IB Chemistry HL Final
1/69
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
70 Terms
ionic bonding
an electrostatic attraction between oppositely charged ions
what must the difference in electronegativities be greater than for a compound to be ionic
(greater than) 1.8
covalent bonding
an electrostatic attraction between a bonding pair of electrons and two positively charged nuclei
metallic bonding
sea of delocalized electrons around a lattice of positively charged nuclei
what is the carbon allotrope of diamond's structure
giant 3D macromolecule (covalently bonded)
what arrangement is diamond
tetrahedral
what arrangement is graphite
trigonal planar
what is the structure of graphite
sheet-like
what is the arrangement of C60
truncated icosahedron (20 hexagons and 12 pentagons)
exothermic
heat loss to surroundings from system
endothermic
heat absorbed by the system from surrounding
heat
total energy of a given amount of a substance
temperature
measure of "hotness" (average kinetic energy)
activation energy
the minimum energy needed to start a reaction
what is the haber process
production of NH3 (ammonia)
what is the contact process
production of SO3 (sulfur trioxide...precursor to H2SO4)
what are the strong acids?
HCl, H2SO4, HNO3
what are the weak acids?
H2CO3, CH3COOH
what are the weak bases?
NH3/NH4OH, C2H5NH2/CH3CH2NH2
what are the strong bases?
NaOH, KOH, Ba(OH)2
bronsted-lowry acid
proton donor
bronsted- lowry base
proton acceptor
strong
completely dissociated into its ion (-->)
concentrated
high # of mol/dm^3
weak
partially dissociated into its ion (equilibrium arrow)
dilute
low # mol/dm^3
lewis acid
electron pair acceptor
lewis base
electron pair donor
isotopes
all atoms of same element with same # of protons/atomic # but differing #s of neutrons
when something is isoelectronic to an element, they have the same # of...?
electrons (ex: Cl- and Ar)
relative atomic mass (Ar)
(relative to 12C) weighted average of naturally occurring isotopes
atomic radius
distance from nucleus to outermost electron
1st ionization energy
energy required to remove an electron from a neutral atom in the gaseous state
electronegativity
attraction of an atom to a shared/bonding pair of electrons
electron affinity
the change in energy when an electron is added to an isolated atom in the gaseous state
alkali are all...
bases!
the nature of the acid/base of Na2O is...
strongly basic (NaOH)
the nature of the acid/base of MgO is...
basic (Mg(OH)2)
the nature of the acid/base of Al2O3 is...
amphoteric
the nature of the acid/base of SiO2 is...
weakly acidic
the nature of the acid/base of P4O10 is...
acid (weak) (H3PO4)
the nature of the acid/base of SO3 is...
strongly acidic (H2SO4)
the nature of the acid/base of Cl2O7 is...
strongly acidic (HCl)
amphiprotic
can either donate/accept an electron
amphoteric
can act as either an acid or base
monoprotic
acid with one H+
diprotic
acid with two H+'s
hybridization
molecular orbital theory of bonding
sigma bonds are...
single bonds
pi bonds are...
