Glycolysis, Citric Acid Cycle, Oxidative Phosphorylation
1/89
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
90 Terms
Phase 1, Reaction 1:
Phosphorylation of D-Glucose
List in the following form:
Reactants:
Products:
Enzyme:
Cofactor:
Delta G:
Reversibility:
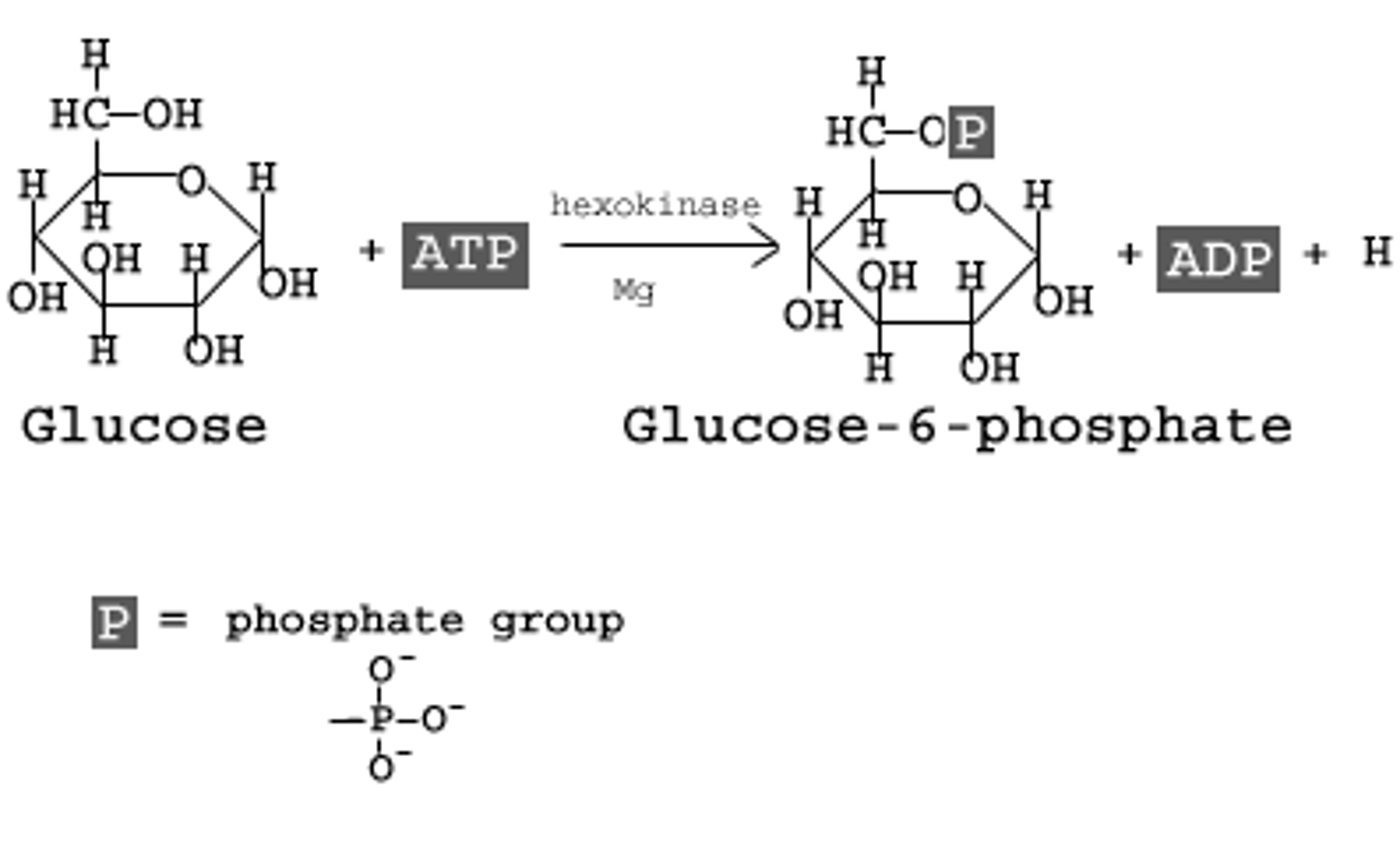
Reactants: Glucose + ATP
Products: Glucose 6-Phosphate + ADP
Enzyme: Hexokinase
Cofactor: Mg2+
delta G: -16.7 kJ/mol
Reversibility: Irreversible
Phase 1, Reaction 2:
Isomerization of Glucose 6-phosphate
List in following form:
Reactants:
products:
Enzyme:
Reversibility:
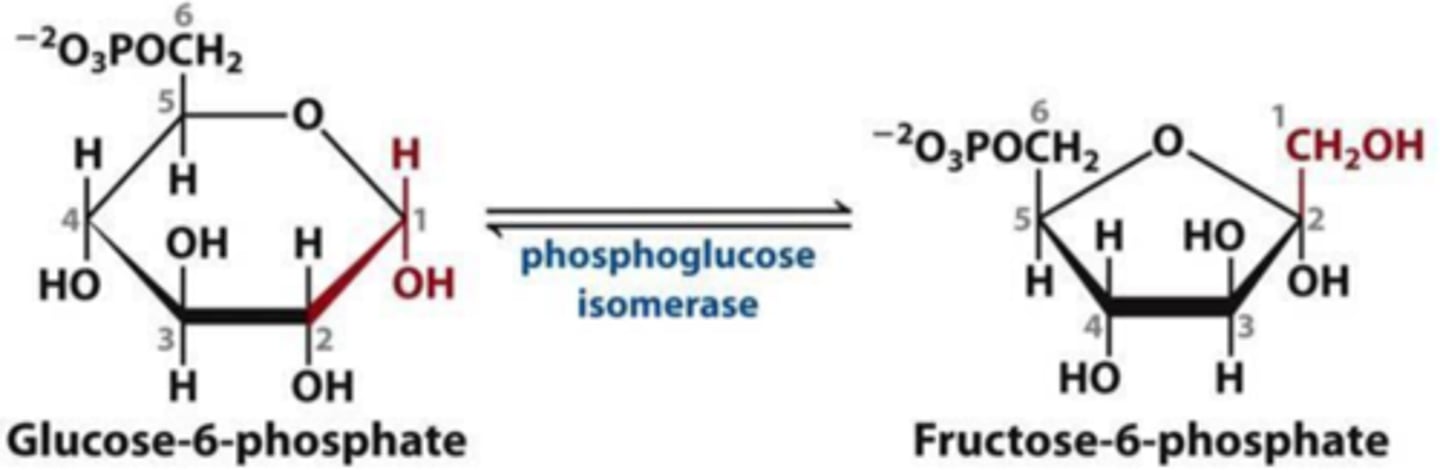
Reactant: Glucose 6-phosphate
Product: Fructose-6-phosphate
Enzyme: Phosphohexose Isomerase
Reversibility: Reversible
Phase 1, Reaction 3:
Phosphorylation of Fructose-6-phosphate
Rate Limiting step!!!
List in following form:
Reactants:
products:
Enzyme:
Reversibility:
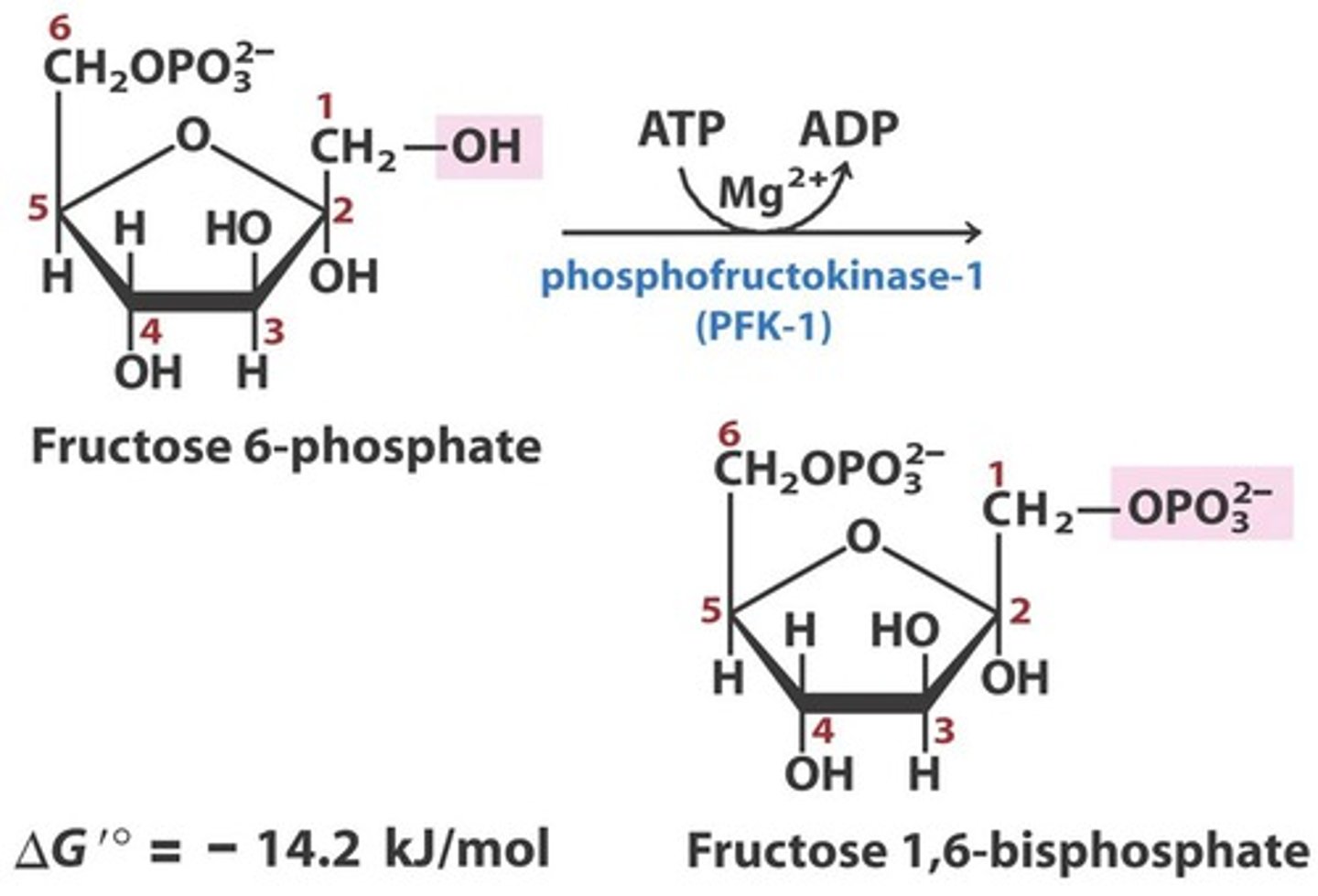
Reactants: Fructose-6-phosphate + ATP
Products: Fructose-1,6- biphosphate + ADP
Enzyme: Phosphofructokinase
Cofactor: Mg2+
Reversibility: Irreversible
How do high concentrations of AMP and ATP allosterically control PFK?
