Topic 6: Carbohydrates: Structure, Function, and Biological Significance
1/99
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
100 Terms
Why Study Carbohydrates?
- Most abundant biomolecules on Earth
- Major global carbon reservoir
What are carbohydrates central to?
- Energy metabolism
- Structural biology
- Cell recognition
- Extracellular matrix organization
Photosynthesis and Carbon Fixation
- Atmospheric CO₂ converted into carbohydrates
- Catalyzed by: Ribulose-1,5-bisphosphate carboxylase/oxygenase (RuBisCO)
- Occurs in chloroplasts
RuBisCO
- Most abundant enzyme on Earth
~50% of total chloroplast protein
Carbohydrates in the Biosphere
- Plants, cyanobacteria, algae synthesize carbohydrates
Non-photosynthetic organisms
Depend on carbohydrates as carbon and energy source
Energy flow through ecosystems depends on what?
carbohydrate synthesis
Primary energy source for most organisms
Glucose, Glycogen, Starch
Where do carnivores obtain significant energy from?
proteins
Structural Functions of Peptidoglycan
Bacterial cell wall
Structural Functions of cellulose
Plant cell wall
Structural Functions of Chitin
Arthropod exoskeleton
Structural Functions of Proteoglycans
Extracellular matrix
Structural Functions of Hyaluronic acid
Lubrication (joints, vitreous humor)
Chemical Definition of Carbohydrates
- Polyhydroxy aldehydes
- Polyhydroxy ketones
- Compounds yielding these upon hydrolysis
Chemical Definition of Carbohydrates- General formula
Often (CH₂O)n
Chemical Definition of Carbohydrates- May Contain
- Nitrogen
- Phosphate
- Sulfate groups
How are carbohydrates are classified?
According to the size of the molecule
Carbohydrate Classification by Size
