thermochem
0.0(0)
Studied by 1 personCard Sorting
1/113
Earn XP
Description and Tags
Last updated 5:25 PM on 10/21/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
114 Terms
1
New cards
Internal energy
molecular composition
2
New cards
what type of energy does a glass of water contain
internal energy
3
New cards
Heat
molecular temperature
4
New cards
is Kinetic energy (KE) is proportional to an object's velocity.
yes
5
New cards
typical definition of energy?
The capacity to do work
6
New cards
What type of energy is increasing when... turning on a light bulb
internal energy
7
New cards
What type of energy is increasing when... An egg falls from a table
Kinetic energy
8
New cards
What type of energy is increasing when...Water begins to boil
potential energy
9
New cards
What type of energy is increasing when... water begins to freeze
Potential energy
10
New cards
What type of energy is increasing when... a skateboard rolls on flat ground
kinetic energy
11
New cards
Potential Energy (PE)
molecular motion
12
New cards
Potential energy is
molecular position
13
New cards
endothermic means
heat enters from surrounds
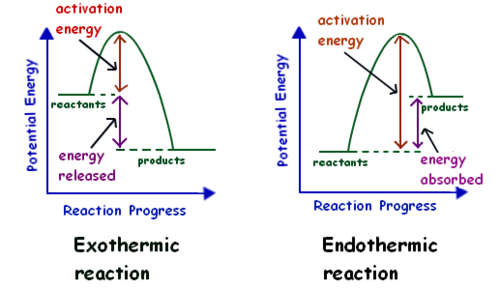
14
New cards
exothermic means
releases heat to surroundings
15
New cards
endothermic is what in the equation
enters as reactant
16
New cards
exothermic in equation is
exits as a product
17
New cards
If energy is produced during a chemical reaction, then the reaction is
exothermic
18
New cards
State functions are known to be
path-independentfunctions.
19
New cards
what describes a state function?
A path-independent function
20
New cards
458 kJ is equivalent to how many calories (cal)?
1.09E5
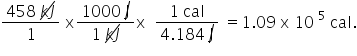
21
New cards
Which quantity of energy is the biggest: 65.4 kJ or 3.21 x 10 kWh?
65.4 kJ
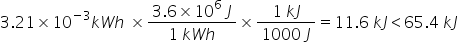
22
New cards
is (1253 Cal > 11553 cal) correct?
true
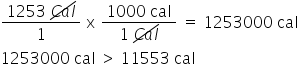
23
New cards
Which of the following is not a unit of energy?
J, K, kJ, cal
J, K, kJ, cal
K
24
New cards
Convert 234 kJ to kcal. Only include the numerical value in your answer.
5.59E1
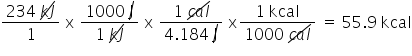
25
New cards
Convert 614 Cal to J. Only include the numerical value in your answer.
2.57E6
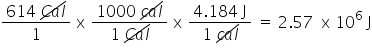
26
New cards
Convert 5.78 x 10 kWh to kJ. Only include the numerical value in your answer.
2.08E6
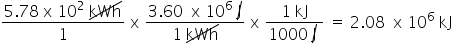
27
New cards
Convert 1.28 x 10cal to kWh. Only include the numerical value in your answer.
1.49E-9
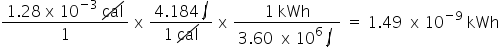
28
New cards
Convert 202 CV to Ws. Only include the numerical value in your answer.
2.02E2
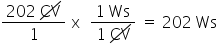
29
New cards
If electricity costs $0.12 per kWh, then what is the total cost for a family of four to illuminate ten 60 watt bulbs each for 30 days? (Round to the nearest dollar)
207.0
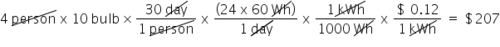
30
New cards
You observe a cup containing ice that is melting inside a room at temperature 25 C. Identify the correct statement.
a The change in heat with respect to the ice is negative
bThe ice cube is the system and gains heat from the higher temperature room
cThe change in heat with respect to the room is positive
dThe ice cube melts because cold energy exits and enters the room
a The change in heat with respect to the ice is negative
bThe ice cube is the system and gains heat from the higher temperature room
cThe change in heat with respect to the room is positive
dThe ice cube melts because cold energy exits and enters the room
B
31
New cards
does A negative heat (-q) implies an exothermic reaction?
yes
A negative sign for q means that the system is losing heat; an exothermic process.