ATP Inhibits
AMP Promotes
Phase 1, Reaction 4:
Asymmetric Cleavage of C3-C4 bond to produce 2 triose phosphates
List in following form:
Reactants:
Products:
Enzyme:
Reversibility:
Delta G:
in vivo delta g and why:
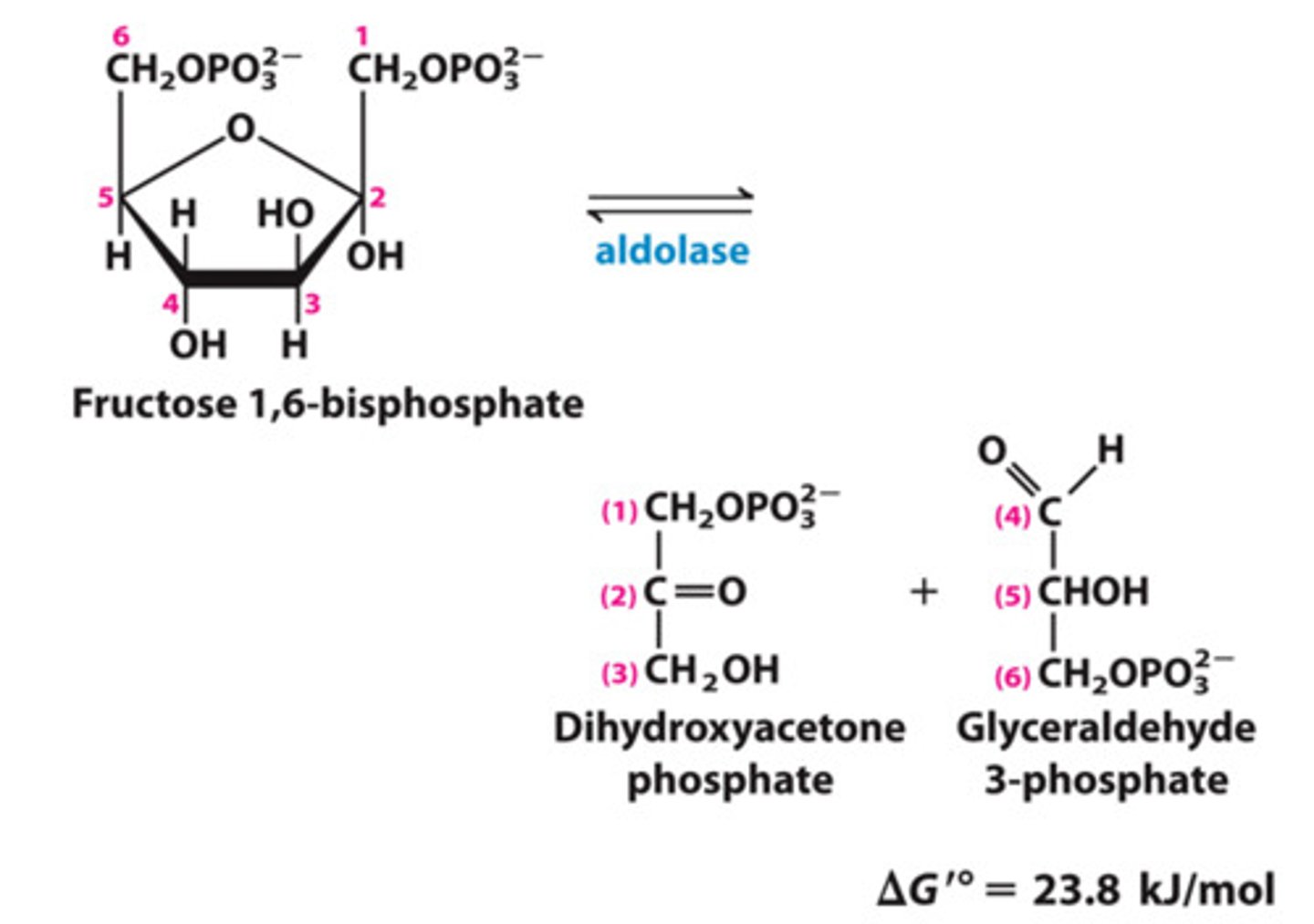
Reactants: Fructose-1,6-biphosphate
Product: Dihydroxyacetone phosphate (DHAP) + Glyceraldehyde-3-phosphate (G-3-P)
Enzyme: Fructose biphosphate aldolase
Reversibility: Reversible
Delta G: 23.9
in vivo delta g and why: -0.23 because product concentrations are kept low
Phase 2, Reaction 1, overall (5):
Conversion of DHAP into G-3-P
List in the following form:
Reactants:
Products:
Enzyme:
Reversibility:
Reactants: Dihydroxyacetone Phosphate (DHAP)
Products: Glyceraldehyde-3-phosphate (G-3-P)
Enzyme: Triose Phosphate Isomerase
Reversibility: Reversible
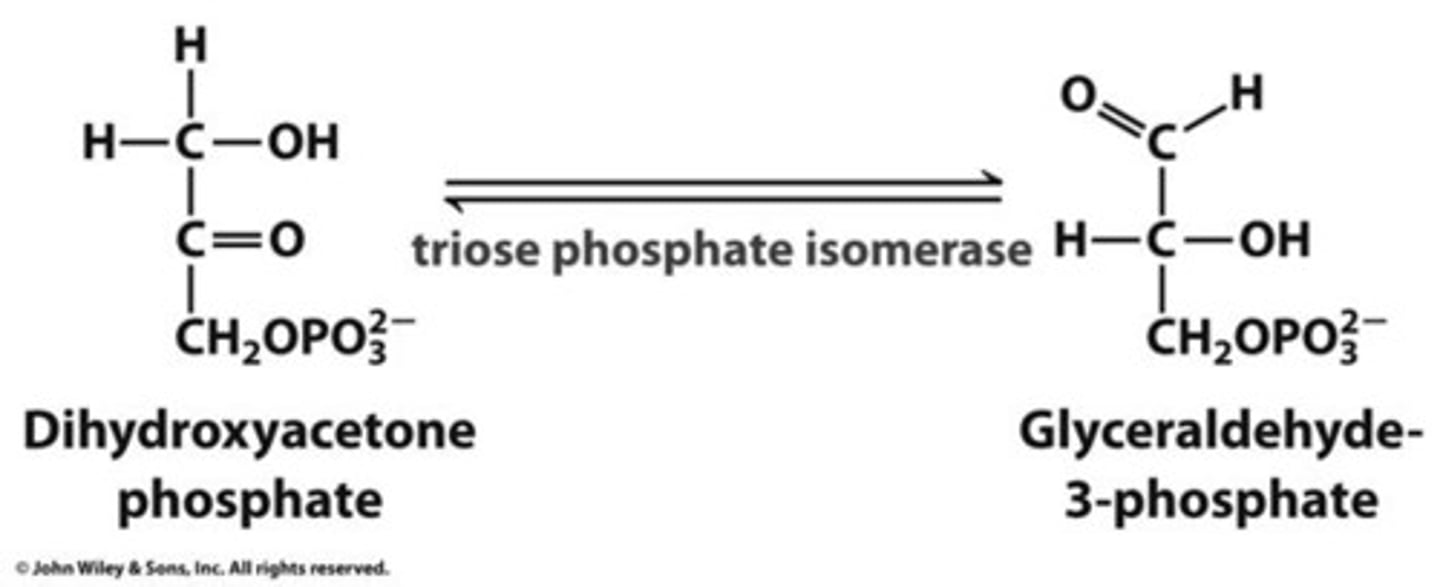
Phase 2, Reaction 2, Overall (6):
Oxidation of G-3-P
List in the following form:
Reactants:
Products:
Enzyme
Delta G:
Reversibility:
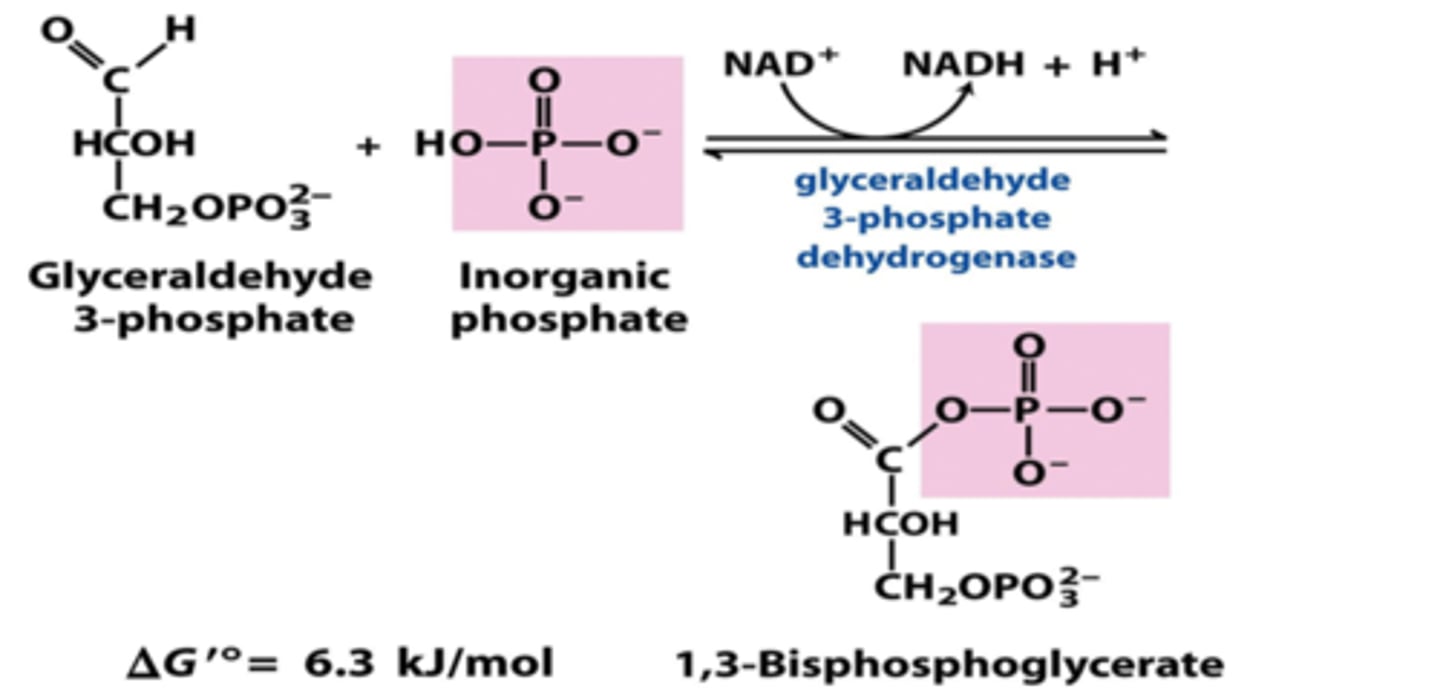
Reactants: G-3-P + NAD+ + H(PO4)2-
Products: 1,3-Bisphosphoglycerate + NADH + H+