1. Monosaccharides - simple sugars
2. Oligosaccharides - made up of two or a few monosaccharide units
3. Polysaccharides - made up of many monosaccharide units
Simple Carbohydrates
monosaccharides (glucose, fructose, galactose) and disaccharides (maltose, lactose, sucrose)
Complex carbohydrates
polysaccharides --> starches & fibres
Monosaccharides
- Simplest carbohydrates
- Cannot be hydrolyzed further
- Water soluble
- Sweet taste
- Unbranched carbon chains
Examples of monosachharides
Glucose, Mannose, Galactose, Fructose
Oligosaccharides
- Polymers of 2-20 monosaccharide units
- Monosaccharides linked by glycosidic bonds
- Oligosaccharides (≥3 units) are often attached to: Proteins → Glycoproteins OR Lipids → Glycolipids
How are oligosachharides named?
Named according to the number of units:
- 2 → Disaccharide
- 3 → Trisaccharide
- 4 → Tetrasaccharide
What carbohydrate are the most abundant?
Disaccharides
- Sucrose (cane sugar)
- Lactose (milk sugar)
Polysaccharides
- 20 monosaccharide units
- Linear or branched
- Storage or structural roles
Examples of polysachharides
- Starch
- Glycogen
- Cellulose
Monosaccharide Chemical Nature
- polyhydroxy aldehydes or ketones
- General formula: (CH₂O)n
- Polymeric carbohydrates yield these, upon hydrolysis
Monosaccharide Characteristics
- Water-soluble
- Crystalline solids
- Usually sweet
- Unbranched carbon chains
Monosaccharide Functional Groups
- Aldoses → contain an aldehyde group (-CHO)
- Ketoses → contain a ketone group (C=O)
- Remaining carbons carry hydroxyl (-OH) groups
Biologically Important Monosaccharide Examples
hexose, glucose, pentose
Hexoses
- most common monosaccharides
