Immunology Exam 3
1/99
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
100 Terms
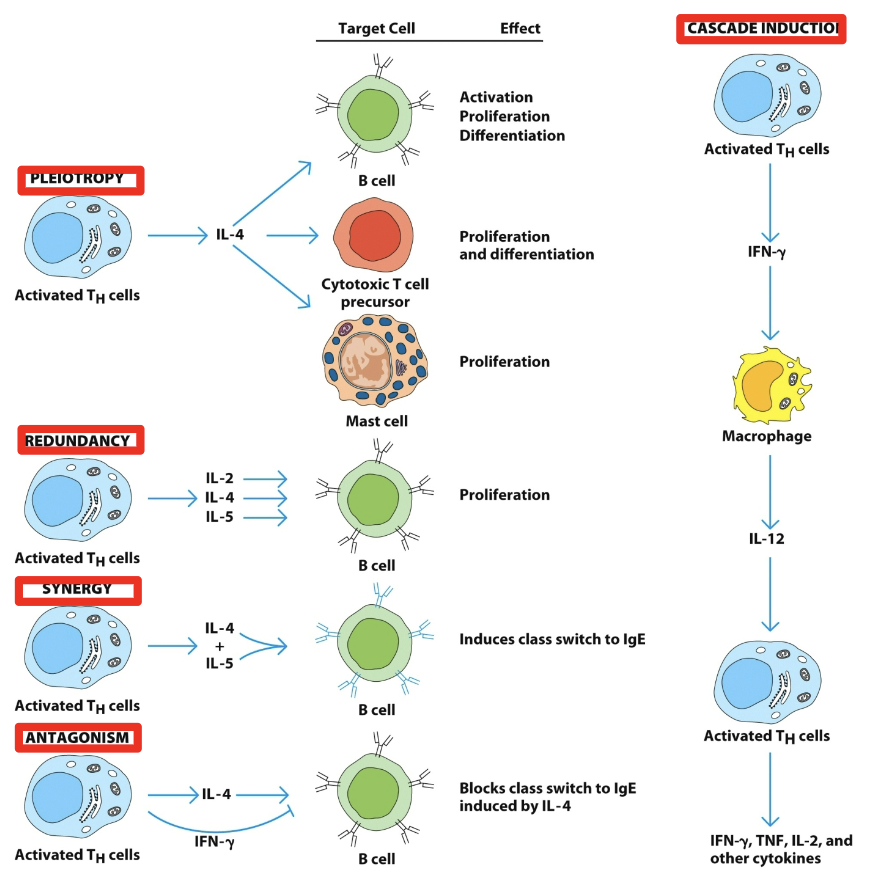
synergy
sum greater than parts
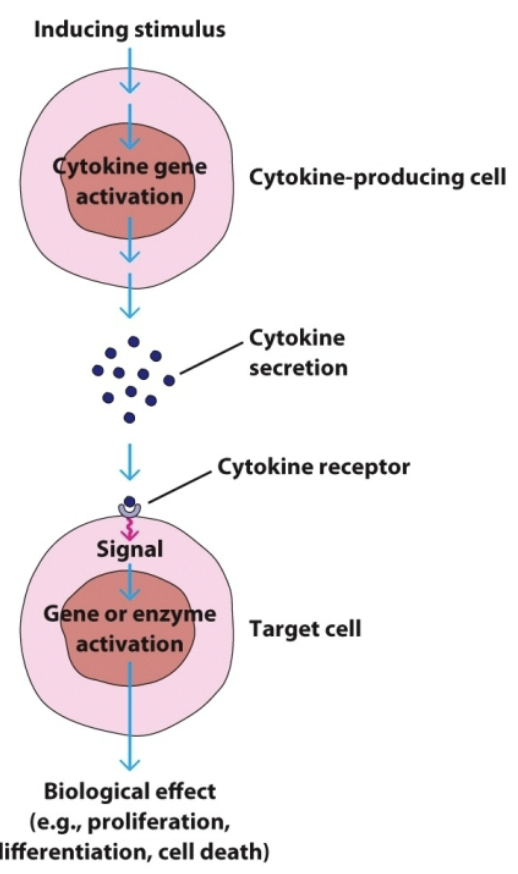
Cytokines
_:
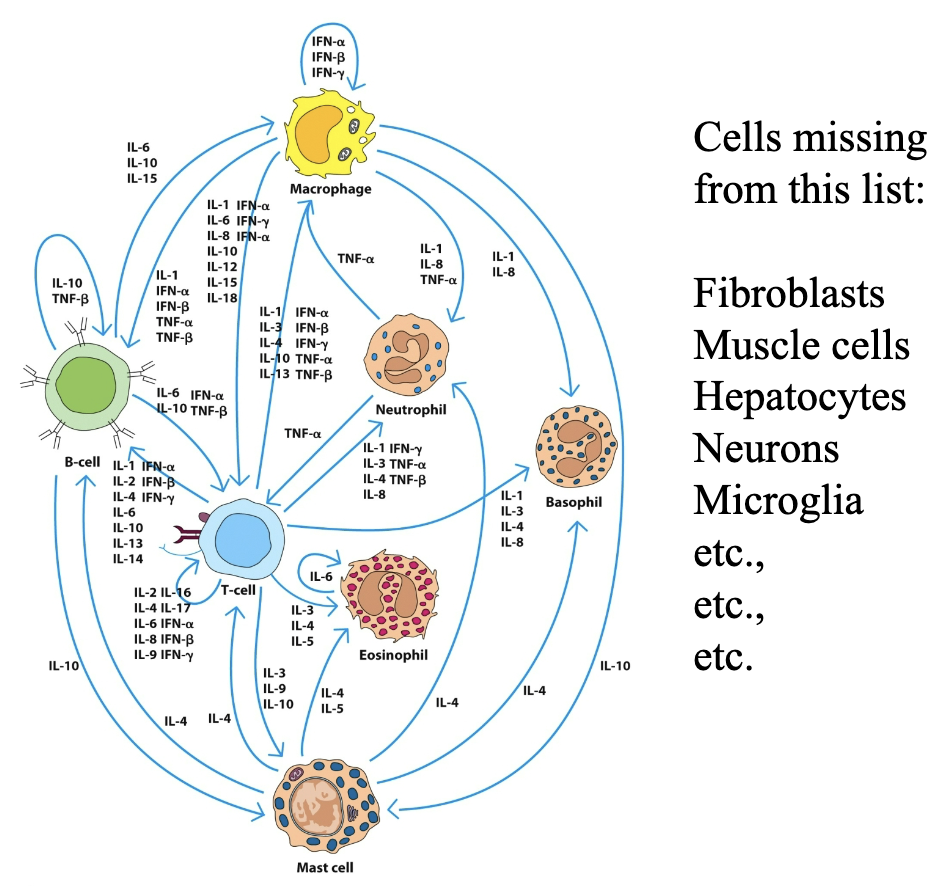
Small signaling proteins secreted by immune and non-immune cells that act as essential chemical messengers to regulate inflammation, immune responses, and cell communication.
Major classes include interleukins (ILs), interferons (IFNs), tumor necrosis factors (TNFs), chemokines, and colony-stimulating factors (CSFs).
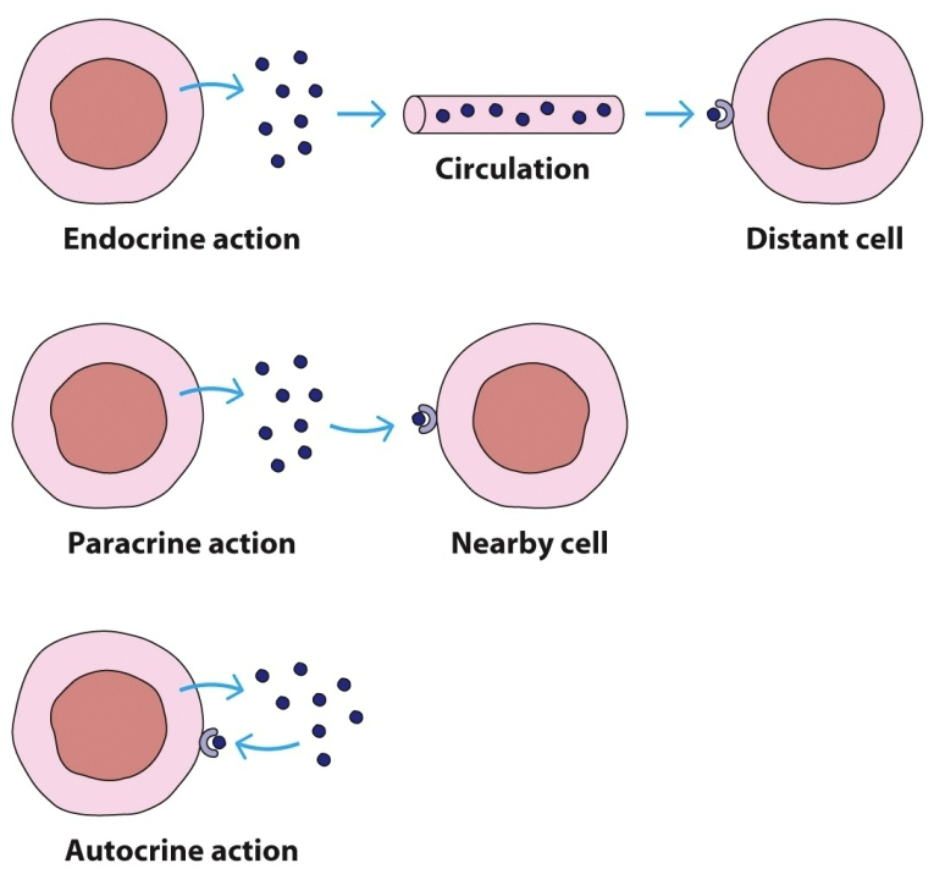
paracrine
Most cytokines act via _ action
multiple
cytokines can have _ biological affects
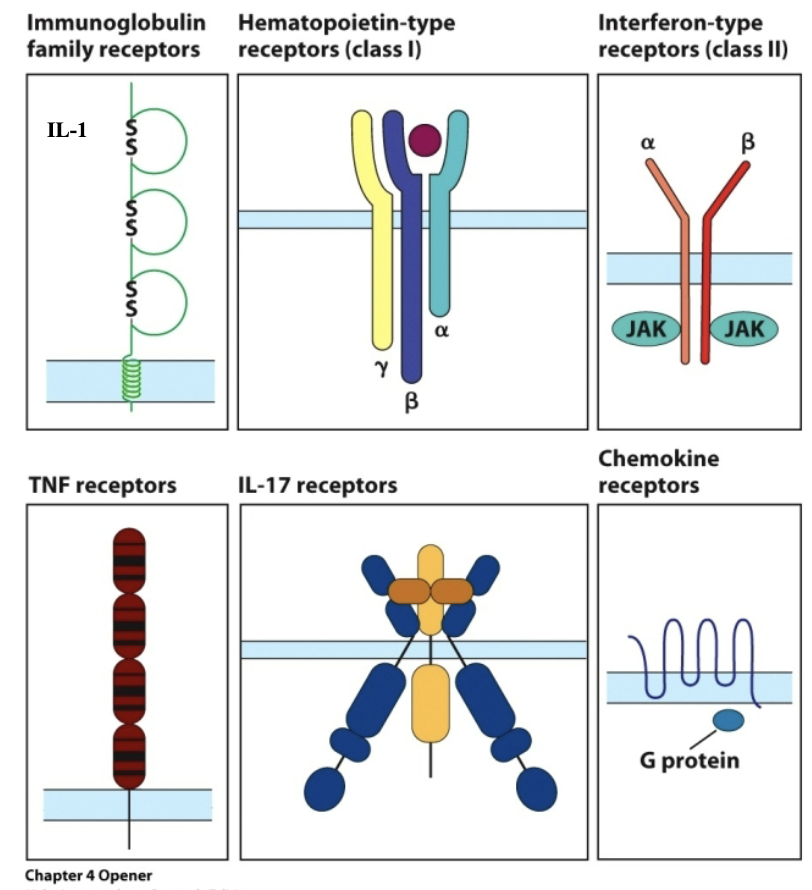
receptors
_
bottom
top
Which row is public chain?
