AP Biology unit 1 - chemistry of life
1/52
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
53 Terms
Elements of life
C, H, N, O, P, S
the four bio molecule families
carbohydrates, lipids, proteins, nucleic acids
Water is a....
polar molecule with unequal electron sharing between oxygen and hydrogen.
The negative and positive parts of water
Oxygen is negative, hydrogen is positive
The kind of bonds water has
Hydrogen bonds which are intermolecular and covalent bonds which are intramolecular
Hydrogen bonds are essential for
DNA and RNA structure
cohesion
hydrogen bonds between water molecules
Cohesion is responsible for
high heat of vaporization, high specific heat/hold a lot of water, high surface tension
Adhesion
hydrogen bonds between water and other polar molecules
transpiration
how water gets pulled up to the top of trees
surface tension
force exerted by the water molecules on the surface of a body of water
Carbohydrates, proteins, nucleic acids are built from
monomers
polymers
chain of monomers
dehydration synthesis
process of putting monomers together to make polymers, water is exerted
hydrolysis
enzymes insert water molecule between the monomers making up a polymer
phosphate
key for energy exchange
methyl
used to silence DNA or turn off genes
hydroxl and carbonyl
makes a molecule hydrophilic/water soluble
carboxyl and amino
essential in amino acids
Sulfhydrl
important in protein structure, creates stabilizing bond that holds proteins in a 3 dimensional shape.
acetyl
used to activate DNA through process called acetylation
monosaccharide
energy sources and building blocks, simple sugars
Disaccharides
energy transfer, two linked monossacharides
polysaccharide
energy storage, starch for plants and glycogen for animals; structure rolls, like cellulose in plant walls
lipids
wholly or partly non polar, not composed of repeating monomers
lipid functions
long term energy storage (fats or oils), wax (waterproofing), membrane function (phospholipid bilayer), signaling, insulation
phospholipid structure
hydrophilic head that is polar and hydrophobic tail that is nonpolar
the four levels of protein structure
primary structure, secondary structure, tertiary structure, Quaternary structure