Enzyme: G-3-P dehydrogenase
Delta G: +6.3 kj/mol
Reversibility: Reversible
Phase 2, Reaction 3, Overall (7):
Phosphoryl transfer to ADP
List in the following form:
Reactants:
Products:
Enzyme:
Cofactor:
Delta G:
Reversibility:
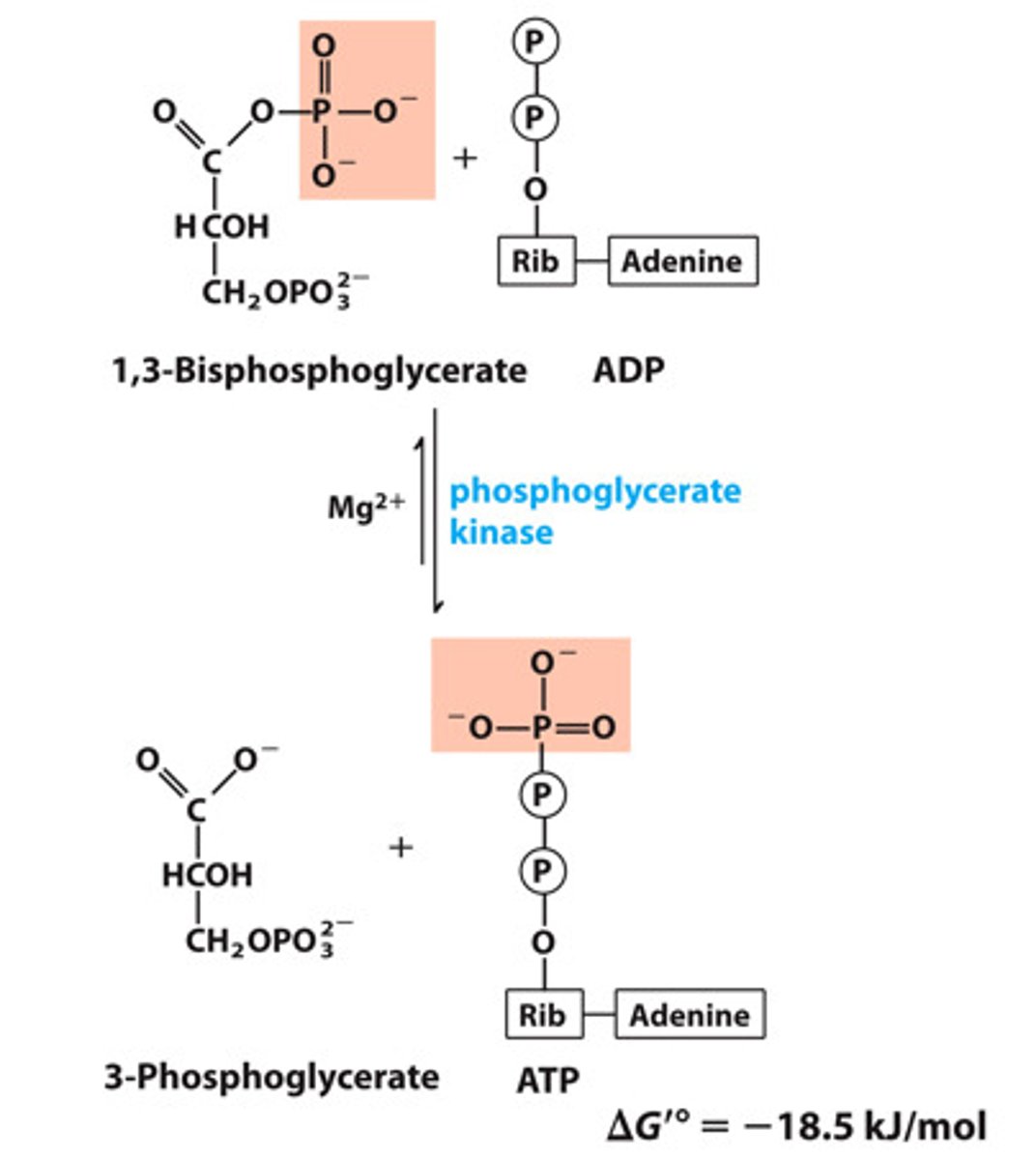
Reactants: 1,3-bisphosphoglycerate (1,3-BPG) + ADP
Products: 3-Phosphoglycerate (3-PG) + ATP
Enzyme: Phosphoglycerate Kinase
Cofactor: Mg2+
Delta G: -18.9 kJ/mol
Reversibility: Reversible
Phase 2, Reaction 4, Overall (8):
Convert 3-PG to 2-PG to set up next substrate level phosphorylation
List in the following form:
Reactants:
Products:
Intermediate:
Enzyme:
Reversibility:
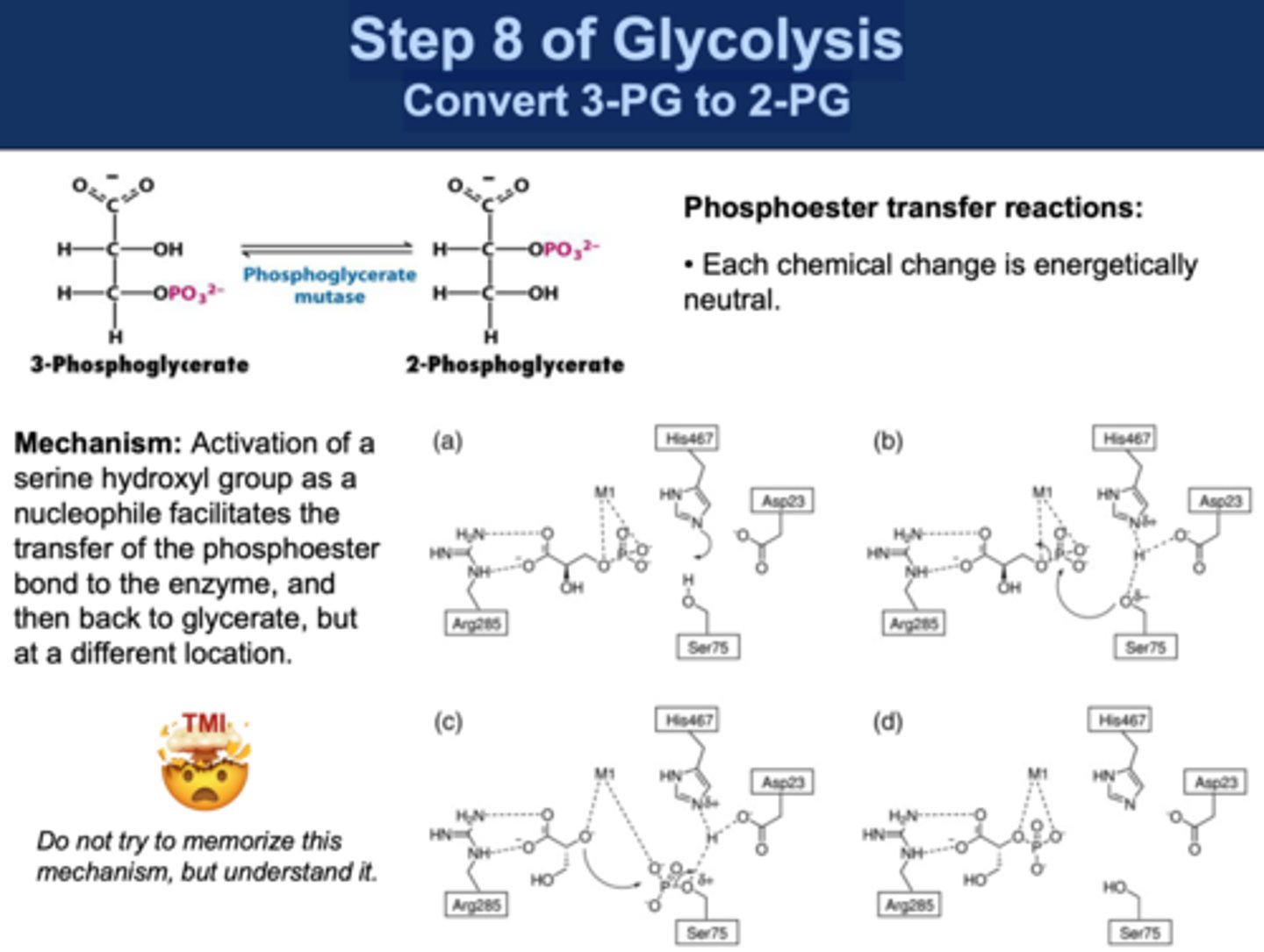
Reactants: 3-Phosphoglycerate (3-PG)
Products: 2-Phosphoglycerate (2-PG)
Intermediate: 2,3-biphosphoglycerate
Enzyme: Phosphoglycerate mutase
Reversibility: Reversible
Phase 2, Reaction 4, Overall (9):
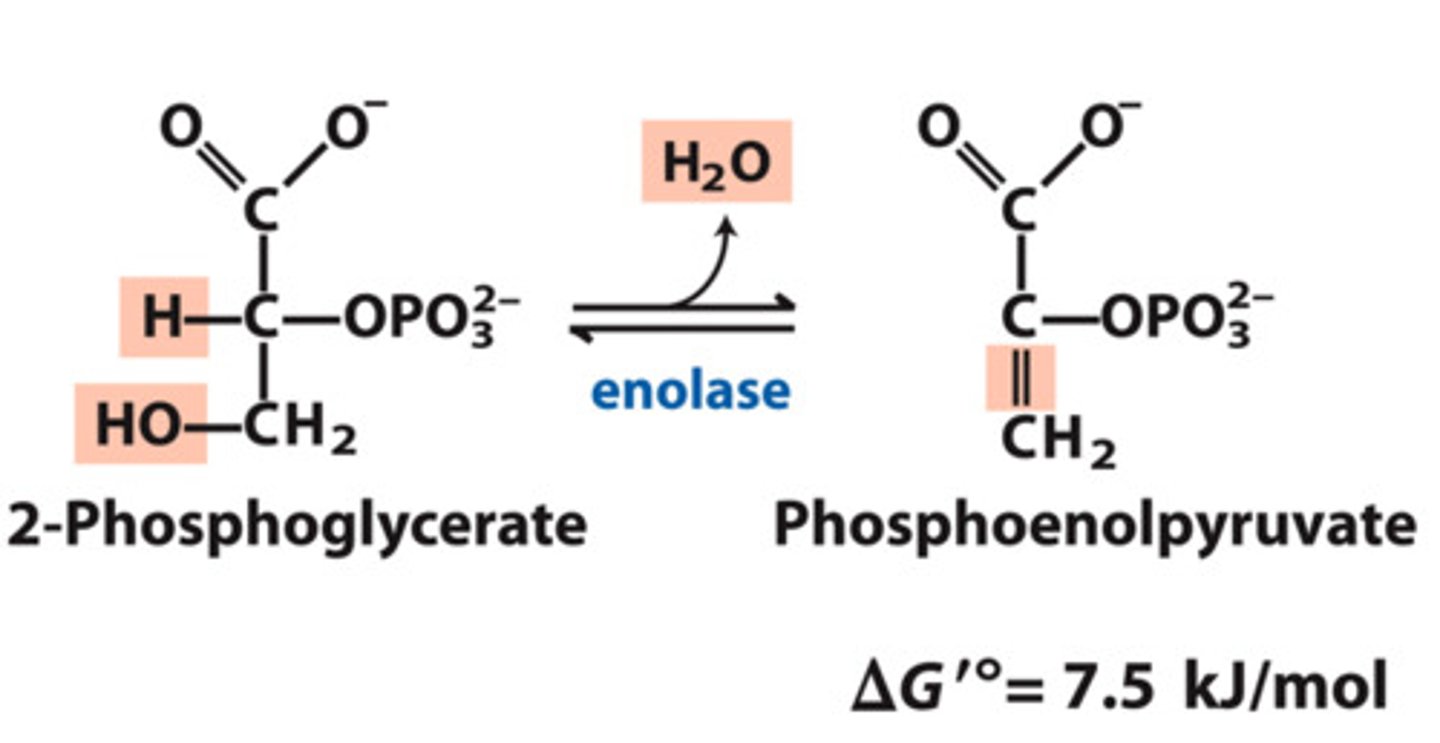
Dehydration of 2-PG into phosphoenolpyruvate
List in the following form:
Reactants:
Products:
Enzyme:
Delta G:
Reversibility:
Reactants: 2-PG
Products: Phosphoenolpyruvate (PEP) + H2O
