ESS W2 L2 biogeochemistry
1/43
Earn XP
Description and Tags
biogeochemistry: carbon and nutrient cycles
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
Biogeochemistry:
How the biosphere functions
Life support systems of the planet
Biological, geological, and chemical processes cycling elements through the earth system
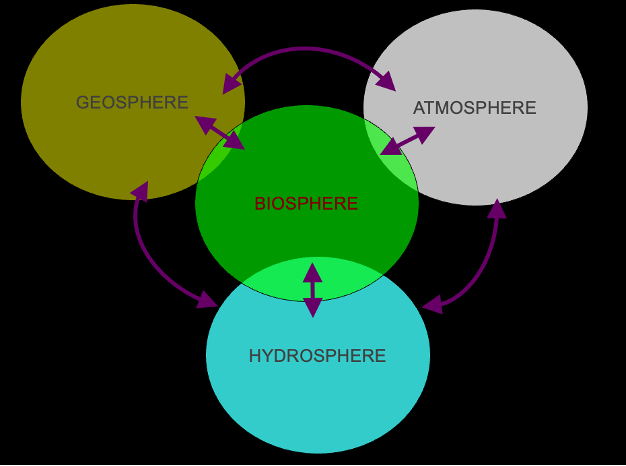
Biogeochemical cycles:
carbon cycle
nitrogen cycle
phosphorous cycle
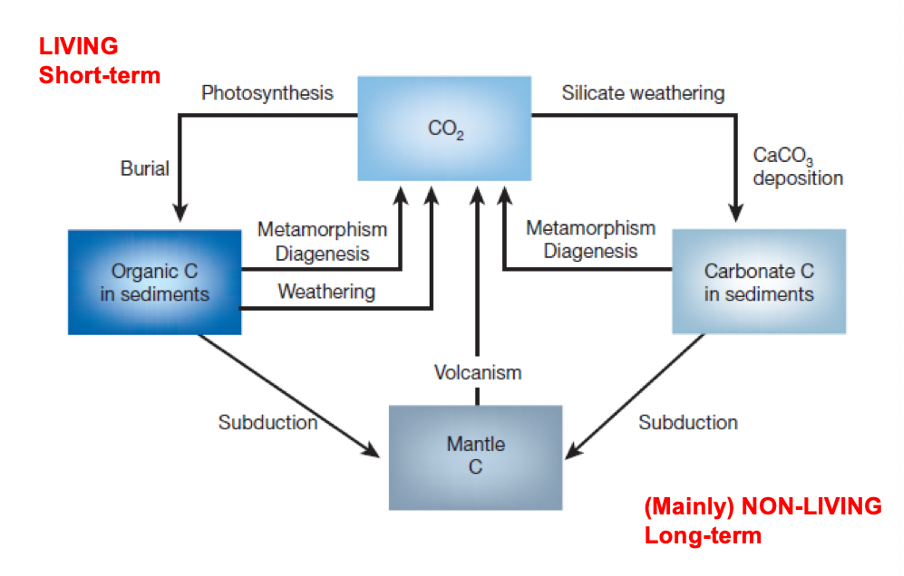
The carbon cycle:
Left – living short term
Right – long term, non-living
Processes within carbon cycle happen on these different time scale
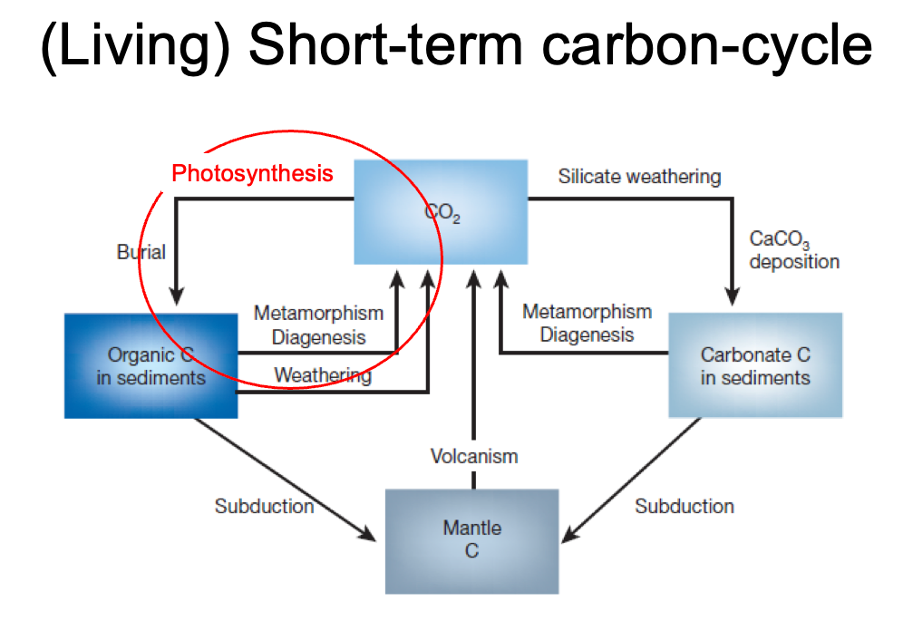
living short term carbon cycle
Short term living part is photosynthesis
