Medchem Final
1/80
Earn XP
Description and Tags
Bagel
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
81 Terms
Molecular Pathology
the study of the molecular basis of disease
‣ Clinical data, genome-wide association study (GWAS)
‣ Cellular models (knockout, transcriptomics / proteomics)
‣ Animal models (knockout, transcriptomics / proteomics)
‣ Pharmacological hypothesis testing with tool compound
Viral RNA encodes for …
One long polyprotein. It has PLpro and MPro chop it up and make it functional.
PCSK9
It is a protease that binds to LDL-Receptors and takes em to lysosome for degredation. (We want less PCSK9 because more receptors means less LDL is the bloodstream)
Assay
are brief, quantitative tests of a biological hypothesis
‣ Does my enzyme transform its substrate with compound X?
‣ Does my receptor signal with compound X?
‣ Does my target bind its partner in the presence of compound X?
‣ Does my compound enter the cell?
Biophysical Assay and Biochemical Assay
Biophysical:
‣ Binding (calorimetry), structure (NMR, X-ray)
Biochemical:
‣ Enzymatic activity, receptor activity, channel conductance
Cell based Assay and Animal Assay
Cell based:
‣ Cytotoxicity, reporter gene, phenotypic
Animal:
‣ Behavior (learning, memory, pain), organ-level, toxicity
Considering factors for the type of assay to use for screening
difficulty
physiological relevance
cost
throughput
Fluorescence concept
‣ High-energy photon is absorbed
‣ Low-energy photon is emitted
‣ Engineer a fluorescence-based sensor of activity
‣ Examples = fluorescent dyes, fluorescent proteins
‣ Potential interferences = fluorescent compounds, quenchers
Chemiluminescence concept
‣ Enzyme (luciferase = luc) transforms substrate (luciferin)
‣ Enzymatic transformation results in light emission
‣ ATP + O2 required, almost no background
‣ Potential interferences = luciferase inhibitors
Normal distribution standard deviation rule
‣ 68-95-99.7 rule
ex) 68% of the data is within 1σ of the µ
Z’ Factor
Z’ = 1 is ideal assay
Z’ = 05 is acceptable assay for screening
Want sharp peaks far apart
Hit Validation steps
‣ Step 1 = synthesize / procure the compound
‣ Step 2 = validate in the primary assay (=confirmation)
‣ Step 3 = dose-response (IC50) in primary assay (=conforming behavior)
‣ Step 4 = secondary assay (orthogonal to primary, biochemical assay or cellular assay)
‣ Step 5 = evaluate common toxicity / nuisance behavior
‣ Step 6 = structure determination (identify functional grps for binding)
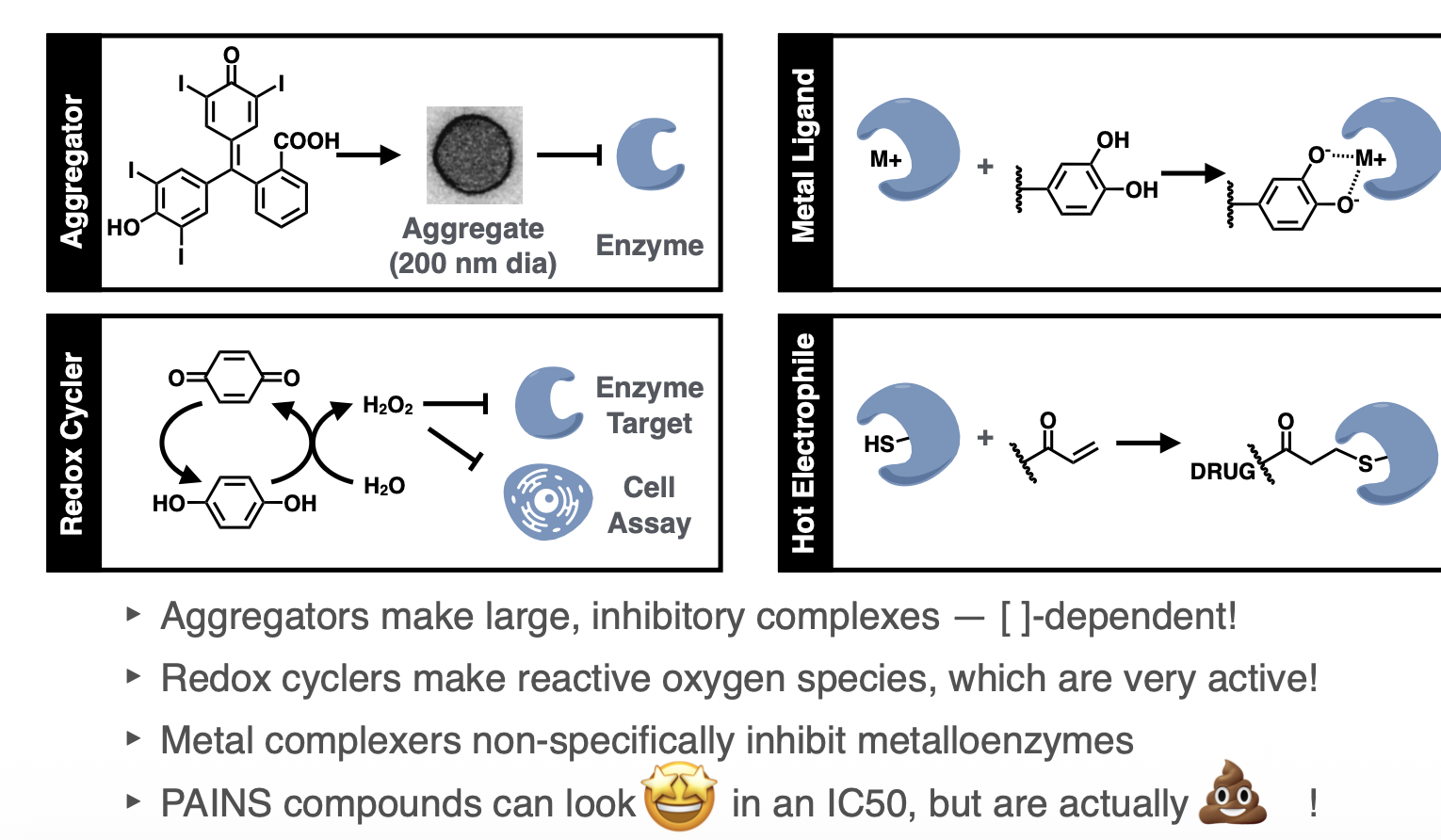
PAINS
Parmaceutic Phase
‣ Administration → distribution
‣ Oral = mouth → stomach → gut
Pharmacokinetic Phase
‣ Distribution → target
‣ Oral admin = gut → liver → target
‣ Metabolism in liver
‣ Transiting cell membranes
‣ Excretion
Pharmacodynamic Phase
‣ Drug acting on target
ADME
A = ABSORPTION
‣ Uptake of drug into circulation (blood)
‣ Usually by mouth or lung
D = DISTRIBUTION
‣ Movement of drug around body
‣ Points of accumulation
M = METABOLISM
‣ Transformation of drug in body
‣ Enzyme action (mostly in liver + stomach)
‣ Usually inactivates, increases polarity for…
E = EXCRETION
‣ Removal of drug from body
‣ Urine (kidneys)
‣ Feces (liver → gut)
Oral Administration Path
1. Mouth
‣ Most drugs are swallowed
‣ Some chewed (absorption)
‣ Enzymes (amylase, lipase)
2. Stomach
‣ H+ (pH 1.5–3 = v acidic!)
‣ Enzymes (peptidases)
3. Gut
4. Liver
‣ More enzymes (MANY!)
‣ Absorption through gut wall
‣ MORE ENZYMES!
‣ phase I metabolism (cytochrome P450)
‣ phase II metabolism (transferases)
‣ Leads to distribution
Bioavailability
the fraction of drug making it to circulation
Intravenous injection by definition is 100% available
logP
Partition coefficient, P
‣ Observe fraction of drug in n-octanol compared to water
(n-Octanol = good approximation of fatty tissues / membranes)
P= [drug] in octanol/ [drug] in water
Higher LogP = more greasy
Lipinski’s Rule of 5
‣ MW < 500 (not very big)
‣ LogP < 5 (not too greasy, but also not too polar)
‣ HBA < 10 (not too much H bonding)
‣ HBD < 5 (not too much H bonding)
Veber Rules
ROTATABLE BONDS (Nrot) ≤ 10
PERMEATION COEFFICIENT = Pe > 10-6 cm/s
or POLAR SURFACE AREA ≤ 140 Å
Liver metabolism
‣ phase I metabolism (cytochrome P450)
‣ phase II metabolism (transferases)
Polar molecules in the blood will be…
excreted by kidneys by dialysis between the renal vein and nephron. Nephron takes polar stuff for piss and Renal vein takes nonpolar stuff for blood.
