporary attractive forces between the different molecules
32
New cards
Random water info
In solution, an ionic compound **dissociates** as water molecules **break** them apart. The **anions** are surrounded by the **positive pole** of the water molecule, and the **cations** are surrounded by the **negative pole** of the water molecule. The sheath of water molecules around an ion in solution is called a **hydration sphere**
33
New cards
What are hydrophilic molecules?
Hydration spheres also form around an organic molecule containing polar covalent bonds. If the molecule binds water strongly, as does glucose, it will dissolve. Molecules that **interact** readily with water are called **hydrophilic**
34
New cards
What are hydrophobic molecules?
Many organic molecules either lack polar covalent bonds or have very few. Such molecules do not have positive and negative poles and are said to be nonpolar. When nonpolar molecules are mixed in water, hydration spheres do not form and the molecules do not dissolve in the water. These molecules are said to be **hydrophobic**
35
New cards
What is interstitial fluid?
Fluid that fills the space between our cells
36
New cards
Is water polar or nonpolar?
Water is very polar. All of the bonds that it has are some of the most polar that exist
37
New cards
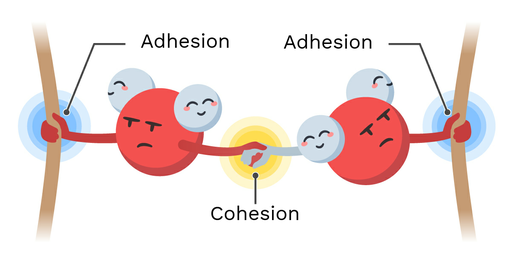
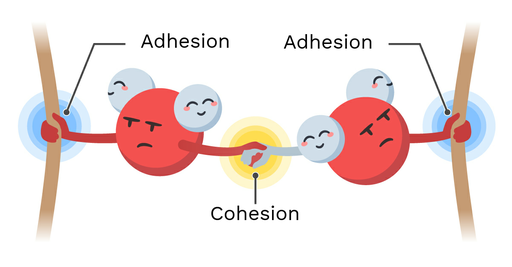
What is cohesion?
Water can stick to itself
38
New cards
What is adhesion?
Water molecules sticking to other polar substances
39
New cards
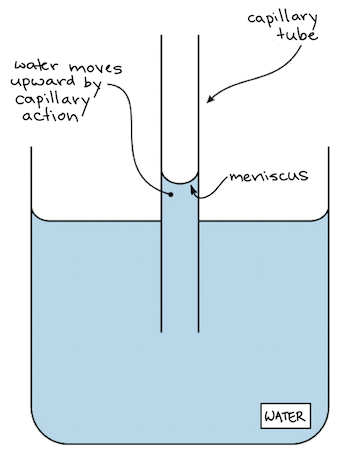
What is capillarity?
Because of adhesion and cohesion you get **capillarity** - water can **climb against gravity** if you give it a optimal situation
40
New cards
What is high surface tension?
Because water likes to stick to other water molecules we get to where it’s a little difficult to fall into water–if you’re small enough you can climb around on water

41
New cards
How is water different from other substances?
Water is the only substance that occurs as a solid (ice), a liquid (water), and a gas (water vapor) at temperatures compatible with life
42
New cards
What is high heat capacity?
The ability to absorb and retain heat
43
New cards
High specific heat
It takes quite a bit of energy to make water change temperature
44
New cards
Liquids
Liquids are going to be **less dense** than solid but as water molecules slow down they are able to stretch out as much as possible and lock into a lattice formation
45
New cards
Cold water
Cold water is going to be **more dense** – critical for things living in a lake during winter (ice will float and won’t crush the organisms underneath during the winter)
46
New cards
What is the universal solvent?
Water. It mixes with pretty much anything (not everything)
47
New cards
Universal solvent
Water can **dissolve** or **dissociate** things that you put into it that have any propensity to become at least partially charged but it won’t dissolve or go through things that are nonpolar
48
New cards
What ions can water form?
Water can naturally breakdown to form two ions - **H+ and OH-** and sometimes H+ binds to a water and becomes H3O+
49
New cards
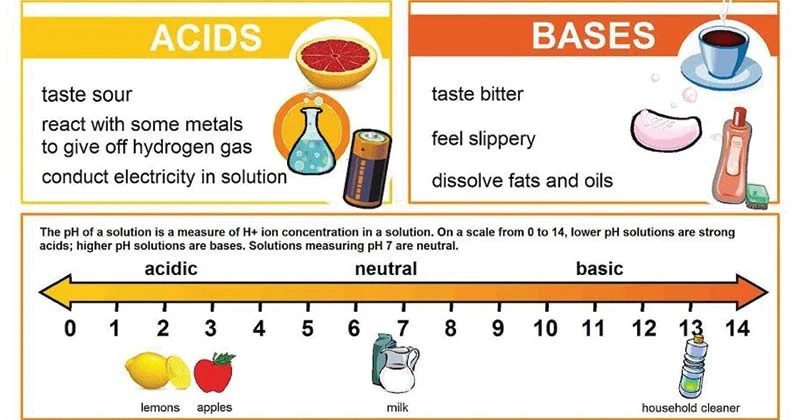
What are acids?