Plants draw co2 out of atmosphere and convert into sugars – glucose, carbohydrates. Also oxygen
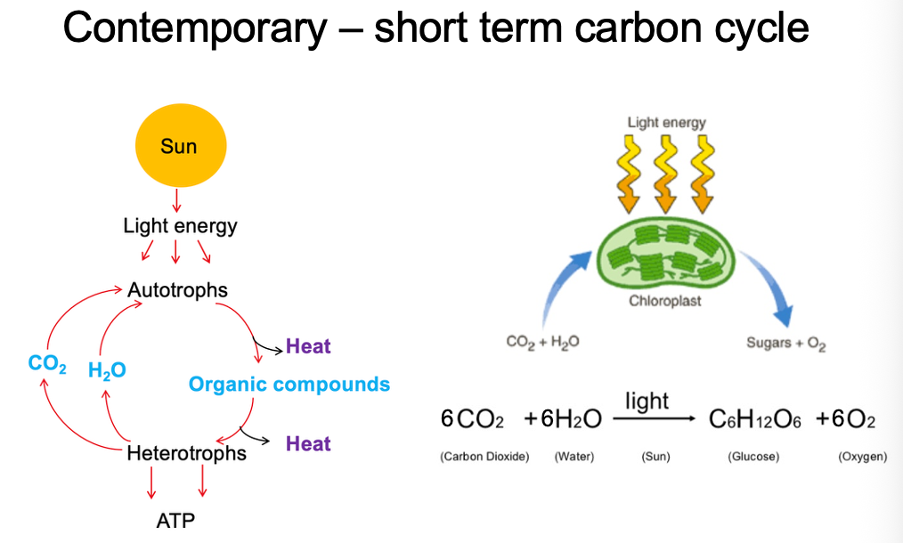
contemporary short term carbon cycle
Heterotrophs produce carbon dioxide and water which is needed by autotrophs
Light/ solar energy converted by chloroplasts/ chlorophyll into chemical energy by ATP. Take carbon dioxide and water and convert it into glucose and oxygen
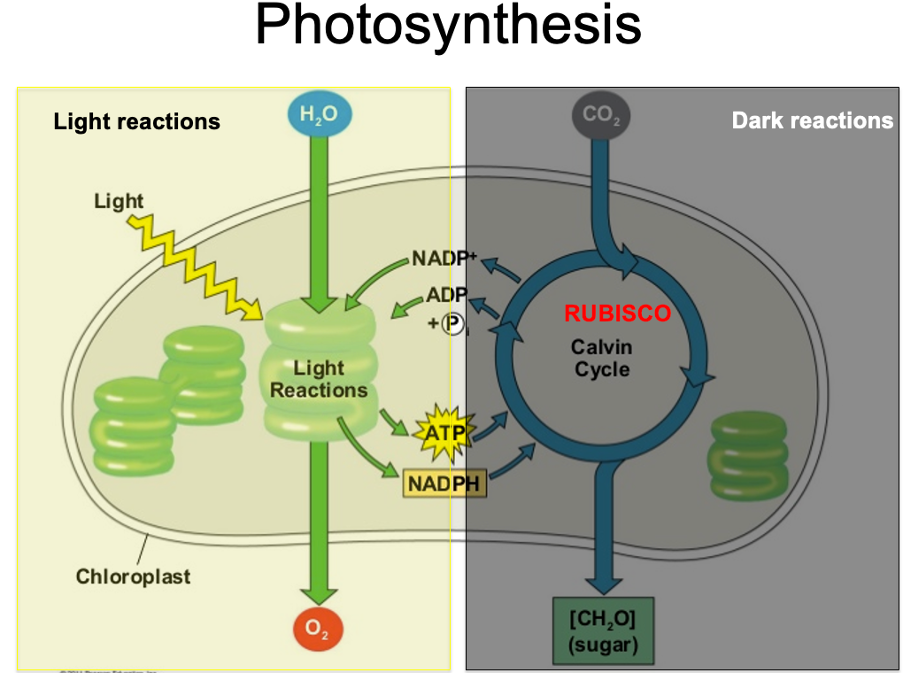
photosynthesis
Enzyme called rubisco draws in carbon dioxide into plant biomass. Essential in converting it into sugar
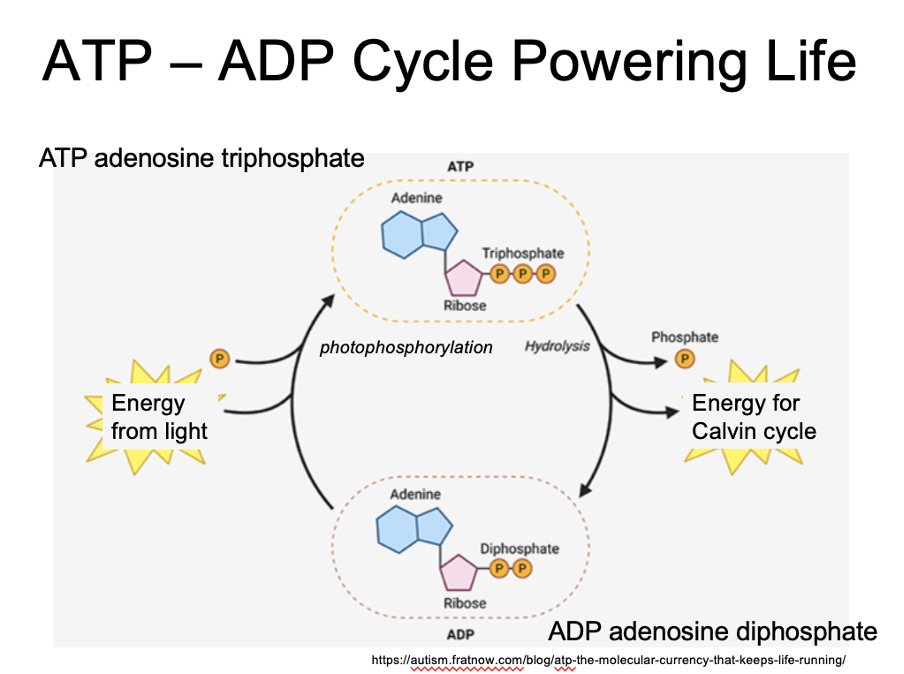
ATP - ADP cycle powering life
Photosynthesis
ATP – three phosphate groups. Energy gets stored in bond between second and third group. When that bond is broken, energy gets released
Atp becomes adp.