Enzyme: Pyruvate Kinase
Delta G: 7.5 kJ/mol
Reversibility: Reversible
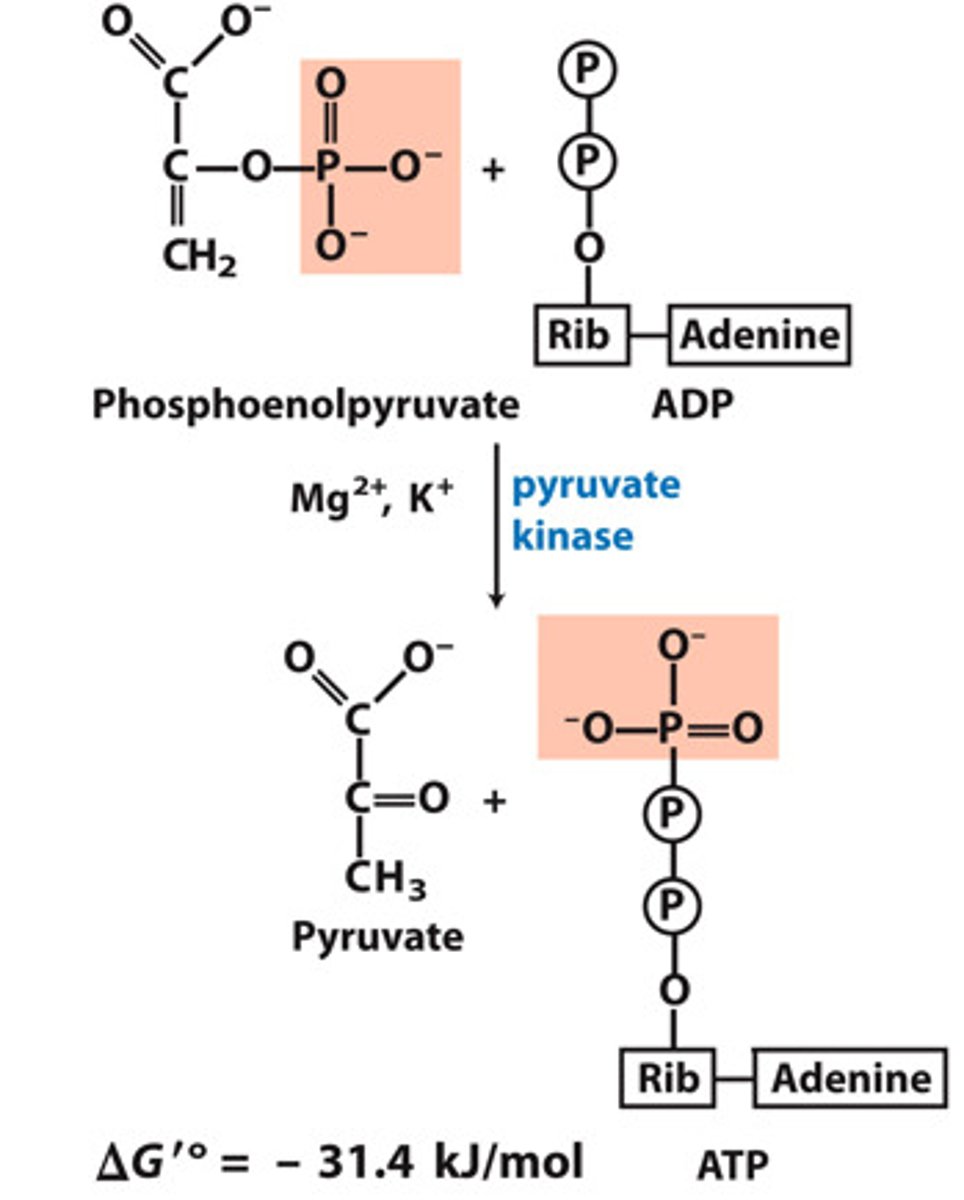
Phase 2, Reaction 5, Overall (10):
Second Substrate-level Phosphorylation
List in the following form:
Reactants:
Products:
Enzyme:
Cofactors:
Delta G:
Reversibility:
Reactants: Phosphoenolpyruvate (PEP) + H+ + ADP
Products: Pyruvate + ATP
Enzyme: Pyruvate Kinase
Cofactors: Mg2+, K+
Delta G: -31.7 kJ/mol
Reversibility: Reversible
Anaerobic Conditions in muscle
Reactants:
Products:
Reactants: 2 Pyruvate + 2 NADH
Products: 2 lactate + 2 NAD+
Anaerobic Conditions Alcoholic Fermentation in Yeast
Reactants:
Products:
Reactants: 2 Pyruvate + 2 NADH
Products: 2 Ethanol + 2 CO2 + 2 NAD+
Investment in Glycolysis: ____
Payoff in glycolysis: ____
2 ATP, 4 ATP (Net 2 ATP) + 2 NADH + 2 Pyruvate
Where is pyruvate imported to for aerobic respiration in cells
Mitochondrial Matrix
Conversion of Pyruvate into Acetyl-CoA
Reactants:
Products:
Enzyme:
Coenzymes:
Delta G:
Reversibility:
Reactants: Pyruvate + CoA-SH + NAD+
Products: Acetyl-CoA + CO2 + NADH