Acids are biological compounds that release hydrogen ions (H+) when placed in solution
* Hydrochloric acid (HCl) is an important compound found in our stomachs and is necessary for the digestion of certain foods
* Acids have a low pH on the pH scale **(1 - 6.9)**. The more H+ ions released by the substance the more acid the substance is (closer to 1)
* Acids are proton **donors**
* Hydrochloric acid (HCl) is an important compound found in our stomachs and is necessary for the digestion of certain foods
* Acids have a low pH on the pH scale **(1 - 6.9)**. The more H+ ions released by the substance the more acid the substance is (closer to 1)
* Acids are proton **donors**
50
New cards
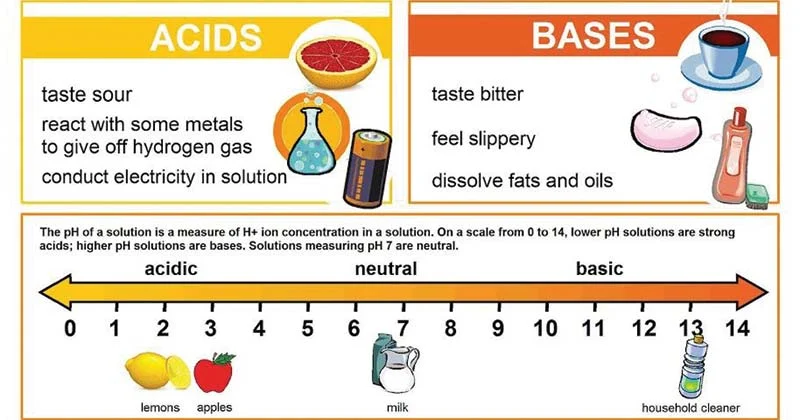
What are bases?
Bases are biological compounds that release hydroxide ions (OH-) when placed in solution. Also known as alkalinity
* Sodium hydroxide (NaOH) reacts with fats in the diet and is used to make soaps in industry
* Bases have a high pH on the pH scale (**7.1 - 14).** The more OH- ions released the more alkaline the substance is (closer to 14)
* Bases are proton **acceptors**
* Sodium hydroxide (NaOH) reacts with fats in the diet and is used to make soaps in industry
* Bases have a high pH on the pH scale (**7.1 - 14).** The more OH- ions released the more alkaline the substance is (closer to 14)
* Bases are proton **acceptors**
51
New cards
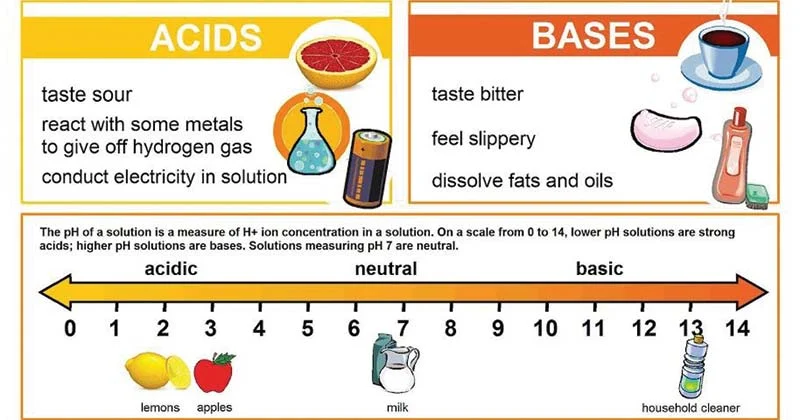
What are neutrals?
Neutrals are biological compounds that release equal numbers of H+ and OH- when placed in solution
* Distilled water is neutral
* Neutrals have a **pH of 7.0** on the pH scale
* Distilled water is neutral
* Neutrals have a **pH of 7.0** on the pH scale
52
New cards
What are buffers?