They cycle through going between atp and adp as they pick up a phosphate then bond is broken
P = phosphorous
Contemporary organic C cycle:
The biosphere is estimated to contain 3170 gigaton (GT)s of carbon, broken down into:
Soils = 2500 GT
Living plants and animals – 560 GT
Atmosphere = 800 GT
In the atmosphere, there are 100,000s GT of O2 in the atmosphere
For every molecule of CO2 fixed one molecule of O2 is released
6CO2 + 6H2O → C6H12O6 + 6O2
So where is the rest of the carbon if not in the biosphere?
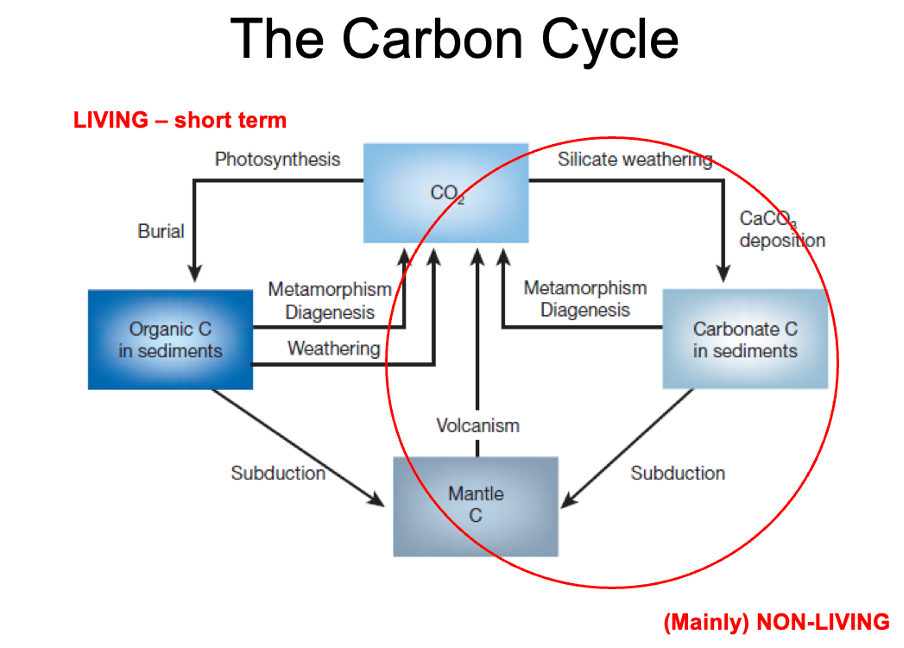
long term carbon cycle
The long term carbon cycle is important to answer it – explains movement of carbon into geosphere
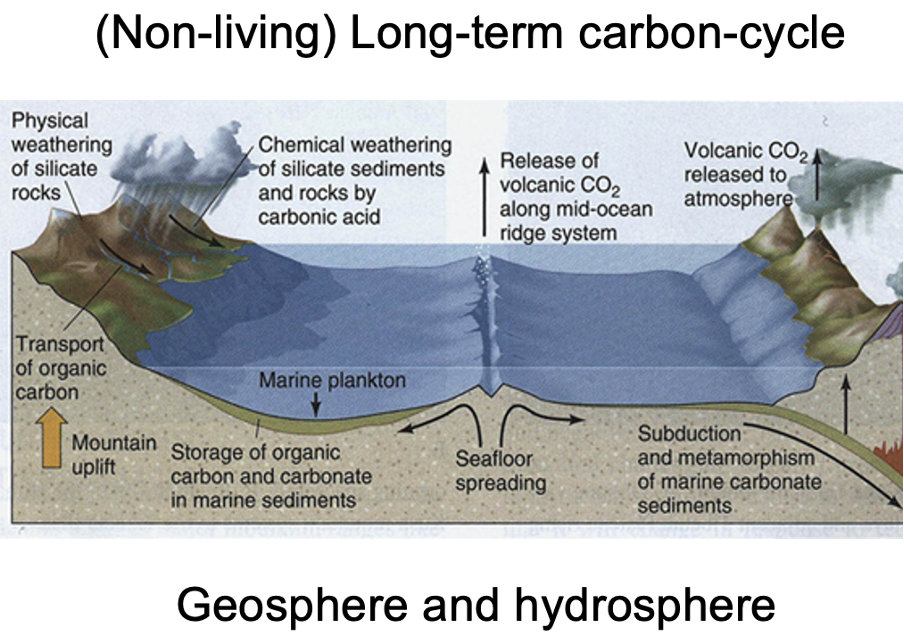
non living carbon cycle
Carbon dioxide in the atmosphere is released by volcanoes (terrestrial and along ocean ridges on the sea floor – where it is spreading) releasing CO2
From lithosphere/ geosphere
Chemical weathering pulls down carbon from the atmosphere to the lithosphere. Atmospheric carbon reacts with water. Forms a weak acid (carbonic acid) that dissolves rocks into bicarbonates. Transport to oceans where marine organisms use calcium carbonate to make shells.
Most of the calcium carbonate in oceans is made by calcifying (shell building) organisms (corals, plankton like coccolithophores).
When they die, these organisms sink to the seafloor, where over time, layers of shells and sediments become compressed together and turn to rock, capturing carbon in the bedrock (limestones, or marble – a form of metamorphic limestone).
The eruption of volcanoes and seafloor spreading releases carbon dioxide and the process starts again.
Peatlands: terrestrial C accumulation and burial:
“Peatlands store vast quantities of carbon – 'locking in' an estimated 3.2 billion tonnes in the UK alone.
Where peat continues to form this helps to offset the effects of human activities (such as fossil fuel burning) that are raising CO2 levels in the atmosphere, leading to climate change.”