Enzyme: Pyruvate Dehydrogenase complex (E1 + E2 + E3)
Coenzymes: TPP, Lipoate, FAD
Delta G: -33.4 kJ/mol
Reversibility: Irreversible
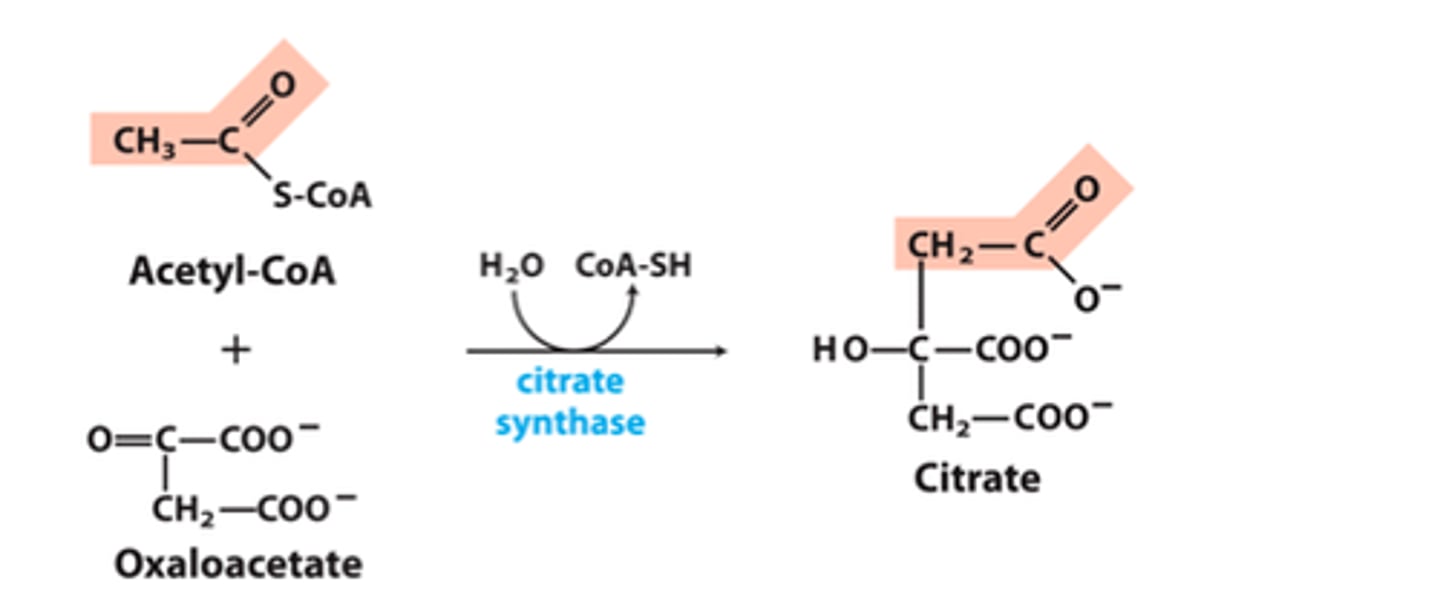
TCA Reaction 1: Claisen Condensation to generate Citrate
Reactants:
Products:
Enzyme:
Delta G:
Reversibility:
Reactants: Acetyl-CoA + Oxaloacetate + H2O
Products: Citrate + CoA-SH
Enzyme: Citrate synthase
Delta G: -32.2 kJ/mol
Reversibility: Irreversible
TCA Reaction 2a: Dehydration/Rehydration of Citrate
Reactants:
Products:
Enzyme:
Overall Delta G:
Reversibility:
Reactants: Citrate
Products: Cis-Aconitate + H2O
Enzyme: Aconitase
Overall Delta G: 13.3 kJ/mol
Reversibility: Reversible
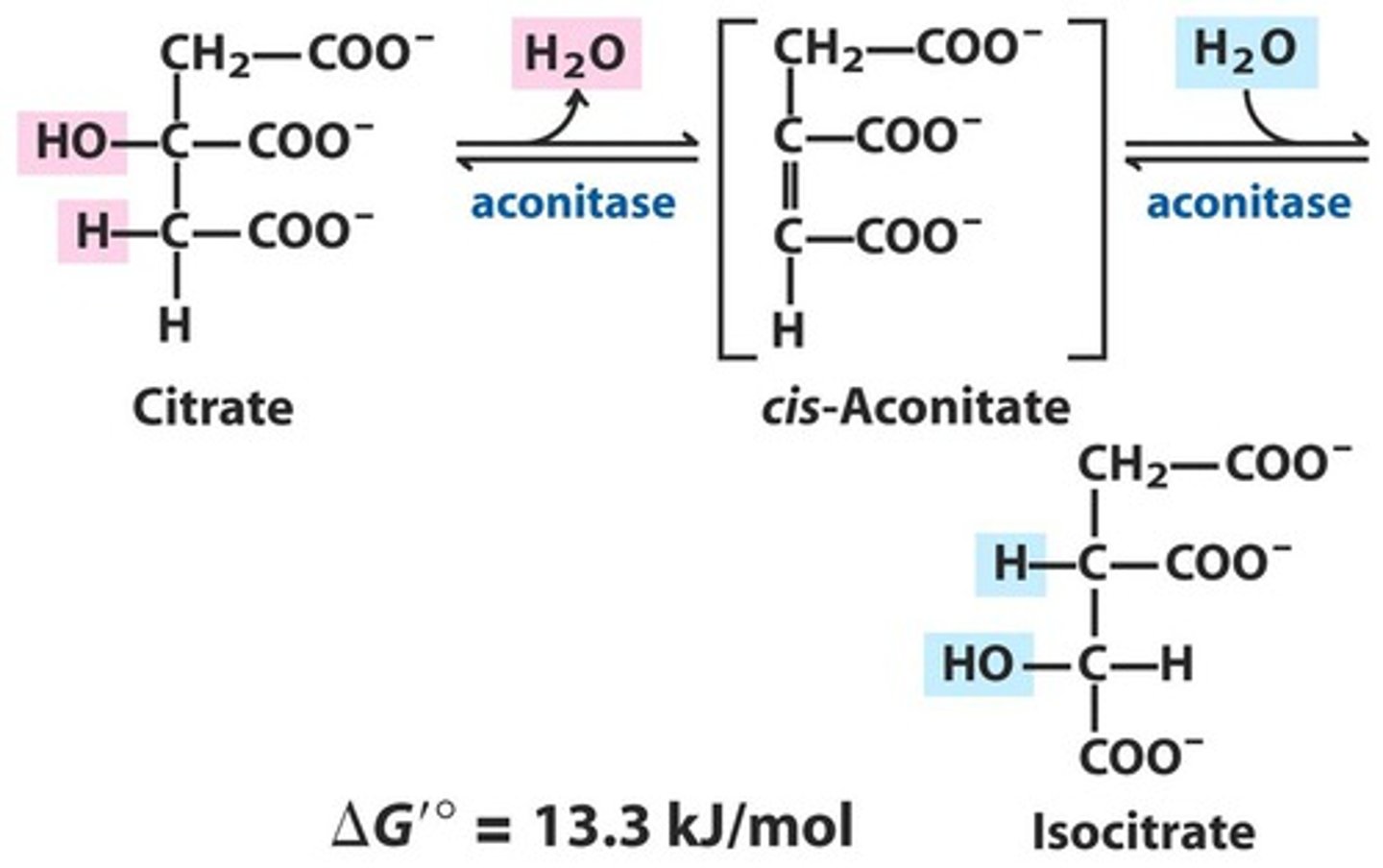
TCA Reaction 2b: Dehydration/Rehydration of Citrate
Reactants:
Products:
Enzyme:
Overall Delta G:
Reversibility:
Reactants: cis-aconitate + H2O
Products: Isocitrate
Enzyme: aconitase
Overall Delta G: 13.3 kJ/mol
Reversibility: Reversible
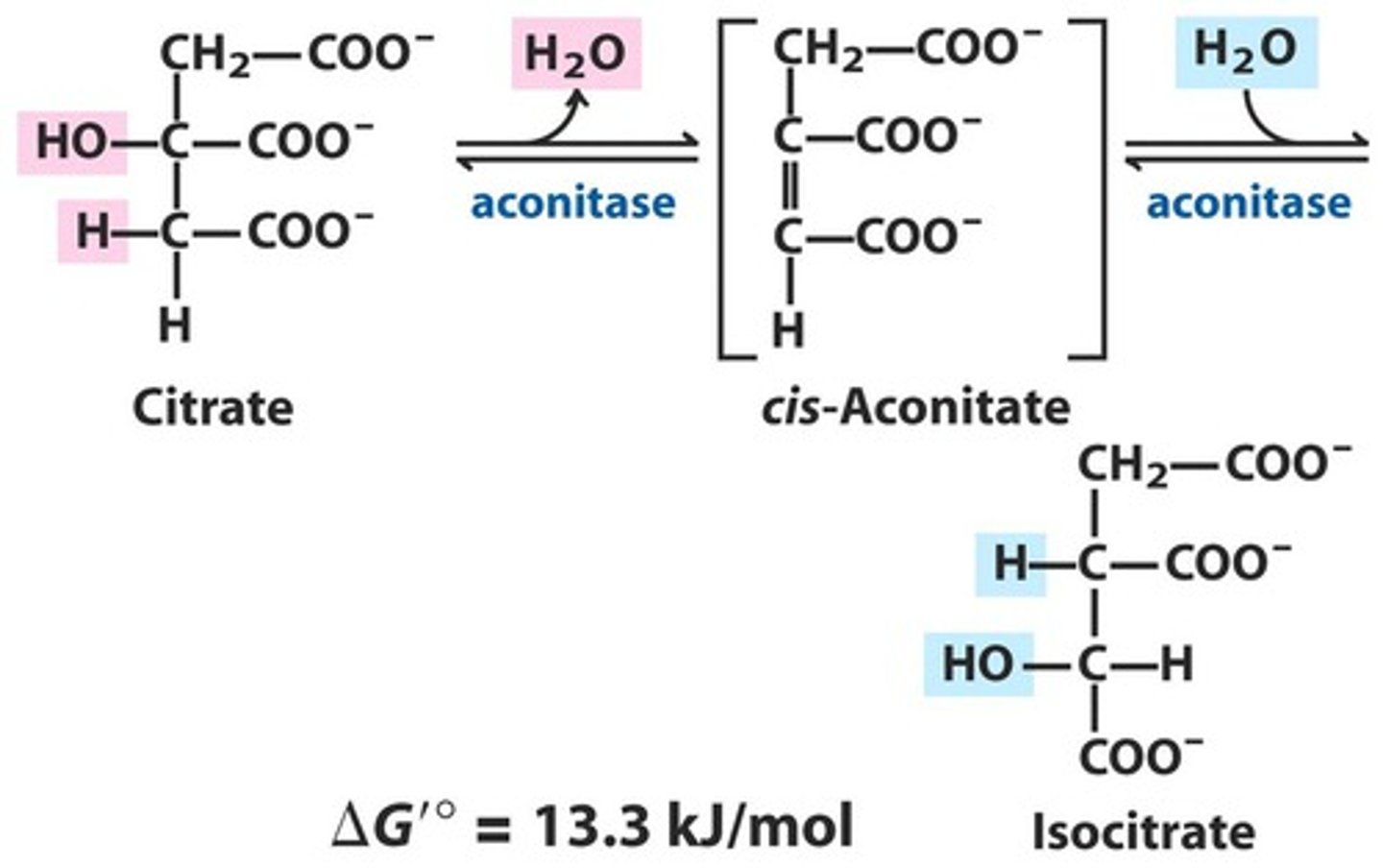
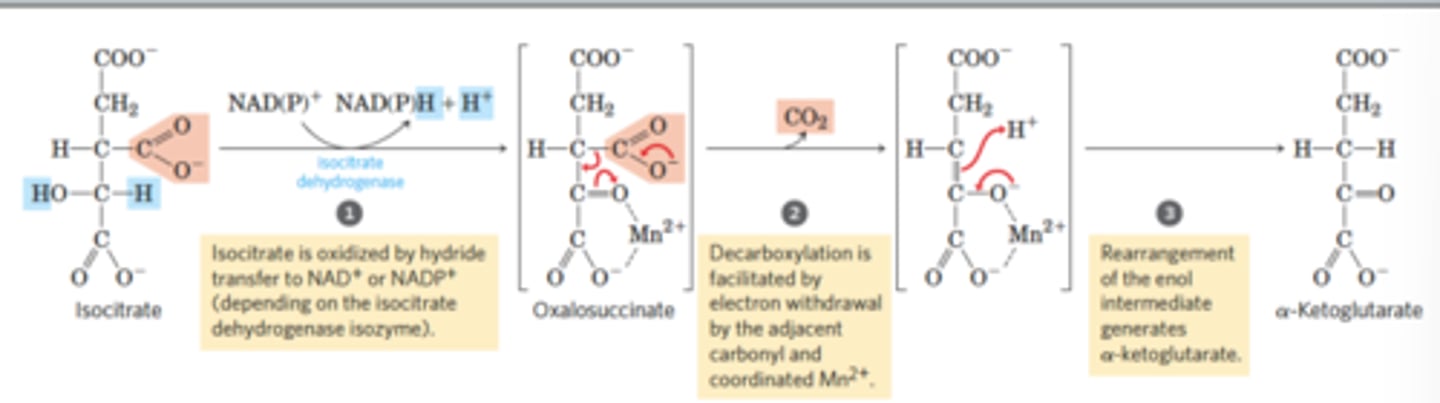
TCA Reaction 3: Oxidative decarboxylation
Reactants:
Products:
Enzyme:
Delta G:
Reversibility:
Reactants: Isocitrate + NAD(P)+
Products: alpha-ketoglutarate + NADH + H+ + CO2
Enzyme: isocitrate dehydrogenase
Delta G: -20.9 kJ/mol
Reversibility: Reversible
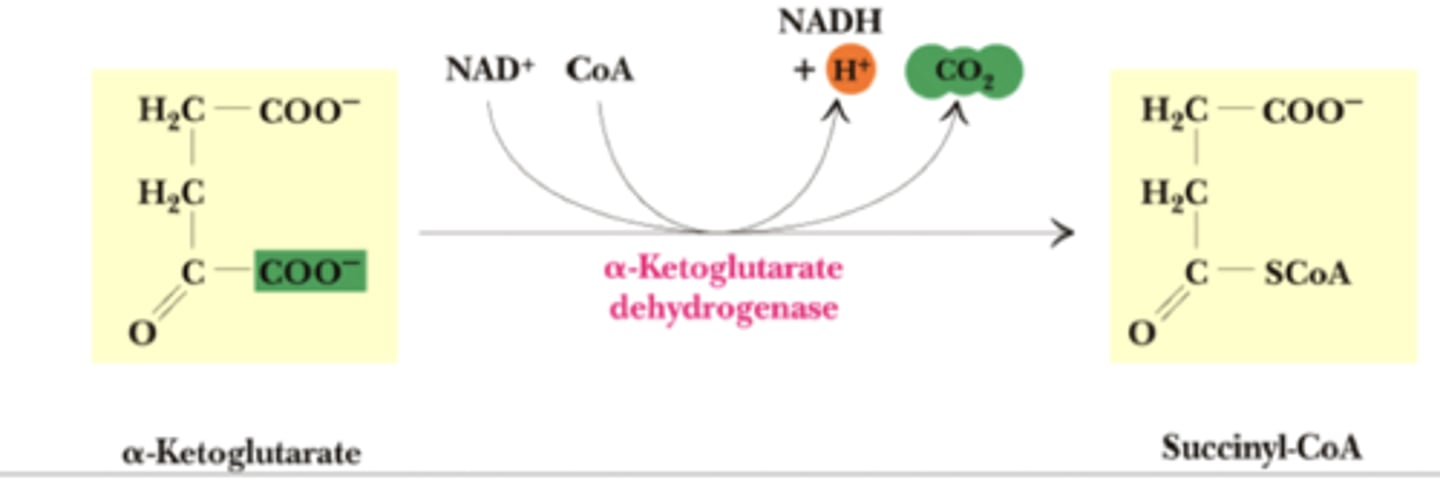
TCA Reaction 4: Oxidative decarboxylation
Reactants:
Products:
Enzyme:
Coenzyme:
Delta G:
Reversibility:
Reactants: Alpha-ketoglutarate + NAD+ + CoASH
Products: Succinyl-CoA + NADH + CO2
Enzyme: Alpha-Ketoglutarate dehydrogenase
Coenzyme: TPP, Lipoate, FAD
Delta G: -33.5 kJ/mol
Reversibility: Irreversible
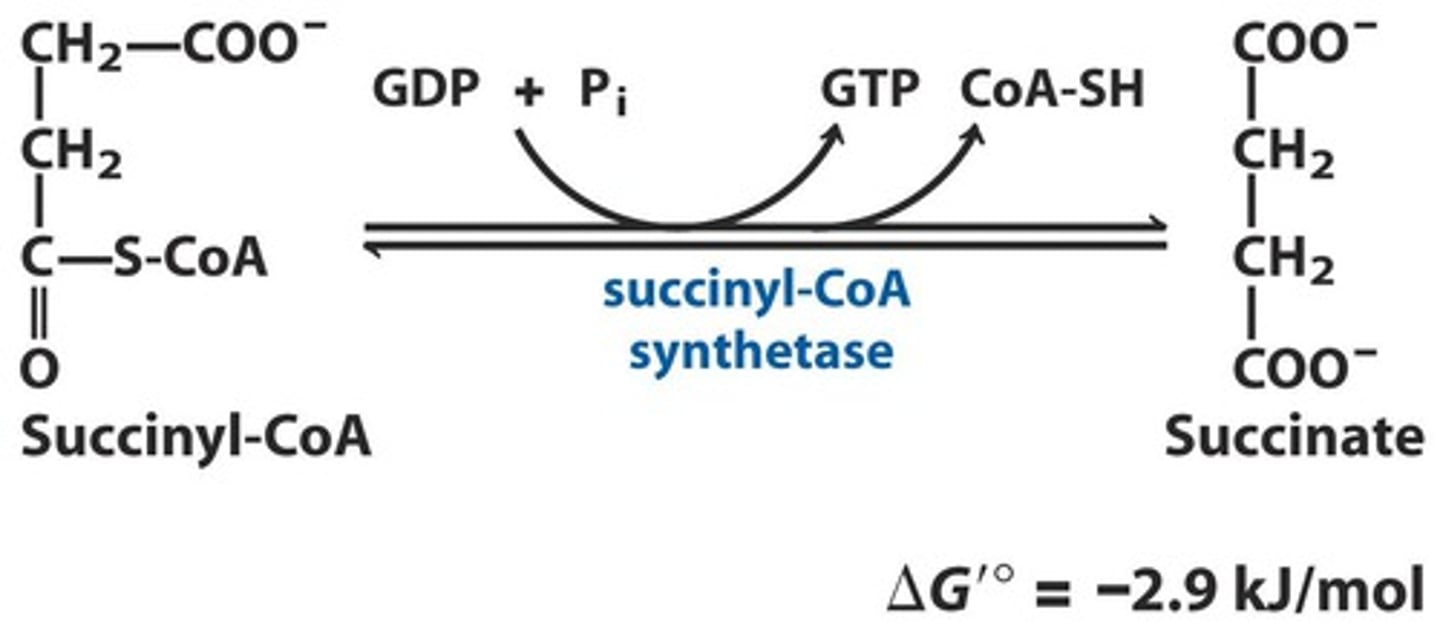
TCA Reaction 5: Substrate level phosphorylation
Reactants:
Products:
Enzyme:
Delta G:
Reversibility:
Reactants: Succinyl-CoA + GDP (ADP) + P
Products: Succinate + GTP (ATP) + CoA-SH
Enzyme: Succinyl-CoA Synthetase
Delta G: -2.9 kJ/mol
Reversibility: Reversible
TCA Reaction 6: Succinate Dehydrogenase
Reactants:
Products:
Enzyme:
Delta G:
Reversibility:
Reactants: Succinate + FAD
Products: Fumarate + FADH2
Enzyme: Succinate Dehydrogenase
Delta G: 0 kJ/mol
Reversibility: Reversible
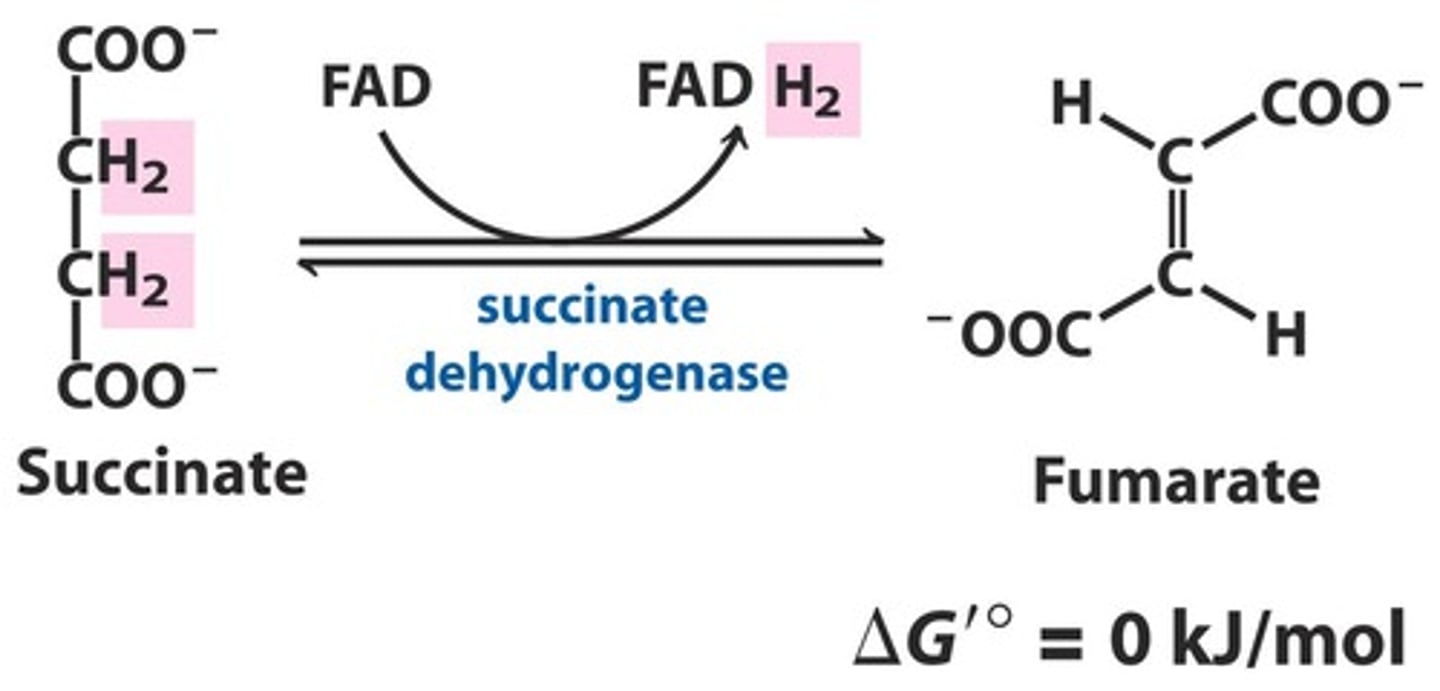
How is FAD linked to proteins?