- Glucose → major metabolic fuel
- Fructose → fruit sugar
Glucose polymers
starch, glycogen, cellulose
Pentoses
- D-ribose → RNA
- 2-deoxy-D-ribose → DNA
Chirality in Monosaccharides
all, except dihydroxyacetone, have a C attached to 4 different groups
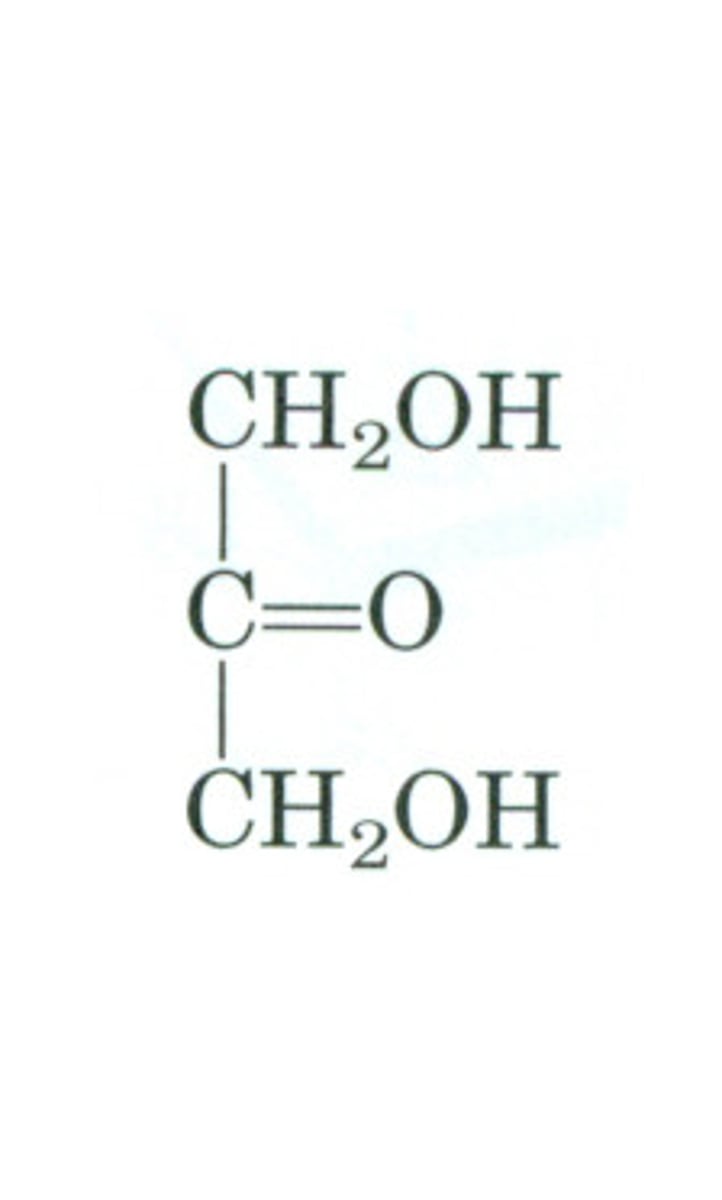
chiral (asymmetric) carbon
Carbon attached to four different groups
# stereoisomers vs. # chiral centers
n chiral centers → 2ⁿ stereoisomers
(1 chiral center → 2 stereoisomers)
enantiomers
Stereoisomers that are mirror images
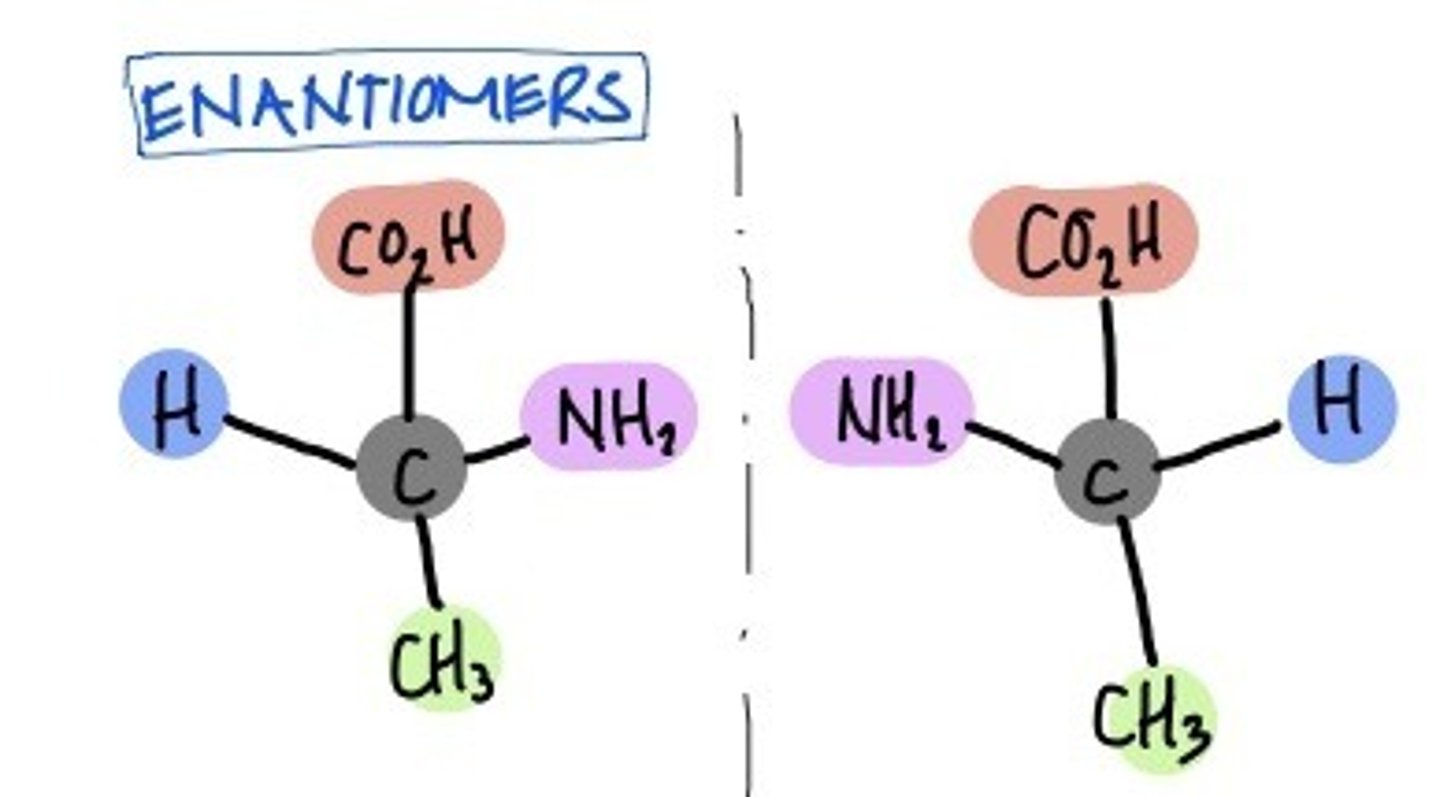
Perspective Structure/ Wedge and Dash Structure
- C chain drawn vertically
- Highest-numbered carbon at bottom
- Vertical bonds → project away from viewer
- Horizontal bonds → project toward viewer
Fischer Projection
- 3D structure projected onto a flat plane
- Widely used for comparing sugar stereochemistry
Glyceraldehyde (The Reference Molecule)
- Smallest chiral monosaccharide
- Contains one asymmetric carbon
- Exists as: D-glyceraldehyde → OH on the right Or L-glyceraldehyde → OH on the left
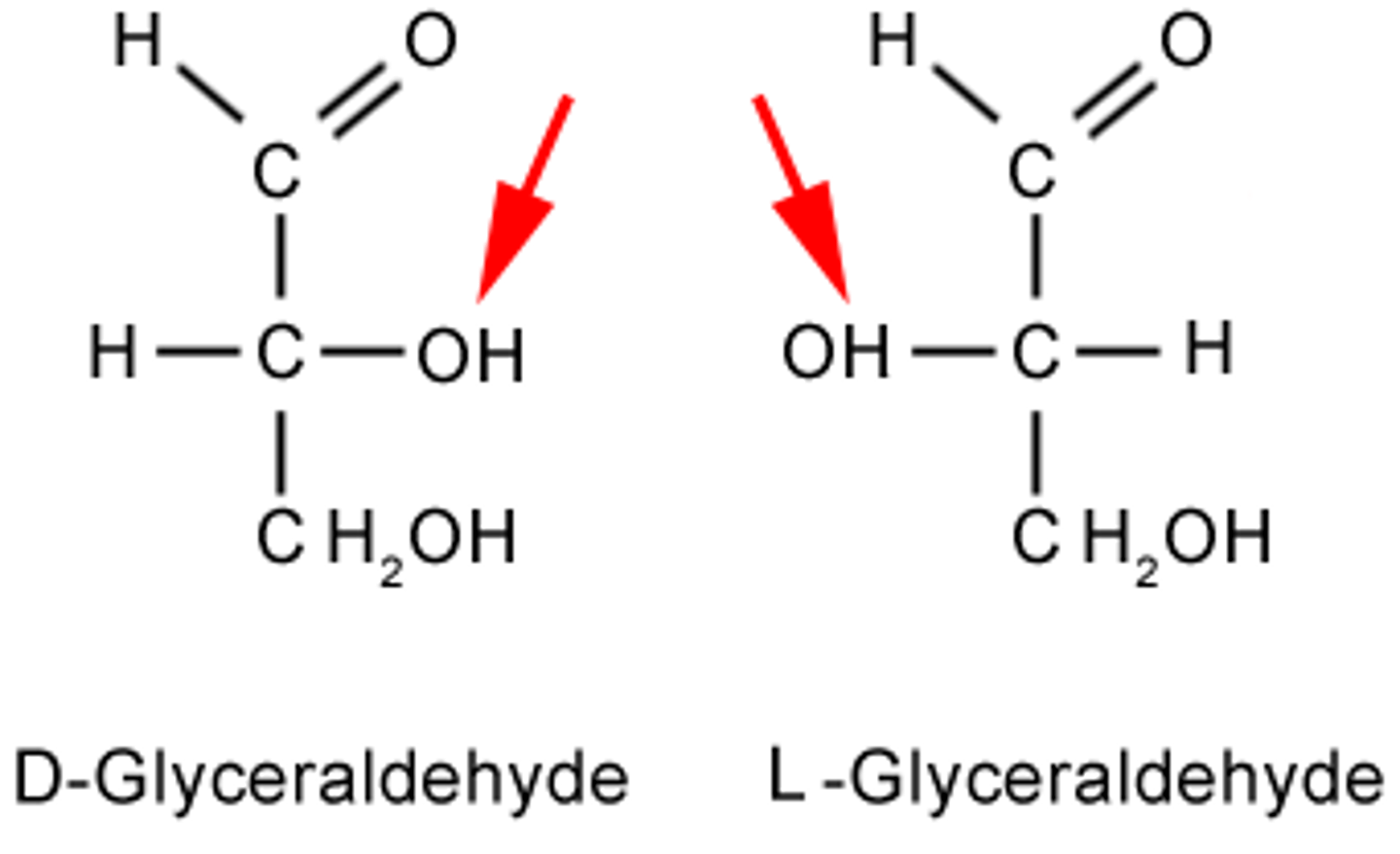
D and L forms of glyceraldehyde
are mirror images
D/L Nomenclature in Higher Sugars
1. Number carbons normally (carbonyl carbon = C1 in aldoses)
2. Examine the highest-numbered chiral carbon
3. Compare its configuration to glyceraldehyde
4. Most naturally occurring sugars → D-configuration
Aldoses
- Polyhydroxy aldehydes
- Example: Aldohexoses -contain 4 asymmetric (chiral) Cs (therefore 2^4 = 16 stereoisomers with 8D & 8D)
- CHO group at top?
How are higher aldoses derived?
by sequential insertion of -CHOH groups adjacent to the aldehyde group, starting from glyceraldehyde.