A negative sign for q means that the system is losing heat; an exothermic process.
32
New cards
Work is done by a gas whenever
For work to be done, the gas must change volume, ∆V≠0.
33
New cards
the equation representing expansion work.
w= - PΔV
34
New cards
are Heat and thermal energy are the same thing?
no
Heat is the energy that flows from a higher temperature object to a lower temperature object as a result of their temperature difference. Thermal energy is the energy associated with the incessant and haphazard motion of atoms or molecules above absolute zero (0 Kelvin).
Heat is the energy that flows from a higher temperature object to a lower temperature object as a result of their temperature difference. Thermal energy is the energy associated with the incessant and haphazard motion of atoms or molecules above absolute zero (0 Kelvin).
35
New cards
A constant volume calorimeter (bomb calorimeter) was calibrated by performing in it a reaction in which 5.23 kJ of heat energy was released, causing the calorimeter to rise by 7.33 °C. What is the heat capacity C of the calorimeter?
7.14E-1

36
New cards
Next, a chemical reaction of interest was conducted in the same constant volume calorimeter. The neutralization reaction of HCl(aq) with NaOH(aq) caused the temperature of the calorimeter to rise by 2.46 °C.What is the change in internal energy ΔU of the neutralization reaction in kJ?
-1.76E0
explanation
△U=qrxn=−Cv△T=(−0.714kJ/K)(2.46K)=−1.76kJ
explanation
△U=qrxn=−Cv△T=(−0.714kJ/K)(2.46K)=−1.76kJ
37
New cards
A typical hamburger has a serving size of 105 g and consists of approximately 20% protein (21.0 g), 33% fat (34.7 g), and 47% carbohydrate (49.3 g). Assume that the heat capacity of the bomb calorimeter is 8.39 kJ K-1, and the increase in water temperature was 13.2 K.
How many Calories (Cal) are in the hamburger described above?
How many Calories (Cal) are in the hamburger described above?
2.65E1
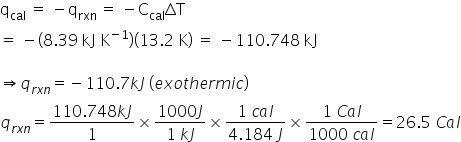
38
New cards
If the same hamburger was placed in a different bomb calorimeter whose is 7.66 kJ K, then what would be the change in temperature of the water bath?
1.45E1
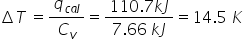
39
New cards
is this statement true(A material with a greater specific heat capacity will experience a greater temperature change than a material with a smaller specific heat capacity per unit of heat)?
false
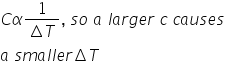
40
New cards
How much heat (in kJ) is needed to heat a 200.0 mL sample of water initially at 25.0 °C to a boiling temperature of 100.0 °C?
6.28E1
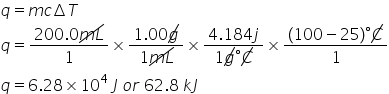
41
New cards
A 6.88 g piece of copper is heated from 20.0 C to 125 C and then submerged in 45.0 mL of water initially at 18.3 C. What is the final temperature of the water?
1.98E1
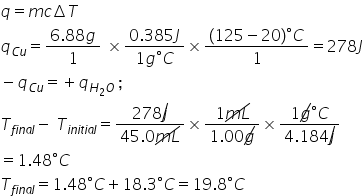
42
New cards
Isobaric calorimetry
is also known as coffee-cup calorimetry.
43
New cards
Isochoric calorimetry
is also known as bomb calorimetry.