Phase 1 metabolism
Oxidation, Reduction, Hydrolysis
Cytochrome p450 (Terminal Aliphatic, oxidize the most exposed)
Benzylic C-H favored
Also demethylation of heteroatoms
Alcohol Dehydrogenase (turns into aldehyde)
Aldehyde dehydrogenase (turns into acid)
Flavin Monooxygenases (heteroatom oxidizers)
Hydrolase activity (proteases and esterases)
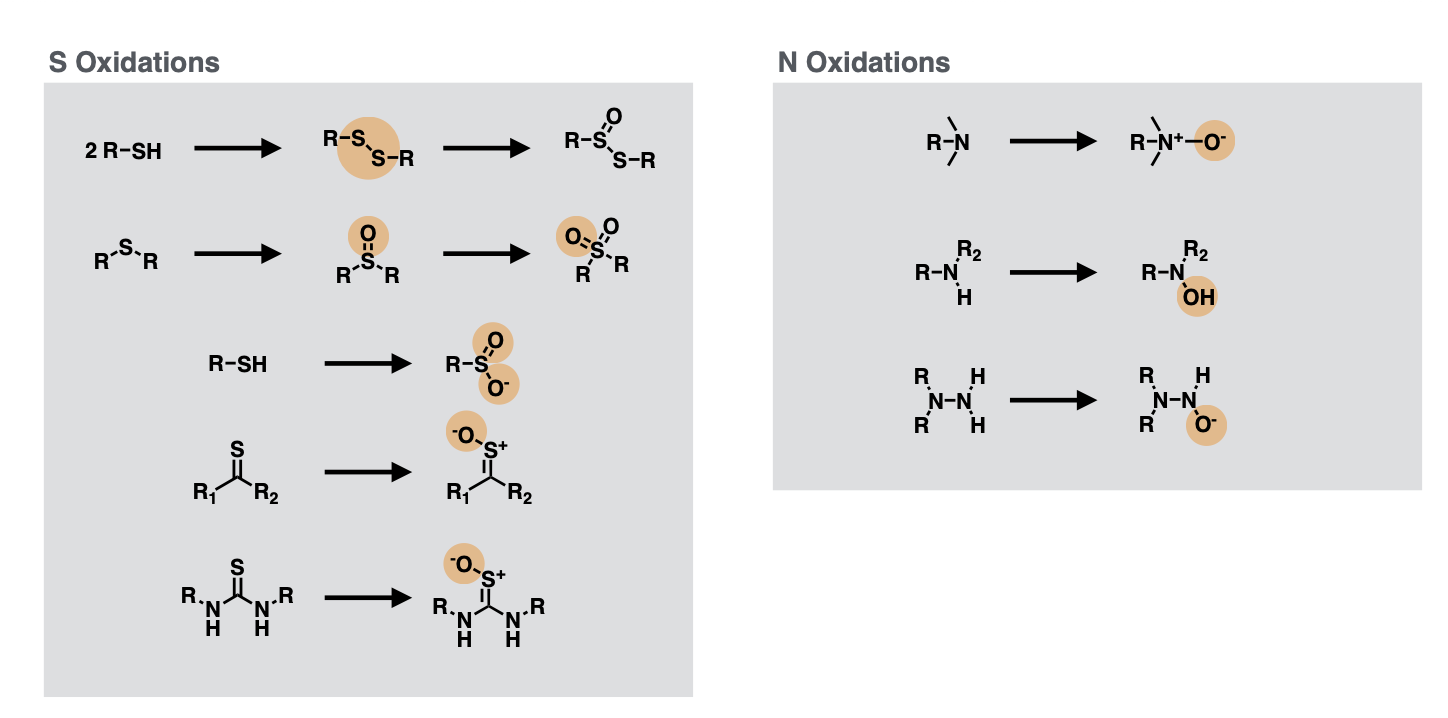
Flavin Monooxygenases
S oxidations
Add =O onto S for R-S-R or (R)2-S=O
Add -O- onto S atom when thiocarbonyl
Add both =O and -O- onto S when -SH
N oxidations
Add -O- onto tertiary amine
Add -OH onto secondary amine
Add -O- onto primary amine bonded to N (N-NH2)
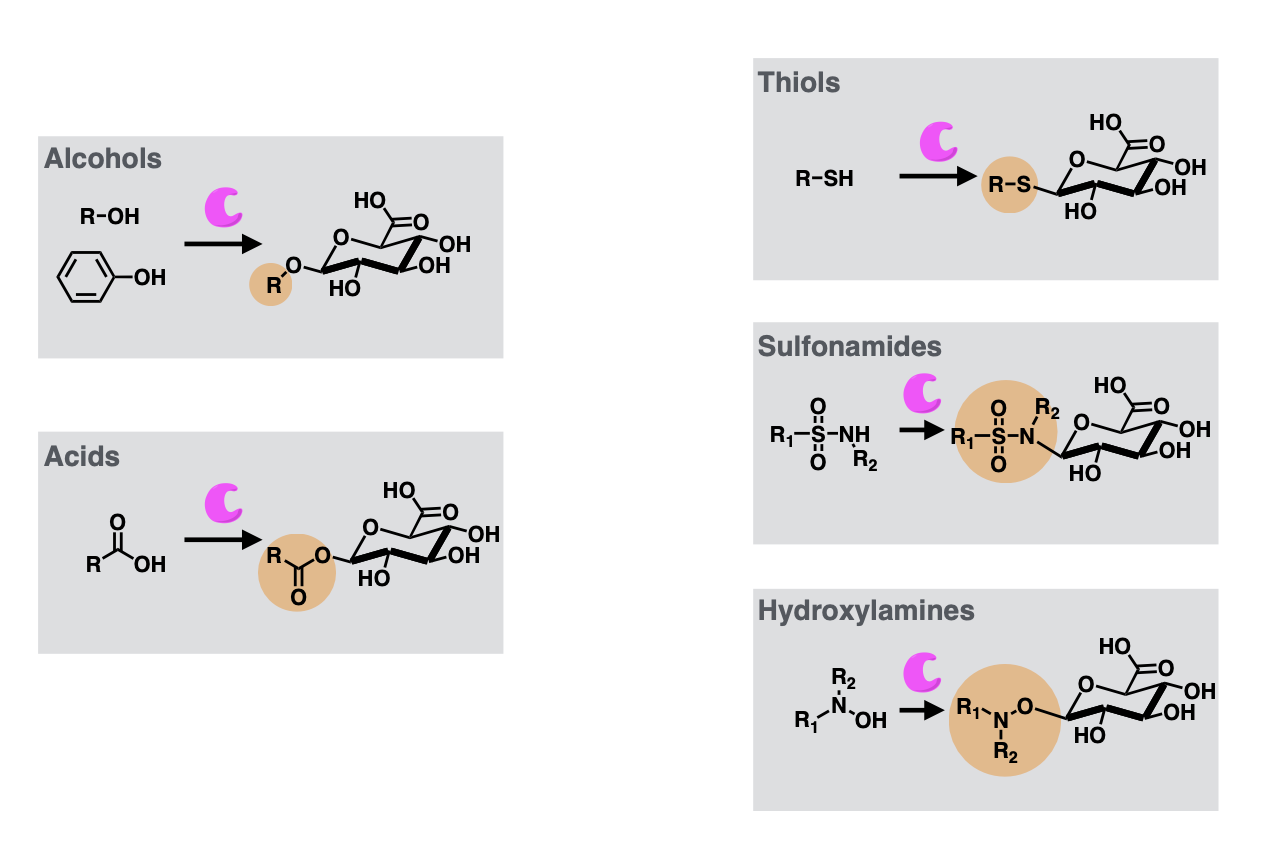
Phase 2 metabolism
Conjugation reactions
Glucaronidation (catalysed by UGT or UDP-glucoronosyltransferase).