Peats from ~ 1m / 1000 years
Carbon sinks due to waterlogged conditions and little oxygen. Also quite acidic where organic matter doesn’t decompose
Help keep carbon locked in geosphere
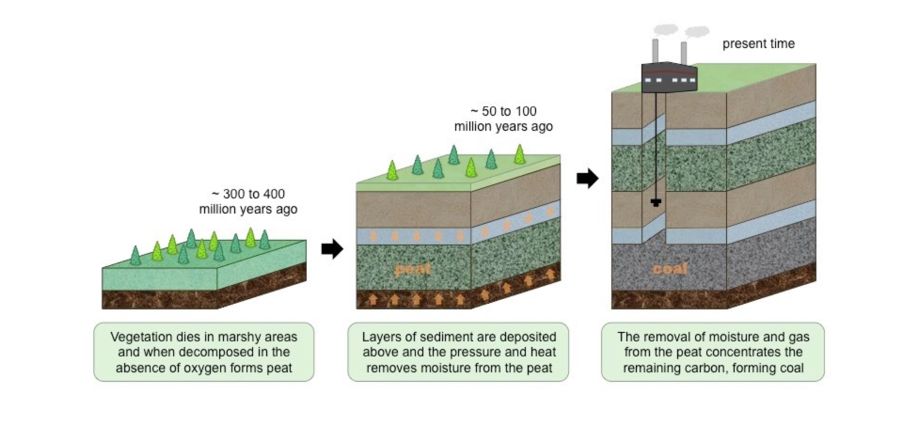
Geosphere: carbon storage:
20 % of carbon-containing rock contains carbon from living organisms (organic carbon) that have deposited in layers of sediments.
Over millions of years, heat and pressure will result in the formation of sedimentary rocks, e.g. shales,
However, where dead organic carbon accumulates faster than it can decay, carbon-rich layers of oil, natural gas or coal form.
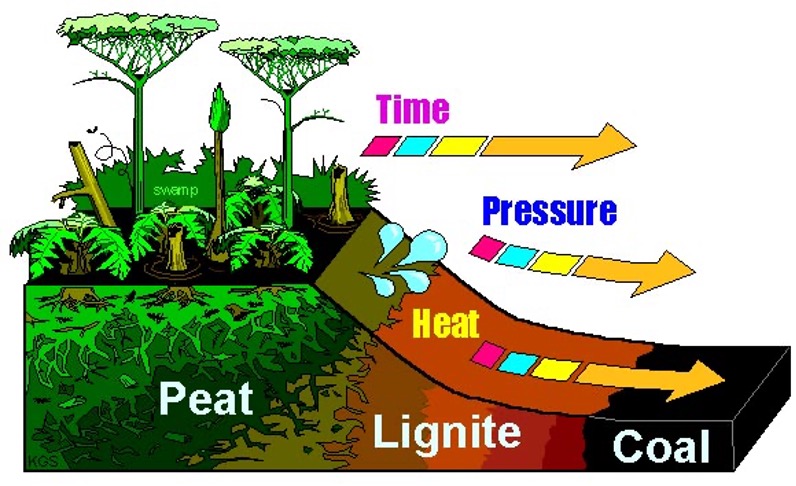
Terrestrial carbon burial: coal:
Over time plant matter accumulates
E.g. from peat
Gets compressed and heated up
Marine carbon burial: oil and gas:
occurs in ocean – not a terrestrial process
Tiny organisms – phytoplankton - autotrophic
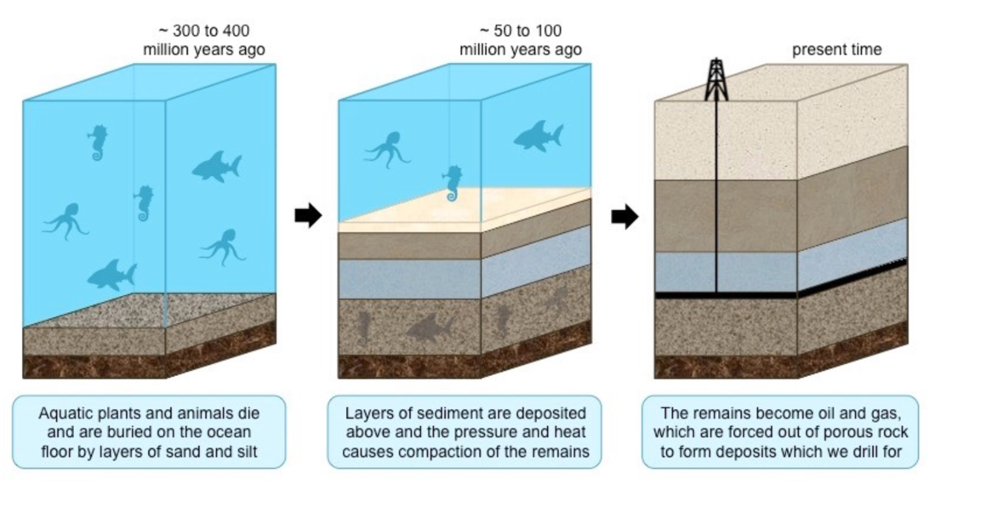
aquatic plants and animals die and are buried on the ocean floor by layers of sand and silt
layers of sediment are deposited above and the pressure and heat causes compaction of the remains
the remains become oil and gas which are forced out of porous rock to form deposits which we drill for
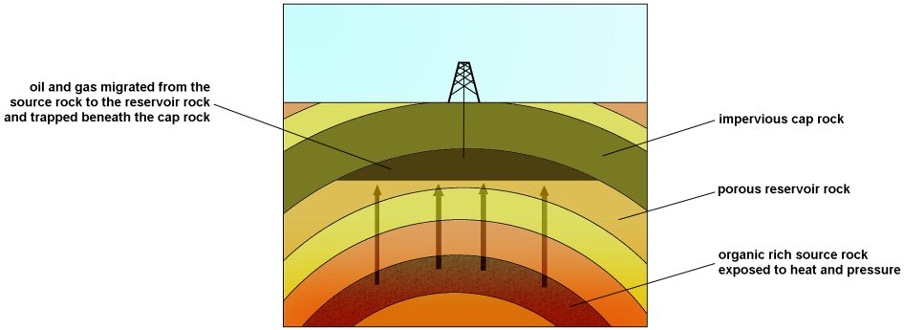
Oil and gas:
Marine sediments: zoo and phytoplankton - buried under anoxic (no oxygen) conditions.