44
New cards
Isobaric condition
takes place under constant pressure
45
New cards
Isochoric condition
takes place under constant volume
46
New cards
Isobaric measurement
the amount of heat transferred is equal to ΔH
47
New cards
Isochoric measurement
all energy is transferred in the form of heat
48
New cards
When hydrogen gas reacts with oxygen gas under certain conditions to produce liquid water, 572.0 kJ of heat energy is released. Is the reaction exothermic or endothermic?
exothermic
We are told that energy is released in the form of heat. By definition, this is an exothermic process.
We are told that energy is released in the form of heat. By definition, this is an exothermic process.
49
New cards
When hydrogen gas reacts with oxygen gas under certain conditions to produce liquid water, 572.0 kJ of heat energy is released. What is the enthalpy change of the reaction when exactly 67.50 grams of water vapor forms?
-1071 kJ
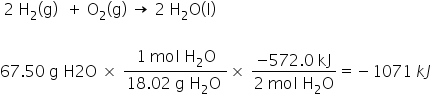
50
New cards
What is the enthalpy of formation (, in kJmol) of Pb(NO)(s) ?
-4.519E2
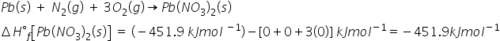
51
New cards
Write an equation for the enthalpy of formation of NaHCO(s).
Na(s), H(g), O(g), C(s, graphite)
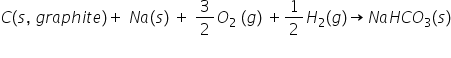
52
New cards
The standard enthalpy of formation () of any element in its standard state is always zero. True or false?
true,The standard state of an element is its states as a solid, liquid or gas to atmosphere of pressure and 25 °C.
53
New cards
Consider the reaction of nitrogen dioxide with molecular hydrogen:2 NO(g) + 7 H(g) → 4 HO(l) + 2 NH(g)
Calculate for this reaction given by manipulation of reactions (i) and (ii):
2 NH(g) → N(g) + 3 H(g) = +92.0 kJ (i)
N(g) + 4 HO(l) → 2 NO(g) + 4 H(g) = +3.40E2 kJ (ii)
Calculate for this reaction given by manipulation of reactions (i) and (ii):
2 NH(g) → N(g) + 3 H(g) = +92.0 kJ (i)
N(g) + 4 HO(l) → 2 NO(g) + 4 H(g) = +3.40E2 kJ (ii)
-4.32E2
Our first task is to find a puzzle - piece containing 2 NO2(g) as a reactant. We see that equation (ii) has 2 NO2 (g) as a product. we know to reverse this reaction to make the product 2 NO2 (g) a reactant instead:
With our first puzzle-piece in place the next piece is 7 H2(g) as a reactant. There are already 4 H2 (g) molecules in the above reaction so we need only 3 more H2(g) to make 7 H2(g) the extra 3 H2(g) come from the reverse of (i):
Summing the reactant and product and allow us to cancel N2(g) as a spectator
Our first task is to find a puzzle - piece containing 2 NO2(g) as a reactant. We see that equation (ii) has 2 NO2 (g) as a product. we know to reverse this reaction to make the product 2 NO2 (g) a reactant instead:
With our first puzzle-piece in place the next piece is 7 H2(g) as a reactant. There are already 4 H2 (g) molecules in the above reaction so we need only 3 more H2(g) to make 7 H2(g) the extra 3 H2(g) come from the reverse of (i):
Summing the reactant and product and allow us to cancel N2(g) as a spectator
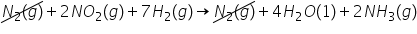
54
New cards
Joule
a derived SI unit of energy equivalent to 1 kg m2 s-2.
55
New cards
Isolated system
- a system that cannot transfer energy or matter.
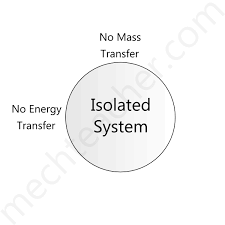
56
New cards
Closed system
a system that can transfer energy but not matter.