Which row is private chain?
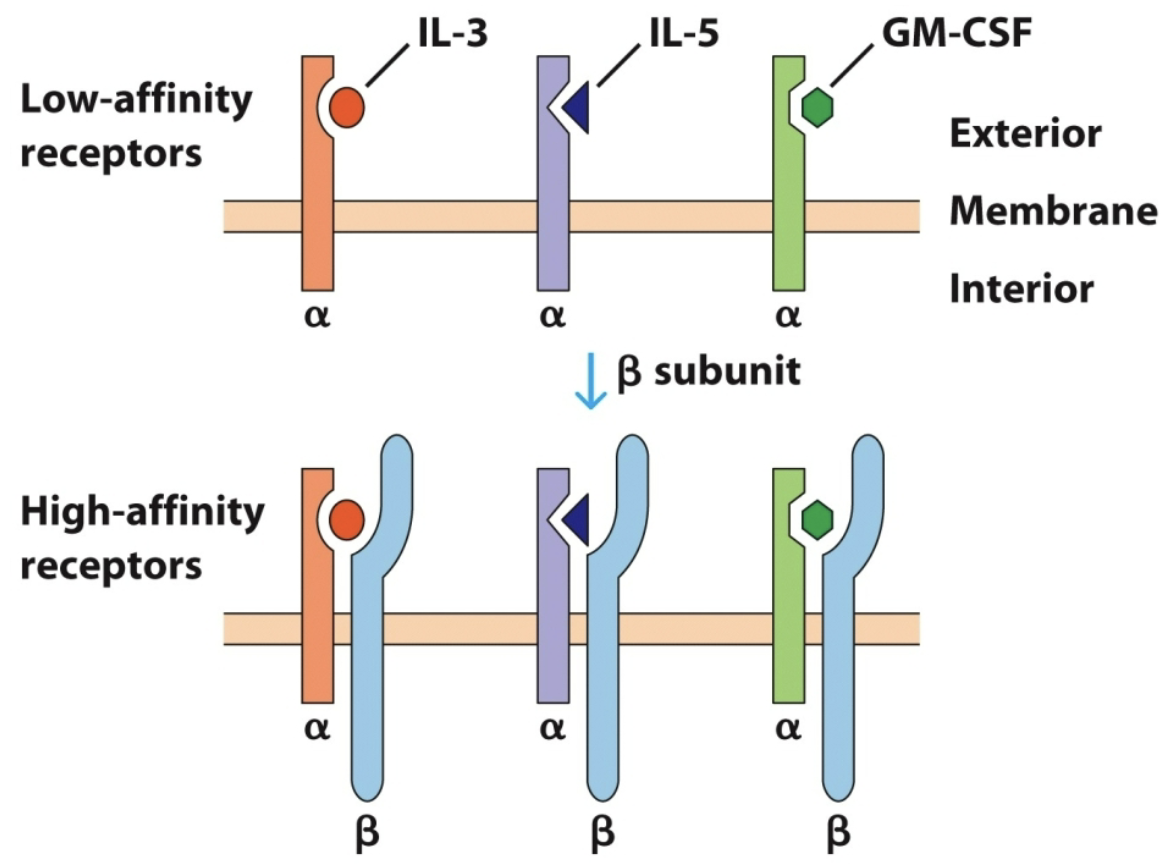
private
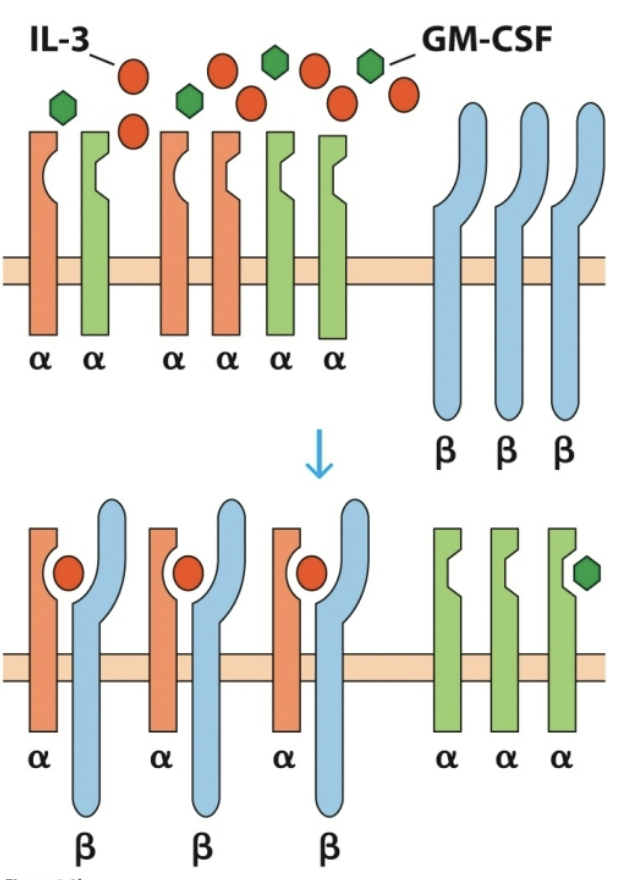
The receptor subunits specific for each cytokine are called _ chains because they define which specific cytokine binds, despite activating the same downstream machinery.
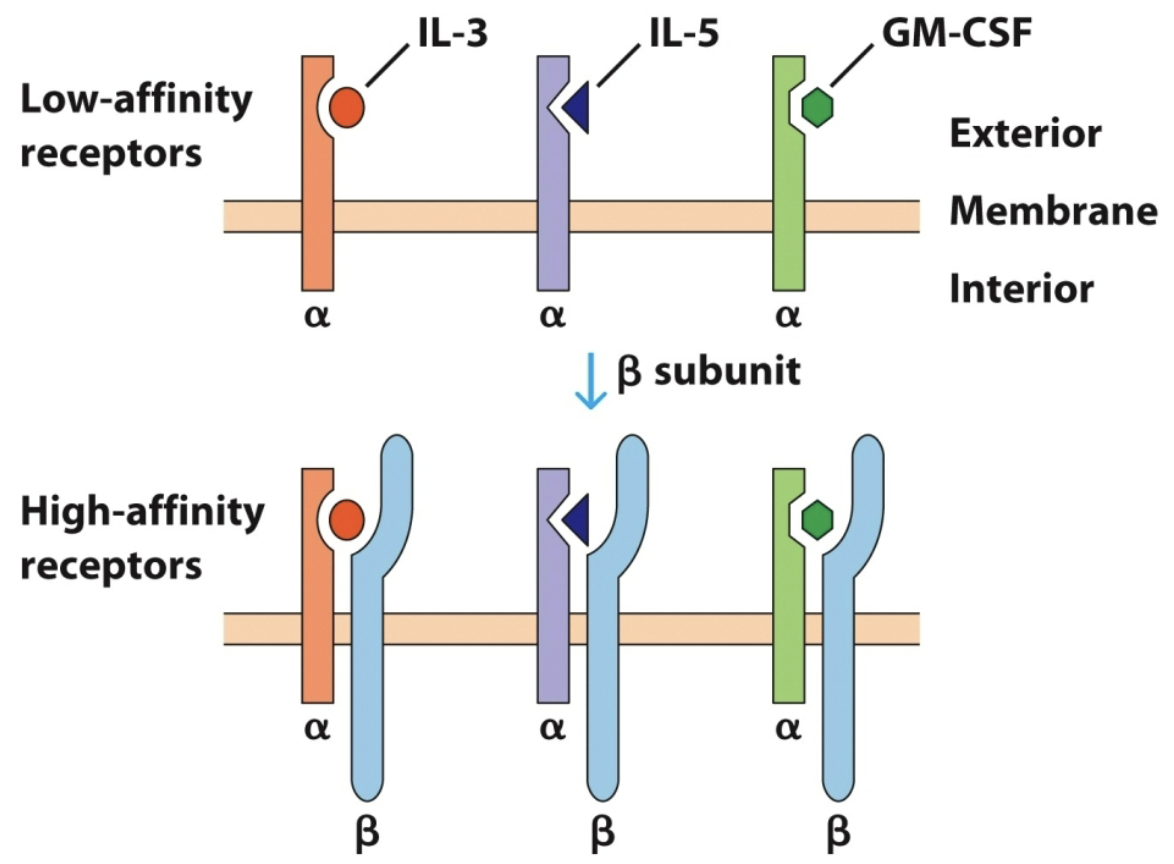
Limited public chain and lots of certain type of cytokine ligand can lead to selective representation of that receptor dimer; competitive inhibition
Purpose of private vs public chain?