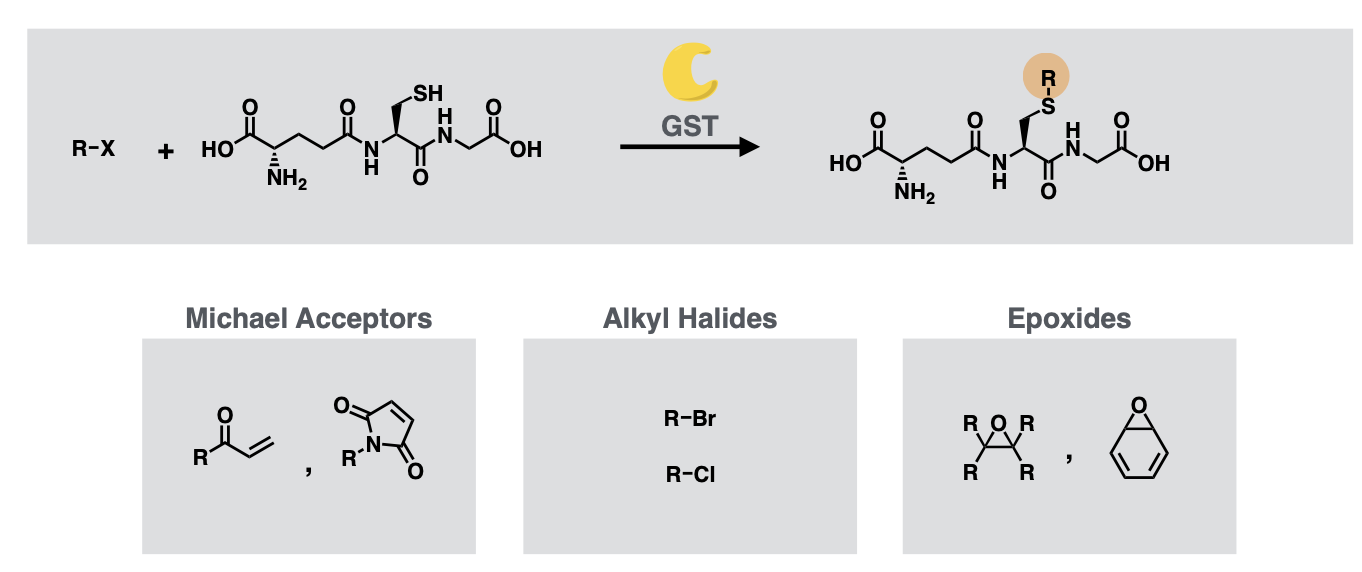
Glutathione (GSH) conjugation (catalysed by GST or Glutathione-S-transferase)
Other enzymes (GGT and CCBL) will cleave GSH to just Cystine
Then N-Acyl Transferase will make the acylate the amine
Glucaronidation substrates
Glutathione Conjugation substrates
Michael acceptors
Alkyl Halides
Epoxides
Drug Elimination
the rate at which drug is removed from the body
What order chemical reaction is the body and what is half life of it
1st order chem rxn
t1/2= ln2/k, (k is first order rate constant)
Theraputic level
above the EC50 of the drug
Toxic level
defines onset of adverse effects
Theraputic window
the range between theraputic level and toxic level
Steady-state drug concentration
Usually achieved after about 7 half lives
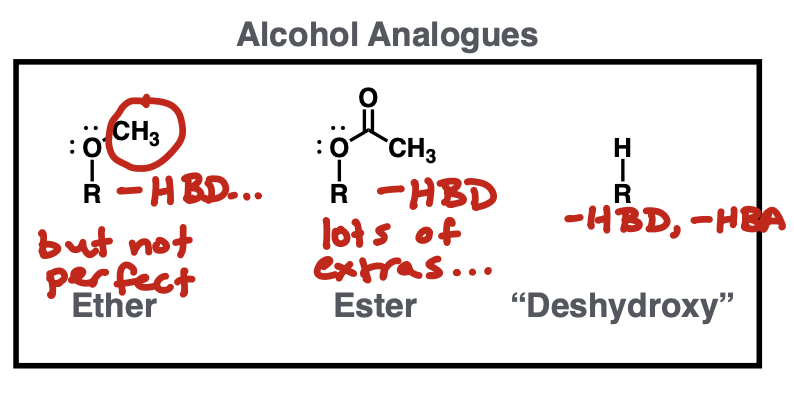
Alcohol analogues
ether, ester or just H
Can you rotate the C-N bond in amide?
No, electrons on N delocalize
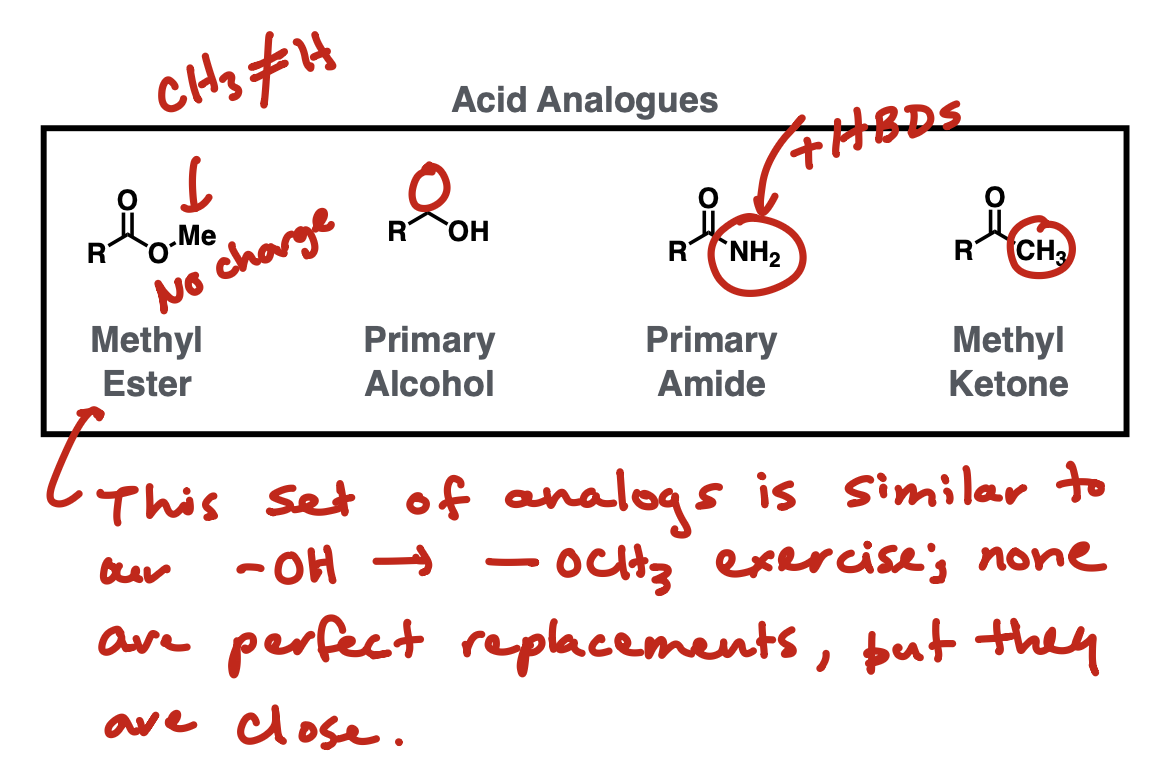
Acid analogues
Methyl ester, primary alcohol, primary amide, methyl ketone
Heteroaryls are dangerous because
they can undergo SNAR reactions (nucleophilic aromatic substitution)
Isostere
same shape or bulk
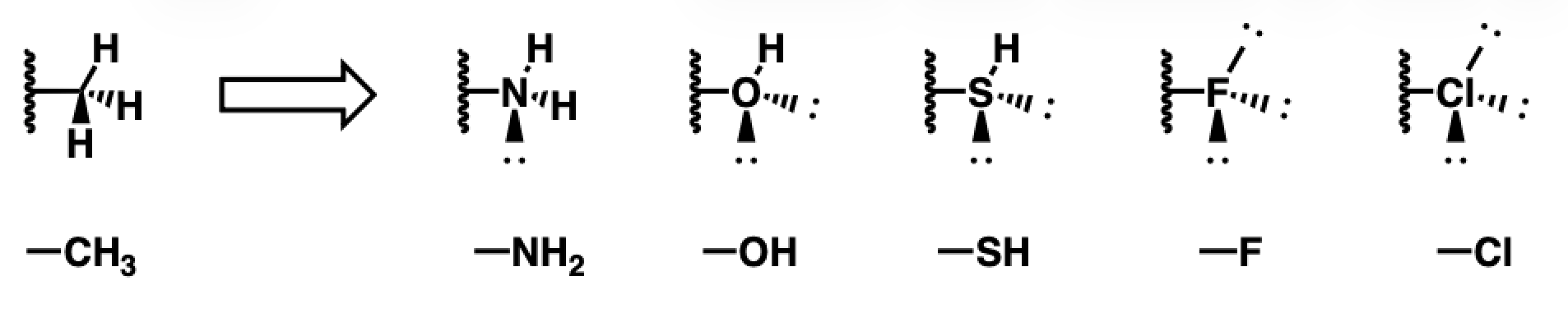
Isosteres for methyl