57
New cards
Open system
a system that can transfer energy and matter.
58
New cards
Internal energy
molecular composition
59
New cards
what type of energy does a glass of water contain
internal energy
60
New cards
Heat
molecular temperature
61
New cards
is Kinetic energy (KE) is proportional to an object's velocity.
yes
62
New cards
typical definition of energy?
The capacity to do work
63
New cards
What type of energy is increasing when... turning on a light bulb
internal energy
64
New cards
What type of energy is increasing when... An egg falls from a table
Kinetic energy
65
New cards
What type of energy is increasing when...Water begins to boil
potential energy
66
New cards
What type of energy is increasing when... water begins to freeze
Potential energy
67
New cards
What type of energy is increasing when... a skateboard rolls on flat ground
kinetic energy
68
New cards
Potential Energy (PE)
molecular motion
69
New cards
Potential energy is
molecular position
70
New cards
endothermic means
heat enters from surrounds
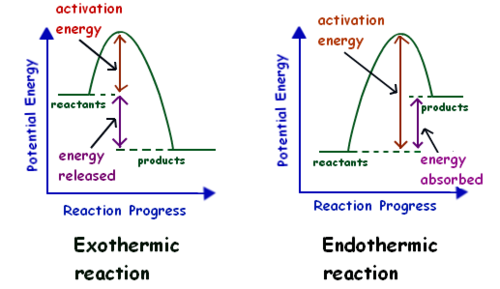
71
New cards
exothermic means
releases heat to surroundings
72
New cards
endothermic is what in the equation
enters as reactant
73
New cards
exothermic in equation is
exits as a product
74
New cards
If energy is produced during a chemical reaction, then the reaction is
exothermic
75
New cards
State functions are known to be
path-independentfunctions.
76
New cards
what describes a state function?
A path-independent function
77
New cards
458 kJ is equivalent to how many calories (cal)?
1.09E5
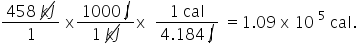
78
New cards
Which quantity of energy is the biggest: 65.4 kJ or 3.21 x 10 kWh?
65.4 kJ
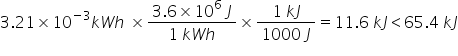
79
New cards
is (1253 Cal > 11553 cal) correct?
true
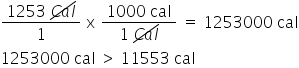
80
New cards
Which of the following is not a unit of energy?
J, K, kJ, cal
J, K, kJ, cal
K
81
New cards
Convert 234 kJ to kcal. Only include the numerical value in your answer.
5.59E1
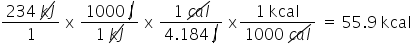
82
New cards
Convert 614 Cal to J. Only include the numerical value in your answer.
2.57E6
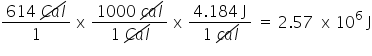
83
New cards
Convert 5.78 x 10 kWh to kJ. Only include the numerical value in your answer.
2.08E6
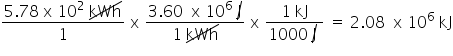
84
New cards
Convert 1.28 x 10cal to kWh. Only include the numerical value in your answer.
1.49E-9
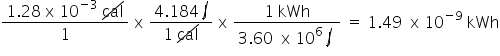
85
New cards
Convert 202 CV to Ws. Only include the numerical value in your answer.
2.02E2
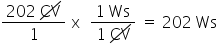
86
New cards
If electricity costs $0.12 per kWh, then what is the total cost for a family of four to illuminate ten 60 watt bulbs each for 30 days? (Round to the nearest dollar)
207.0
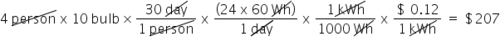
87
New cards
You observe a cup containing ice that is melting inside a room at temperature 25 C. Identify the correct statement.
a The change in heat with respect to the ice is negative
bThe ice cube is the system and gains heat from the higher temperature room
cThe change in heat with respect to the room is positive
dThe ice cube melts because cold energy exits and enters the room
a The change in heat with respect to the ice is negative
bThe ice cube is the system and gains heat from the higher temperature room
cThe change in heat with respect to the room is positive
dThe ice cube melts because cold energy exits and enters the room
B
88
New cards
does A negative heat (-q) implies an exothermic reaction?
yes
A negative sign for q means that the system is losing heat; an exothermic process.