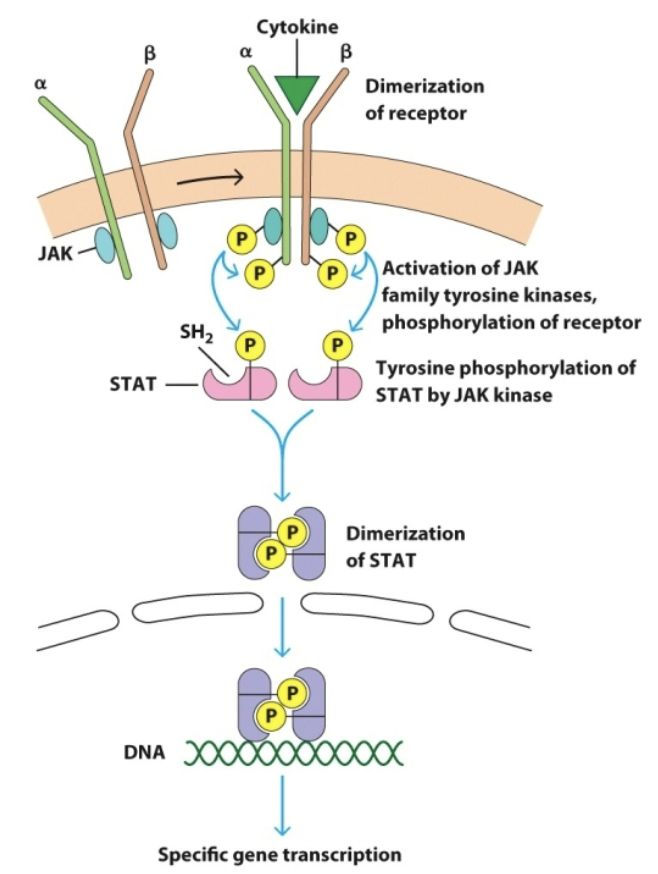
phosphorylated tyrosines
SH2 binds to _W
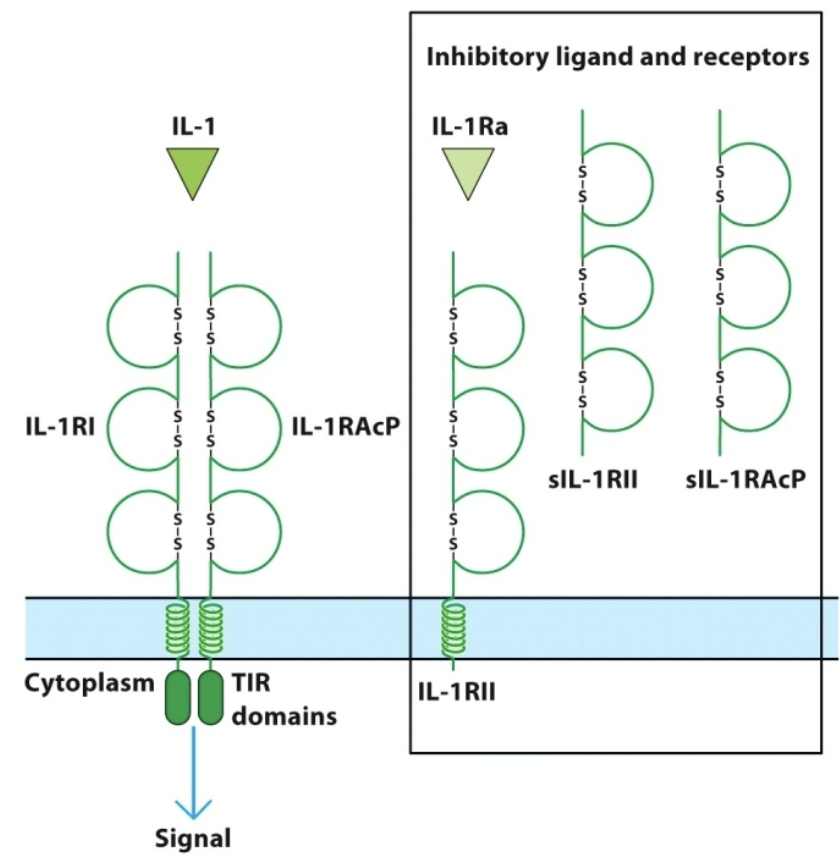
No more binding domain
Acts as a soluble inhibitor to other receptors (by binding to them)
Decoy receptor (ex. sIL-1RII decoy can bind IL-I but iC domain can’t signal)
Why chop off extracellular (EC) part (3 reasons)?
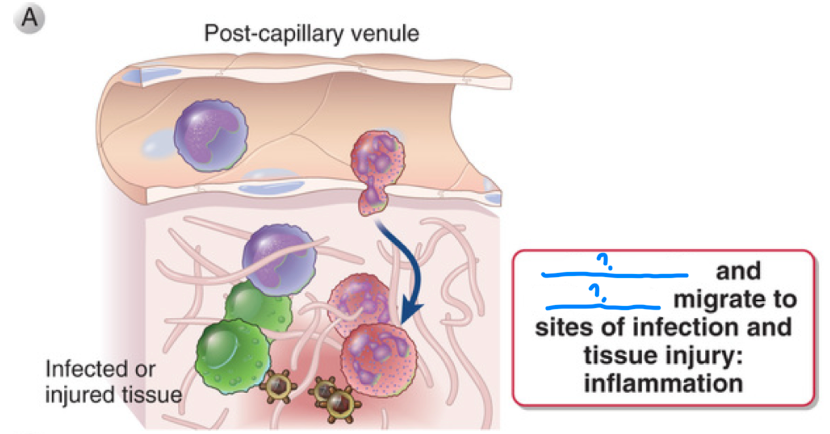
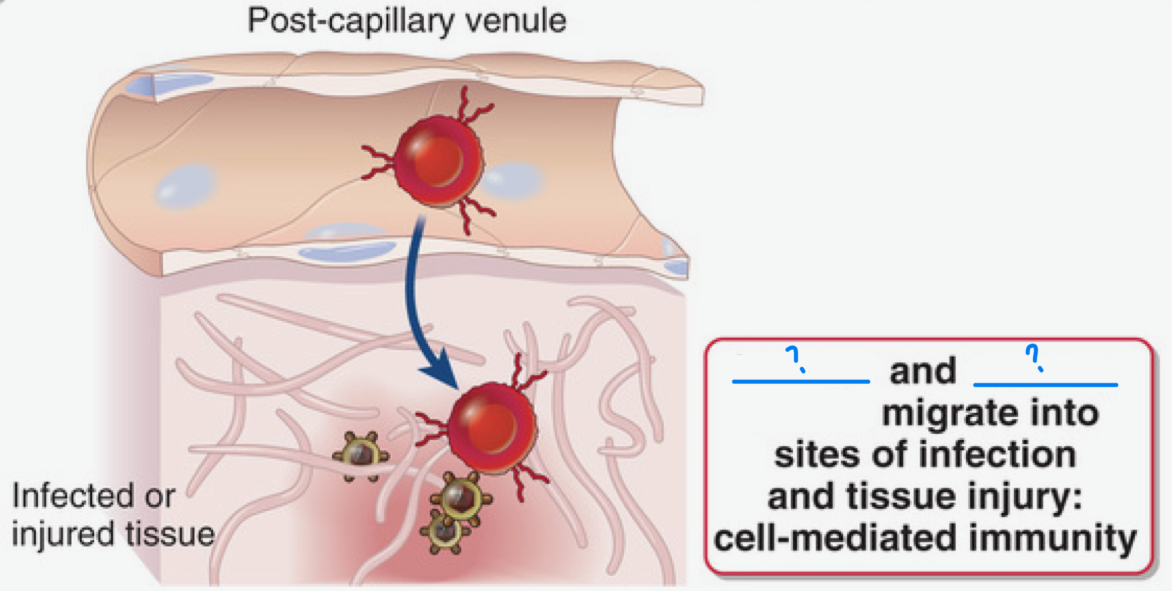
neutrophils ; monocytes
Inflammation is when _ and _ migrate to sites of infection and tissue injury
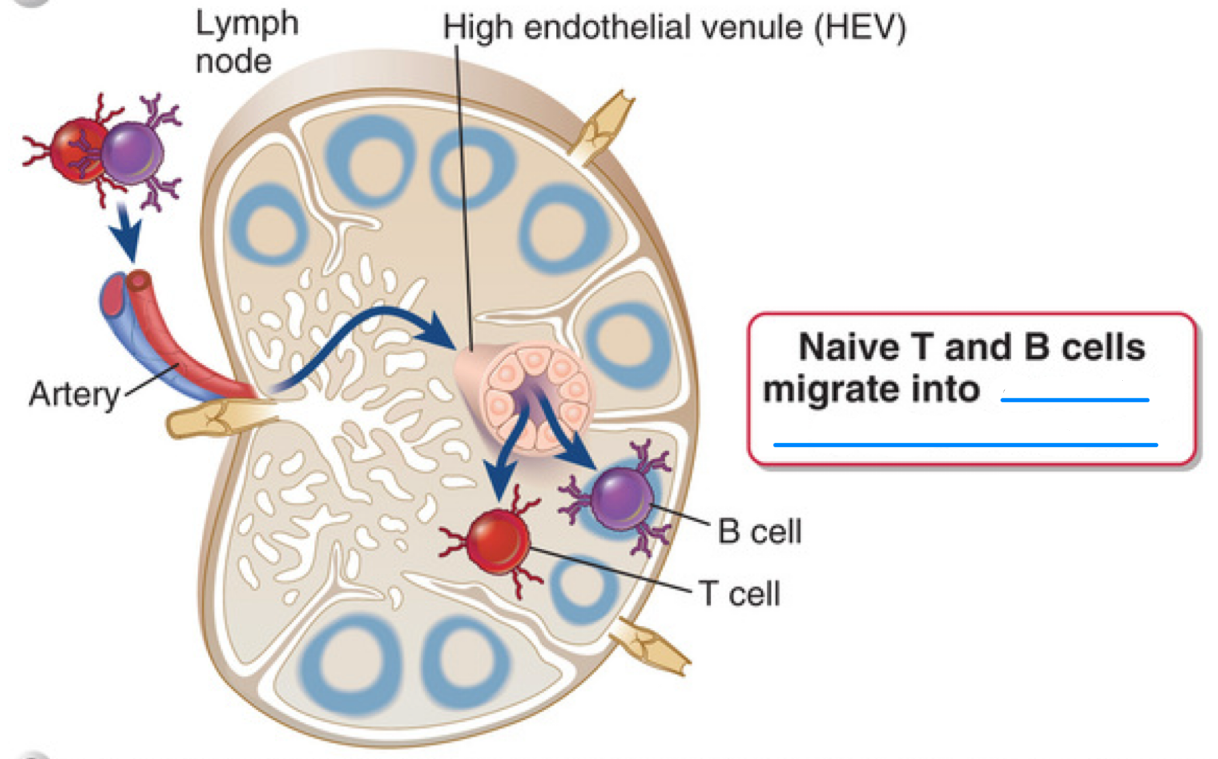
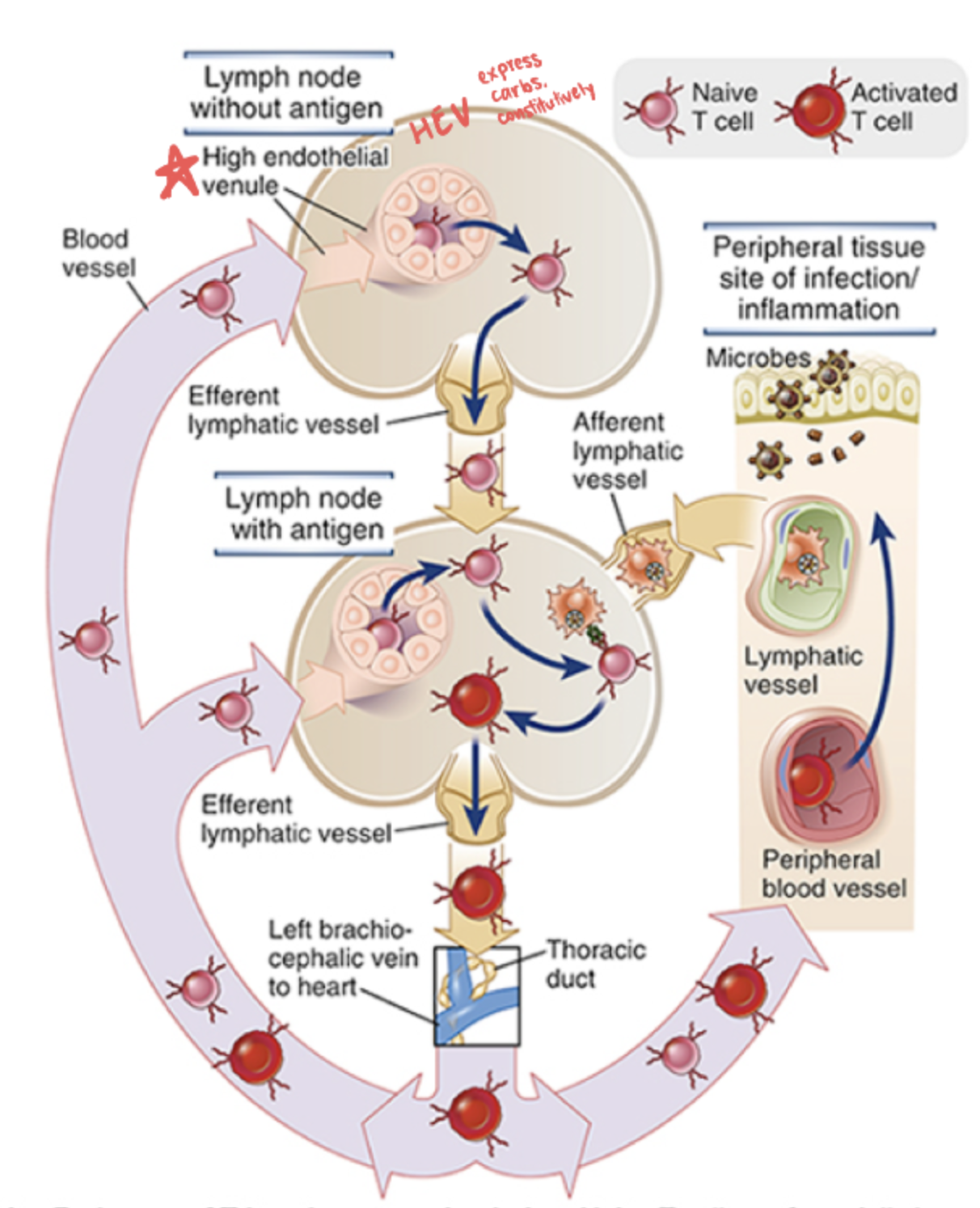
secondary lymphoid organs
Naive T and B cells migrate into ___
Effector ; Memory T Cells
Cell-mediated immunity is when _ and _ migrate into sites of infection and tissue injury
Pain (dolor)
Heat (calor)
Redness (rubor)
Swelling (tumor)
Loss of function (functio laesa)
5 Hallmarks of Inflammation:
WBCs are metabolically active, releasing energy
Increased blood flow to the injured or infected area
Why does heat happen during inflammation?
