Organic Chem chapter 5
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
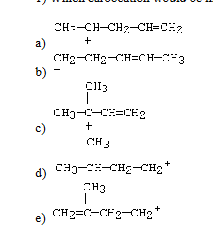
What Carboation would be most stable
C
what carbocation would be least stable
D
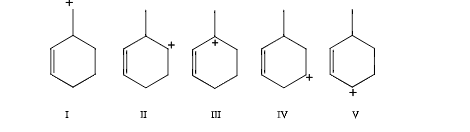
Which carbocation would be most stable
|||
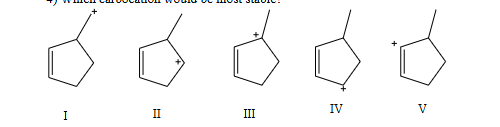
which carbocation would be most stable
C
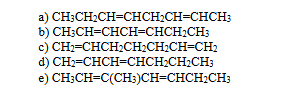
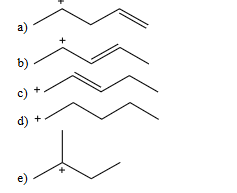
which of the following dienes would you expect to be most stable
B
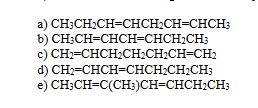
which of the following dienes would you expect to be the least stable?
C
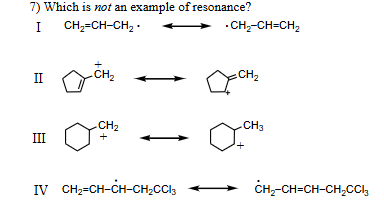
Which is not an example of resonance
III
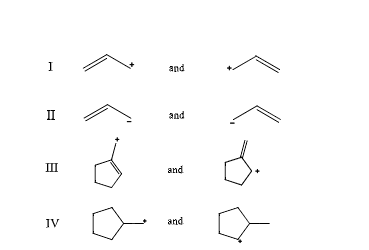
which pair does not represent a pair of resonance structures
IV
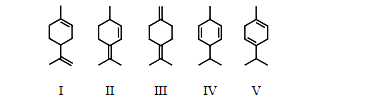
selected the structure of the conjugated diene
II
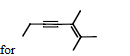
what is an IUPAC name for
2,3-dimethyl-2-hepten-4-yne
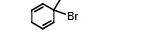
A correct IUPAC name of the compound is
3-Bromo-3-methyl-1,4-cyclohexadiene
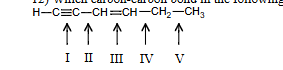
Which carbon-carbon bond in the following compound would you expect to be the shortest
I
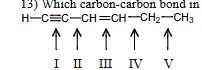
Which carbon-carbon bond in the following compound would you expect to be the longest
V
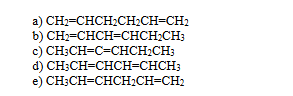
which of the following dineses might react with bromine CCl4 to yeild 2,5-dibromo-3-hexene
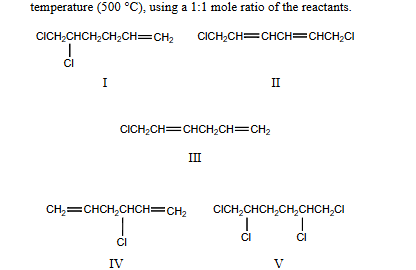
Indicate which products would be obtained from the chlorination of 1,5-hexadiene at high temperature (500 C), using a 1:1 mole ratio of the reactants
III and IV
which reagent would convert 1,3- pentadiene into 3-penten-2-ol
H3O+
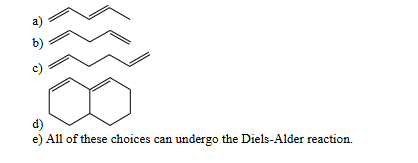
which of these dienes can undergo the diels-alder reaction
A
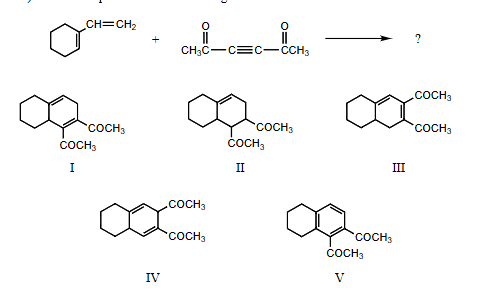
What is the product of the following reaction
I
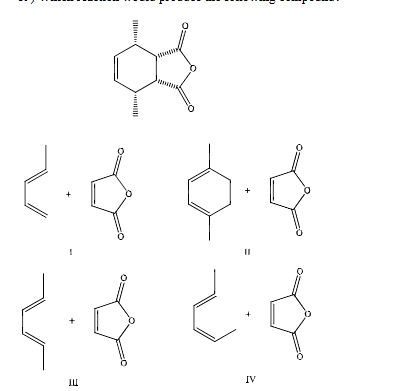
which reaction would produce the following compound
III
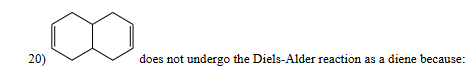
The structure above does not undergo the diels-alder reaction as a diene because
a ring system with isolated alkenes cannot function as the diene component