amine, alcohol, thiol, fluorine, chlorine
Isostere for isopropyl
Bromine
Isostere for butyl
Iodine
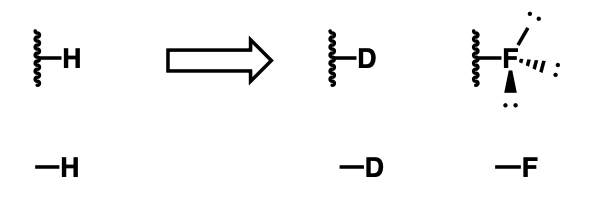
Isostere for proton
deuterium, fluorine
Bivalent Carbonyl Connector Isosteres
ketone, amide, ester, thioester
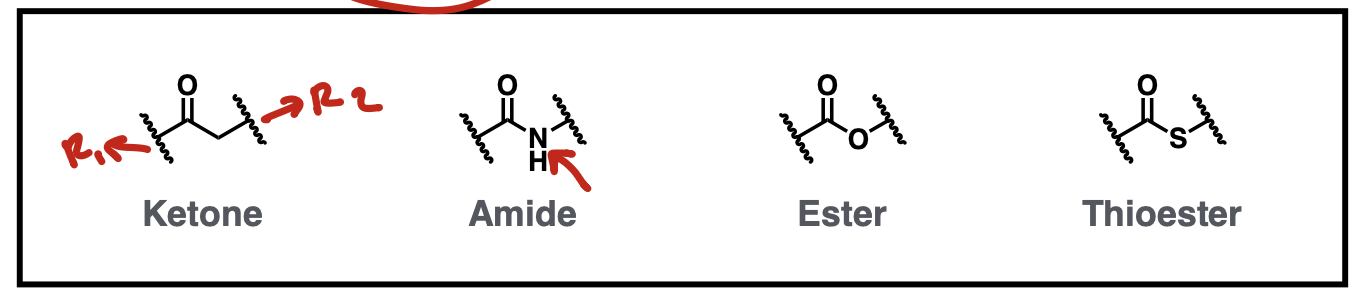
Bivalent one atom connector isosteres
methylene, amine, ether, thioether
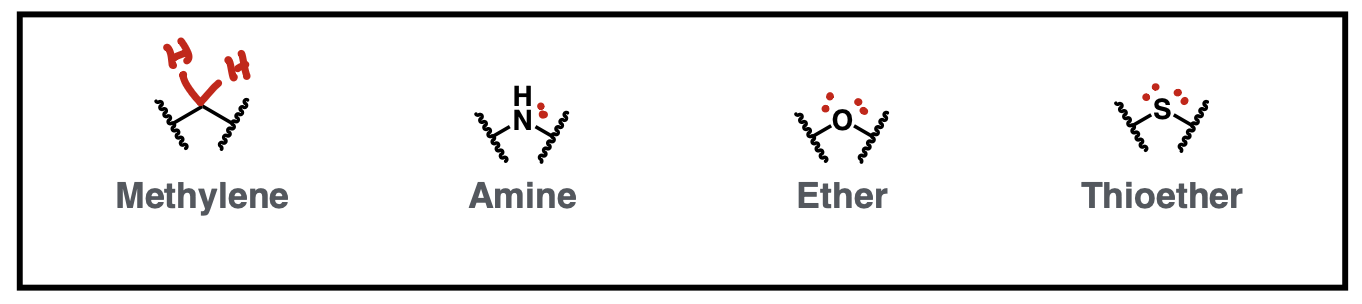
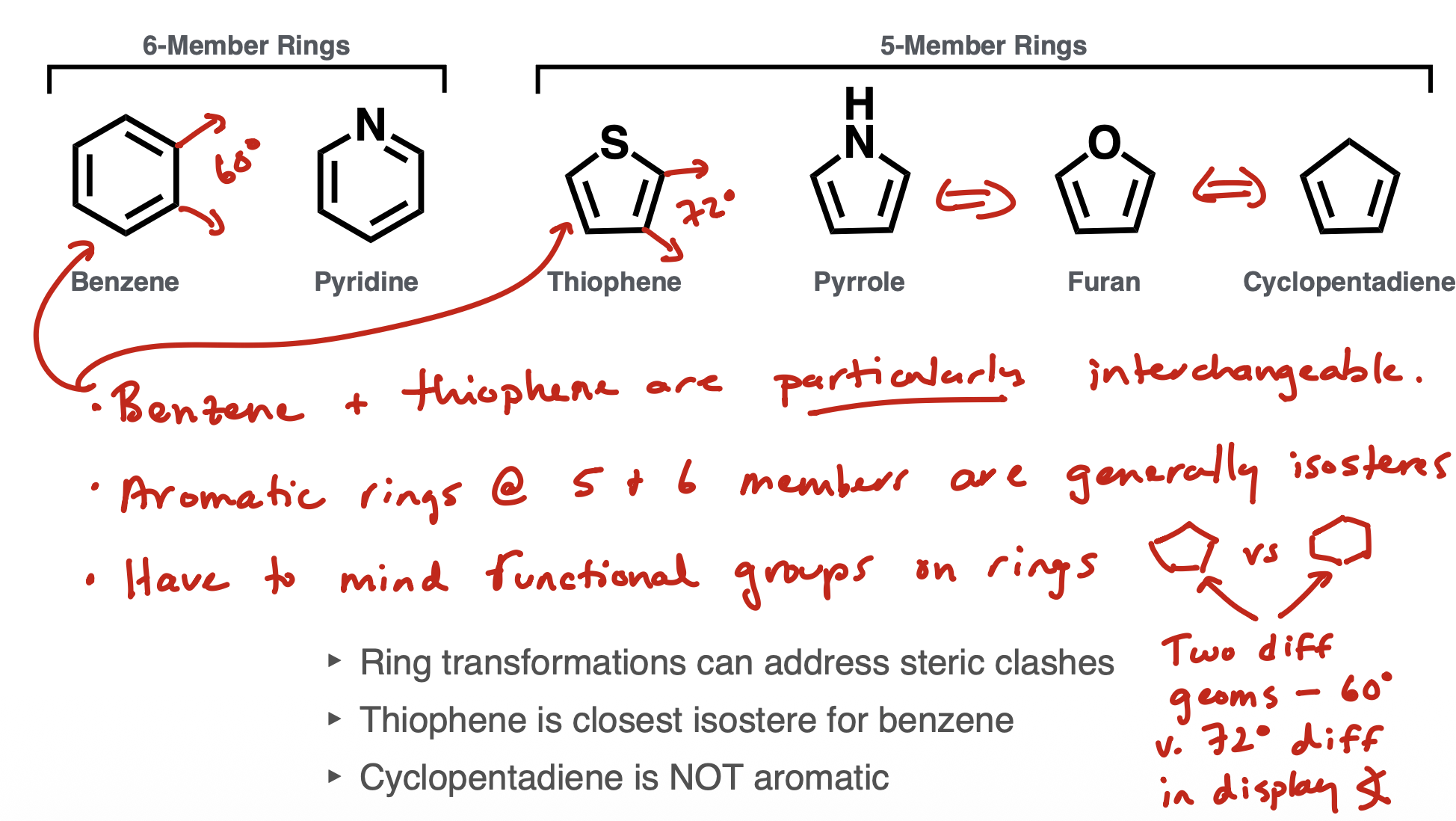
Ring isosteres
Benzene and thiophene are very interchangable
Others are pyridine, pyrrole, furan and cyclopentadiene
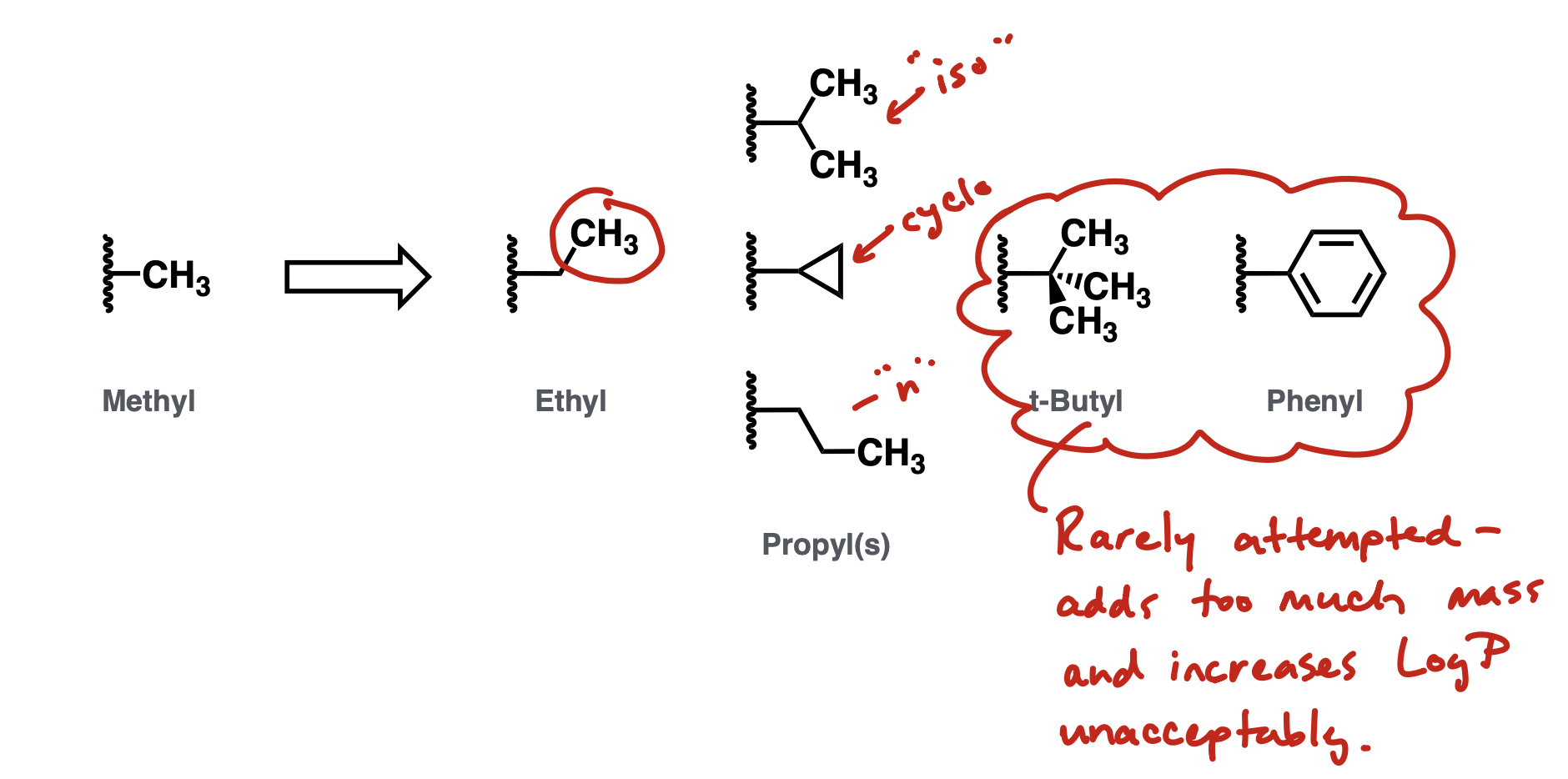
Exploring Hydrophobic pocket
cyclopropyl is especially interesting bc it investigates with minimal increase in logP and no Nrot
Simplifying Molecules for production
Eliminate unnecessary chiral centers (ease synthesis / scale-up)
Enantiomers are difficult to separate and must be tested for activity
Can do this by introducing symmetry
Why no floppy
Binding free energy depends on entropy, so more entropy means its not gonna wanna just snap into place
can fix with cyclization
Morpholine
the goat
aliphatic amine with pKa= 7.4
No exposed C-H for metabolism
Polar oxygen but not nucleophilic
Desolvation