multiple bonds
sigma bonds
formed by axial overlap of atomic overlap (same orientation) (head-on/axial)
pi bonds
2 p orbitals that overlap side-ways/parallel
bond angle for sp3
109.5
bond angle for sp2
120
bond angle for sp
180
resonance
equal probability of double bond in two or more directions
delta H f theta
change in enthalpy when one mol of a compound is formed from its elements under standard conditions (298K) (1 atm)
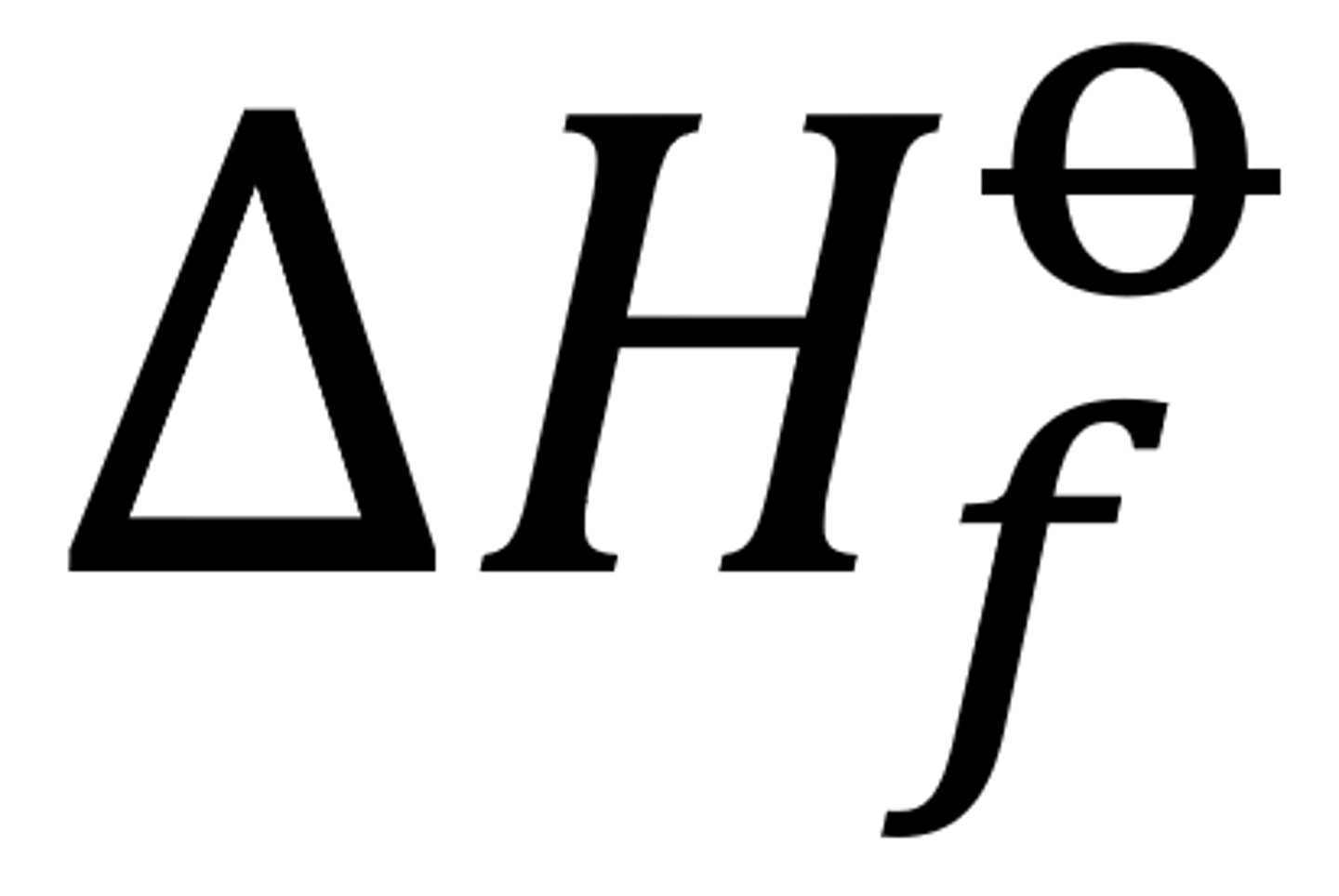
delta H c theta
change in enthalpy when one mol of compound is combusted (completely) in O2 under standard conditions (298K) (1 atm)
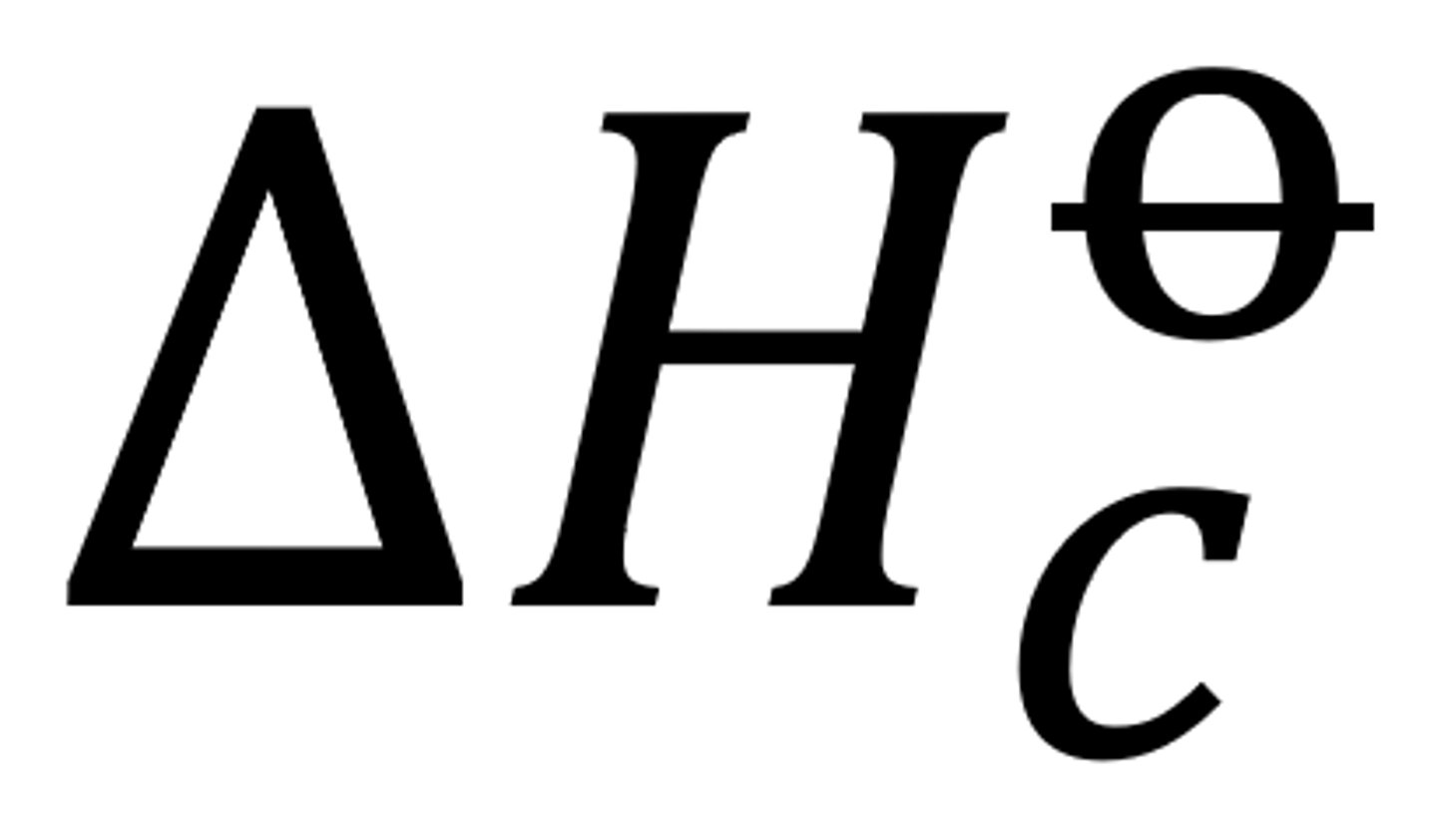
delta H at theta
change in enthalpy of atomization when 1 mol gaseous atom is formed under standard conditions (298K) (1 atm)
delta H ea theta
change in enthalpy of electron affinity when one electron is addd to an isolated atom in the gaseous state under standard conditions (298K) (1 atm)
delta H latt theta (endothermic)
change in lattice enthalpy when a crystalline solid is turned into gaseous ions under standard conditions (298K) (1 atm)
delta H latt theta (exothermic)
change in lattice enthalpy when gaseous ions are turned into a crystalline solid under standard conditions (298K) (1 atm)
delta H solution theta
change in enthalpy when one mol of ionic compound dissolves in water to make an infinitely dilute solution under standard conditions (298K) (1 atm)
delta H hydration theta
change in enthalpy when one mol of gaseous ions dissolve in water to give an infinitely dilute solution under standard conditions (298K) (1 atm)
entropy
(S) disorder
delta S reaction theta
S products theta - S reactants theta
spontaneity
will the reaction want to happen?
gibbs free energy
spontaneous if negative
equation for gibbs free energy
delta g theta = delta h theta - T (delta s theta)
isomerism
same MF, different structural formulas