Buffers **resist change** in pH. This can help organisms maintain homeostasis and stay alive – they can help balance out the concentration of H+ to prevent the pH from spiking and becoming too acidic or becoming too basic
53
New cards
Characteristics of carbon
Carbon has **four valence electrons** – it wants to form **four covalent bonds** (max that you can do as an element) so it’s very good at connecting to other things (gives it lots of possibilities)
It can **bond with itself** – it forms large carbon compounds (graphite, diamond) because a lot of elements don’t really like to bond with themselves OR they’ll bind together but only about two of them
Carbon doesn’t just form single bonds and double bonds but it **can form triple bonds** – this allows for it to have all of the bonding options that there are
It can **bond with itself** – it forms large carbon compounds (graphite, diamond) because a lot of elements don’t really like to bond with themselves OR they’ll bind together but only about two of them
Carbon doesn’t just form single bonds and double bonds but it **can form triple bonds** – this allows for it to have all of the bonding options that there are
54
New cards
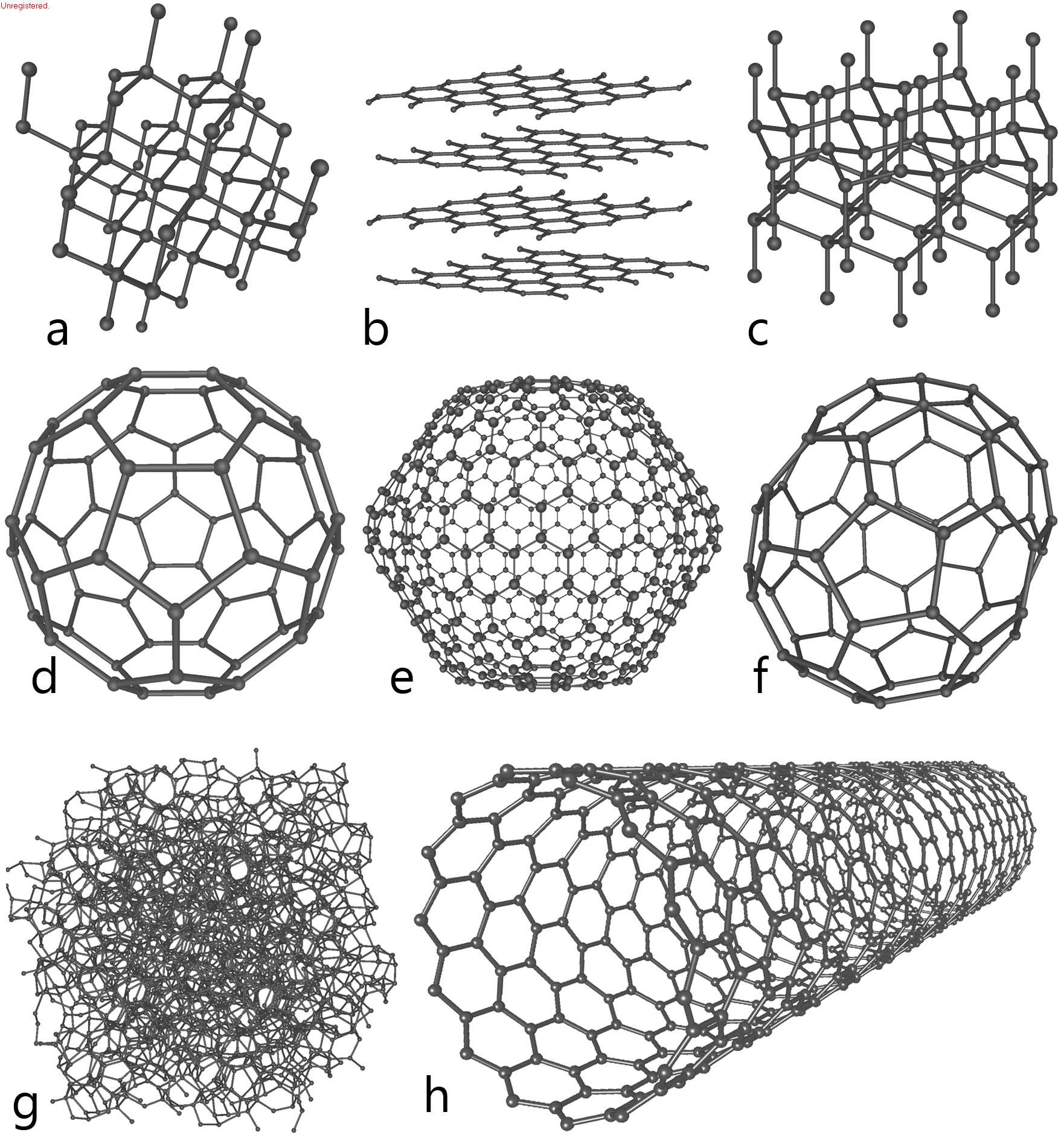
What are allotropes?