Fluids leak out due to increased vascular permeability
Why does swelling happen during inflammation?
Increased blood flow to infection site —> blood = red
Why does redness happen during inflammation?
dead cells
neighboring cells
IL-1 alpha
Injury in big toe:
Who FIRST cries for help?
_
Second?
_
_ is never secreted in healthy cells; Dead cells secrete it.
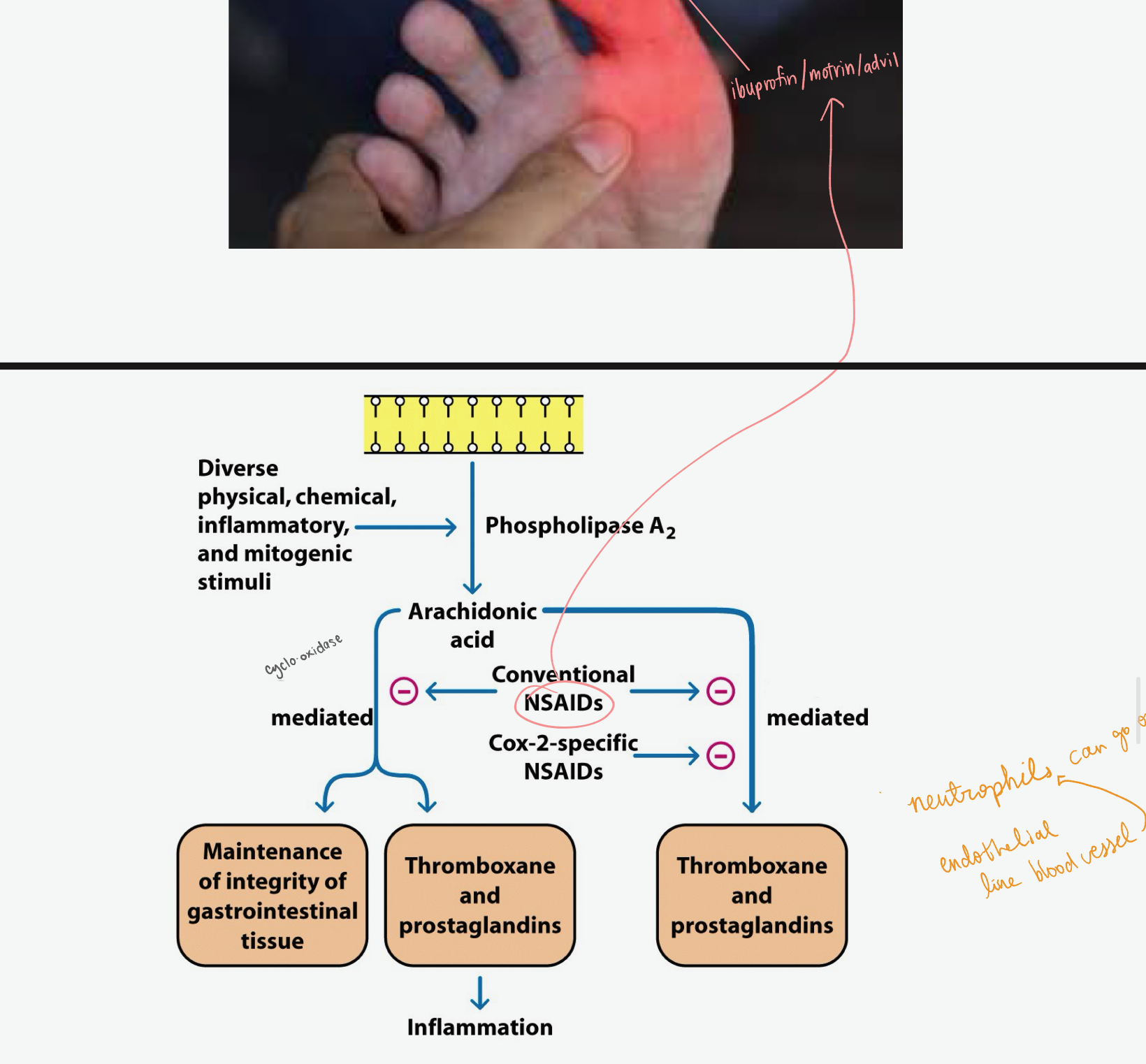
NSAIDS
_, like ibuprofen/motrin/advil block Cox-1/2 pathways leading to inflammation