There is an energetic penalty for having to remove H2Os from enzyme or ligand for binding
Bioisosteres
bioactive functional group replacements
Bioisostere of COOH
tetrazole, same pKa but much better cLogP
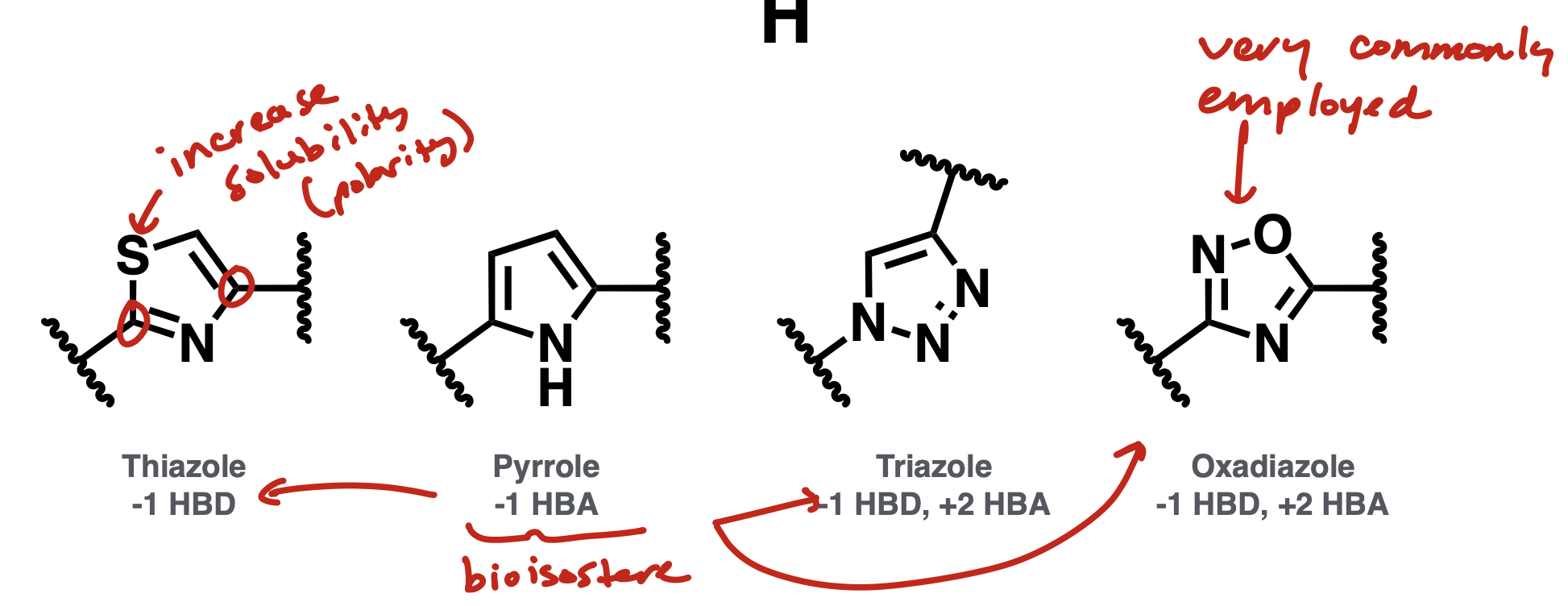
Amide bioisostere
pyrrole ring, thiazole, triazole, oxadiazole
Oligonucleotide drugs
For gene silencing
go in a ds-oligo and form RNA-Induced Silencing Complex (RISC) which bind to complementary mRNA and cleave it. Leaving it for degredation
Will need to modify phosphate backbone and sugars for solubility and selectivity
Gram positive and Gram negative bacteria
+ Bacteria that have a thick cell wall
- Bacteria have thin cell wall
Bacterial Cell wall Biosynthesis
NAG and NAM sugars bound to L-Ala + D-Glu (where the chain continues from Glu side chain) + L-Lys + D-Ala and crosslinks from there to the Lys behind.
Origionally it is a D-Ala + D-Ala that transpeptidaese acts on to crosslink em
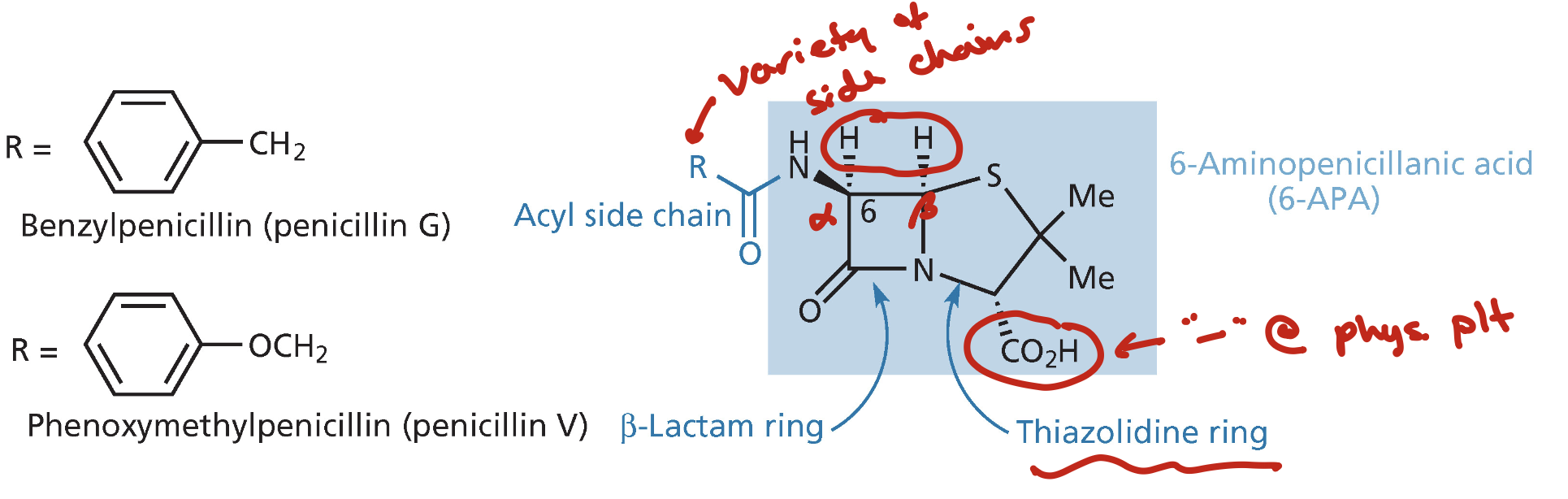
Beta-Lactam Antibacterials
4 membered ring with amide in it is a beta lactam ring
these inhibit cell wall biosynthesis by forming a covalent link to transpeptidase
Beta lactamases
inactivate beta lactam by cleaving the amide bond in the beta lactam ring
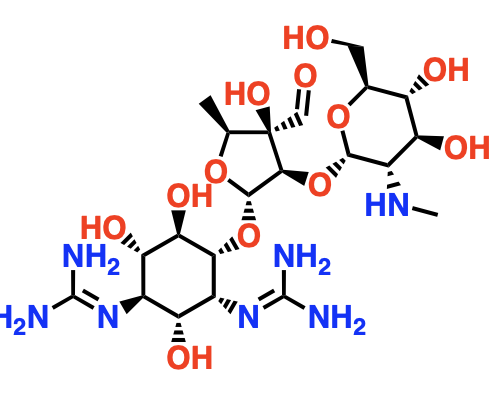