Sedimentation increased the pressure and temperature
Organic matter - converted into oil & gas
Get buried under anoxic conditions
Sedimentation increased the pressure and temperature
Organic matter – converted into oil and gas
Burial of organic matter:
Huge amounts of carbon in these sediments (100,000s of giga tonnes)
Burying the carbon that became fossil fuels was key to advanced animal life on Earth as allowed oxygen to build up in the atmosphere.
Without burial, decomposition would have released almost all the carbon as CO2 and consumed the oxygen in the atmosphere
Link between biological and geological processes is key = Biogeochemistry
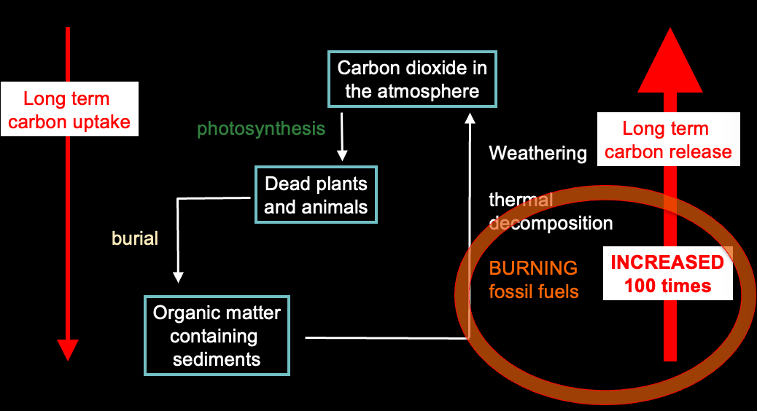
Human influence on the carbon cycle:
Humans releasing carbon thats been locked away for millennia in geosphere and releasing it into the atmosphere.
Causes an imbalance in carbon cycle
when last uk coal fired power station closed
30th September 2024
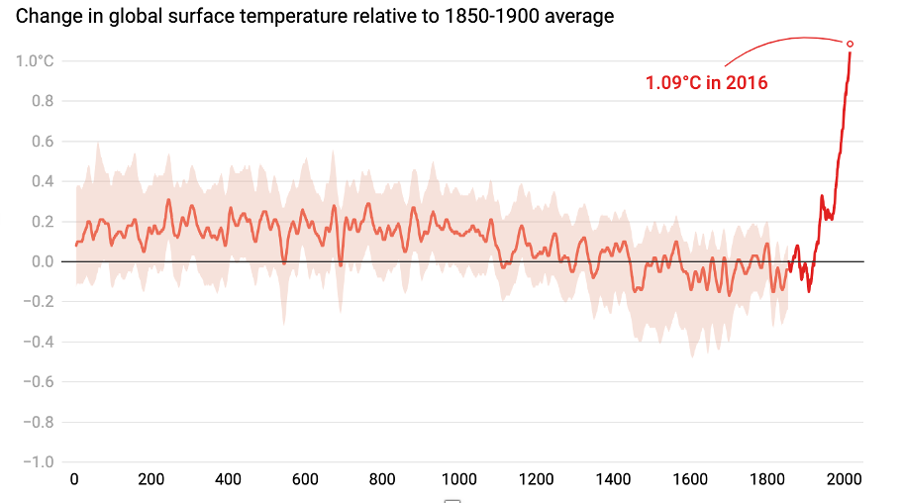
CO2 and global warming:
Known as the Hockey stick graph
Shows long-term cooling trend interrupted by rapid rise in temperature during the 20th century
Human –produced green houses gases (including CO2) are causing global warming
Change in global surface temperature over time.
Data from 1902-1998 – observed data
Before that – reconstructed from proxy records: e.g., ice cores, tree rings
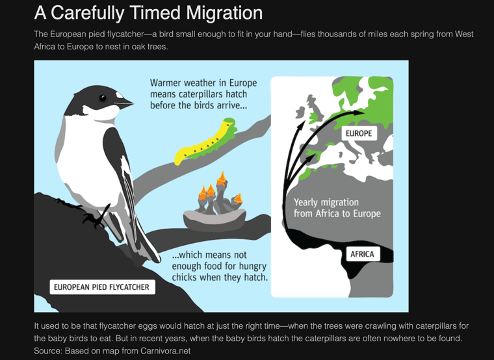
How is global warming impacting the biosphere:
- Some examples:
Extinction- rates higher than natural, particularly impacting vulnerable species (limited range or isolated populations).
Loss of habitat
Ecosystem changes (e.g,. ocean acidification)
Range shifts - forcing plants and animals to migrate to escape temperature increases.