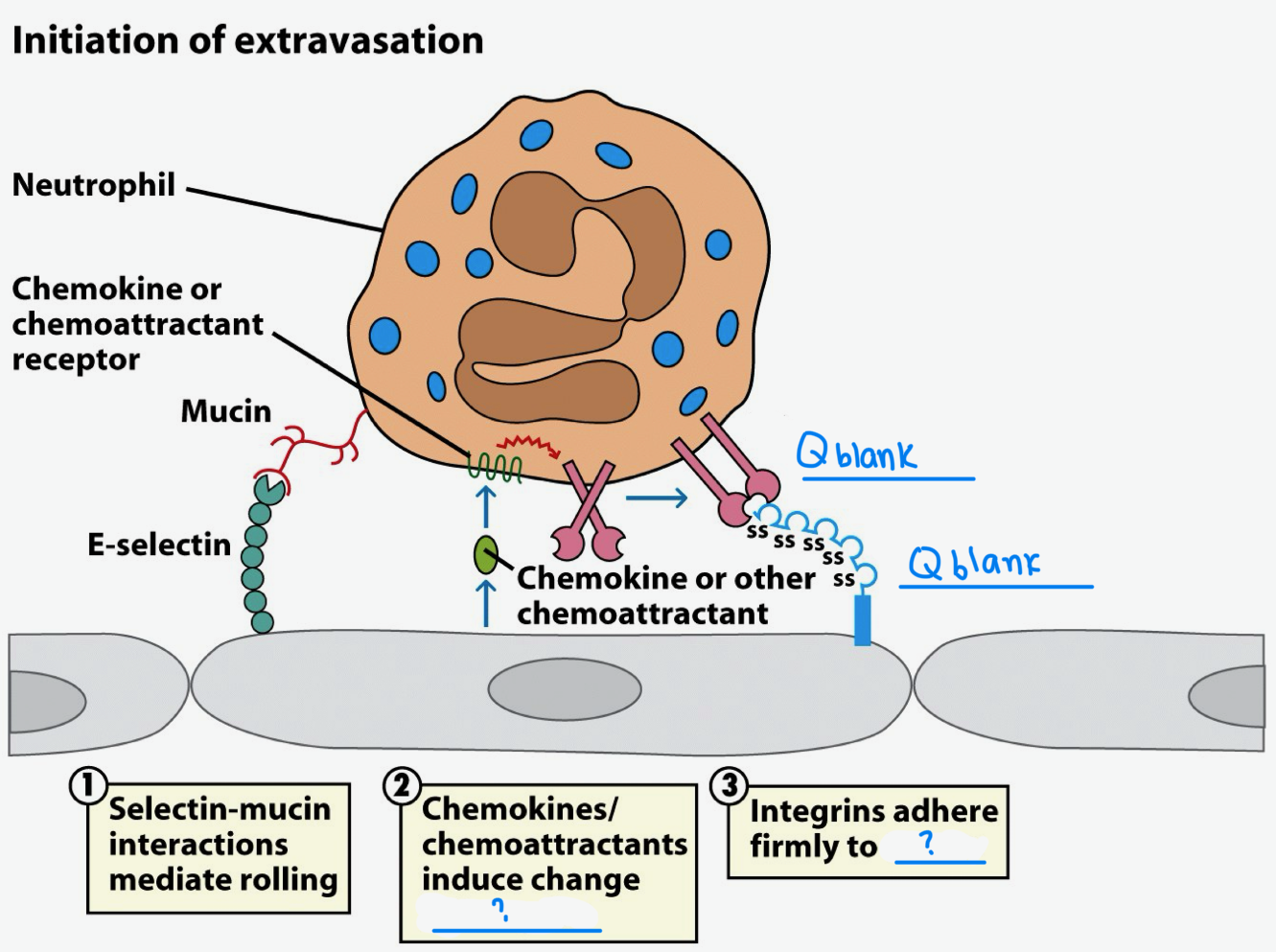
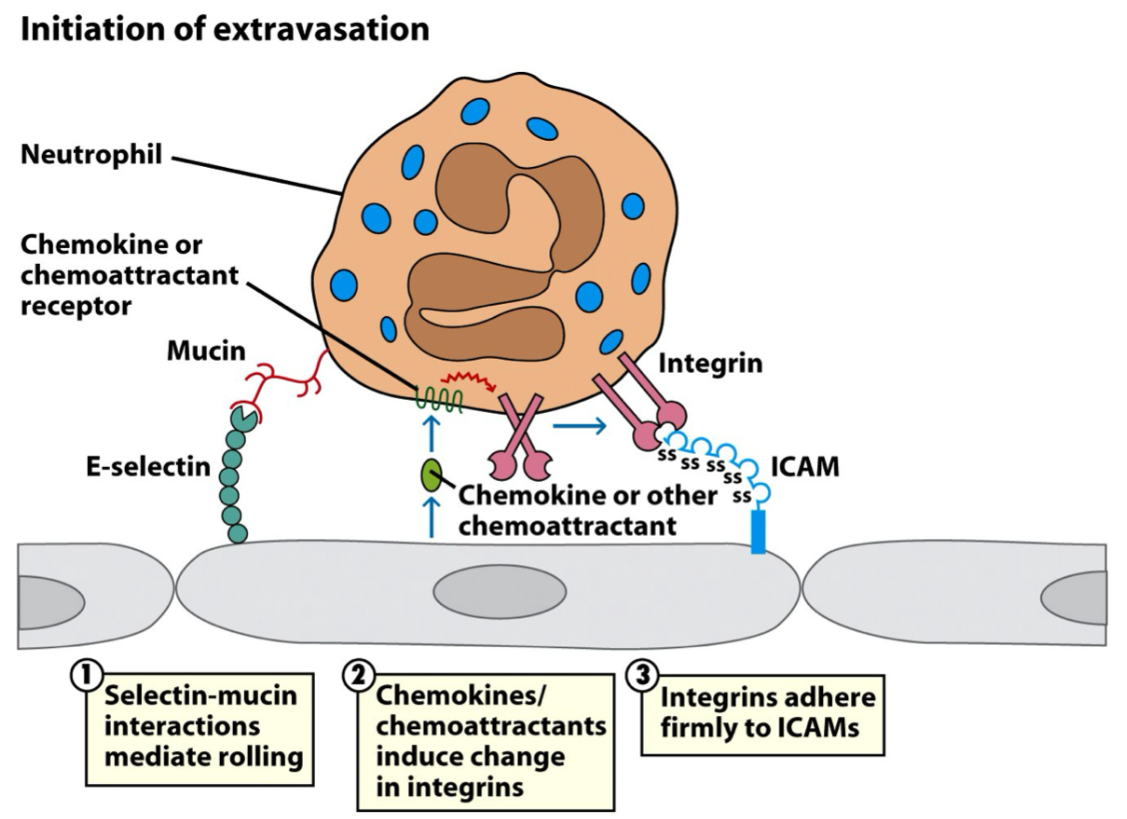
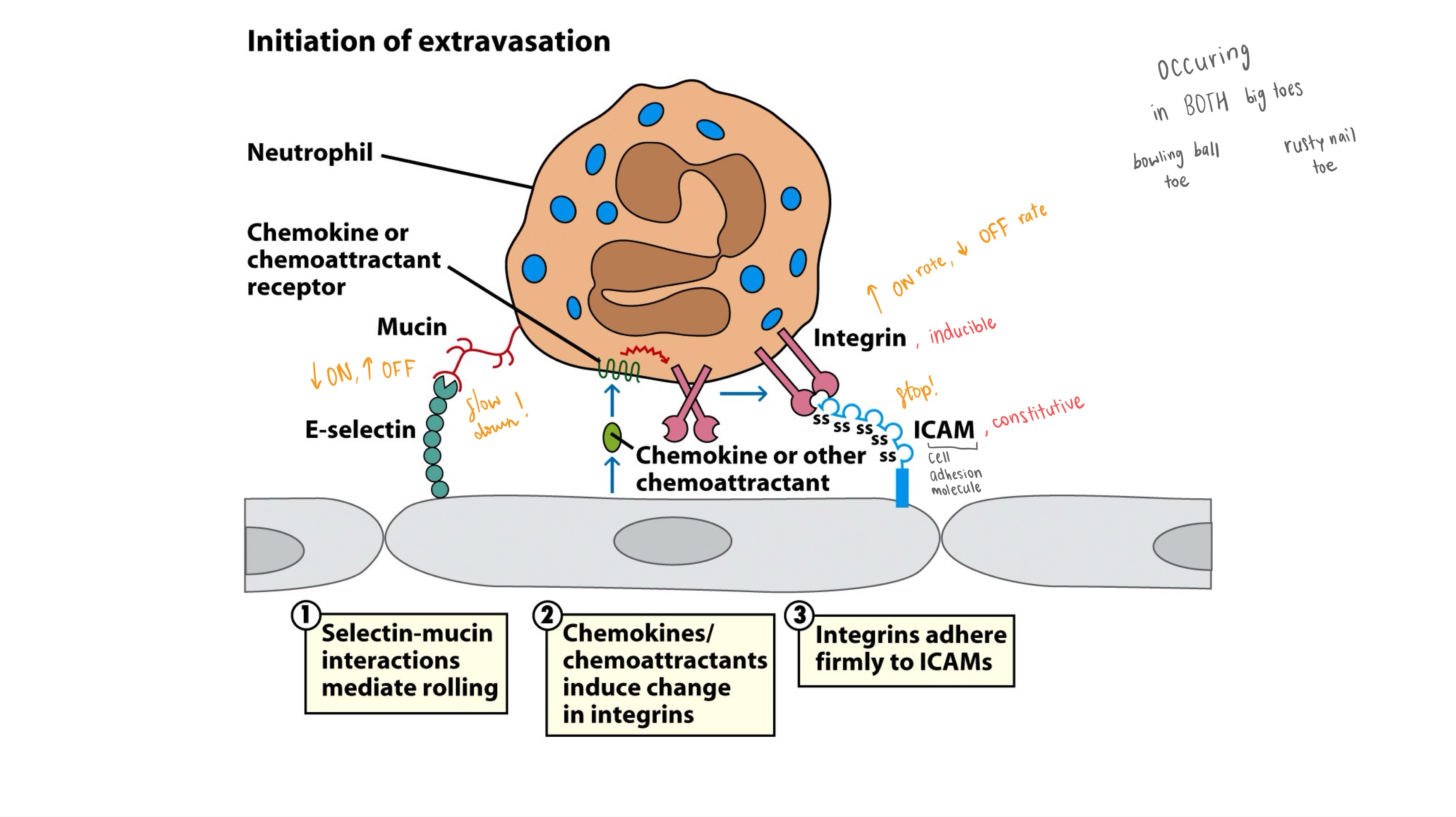
Selectin
_ are critical adhesion molecules that initiate the immune response by mediating the initial "hopping" of leukocytes along the vascular endothelium.
(image fill in blank)
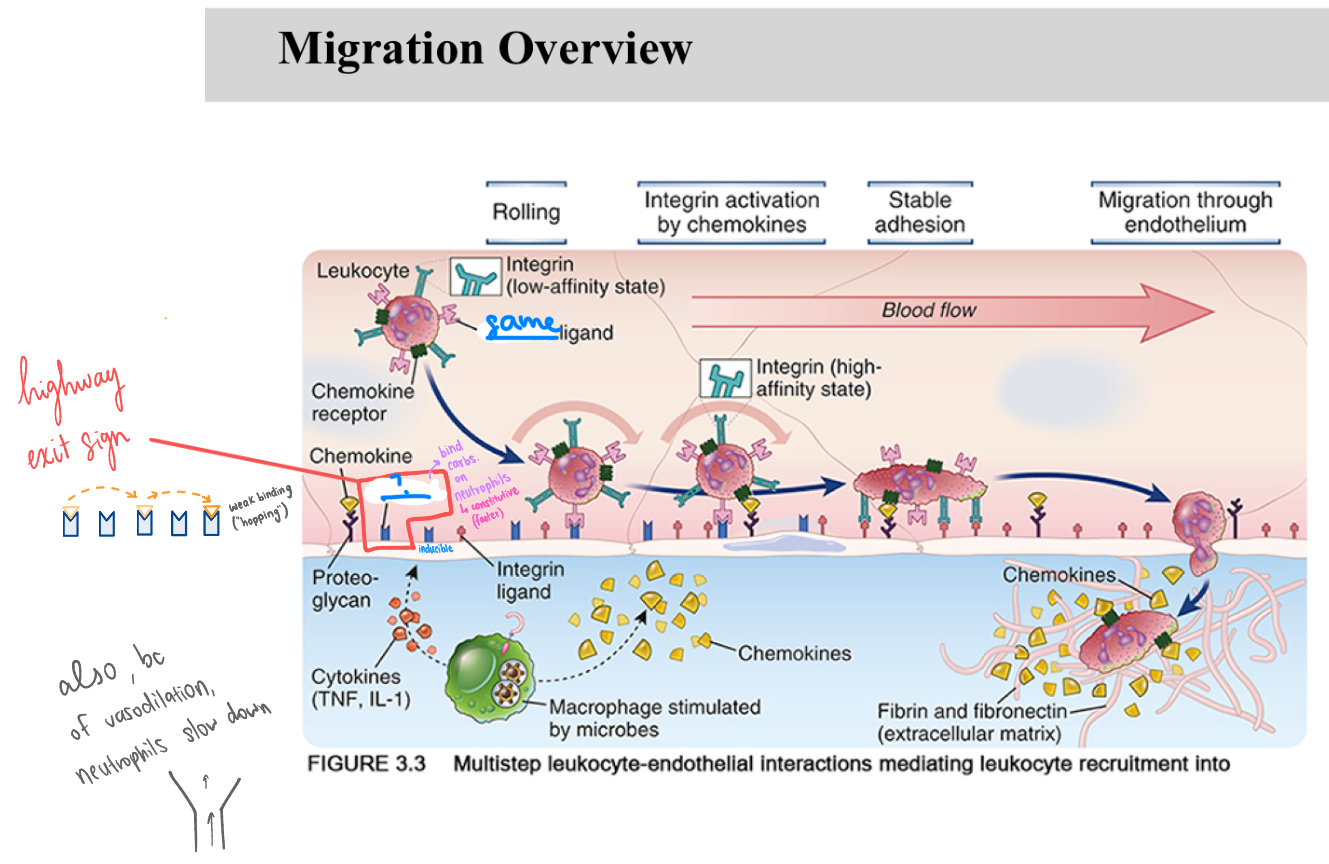
inducible
constitutive
Selectins are inducible on endothelial cells to prevent indiscriminate, potentially harmful neutrophil adhesion in healthy blood vessels.