Epimers
- Molecules that differ in configuration at only one chiral carbon Examples:
• D-Glucose & D-Mannose → C-2 epimers (where the C differs)
• D-Glucose & D-Galactose → C-4 epimers
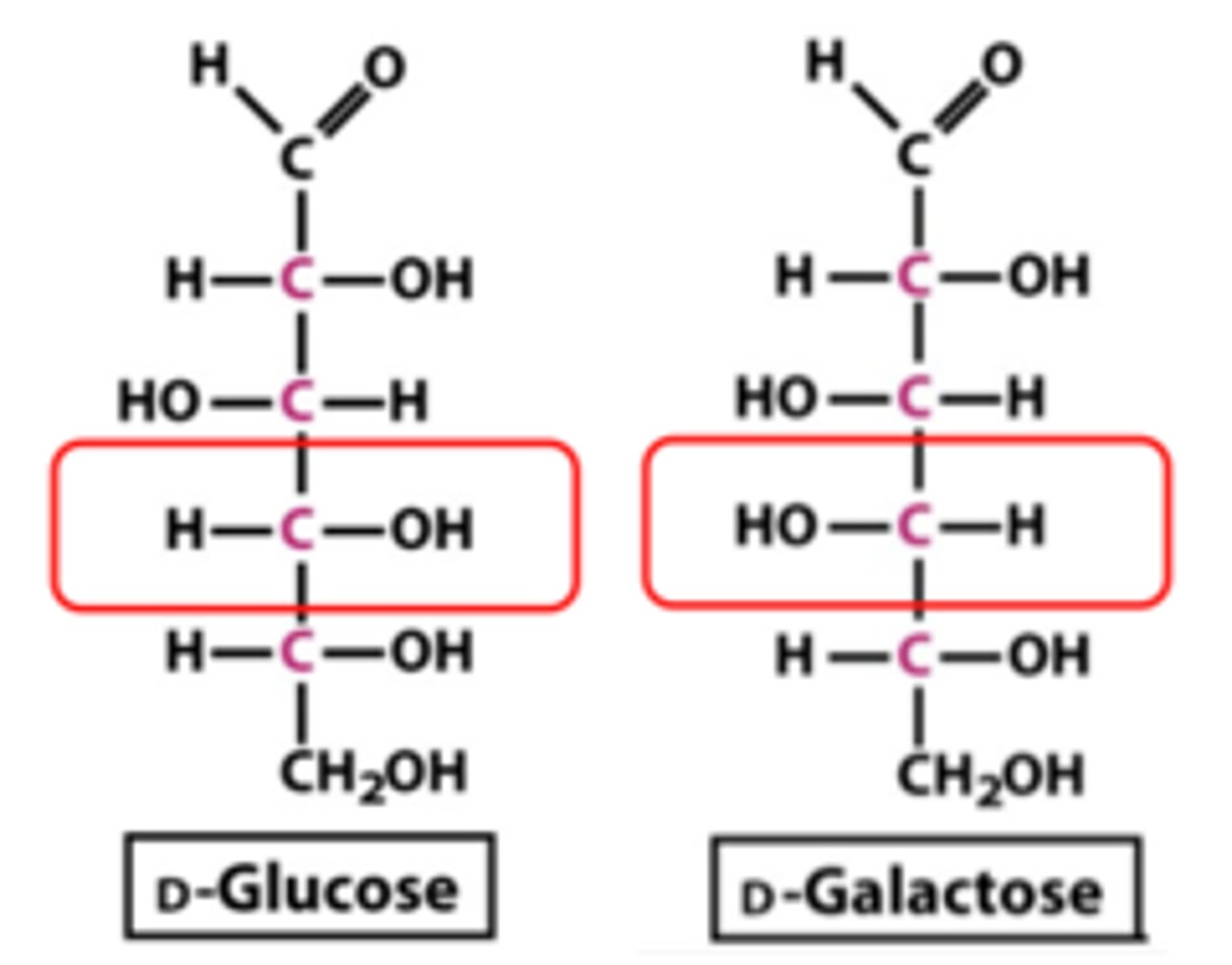
What does small stereochemical change result in
major biochemical consequences
Ketoses
- Polyhydroxy ketones
- Carbonyl group located internally (usually C-2)
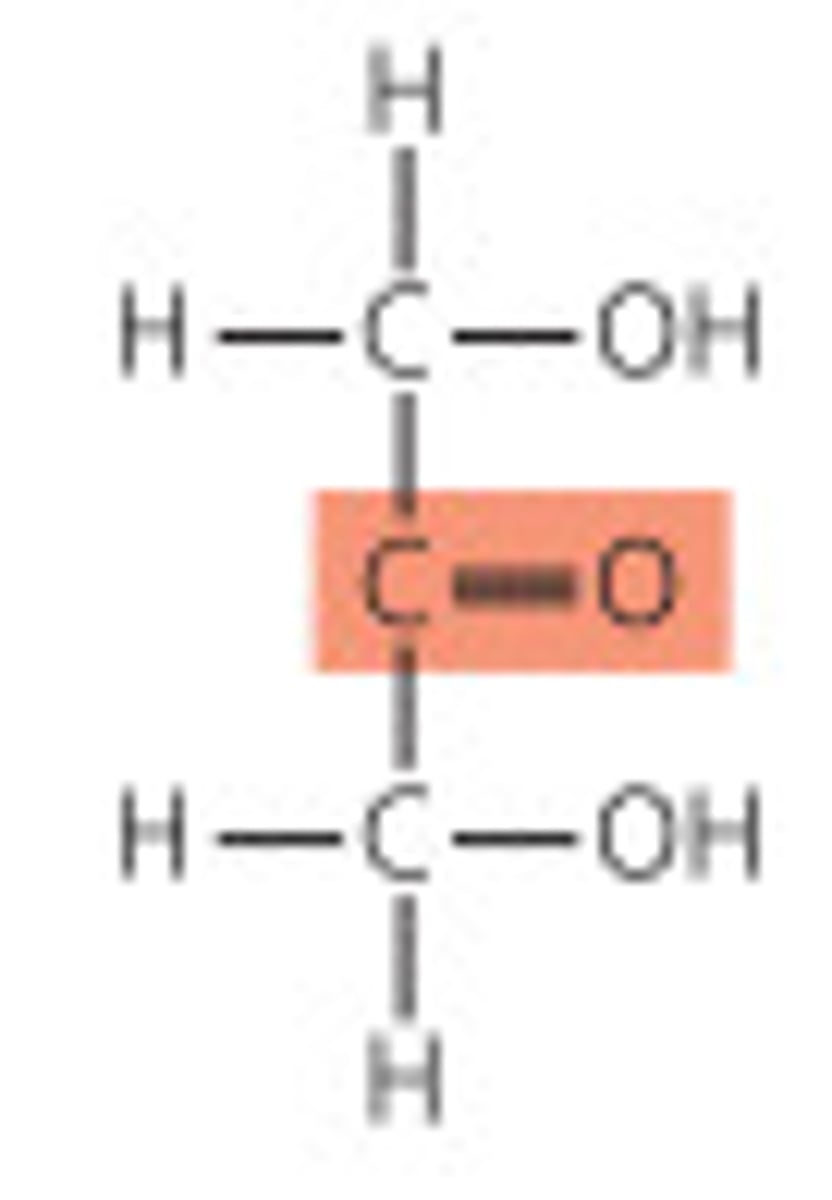
Naming Ketoses
Naming rule (4- and 5-C ketoses):
- Derived from corresponding aldose by inserting "ul"
Erythrose → Erythrulose; Ribose → Ribulose; Xylose → Xylulose
- exceptions: named by source (fructose -fruit sugar)
Cyclic Structures of Monosaccharides
In aqueous solution:
- Aldoses (≥4 carbons) and ketoses (≥5 carbons) predominantly exist in cyclic form
Why Sugars Form Rings
- Ring formation occurs via Intramolecular hemiacetal formation (aldoses) and intramolecular hemiketal formation (ketoses)
- Rxn bw carbonyl group (aldehyde/ketone) and internal hydroxyl group
Pyranose Structures (6-membered rings)
- cyclic carb molecules resemble pyran ring → called pyranoses
- 2 conformations: Chair (more stable) Boat
- Formation creates new chiral center at Anomeric carbon (C-1 in aldoses; C-2 in ketoses)
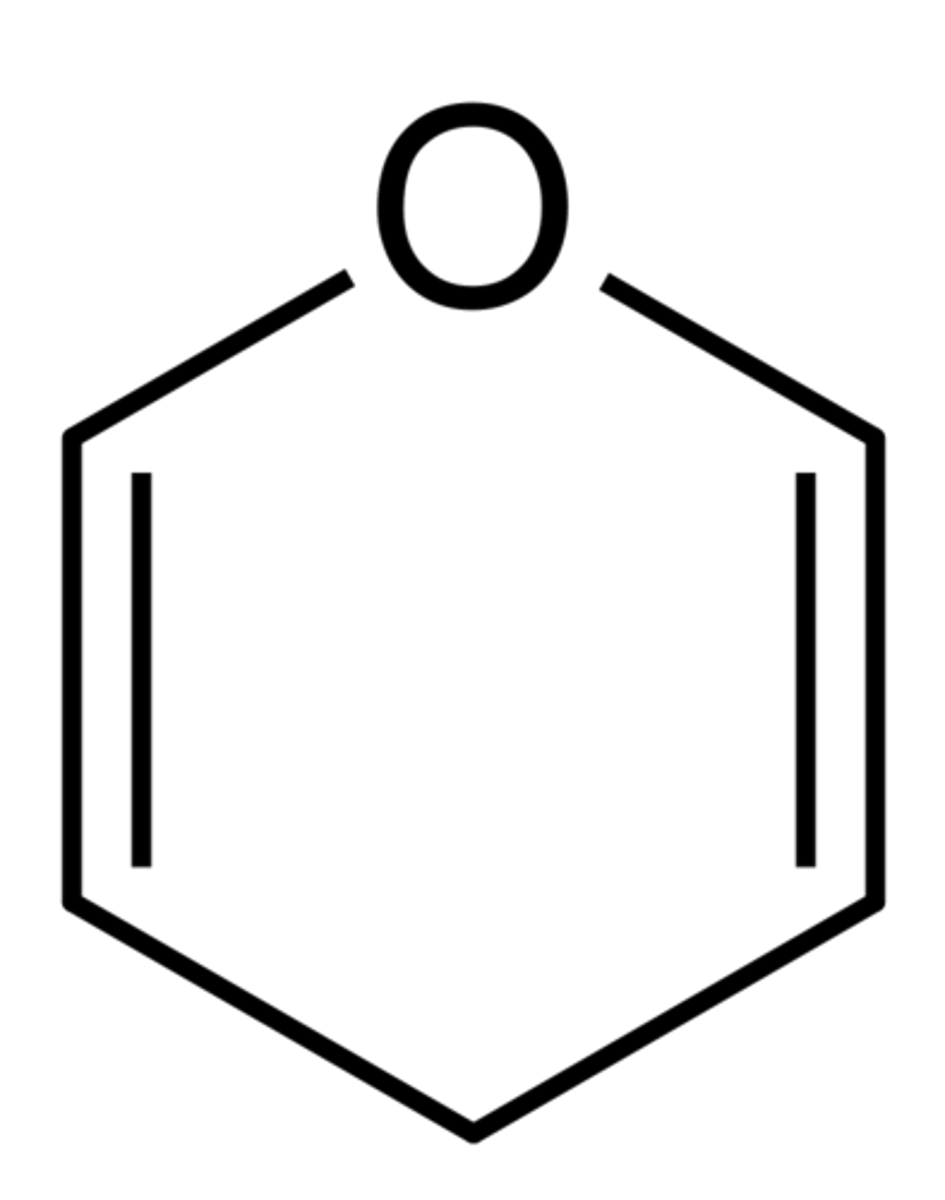
Chair conformation stability
- more stable than boat
- Bulky groups prefer equatorial positions
anomeric carbon