Timings of bird migration and caterpillar hatchings not working properly due to warming but not having resources available for offspring
Nitrogen cycle:
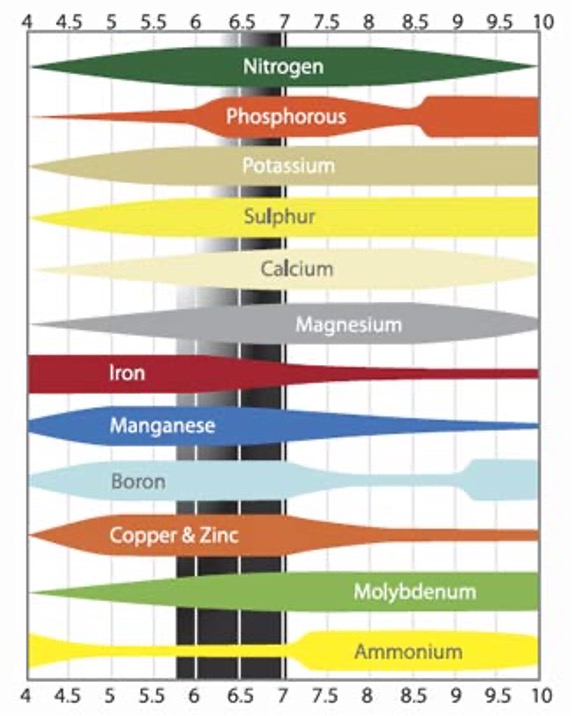
Key nutrients for life:
Nitrogen
Proteins, nucleic acids (DNA)
Phosphorous
Nucleic acids, ATP, membranes
Potassium
Osmosis, transport
Sulphur
Proteins
Calcium
Membrane, and enzyme function
Magnesium
Chlorophyll
Iron
Chlorophyll synthesis, oxygen transport
Others:
Sodium, manganese, boron, copper, zinc, molybdenum
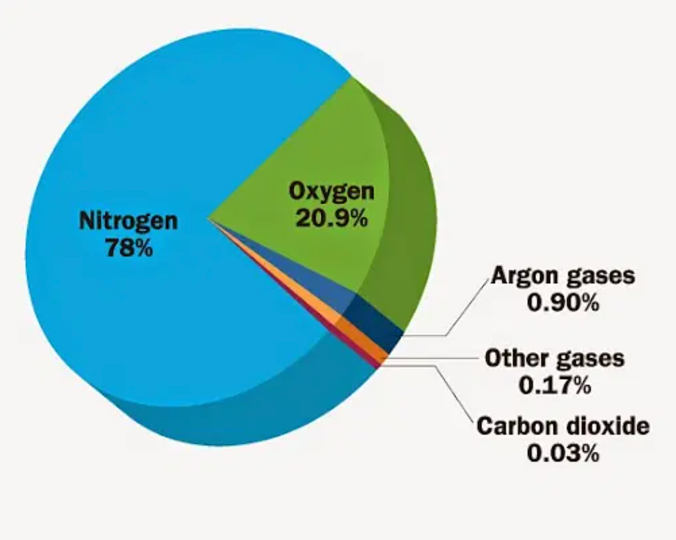
nitrogen gas abundance
Most abundant gas in atmosphere – 78% air
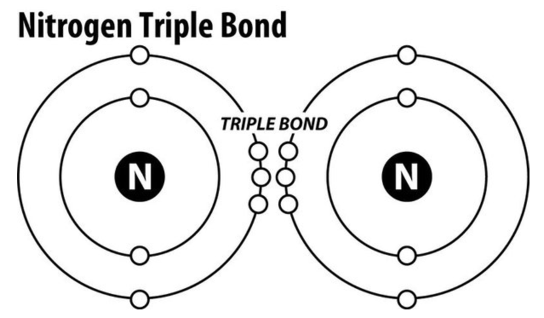
nitrogen triple bond
In atmospheric form – 2 nitrogen atoms bonded together – covalent bond
Very strong bond
Plants and animals can’t break and use it
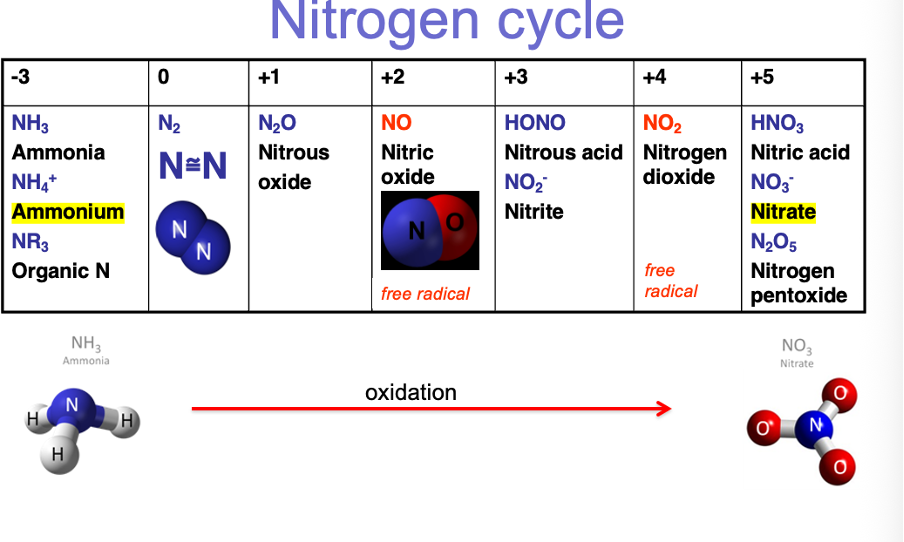
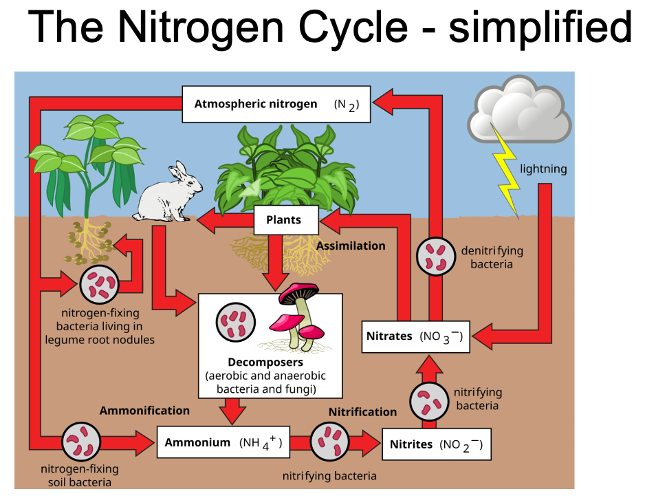
Nitrogen cycle: soil microorganisms:
Conversion can only happen by nitrogen fixing bacteria (in soil)
Bacteria take nitrogen from atmosphere and fix it into soil making it available to plants
Atmospheric nitrogen into ammonia
Ammonia can pick up extra nitrogen atoms in soil to make ammonium
Ammonium taken up by plants to make dna etc.