Carbohydrates (selectin ligands) on neutrophils are constitutive to allow them to respond instantly to inflammatory signals.
Selectins are _
Carbs on Neutrophils are _
WHY?
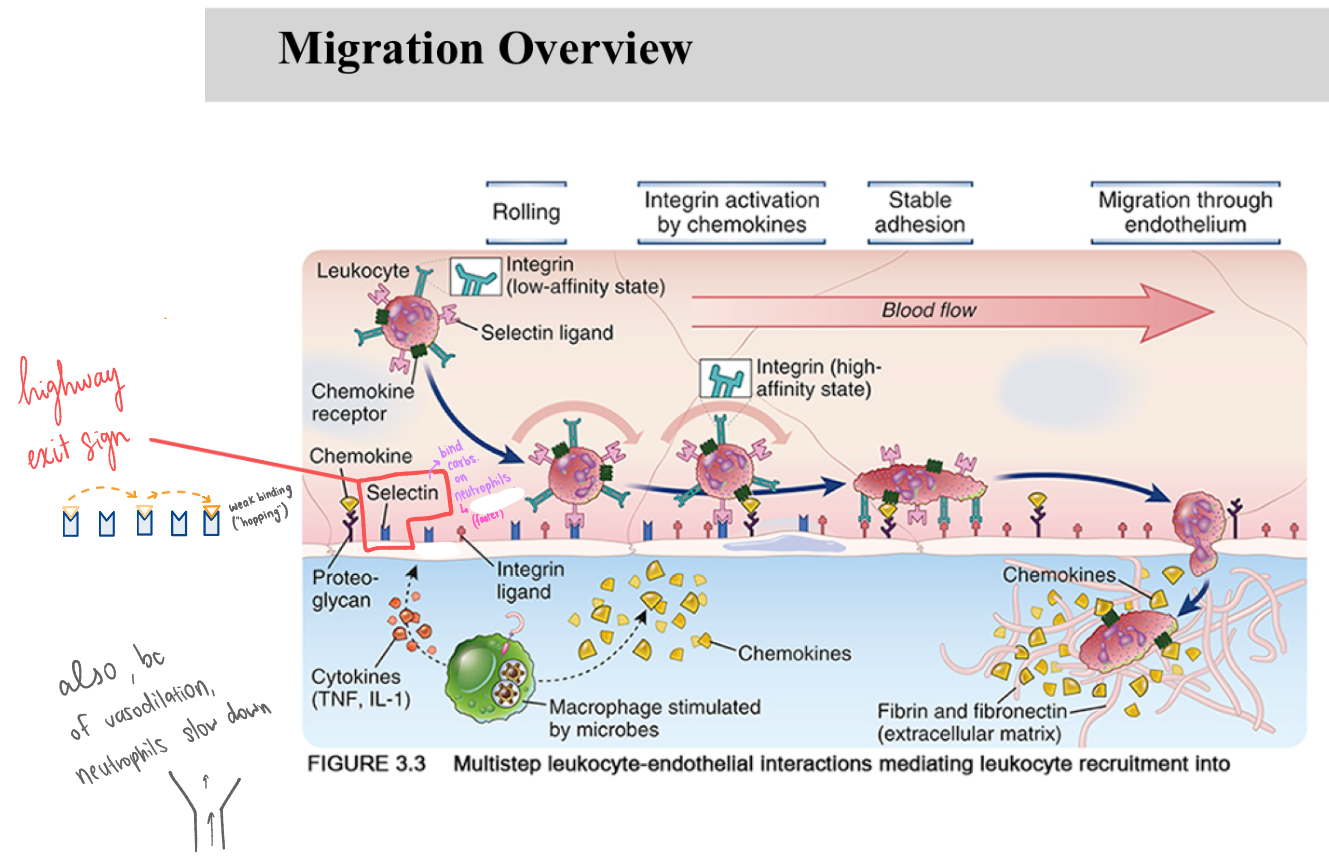
vasodilation
Neutrophils can slow down (for migration into the tissue) bc of selectin hopping and ALSO because of _
(image fill in blank)
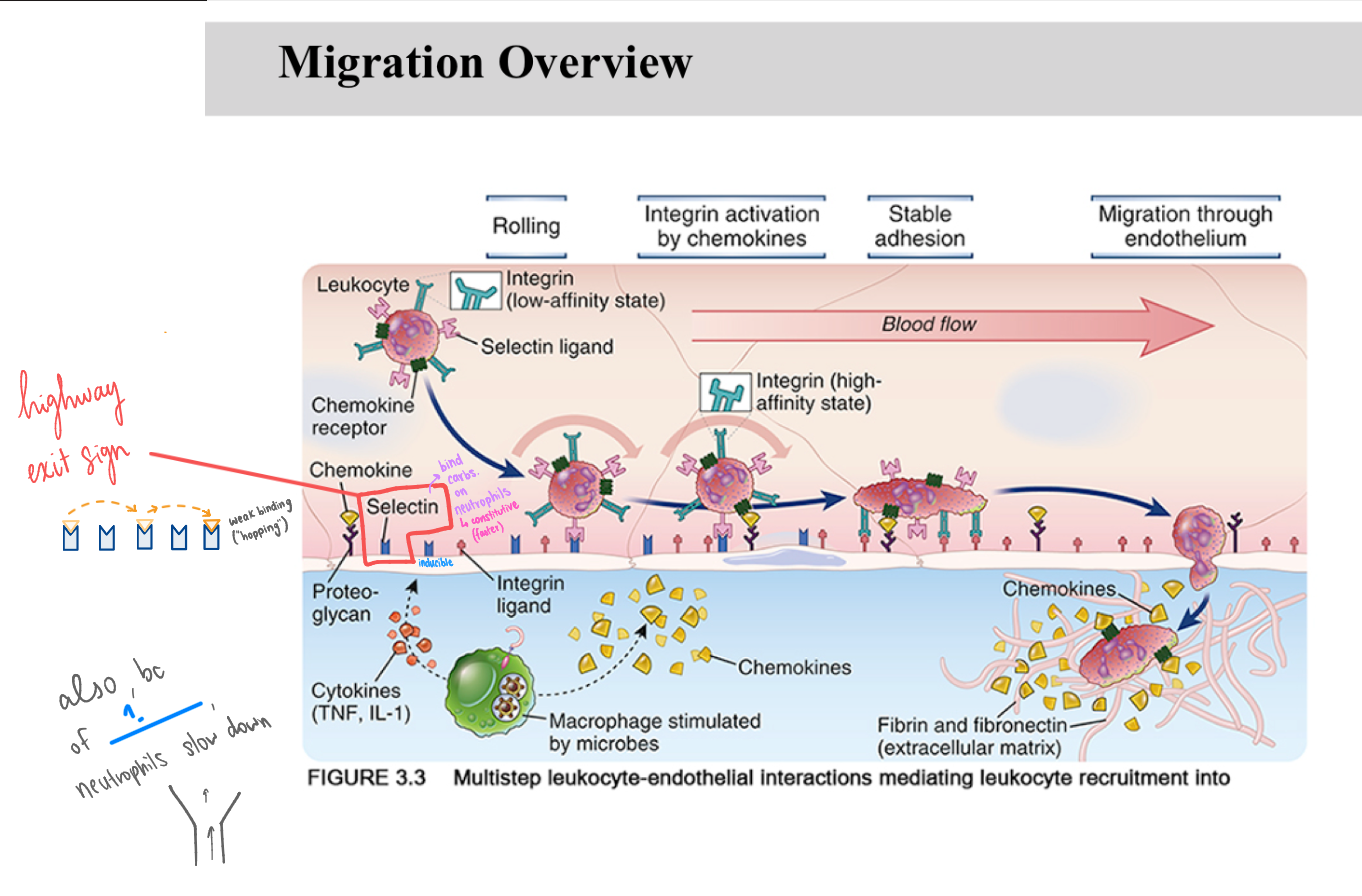
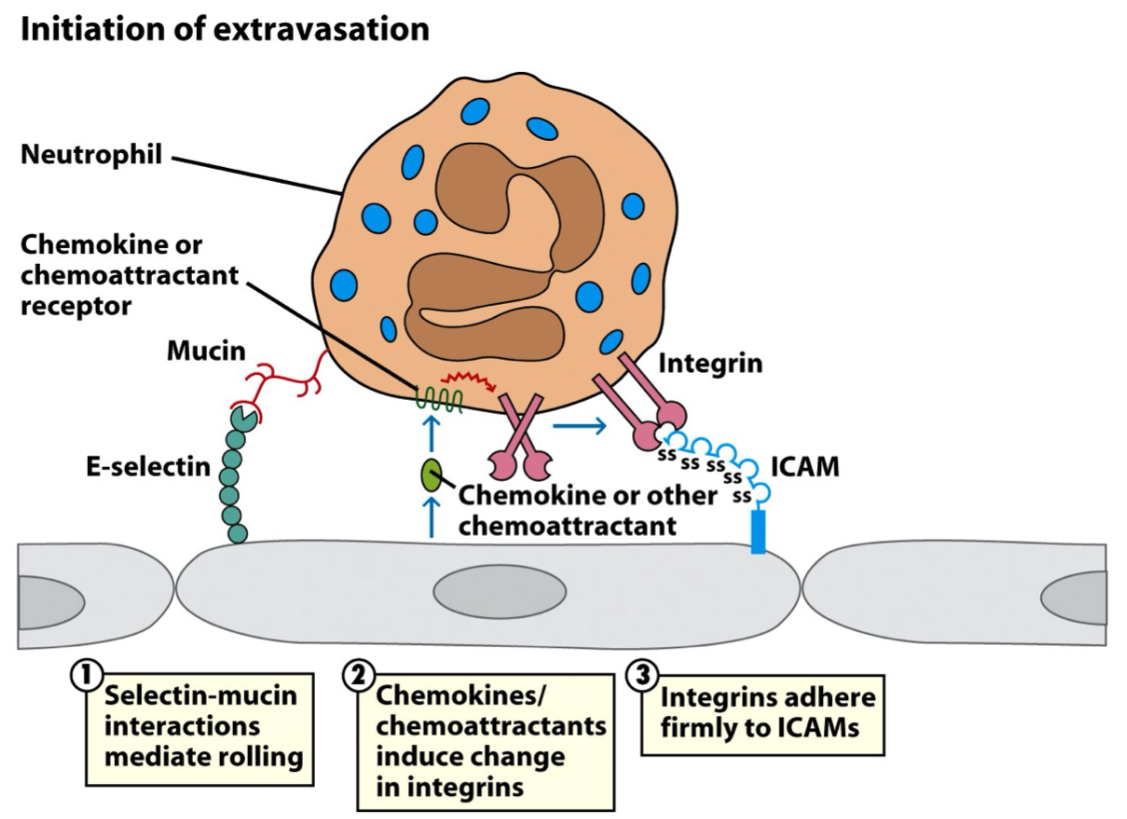
integrins ; ICAM
in integrins ; ICAM
After selectin binds carb on neutrophil, _ on neutrophil binds to _ on endothelial cell.