Aminoglycosides
Protein synthesis inhibitors for bacteria
They have alot of -OH and positively charged functional groups
They bind to the 30S/mRNA complex near the decoding site
Block translocation
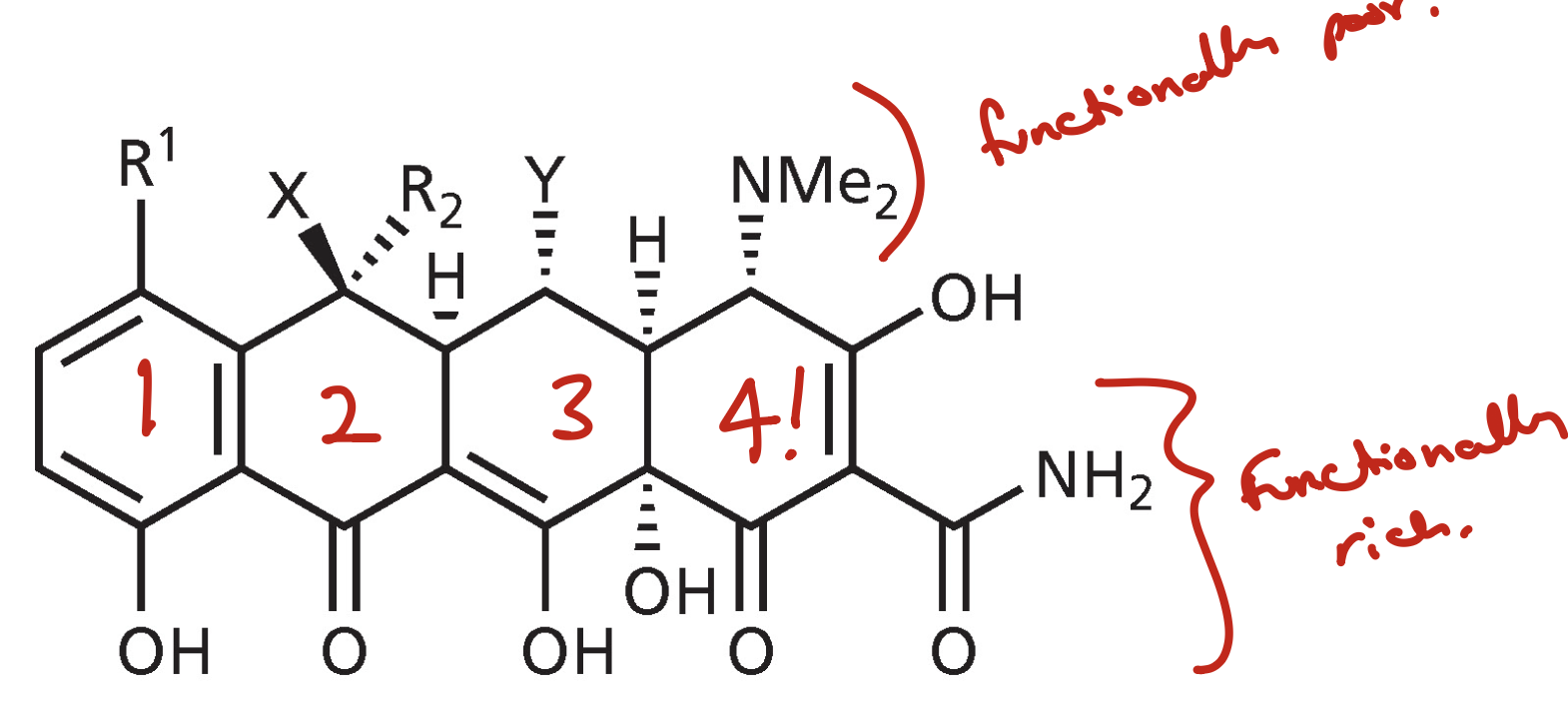
Tetracycline
Protein synthesis inhibitors for bacteria
They have a functionally rich and a functionally poor side
They bind to the decoding site and inhibit tRNA binding
Binds exclusively to backbone and nucleobases of rRNA
Fluoroquinolones
They stabilize topoisomerase which pause DNA in the ds break form
Quinolone group
Its a benzene ring attatched to a pyridine with a carbonyl

Viral Life Cycle (Influenza)
Adsorption
Fusion & Entry
vRNA Replication
Virion Assembly
Budding & Release
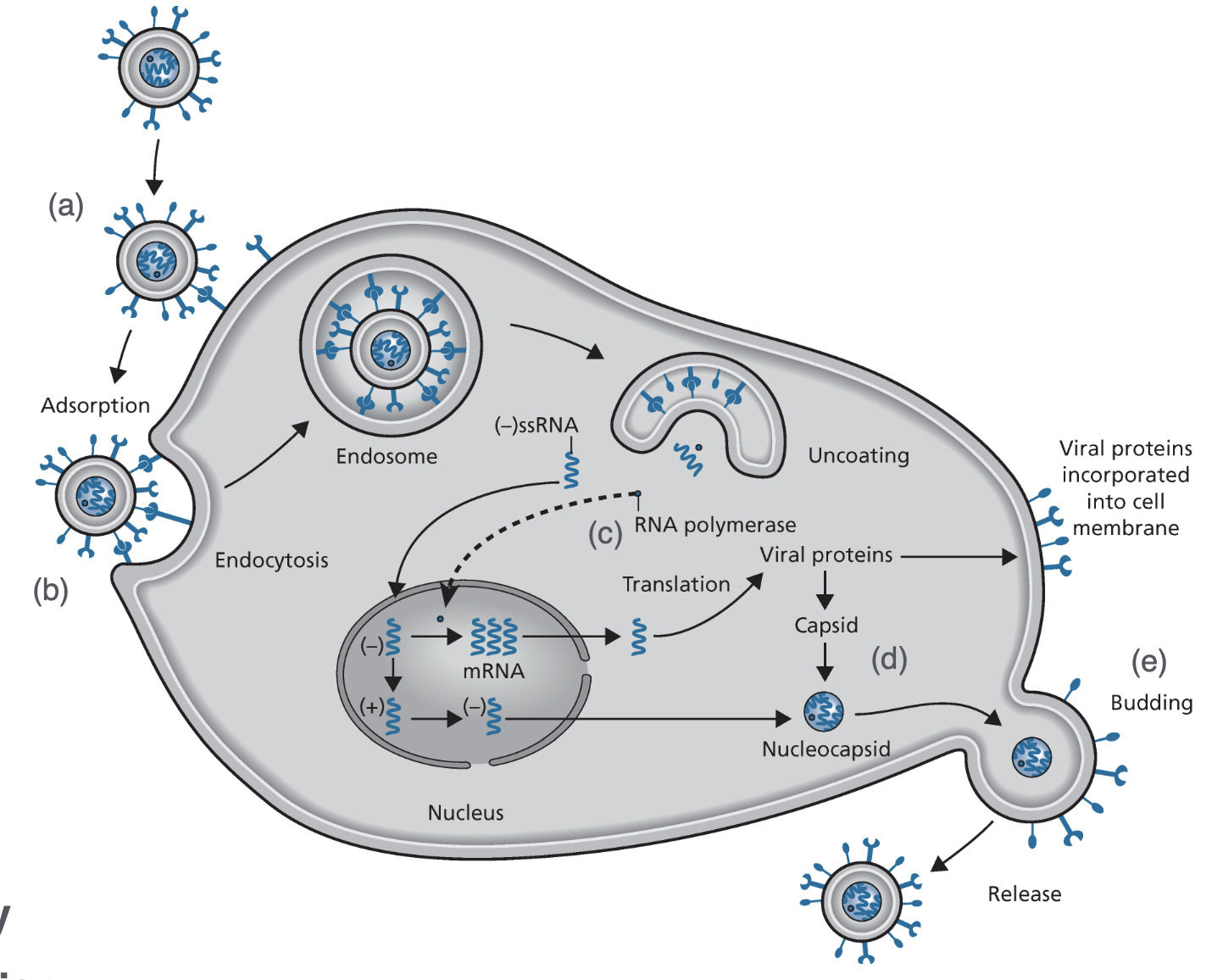
Viral Life Cycle (HIV)
Adsorption & Fusion
Reverse Transcription
Integration
Transcription
Polyprotein Synthesis & Processing
Virion Assembly, Budding, & Release
Nucleoside Reverse Transcriptase Inhibitors
They will mimic a nucleoside but will have chain terminating groups like an azide or sulfur instead of a 3’ OH
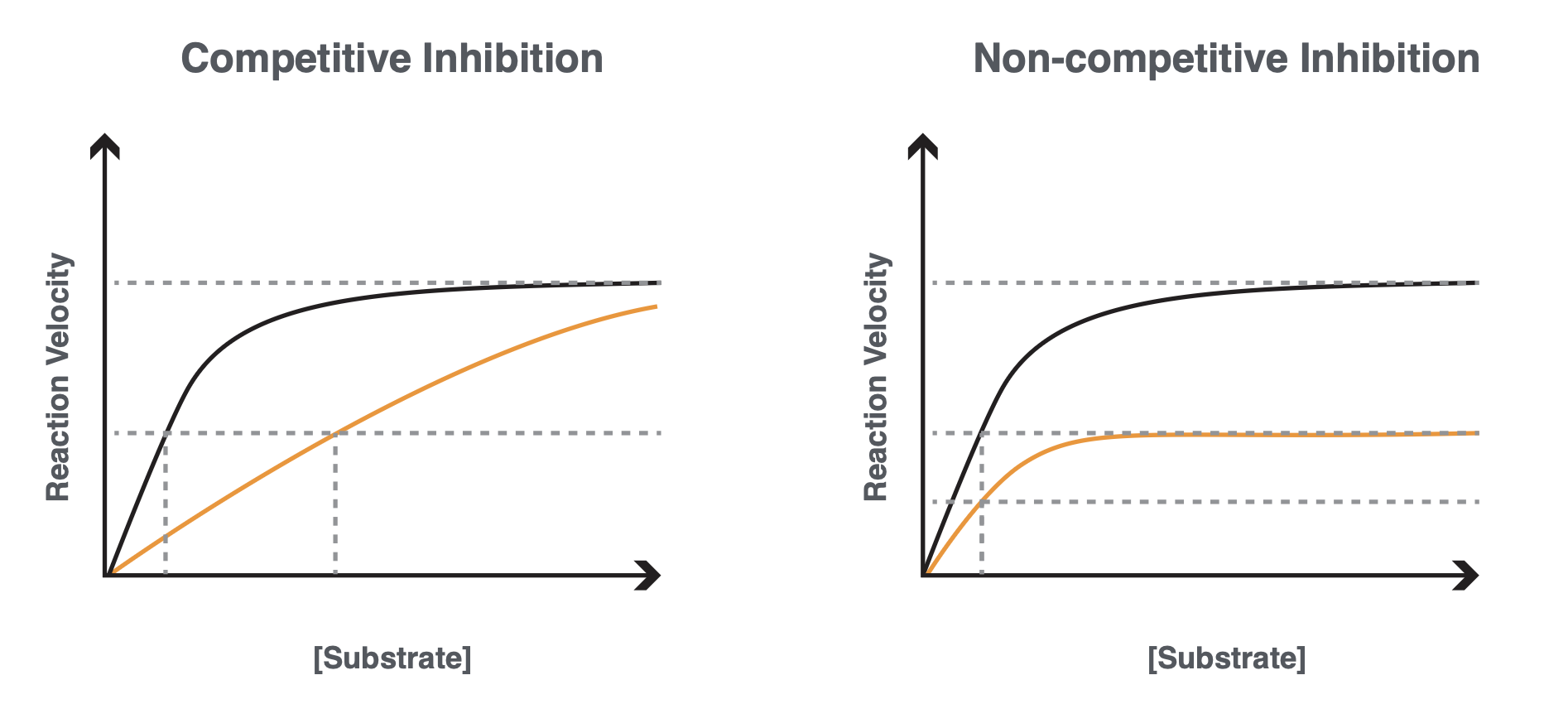
Non-Nucleoside Reverse Transcriptase Inhibitors
Allosteric binders to reverse transcriptase