Lightning and wildfires play important role in nitrogen cycle as the energy can break apart the atmospheric nitrogen bonds.
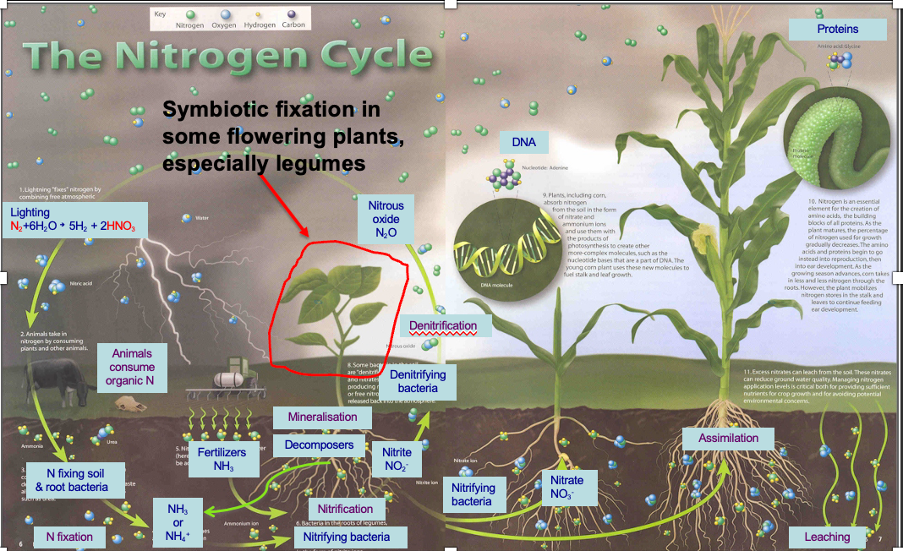
ammonium in nitrogen cycle
Ammonium is positively charged (soil is negatively charged)
They are attracted to soil particles so are retained din soil – not vert mobile
Nitrate is negatively charged – repelled by soil particles – is highly soluble – leeches out of soils
As they aren’t retained well humans add them back in
Human activity and the N cycle:
Human activity is having a major impact on the nitrogen cycle: fertilizers
We can now make liquid nitrogen (nitrate) from N2 (gaseous nitrogen)
Nitrogen fertiliser (using chemicals using the Haber process)
Has become an important part of the N cycle
80% of the nitrogen in your body comes from an artificial fertiliser source.
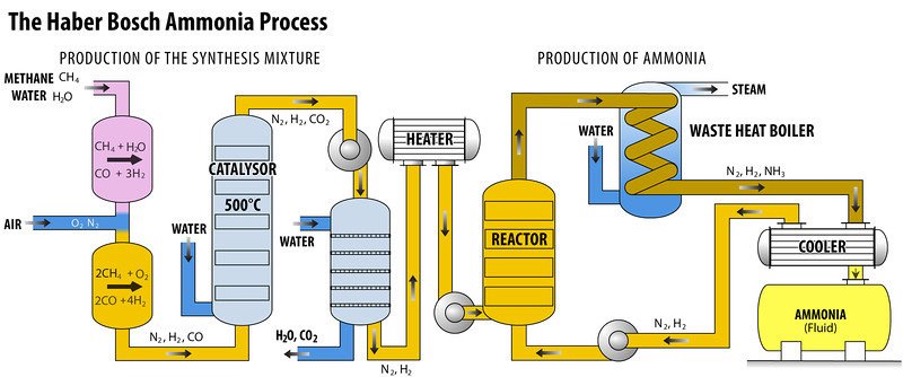
The Haber process:
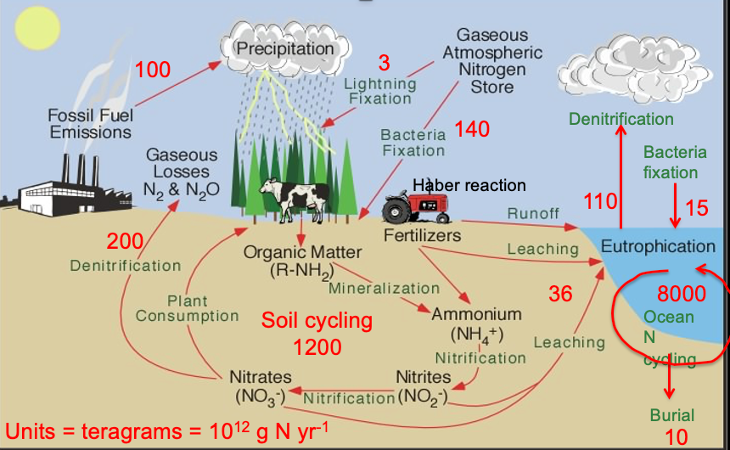
Nitrogen accumulation in oceans:
quantities of nitrogen
Accumulates in soils/ lakes
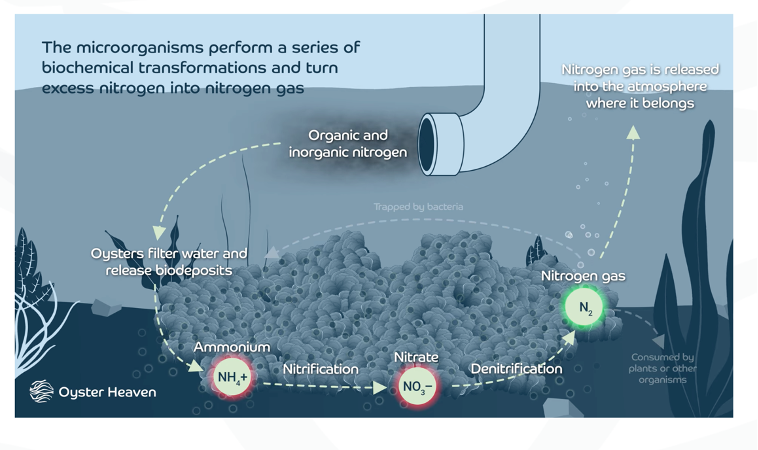
Eutrophication:
Causes oceans to acidify
Nature based solution using oyster reefs to remove nitrogen from oceans
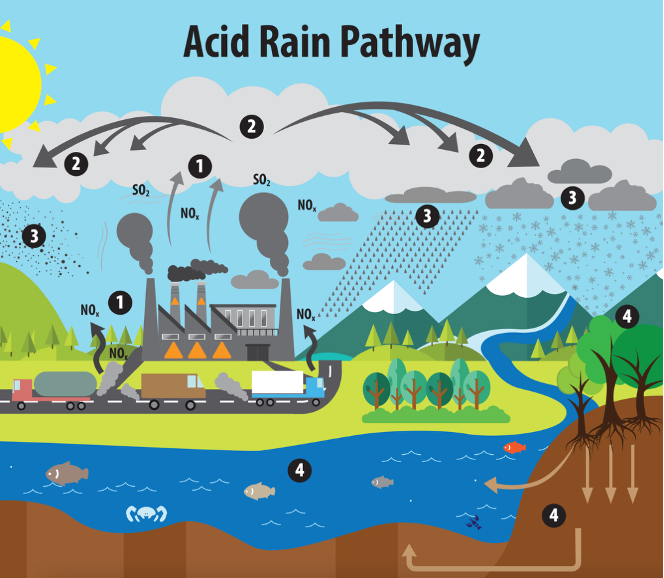
Acid rain:
1 - Emissions of SO2 and NOx are released into the air
2 - pollutants are transformed into acid particles that may be transported long distances
3- These acid particles then fall to the earth as wet and dry deposition (dust, rain, snow, etc.)