Now fill in image _’s
OFF ; ON
ON ; OFF
Carb / selectin has: High _ rate; Low _ rate
Integrin / ICAM has: High _ rate; Low _ rate
inducible
constitutive
Integrin is _
ICAM is _
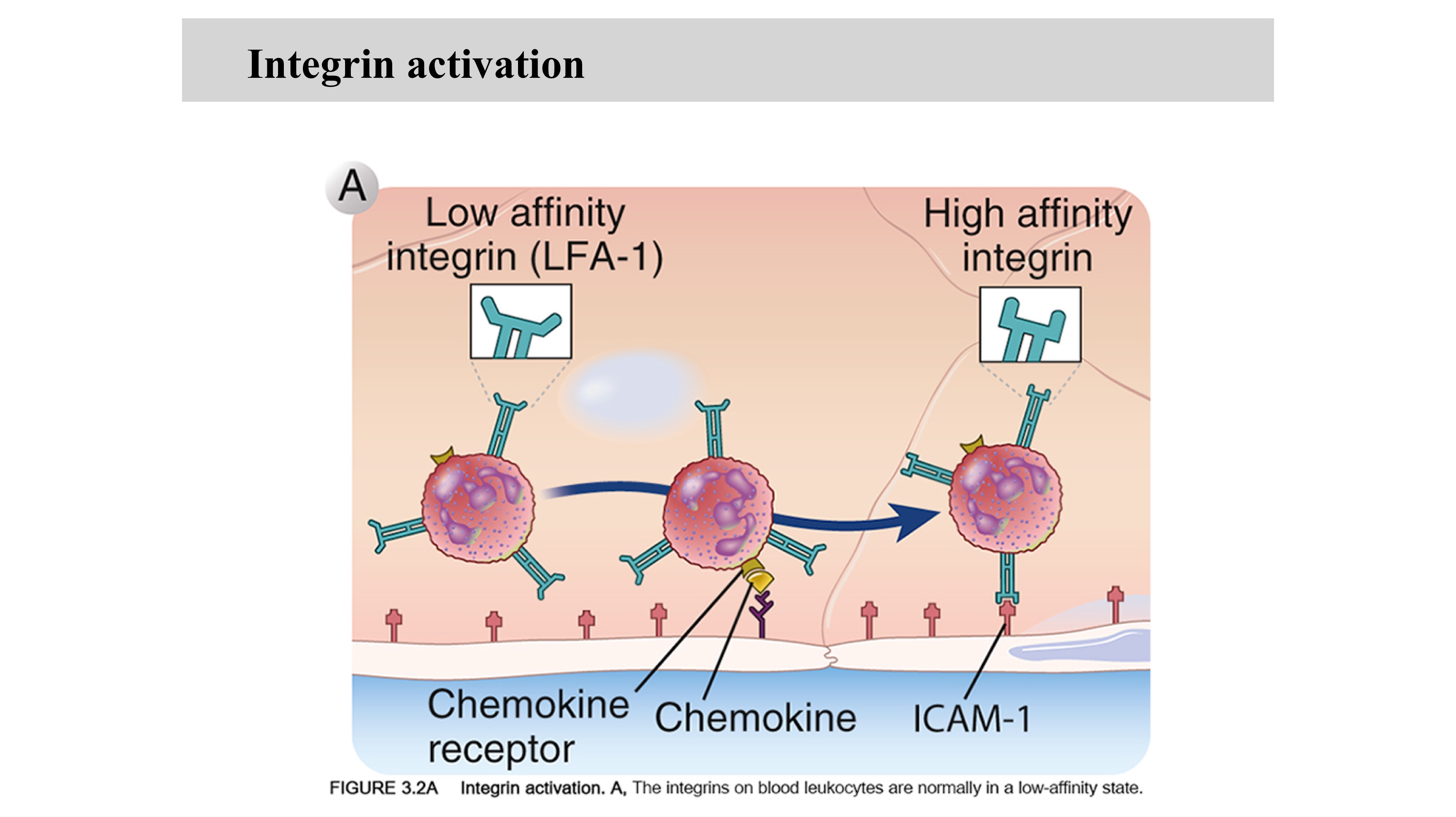
constitutive
In lymph nodes, everything is _
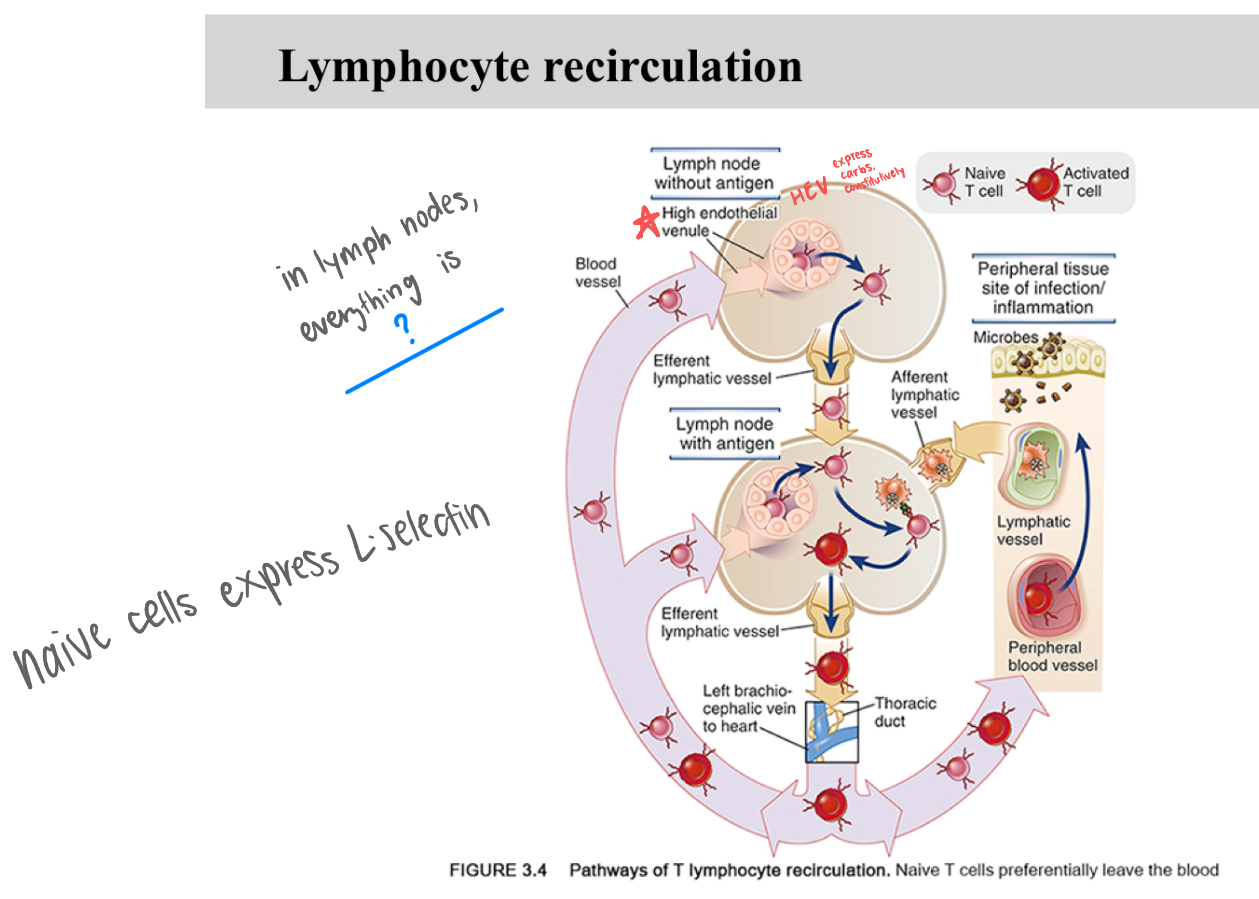
HEV
_ are the endothelial cells in blood vessels and lymph nodes that express a carb all the time that binds L-selectin
T cell progenitors
B cell progenitors
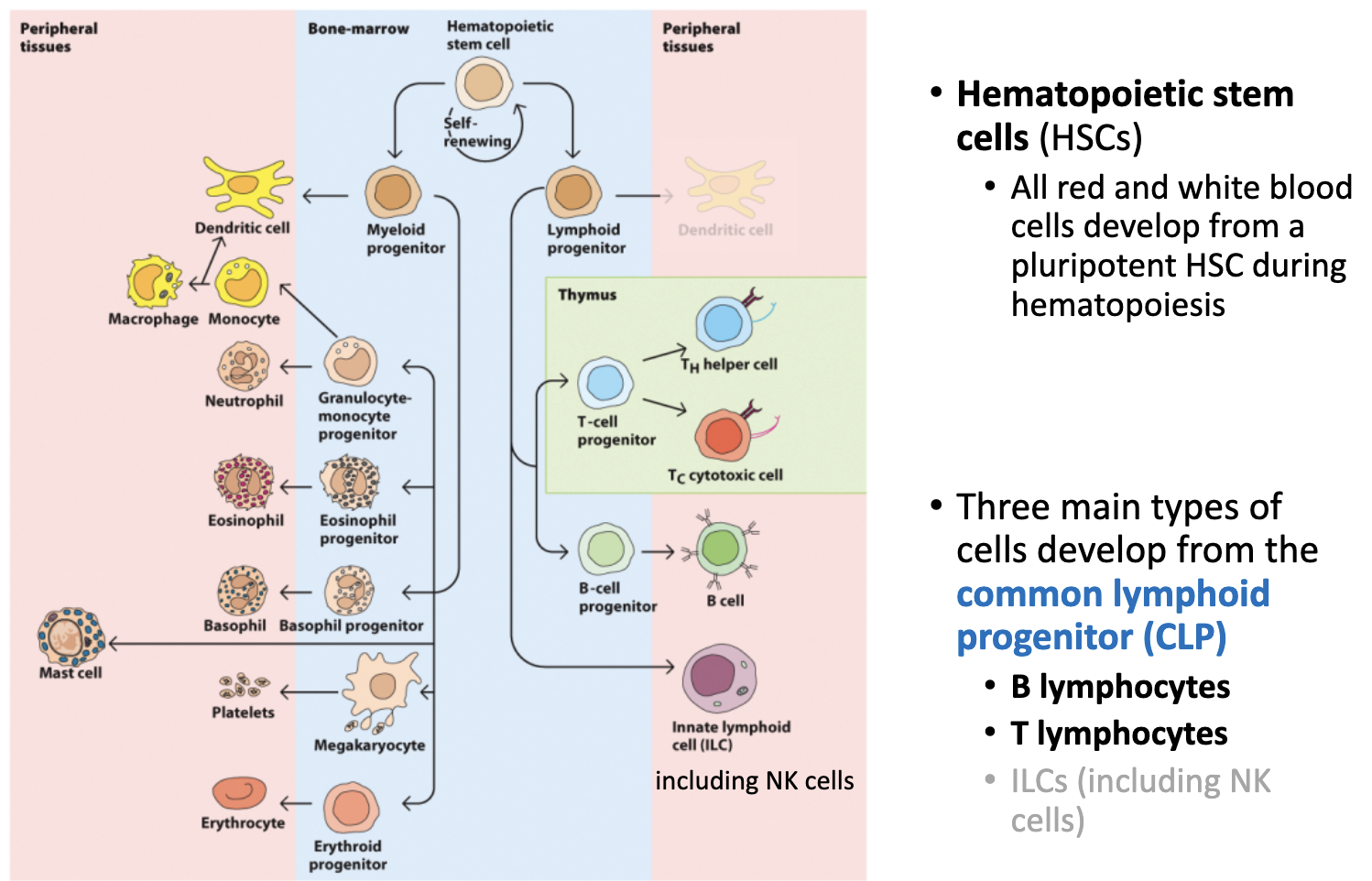
In bone marrow, lymphoid progenitors into..