The structures of carbon that bond together are called **allotropes** – the ability to swap out small numbers of other things that are mixed in there gives us brand new molecules and brand new characteristics
55
New cards
What are the four types of biological macromolecules?
There are four types of biological macromolecules: **carbohydrates, lipids, proteins and nucleic acids**
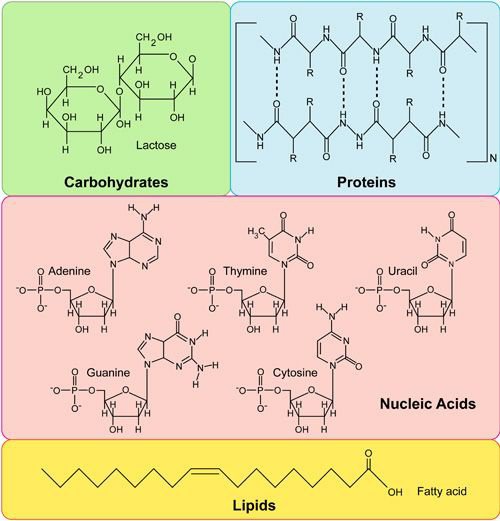
56
New cards
What are hydrocarbons?
Hydrocarbons are organic molecules consisting entirely of carbon and hydrogen, such as methane (CH4)
They are **nonpolar** (oil)
The bonds are **high energy bonds** – so when we break/reform them we give off a lot of energy
They are **nonpolar** (oil)
The bonds are **high energy bonds** – so when we break/reform them we give off a lot of energy
57
New cards
What are isomers?
Isomers are molecules that share the same chemical formula but differ in the placement (structure) of their atoms and/or chemical bonds (glucose and fructose)
58
New cards
What are structural isomers?
**Structural isomers** (ex: butane and isobutene) differ in the placement of their covalent bonds: both molecules have four carbons and ten hydrogens (C4H10), but the different arrangement of the atoms within the molecules leads to differences in their chemical properties
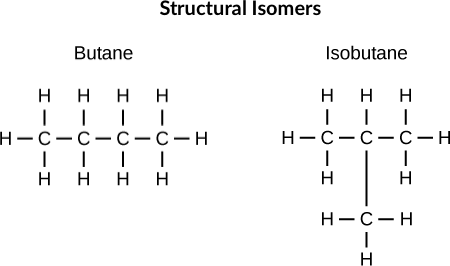
59
New cards
What are geometric isomers?
**Geometric isomers** usually have a double bond and wherever that double bond, is rotated
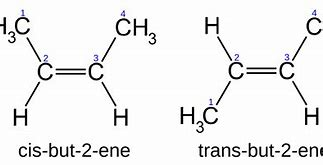
60
New cards
What are enantiomers?
**Enantiomers** are molecules that share the same chemical structure and chemical bonds but differ in the three-dimensional placement of atoms (mirror images) – these differences will have different properties and cause major issues
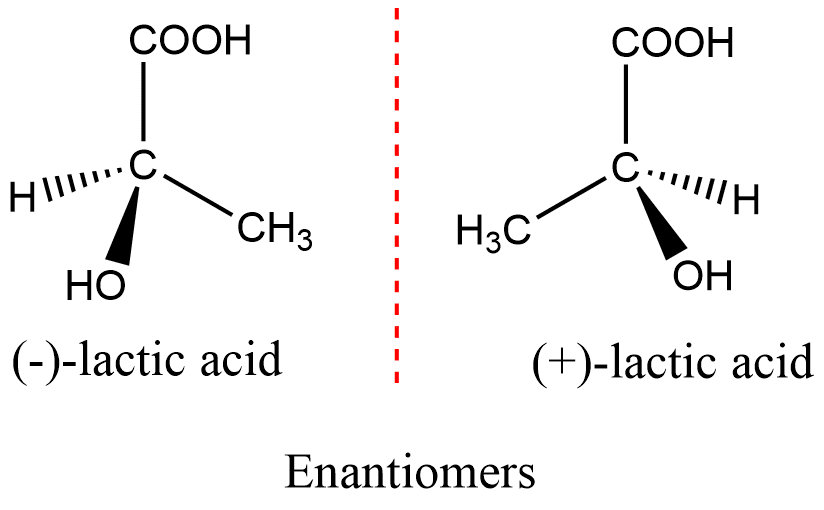
61
New cards
What are functional groups?
Groups of atoms found along the “carbon backbone” that confer specific chemical properties to those molecules. Molecules with other elements in their carbon backbone are substituted hydrocarbons