Covalently linked to protein; Limited mobility
What is the redox potential of FAD compared to NAD+?
Carries a lower redox potential than NAD+
Is FAD easier or harder to reduce compared to NAD+?
Easier to reduce (Source is not as easily oxidized)
Is NAD+ water soluble?
Yes
Does NAD+ have a higher or lower redox potential than FAD?
Higher
What type of reaction is needed to reduce NAD+?
A highly exergonic reaction
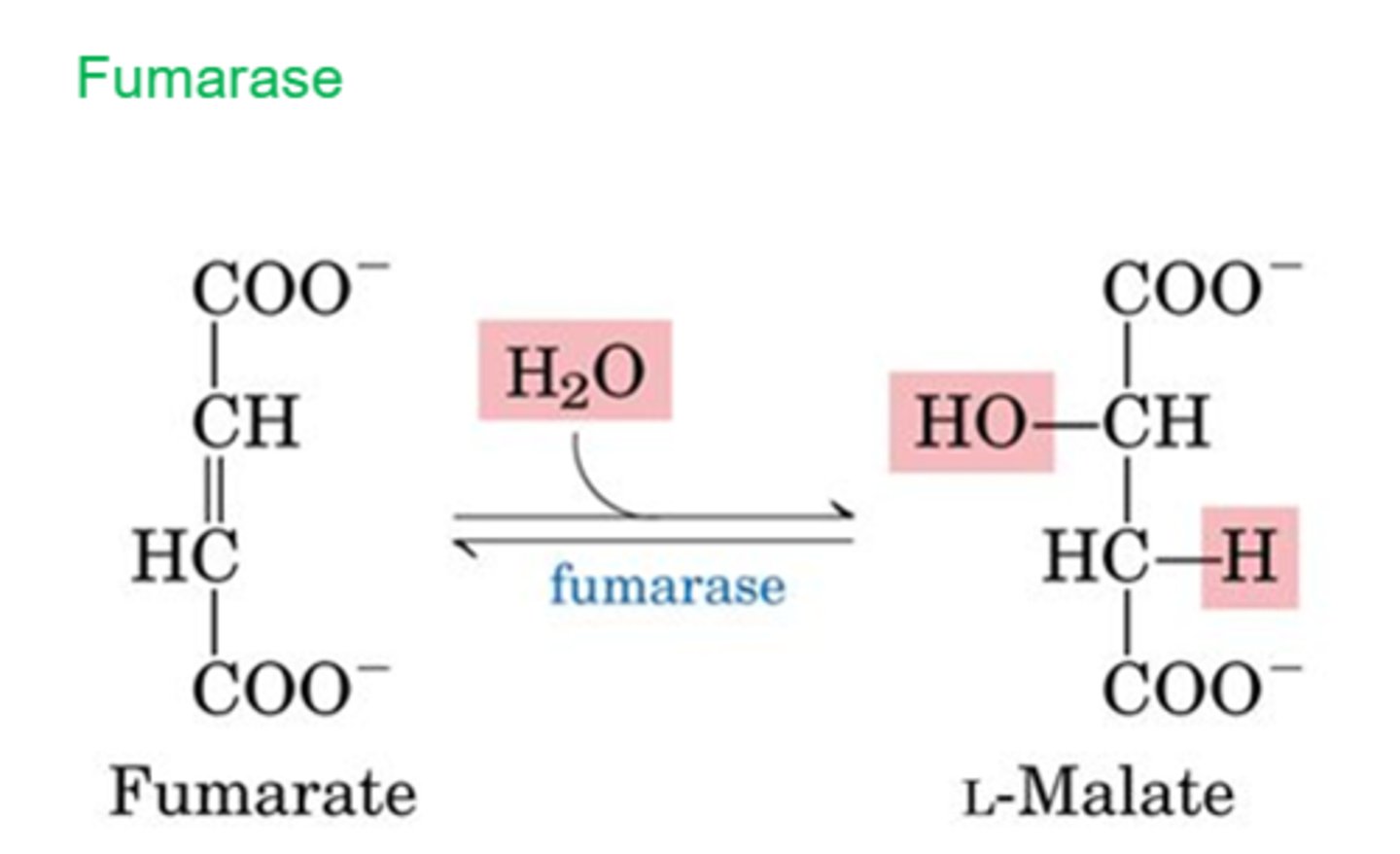
TCA Reaction 7: Hydration of Fumarate
Reactants:
Products:
Enzyme:
Delta G:
Reversibility:
Reactants: Fumarate + H2O
Products: L-Malate
Enzyme: Fumarase
Delta G: 0 kJ/mol
Reversibility: Reversible
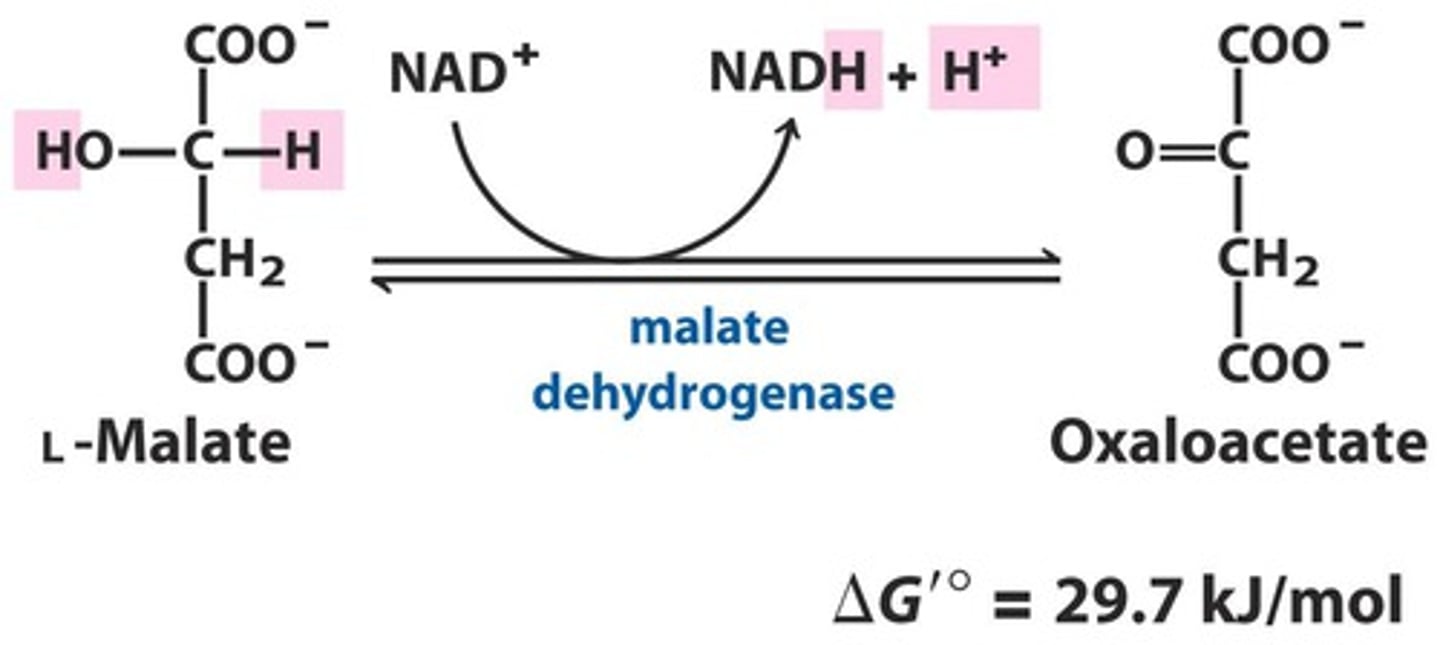
TCA Reaction 8: Malate Dehydrogenase catalyzes the final oxidation and produces oxaloacetate
Reactants:
Products:
Enzyme:
Delta G:
Reversibility:
Reactants: L-Malate + NAD+
Products: Oxaloacetate + NADH + H+
Enzyme: malate Dehydrogenase
Delta G: 29.7 kJ/mol
Reversibility: Reversible
Is the concentration of Oxaloacetate from step 8 of TCA high or low
Low
Products of 1 Trip around TCA
3 NADH
1 FADH2
1 GTP (ATP)
2 CO2
How many trips around TCA occurs per molecule of glucose
2
Where does Oxidative Phosphorylation Occur?
mitochondria
Coenzyme Q (CoQ)
-Ubiquinone (UQ or Q)
-Quinone head group with long hydrocarbon tail
-Transfer one of two electrons
What enters Complex I of the oxidation process?
NADH + H+
What is formed when 2 electrons are transferred to Q in Complex I?
QH2
What happens during the electron transfer in Complex I?
A conformation change occurs, releasing 4 H+ into the intermembrane space
What is generated as a result of the reactions in Complex I and pumped into the IMS?
A pool of UQH2
What is the function of Complex II in the electron transport chain?
Complex II produces enough energy to reduce UQ into UQH2.
What does FADH2 donate in the process of fumarate formation in the TCA cycle?
FADH2 donates H2 for QH2.
Does Complex II pump protons into the intermembrane space (IMS)?
No, Complex II does not pump H+ into IMS.
What does Complex II generate in terms of ubiquinone in the IMS?
Complex II generates a pool of UQH2.
What is the function of Complex III in the electron transport chain?
Oxidizes UQH2, transferring 1 electron at a time via cytochrome c
How many subunits does Complex III have?
9-10 subunits.
What mechanism does Complex III use to drive proton transport?
The Q cycle.