- initially not chiral carbon, but becomes one after taking cyclic form
- C in cyclic sugar that was the carbonyl C in linear form
(C-1 in aldoses; C-2 in ketoses)
anomers
- alpha-anomers: OH down
- beta-anomers: OH up
- type of stereoisomer in cyclic sugars that differ only in the configuration around the anomeric carbon
(C-1 in aldoses; C-2 in ketoses)
Anomers Examples
- α-D-glucopyranose
- β-D-glucopyranose
- Differ only at the anomeric carbo
Furanose Structures (5-membered rings)
- Resemble furan ring → called furanoses
- usually attack 2nd C
- Common in Fructose & Ribose
- Aldohexoses can form furanoses, but pyranoses are usually more stable
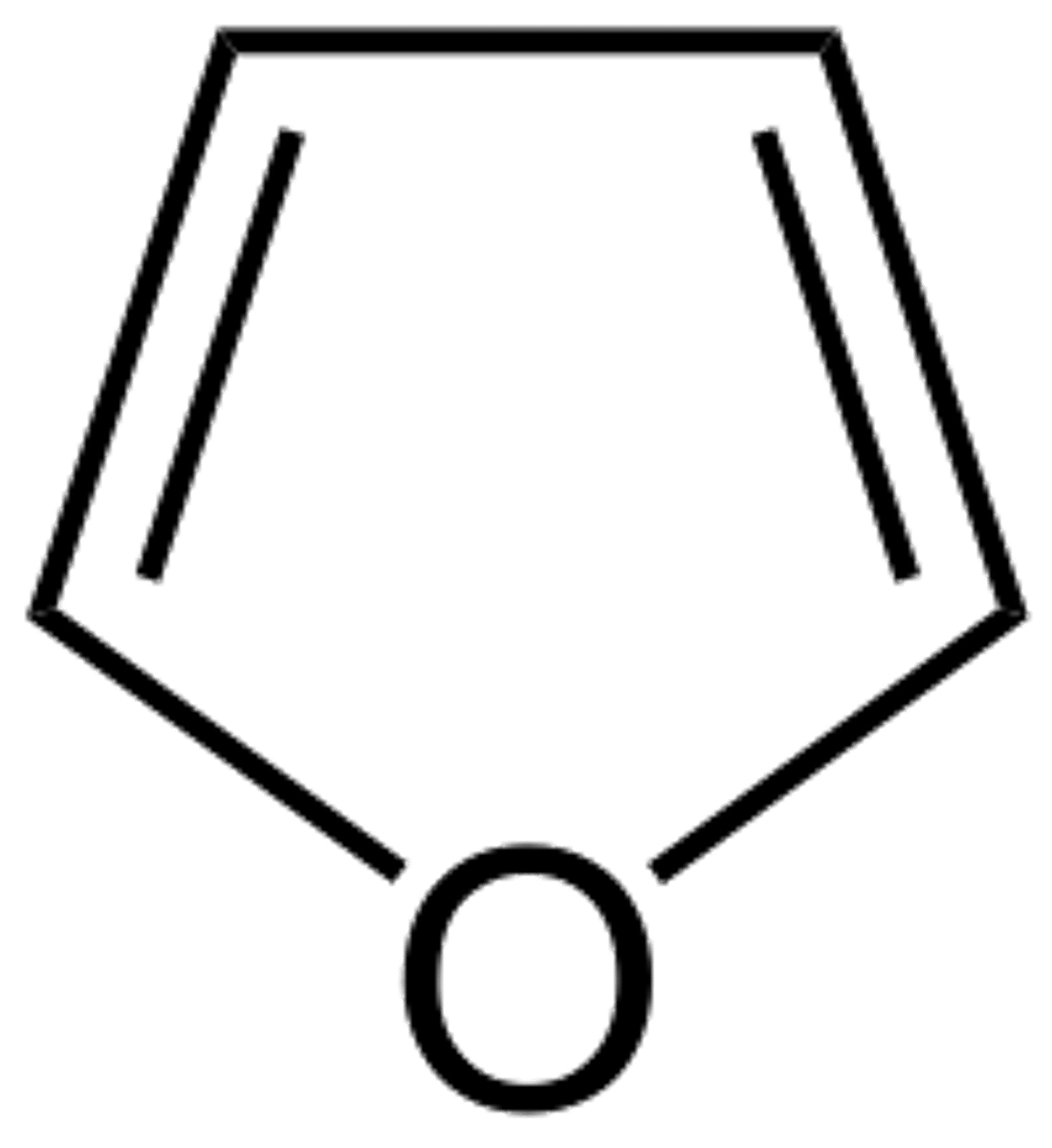
The same monosaccharide can either create what?