they are non-competitive binders as a result of allosteric binding
Viral protease inhibitors
They make it so that the polyprotein remains inactive
Lenacapavir
Accelerates the capsid assembly and will yield malformed capsids that will clump everything together making even drug resistant capsids stuck using the druggable capsids
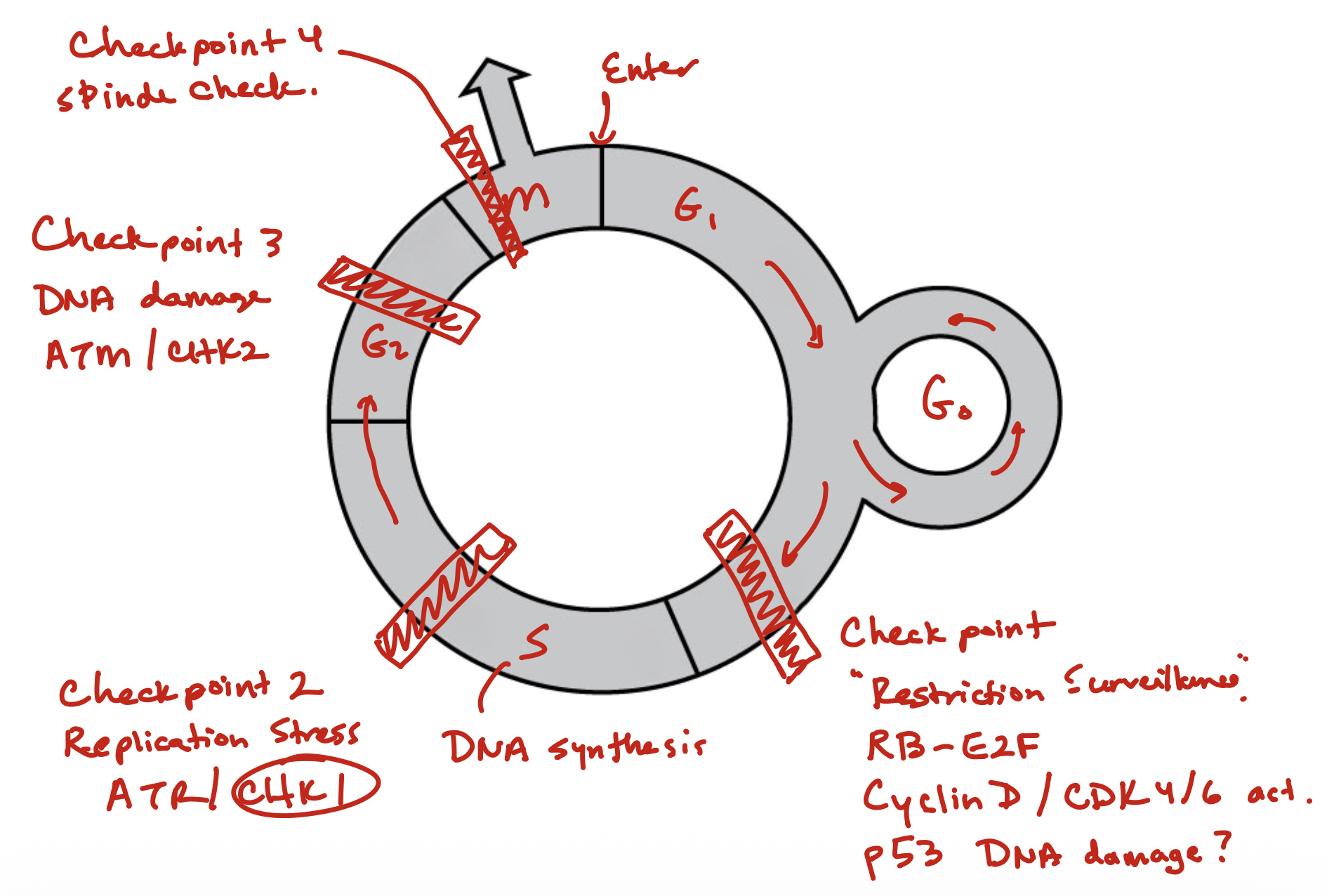
Cell Cycle
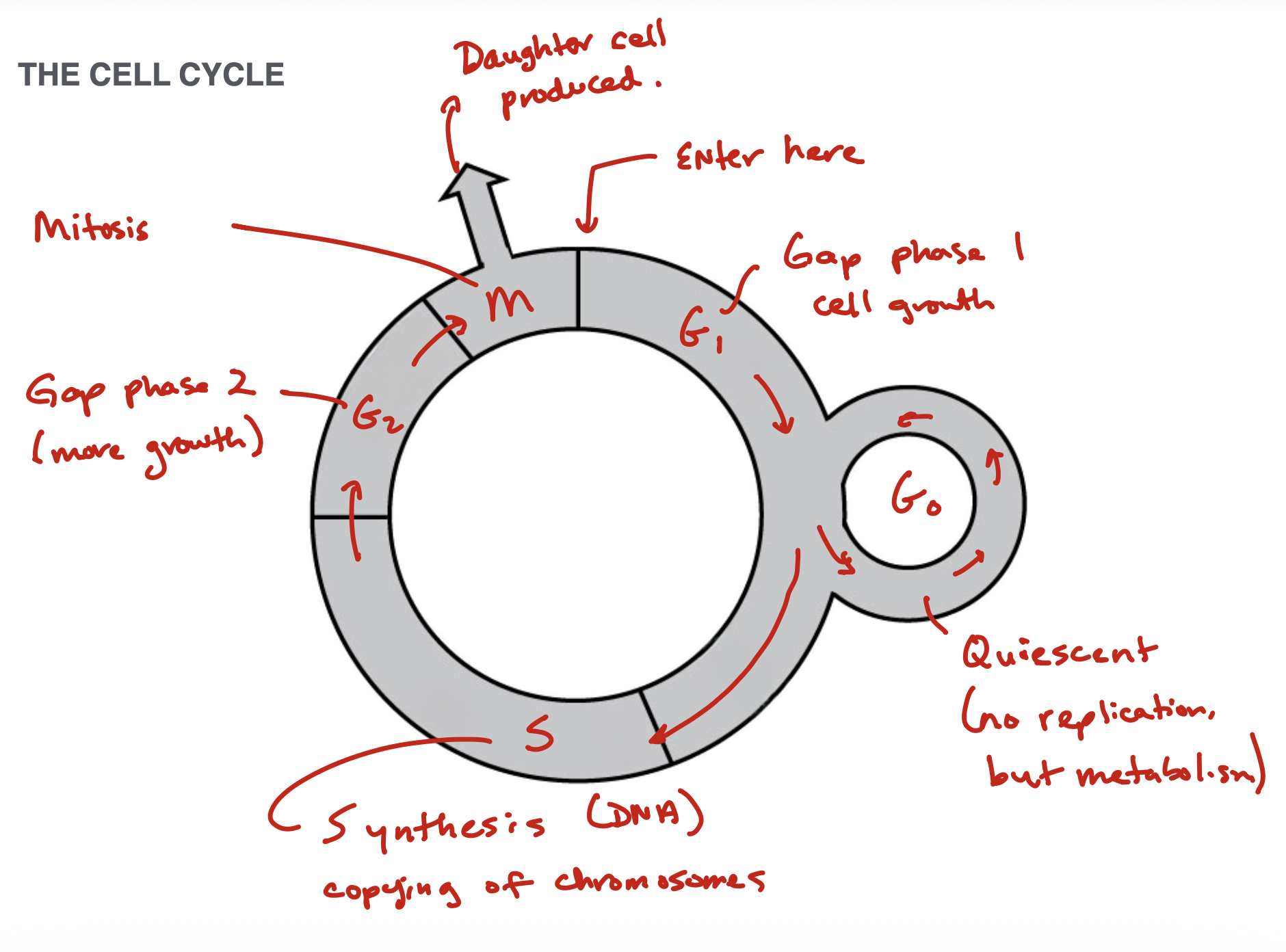
Checkpoints in cell cycle
Foot on the Accelerator
Epidermal Growth Factor will interact with its receptor (which is an RTK)
EGFR will autophosphorylate
Grb2 and SOS will interact with the phosphorylated EGFR and RAS which is bound to Grb2 and SOS will release a GDP
GTP will bind to Ras and activate Raf
Cascade down to activate Cyclin D and CDK4/6
This activates RB by phosphorylating it and releases the E2F which tells cell cycle to go
Foot on brakes
DNA Damage
will activate p53, then activate p21, which will inhibit Cyclin D
Transforming Growth Factor
Will bind to TGF beta receptor
SMAPS activate p15, p27, and p21
These inhibit CDK4/6
Foot on cell’s neck (cell death aka apoptosis)
Immunologic T cells
Inject Granzyme
increase permiability of mitochondria
Cytochrome C will come out of mitochondria, activating Casp 9