primary structure
linear sequence of amino acids that is genetically determined
secondary structure
interactions between carbonyl group and amino groups within a polypeptide backbone, forms hydrogen bonds which stabalizes shapes
tertiary structure
emerges from interactions between amino acid side chains/r groups. Can either have a alpha helix shape or beta pleated sheet shape.
alpha helix
a corkscrew shape stabalized by internal hydrogen bonds within the polypeptide backbone
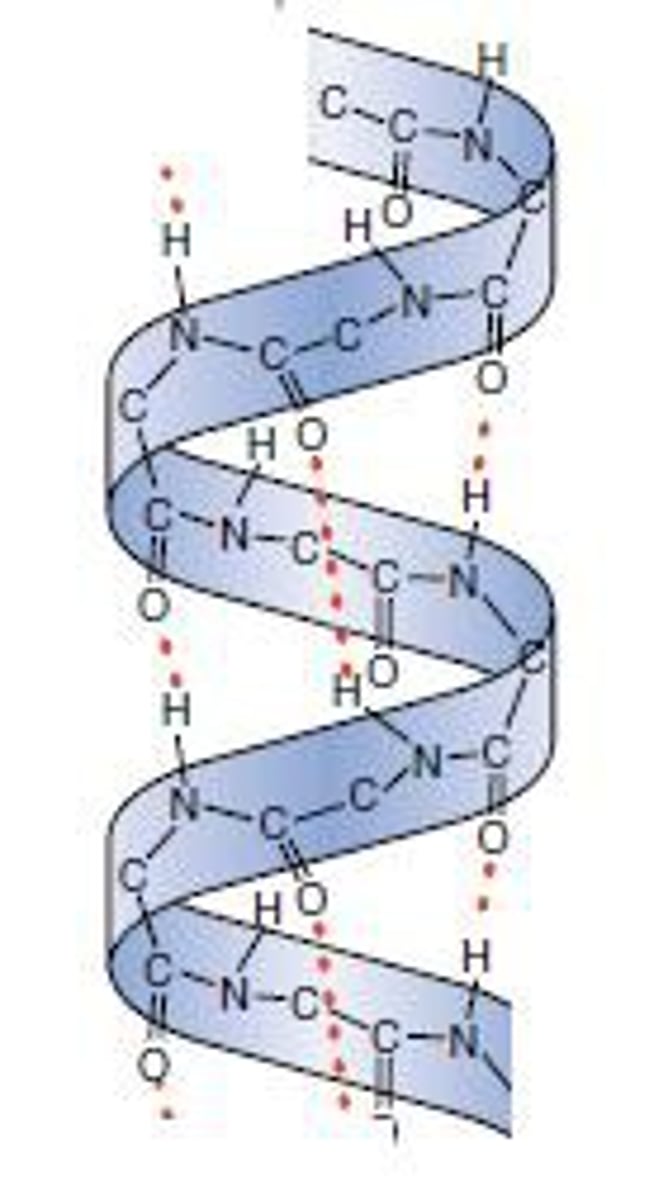
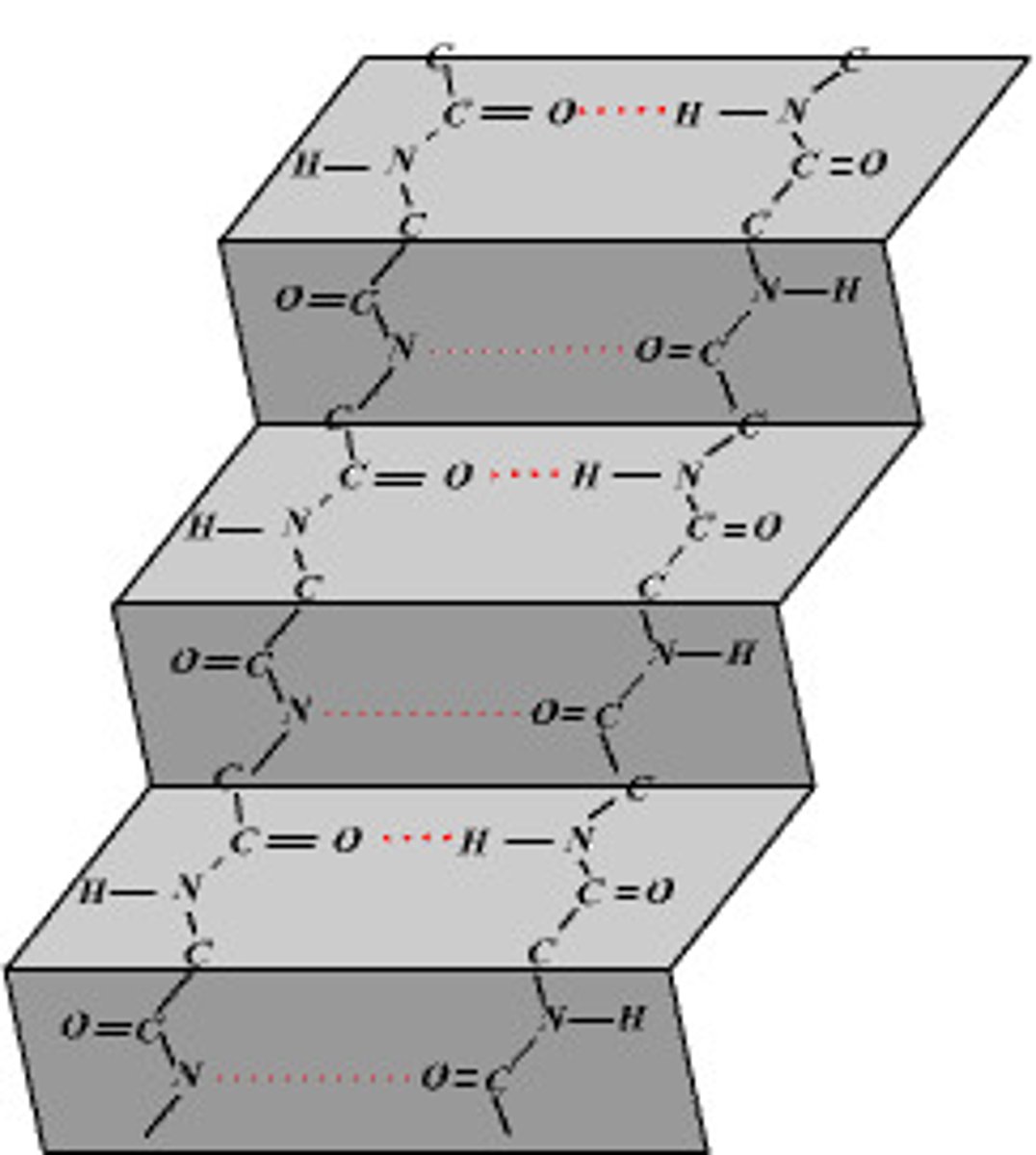
beta pleated sheet
stabilized by hydrogen bonds within the polypeptide backbone
Quaternary structure
involves multiple folded polypeptide chains that bond together, can involve hydrogen bonds, ionic bonds, hydrophobic interactions, and rarely covalent bonds .
nucleic acids
molecules of genetic information
DNA
Molecule of hereditary
RNA
information transfer which can fold, bend into variety of shapes which leads to variety of functions
nucelotides
monomer of nucleic acids
amino aicd
monomer of protein
Sugar in DNA
deoxyribose
DNA bases
adenine, thymine, cytosine, guanine
Sugar in RNA
ribose
RNA bases
adenine, uracil, cytosine, guanine
structure of DNA
two nucleotide strands, within each strand the nucleotides are connected by sugar phosphate bonds. Strands connect to one another by hydrogen bonds. The two strands area antiparallel
DNA Base matches
Adenine --> thymine
Guanine --> cytosine
polymerases
enzymes that build DNA which can only add new nucleotides at the 3' end of growing strand
RNA base matches
Adenine --> Uracil
Guanine --> cytosine
carbohydrate functions
short term energy storage, main source of energy, excess is converted to fat, structural base on many organisms
Elements that are in carbohydrates
CHO, Carbon, Hydrogen, Oxygen
elements that are in lipids
CHO, Carbon, Hydrogen Oxygen
protein functions
makes up things like muscle tissues, hair, collagen, protein channels, can be receptors in cell signaling
elements that are in proteins
CHON, Carbon, Hydrogen, Ozygen, Nitrogen
Elements in nucleic acids
CHONP, Carbon, hydrogen, oxygen, nitrogen, phosphorus