Q Cycle Step 1:
- Oxidation of the first UQH2 to UQ
- 1 electron goes to cytochrome C
- 1 electron goes back to UQ to form UQdot (semiquinone intermediate)
- 2 H+ released
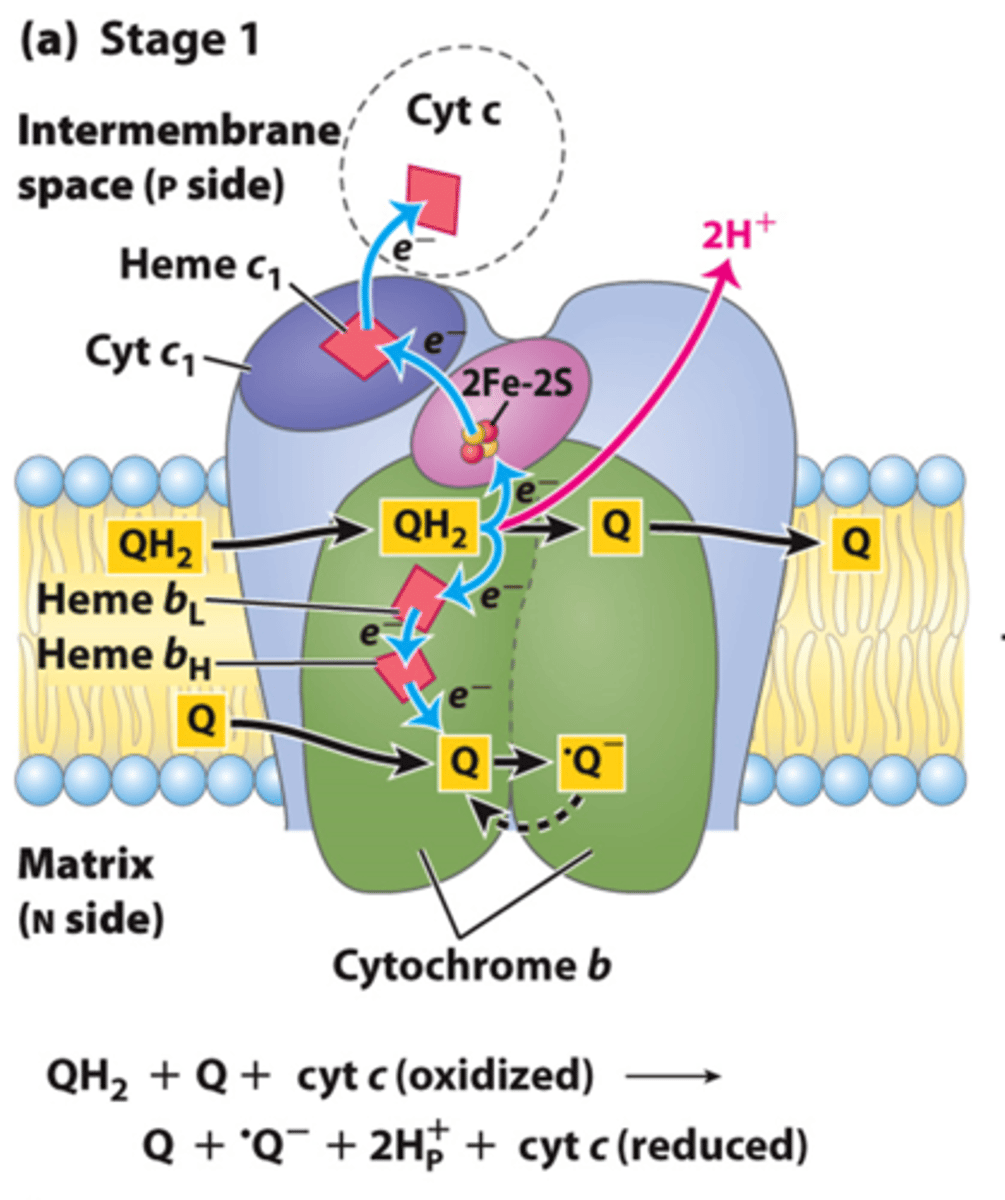
Q Cycle Step 2:
- Oxidation of the second UQH2 to UQ
- 1 electron goes to cytochrome c
- 1 electron goes to UQdot (semiquinone intermediate) to regenerate UQH2
- 2 H+ released
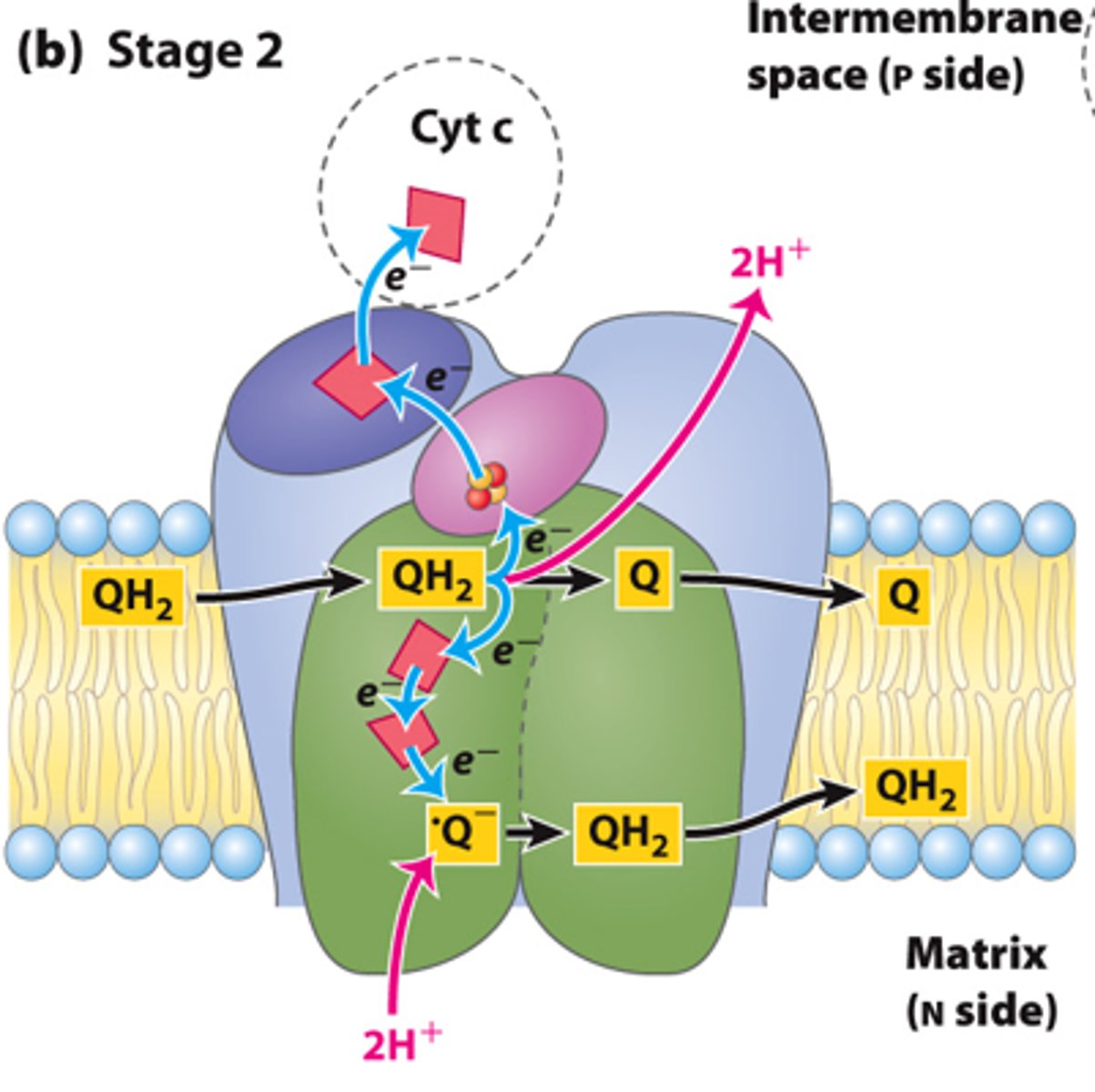
How many H+ Released by compelex III?
4 H+
What is Complex IV also known as?
Cytochrome C oxidase
What does Complex IV accept electrons from?
Cytochrome c
What is the result of the 4 electron reduction of O2 in Complex IV?
Formation of 2 H2O
What has the highest affinity for electrons in the electron transport chain?
Oxygen
How many protons are pumped across the inner membrane per 4 electrons transferred from cytochrome c to O2?
4 H+
Which metal ions are involved in transferring electrons to stabilize oxygen in Complex IV?
Copper (Cu) and Iron (Fe)
What does Complex IV minimize the formation of?
Reactive oxygen species (H2O2, O2-)
How many H+ are pumped from NADH Oxidation?
10 H+
How many H+ are pumped from FADH2 Oxidation?
6 H+
Chemiosmosis
The movement of protons down its concentration gradient is coupled with ATP Synthesis
What is the result of uncoupling O2 consumption and ATP Synthesis?
O2 consumption skyrockets
ATP Synthesis decreases
What drives the rotation of the F0 rotor in ATP Synthase?
Proton gradient
What happens when the F0 rotor and rotor shaft rotate in ATP Synthase?
It causes a conformational change in the F1 subunit.
What do conformational changes in the F1 subunit of ATP Synthase drive?
ATP synthesis
What is the structure of the F0 subunit in ATP synthase?
It has a hydrophobic core embedded within the mitochondria inner membrane
What is the function of the stator (b2) in the F0 subunit?
It is the stationary component that anchors the entire enzyme.
What is the role of the rotor in the F0 subunit?
It consists of 8-15 c subunits that rotate as H+ flow into subunit a.
What is the function of the rotor shaft or stalk (gamma) in ATP synthase?
It translates rotational movement in F0 to cause conformational changes in F1.
What causes F0 rotation in ATP synthase?
The flow of H+ ions
What happens when a proton enters from the IMS in ATP synthase?
It protonates an Aspartate
What occurs after the Aspartate is protonated in ATP synthase?
The c-subunit becomes neutral and can rotate into the hydrophobic membrane
What effect does the rotation of the c-subunit have on the entire ring in ATP synthase?
It pushes the entire ring forward by one subunit.
What happens when a c-subunit completes the circle and hits the Matrix channel?
The proton dissociates
Where does the changed Aspartate stay until it is protonated again?
At the aqueous interface.
What is the structure of the F1 subunit?
Hexamer of alpha and beta subunits
Which subunits of the F1 subunit catalyze ATP synthesis?
Only beta subunits
What type of change do the beta subunits undergo?
Significant conformational change
How many states are the beta subunits found in?
3 states
What is the shape of the rotor shaft in the F1 subunit?
Asymmetrical
What effect does the asymmetrical rotor shaft have on the beta subunits?
Causes each beta subunit to be in different conformations
What causes the Beta-subunit conformational change?
Rotor Shaft Rotation
Which subunit interacts strongly with the Open beta subunit form?
Gamma subunit
How often does the gamma subunit come into contact with a different ATP-containing beta subunit?
Every 120 degrees
What state is the beta subunit forced into when the gamma subunit interacts with it?
Beta-empty state
What is required to move the gamma subunit from the empty beta subunit to the next ATP-containing beta subunit?
Energy
What is the affinity of the Open (O) state?
Lowest Affinity
What is the affinity of the Loose (L) state?
Affinity for ADP + Pi
What is the affinity of the Tight (T) state?
High affinity for ATP
How much ATP is produced from NADH Pumps?
2.5 ATP
How much ATP is produced from FADH2 Pumps?
1.5 ATP