Casp9 activates Casp 3 and Casp 7.
Casp 3 and Casp 7 will lead to Apoptosis
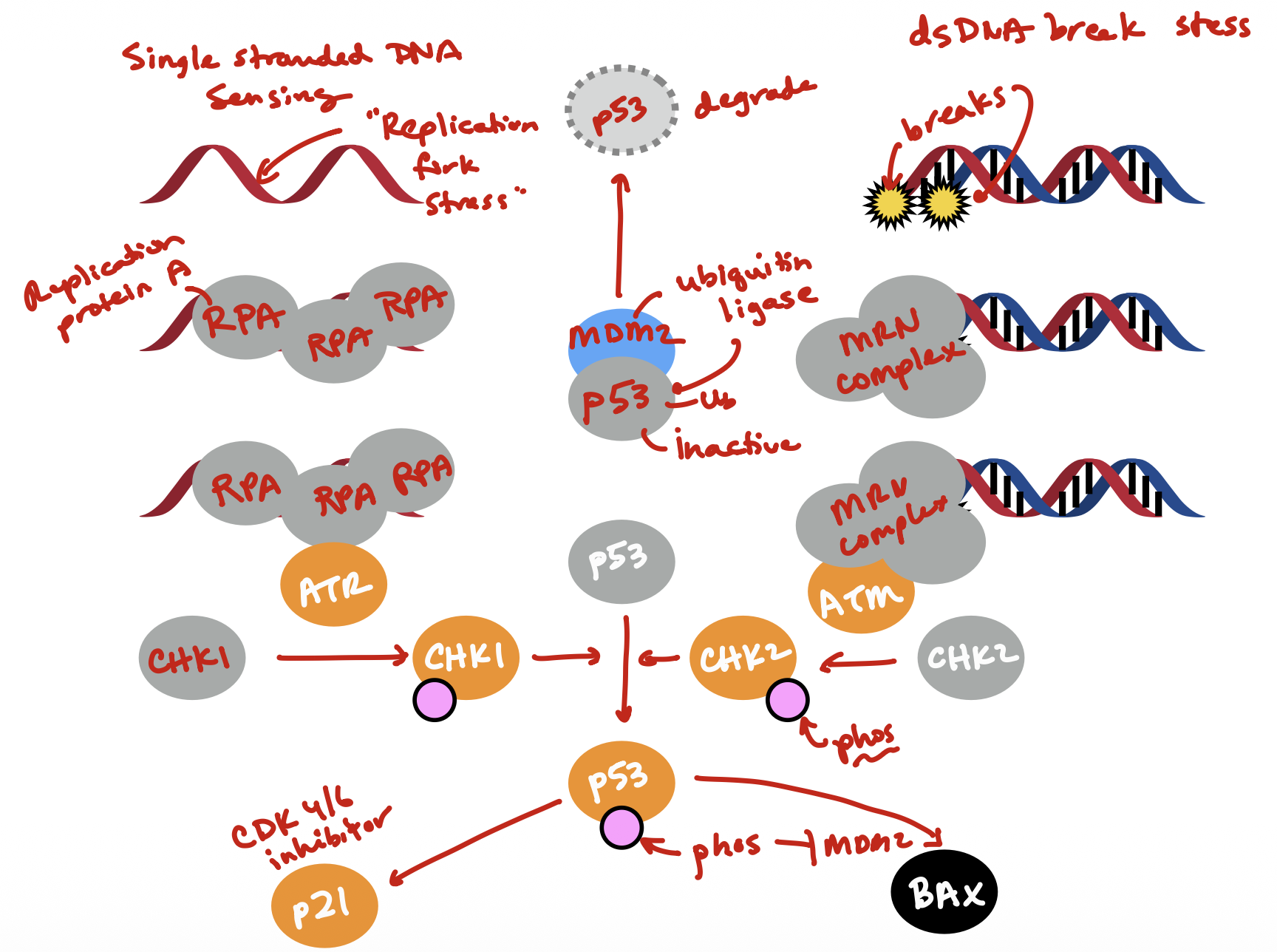
DNA Damage
p53 activates
BAX is activated by p53 and will increase permiability of mitochondria
…
Tumor Necrosis Factor
Will interact with its recepor (a trimer)
Activate scaffolding proteins that will activate Casp9
p53
aka Guardian of the Genome
EGFR inhibitors
will stop Ras from being activated and the cascade that leads to cell replication
cancer can counter by just upregulating EGFR
This is hard because Kinases are all very similar, so this can be doing bad things to yo body
Paclitaxel
Binds to tubulin and stabilizes it (freezing it)
WIll induce mitotic arrest and apoptosis
Quasispecies
Host cells are infected with multiple viral genomes
Monoclonal antibodies (mAbs) and antibody-drug conjugates (ADCs)
‣ Design antibodies to bind overexpressed receptors on cancer
‣ Antibody binding flags cell for immunological disposition
‣ Conjugate cytotoxin to mAb = ADC; silver bullet to cancer cells
What are the liabilities and optimizations of peptide-derived drugs
Liabilities:
human proteases potentially cleave the drug
lots of HBA, HBD and high MW
Optimizations:
steric shields, isosteres
removing chirality