A negative sign for q means that the system is losing heat; an exothermic process.
89
New cards
Work is done by a gas whenever
For work to be done, the gas must change volume, ∆V≠0.
90
New cards
the equation representing expansion work.
w= - PΔV
91
New cards
are Heat and thermal energy are the same thing?
no
Heat is the energy that flows from a higher temperature object to a lower temperature object as a result of their temperature difference. Thermal energy is the energy associated with the incessant and haphazard motion of atoms or molecules above absolute zero (0 Kelvin).
Heat is the energy that flows from a higher temperature object to a lower temperature object as a result of their temperature difference. Thermal energy is the energy associated with the incessant and haphazard motion of atoms or molecules above absolute zero (0 Kelvin).
92
New cards
A constant volume calorimeter (bomb calorimeter) was calibrated by performing in it a reaction in which 5.23 kJ of heat energy was released, causing the calorimeter to rise by 7.33 °C. What is the heat capacity C of the calorimeter?
7.14E-1

93
New cards
Next, a chemical reaction of interest was conducted in the same constant volume calorimeter. The neutralization reaction of HCl(aq) with NaOH(aq) caused the temperature of the calorimeter to rise by 2.46 °C.What is the change in internal energy ΔU of the neutralization reaction in kJ?
-1.76E0
explanation
△U=qrxn=−Cv△T=(−0.714kJ/K)(2.46K)=−1.76kJ
explanation
△U=qrxn=−Cv△T=(−0.714kJ/K)(2.46K)=−1.76kJ
94
New cards
A typical hamburger has a serving size of 105 g and consists of approximately 20% protein (21.0 g), 33% fat (34.7 g), and 47% carbohydrate (49.3 g). Assume that the heat capacity of the bomb calorimeter is 8.39 kJ K-1, and the increase in water temperature was 13.2 K.
How many Calories (Cal) are in the hamburger described above?
How many Calories (Cal) are in the hamburger described above?
2.65E1
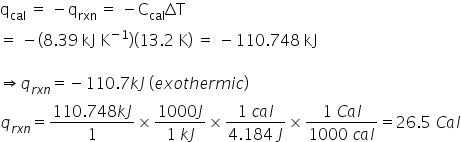
95
New cards
If the same hamburger was placed in a different bomb calorimeter whose is 7.66 kJ K, then what would be the change in temperature of the water bath?
1.45E1
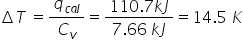
96
New cards
is this statement true(A material with a greater specific heat capacity will experience a greater temperature change than a material with a smaller specific heat capacity per unit of heat)?
false
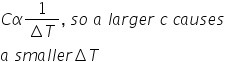
97
New cards
How much heat (in kJ) is needed to heat a 200.0 mL sample of water initially at 25.0 °C to a boiling temperature of 100.0 °C?
6.28E1
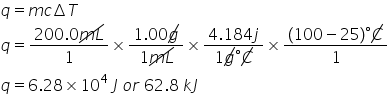
98
New cards
A 6.88 g piece of copper is heated from 20.0 C to 125 C and then submerged in 45.0 mL of water initially at 18.3 C. What is the final temperature of the water?
1.98E1
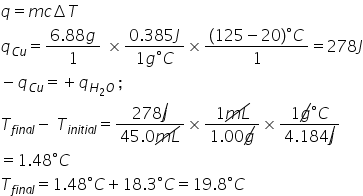
99
New cards
Isobaric calorimetry
is also known as coffee-cup calorimetry.
100
New cards
Isochoric calorimetry
is also known as bomb calorimetry.