4 - may cause harmful effects on soil, forests, streams, and lakes.
Phosphorous cycle:
Phosphorous: PO3-3
Essential in living organisms:
phospholipids in cell membranes
DNA and RNA
ATP, NADPH
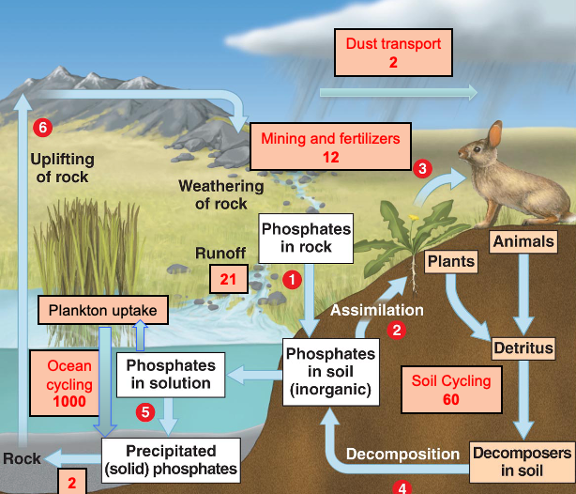
Phosphorous cycle: the slow cycle:
Units = teragrams = 1012 g P yr^-1
Phosphorous is a very slow cycle
Saharan dust and the phosphorous cycle:
Dust blown off the Sahara crosses the Atlantic and fertilises the Amazon – where amazon gets its phosphorous from
The most productive ecosystem on the planet is dependent on one of the least productive for key nutrients
Amazon rainforest – phosphorous poor – doesn’t have that bedrock. Heavy rainfall means that P is lost rapidly
Nitrogen versus phosphorous:
Similarities
Effective recycling within terrestrial ecosystems
Transport to oceans through rivers
Human addition through fertilizers
Long-term burial in sediments
Nitrogen versus phosphorous: differences
No gaseous phase in phosphorus cycle
Main inputs differ
Biological nitrogen fixation
Geological rock weathering
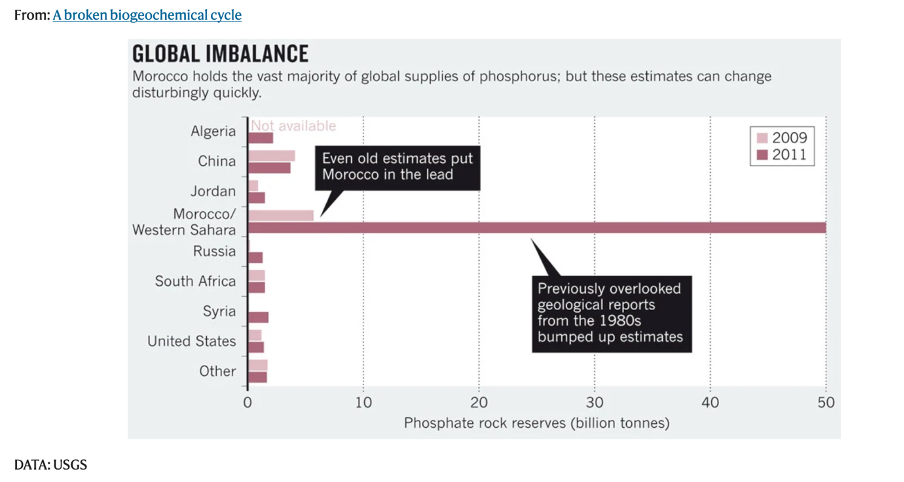
What happens if all the phosphorous is mined:
Summary:
Biogeochemistry = study of the biological, geological and chemical processes cycling elements through the Earth System
Carbon, nitrogen and phosphorus cycles are essential to life on Earth
All cycles are being affected by human activity