pyranoses and furanoses (more stable b/c can handle bulky groups)
- riboses (5C) like furanose form
Reversibility & Reducing Sugars
- Hemiacetal/hemiketal formation is reversible
- α and β forms exist in equilibrium in solution
- Rings can reopen → regenerate free aldehyde/ketone
- Therefore, monosaccharides can be oxidized
Benedicts test
1. Glucose reduces Cu²⁺ → Cu⁺
2. Forms brick red Cu₂O precipitate
3. Basis of reducing sugar tests (e.g., Benedict's test) - find out if sugas there in urine, normally glucose not there. then diabetic
- benedict solution contains Cu ions (blue), then green, orange, red.

Derivatives of monosaccharides
In addition to simple sugars, living organisms contains sugar derivatives in which OH groups are replaced by other groups
Glycosidic Bonds
- link oligosaccharide monomers by O-glycosidic bonds
- Formed when anomeric Cof one sugar reacts w hydroxyl of another sugar. Water is eliminated (condensation rxn)
Reducing Sugar and disaccharide with 2 glucose residues
The first glucose:
- Its anomeric C is involved in glycosidic bond. Therefore, it's not free, so not a reducing sugar
- The second glucose
- Its anomeric C remains free. Therefore, it is a reducing sugar
Reducing sugars
- ribose and lactose
- sucrose is non-reducing because doesn't have any reducing free ends, therefore cannot work in benedicts solution
Reducing Properties In oligosaccharides and polysaccharides:
- end w free anomeric C→ Reducing end
- end w anomeric C involved in bonding → Nonreducing end
What is the reducing concept important for? (3)
1. Enzyme recognition
2. Metabolism
3. Polysaccharide BD
Oligosaccharides Common abbreviations
Glc = Glucose
Gal = Galactose
Man = Mannose
Fru = Fructose
Fuc = Fucose
GlcN = Glucosamine
GlcNAc = N-acetylglucosamine
Naming Rule
- Sugars written from right to left
- Linkage positions indicated by carbon numbers
- anomeric form (alpha or beta)
Examples:
Gal(β1→4)Glc → Lactose
Glc(α1→4)Glc → Maltose
Glc(α1 β2)Fru → Sucrose
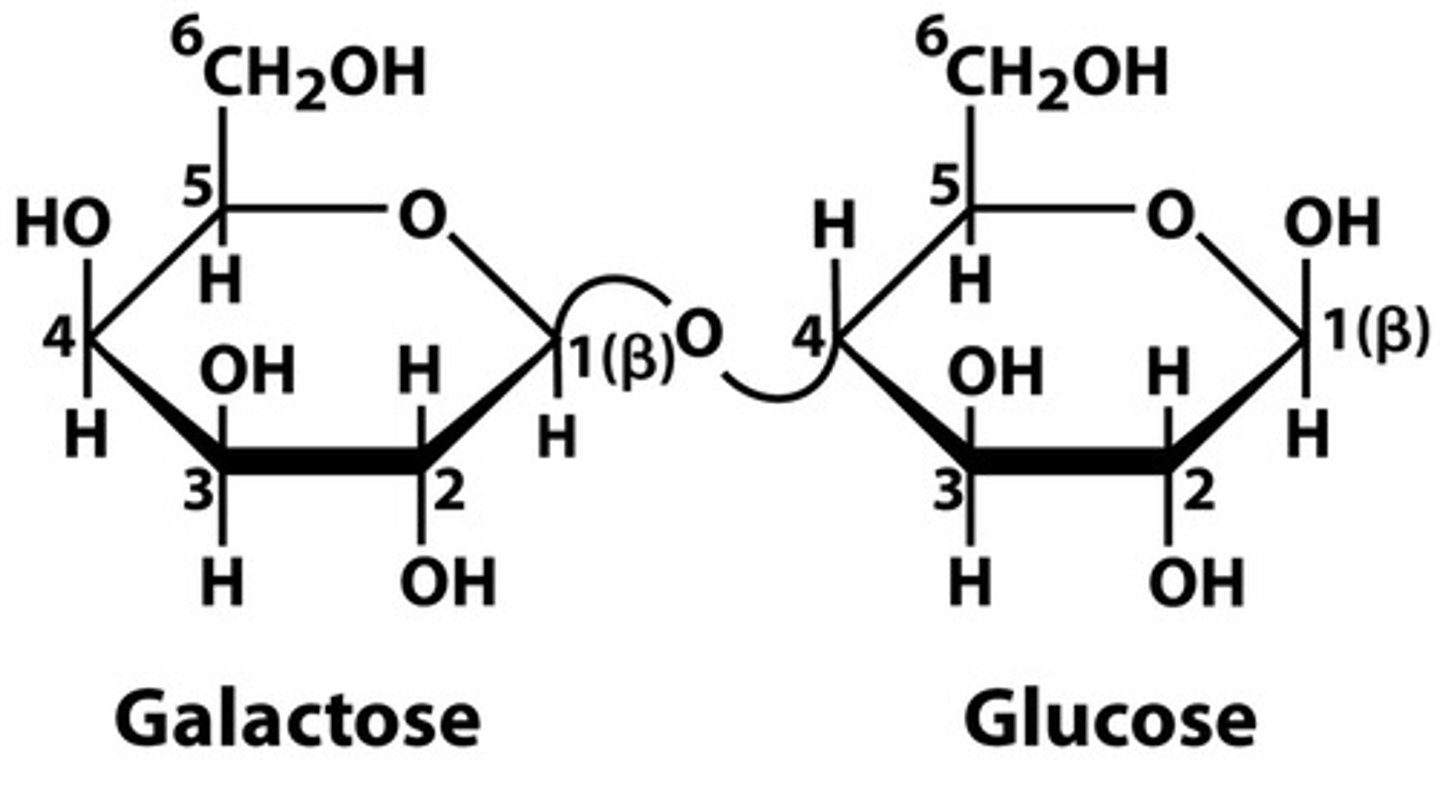
Structural Diversity of Oligosaccharides
- may be Linear or Branched
- Frequently attached to Proteins → Glycoproteins OR Lipids → Glycolipids (mostly in outer cell MB)
Polysaccharides Definition
- Polymers containing >20 monosaccharide units
- Also called glycans: Mannans → polymers of mannose; Galactans → polymers of galactose
- may be linear OR branched
Homopolysaccharides (classification of polysaccharides)
one type of monosaccharide
Heteropolysaccharides (classification of polysaccharides)
more than one type
Plant Storage Form of Glucose
starch
Starch
- storage polysaccaride in plants
- composed of 2 polymers
- 2 types: amylose & amylopectin
Amylose
- Linear polymer/unbranched
- α(1→4) linkages
- Molecular weight: thousands to millions
- Helical structure
Amylopectin
- Branched polymer: Branch points α(1→6) and branch every 24-30 glucose units
- Backbone: α(1→4)
- Up to 10⁶ glucose residues
Ratio of amylose:amylopectin
varies by plant source
Amylopectin storage
Stored as granules in chloroplasts & tubers (e.g., potato, yam)
Animal Storage Form of Glucose
Glycogen
Glycogen
- storage of starch in animals
- similar structure to amylopectin
glycogen structure
- Backbone: α(1→4)
- Branches: α(1→6), More highly branched: every 8-12 residues
- Up to 50,000 glucose residues
- always attach to protein molecule (like enzyme)
glycogen abundance
Especially abundant in liver (~7% liver weight)
Glycogen Functional Significance of Branching
-One reducing end & Many nonreducing ends
- Enzymes (Glycogen phosphorylase) remove glucose from nonreducing ends, allowing rapid, simultaneous glucose release
Glycogen Structural Organization
- Exists as granules in liver cells
- Reducing end attached to Glycogenin
Glycogenin
- Structural anchor
- Enzyme involved in synthesis of glycogen
Why Store Glucose as Glycogen?
- Free glucose at equivalent levels → ~0.4 M concentration
- Would cause: High osmolarity, Water influx, Cell rupture
solution to cell rupturing
Glycogen: Insoluble, Low osmotic impact (conc goes down)
- Facilitates glucose uptake from blood (~5 mM extracellular)
Cellulose
Linear glucose polymer
- structural polysaccharide
cellulose structure
- β(1→4) linkages
- 10,000-15,000 glucose units
- Strong hydrogen bonding bw chains
cellulose characteristics
- Water insoluble
- Major component of plant cell wall
- Most abundant polysaccharide in nature
- Cotton = nearly pure cellulose
- Most animals cannot digest (lack enzyme for β-linkage cleavage)
Chitin
Linear homopolysaccharide
- structural polysaccharide
chitin structure
- Monomer: N-acetylglucosamine
- β(1→4) linkage
Chitin characteristics
- Structural component of insect exoskeleton & crustacean shells
- Provides rigidity and protection
Bacterial & Extracellular Matrix Carbohydrates
Peptidoglycans, Proteoglycans
Peptidoglycans
- Linear heteropolymers
- Bacterial Cell Wall
Peptodoglycan structure
- Alternating N-acetylglucosamine (GlcNAc) & N-acetylmuramic acid (MurNAc)
- Linked by β(1→4) glycosidic bonds
- Cross-linked by short peptide chains
peptidoglycan functions (3)
1. Provide structural strength
2. Prevent osmotic swelling and lysis
3. Especially important in Gram-positive bacteria
Proteoglycans structure
- Core protein + glycosaminoglycan (GAG) chains
- GAGs = long, unbranched heteropolysaccharides
proteoglycans characteristics/functions
- Major components of extracellular matrix (ECM)
- Highly hydrated → provide cushioning & lubrication