_
_
IgM
Naïve
self-reactive
LN to LN
Mature B cell in periphery
Expresses surface _
_ (no effector function)
Ideally, no _ cells
Circulate from _, “looking” for cognate antigen
HC and LC
Ig
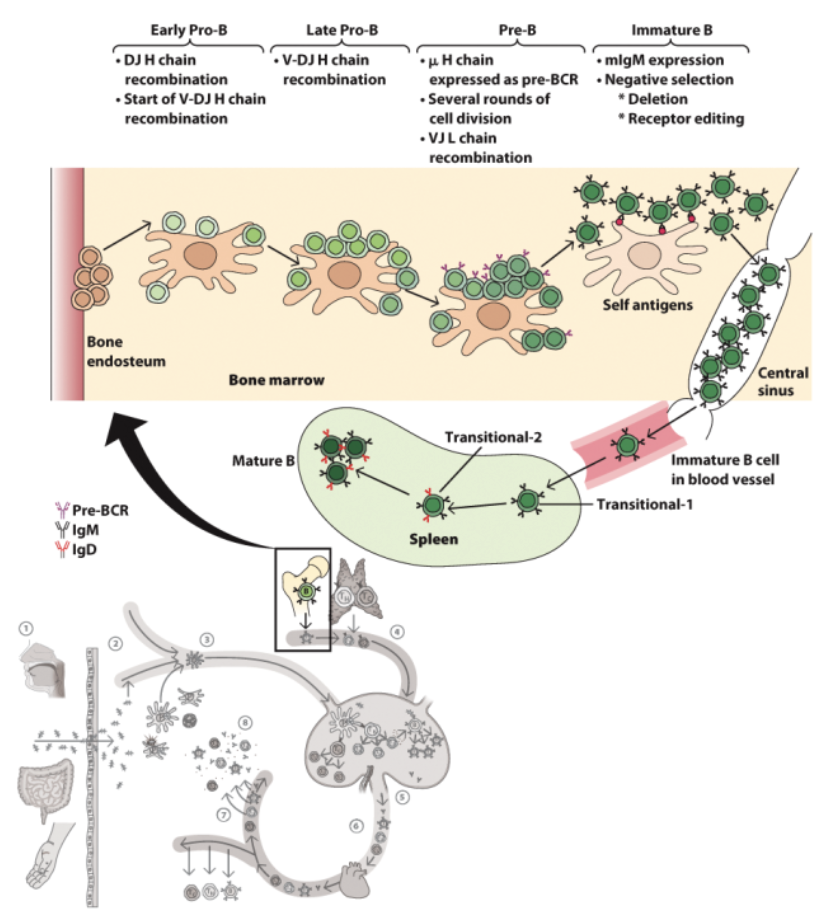
What needs to happen during B cell development ….
Express proteins needed for gene rearrangement, B cell signaling, and B cell function.
Rearrange _ and _ genes
Test rearranged _
Eliminate cells with non-productive RAR
Tolerance – eliminate cells with Ig that recognizes self
spleen
B-cell development begins in the bone marrow and is completed in the _.
Commit to B cell lineage
RAR (rearrangement) HC
Test HC
RAR LC
Test HC/LC
Negative selection
More negative selection
Bone marrow
Stem cells
Stromal cells
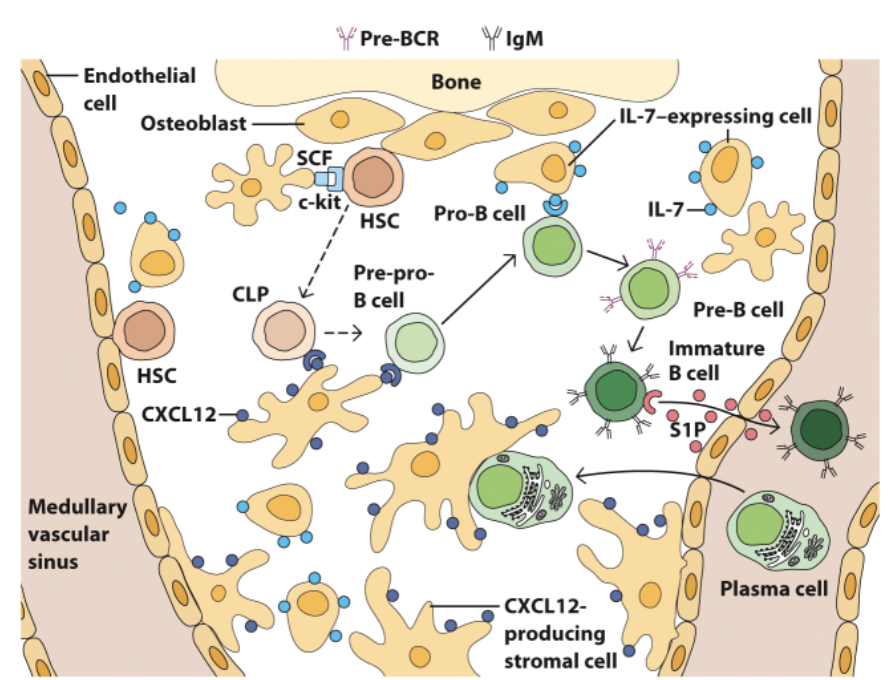
B-cell development in the bone marrow
_ structure is dynamic & complex.
_ differentiate into many cell types
_ provide support & growth factors
Soluble mediators
Cell surface molecules
Examples for EARLY B cell development
SCF (stem cell factor) on stromal cells binds to c-kit on HSCs.
CXCL12 on stromal cells -> supports differentiation to pro-B cell stage.
IL-7 – growth factor for lymphoid cells -> supports differentiation to pre-B cell stage.
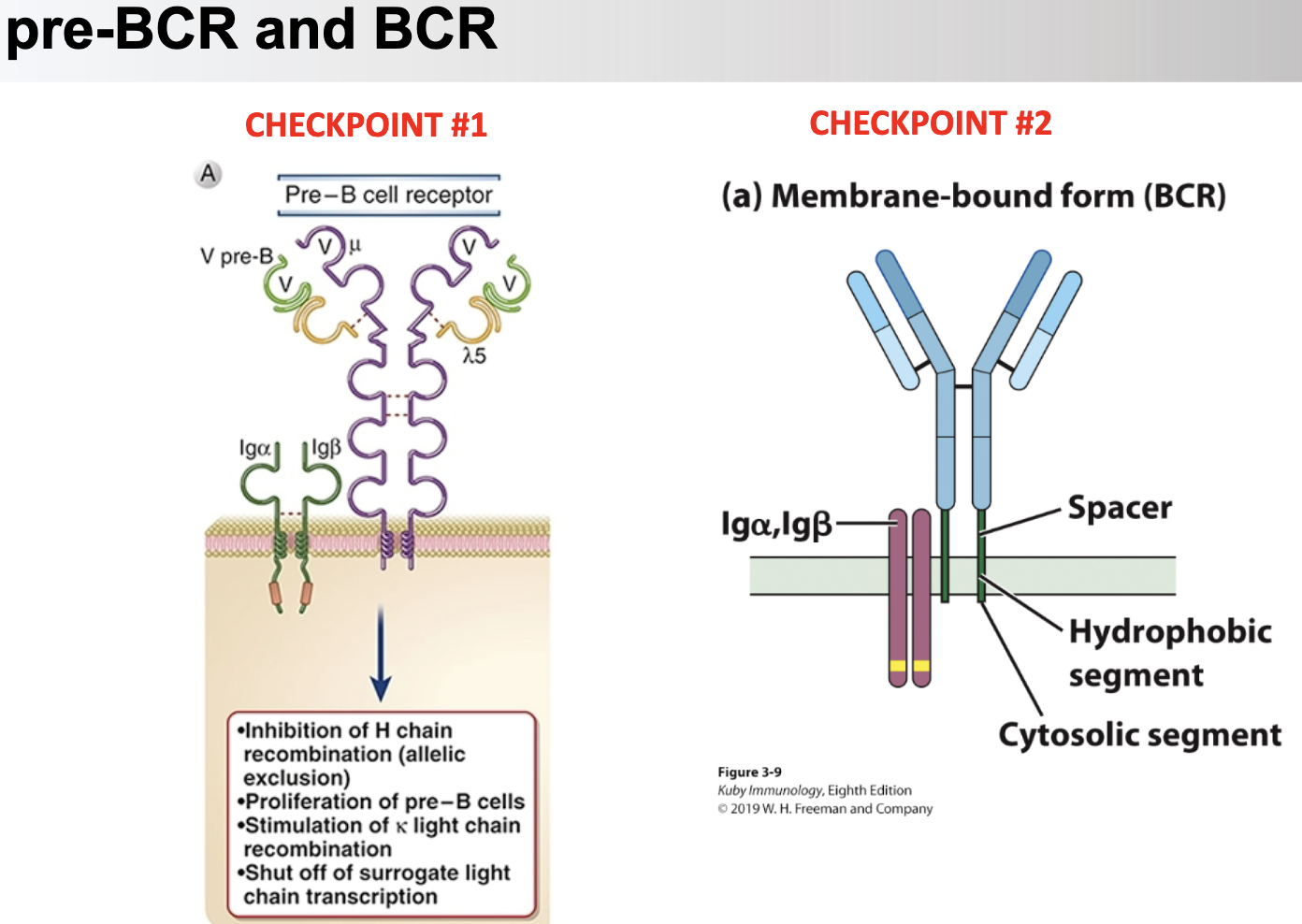
V pre-B, Lambda 5, productively rearranged heavy chain, and signaling subunits Ig-alpha and -beta.
pre-BCR made of?
germline (big toe)
RAG1/2 ; TdT —> HC RAR
Pre-BCR
2 ;
RAG1/2 ; TdT ; proliferation —> Large pre-BRAG1/2
VpreB and lambda5 ; BCR ;
IMMATURE B CELL
5Blood
Receptor-editing
6 (checkpoint 2)
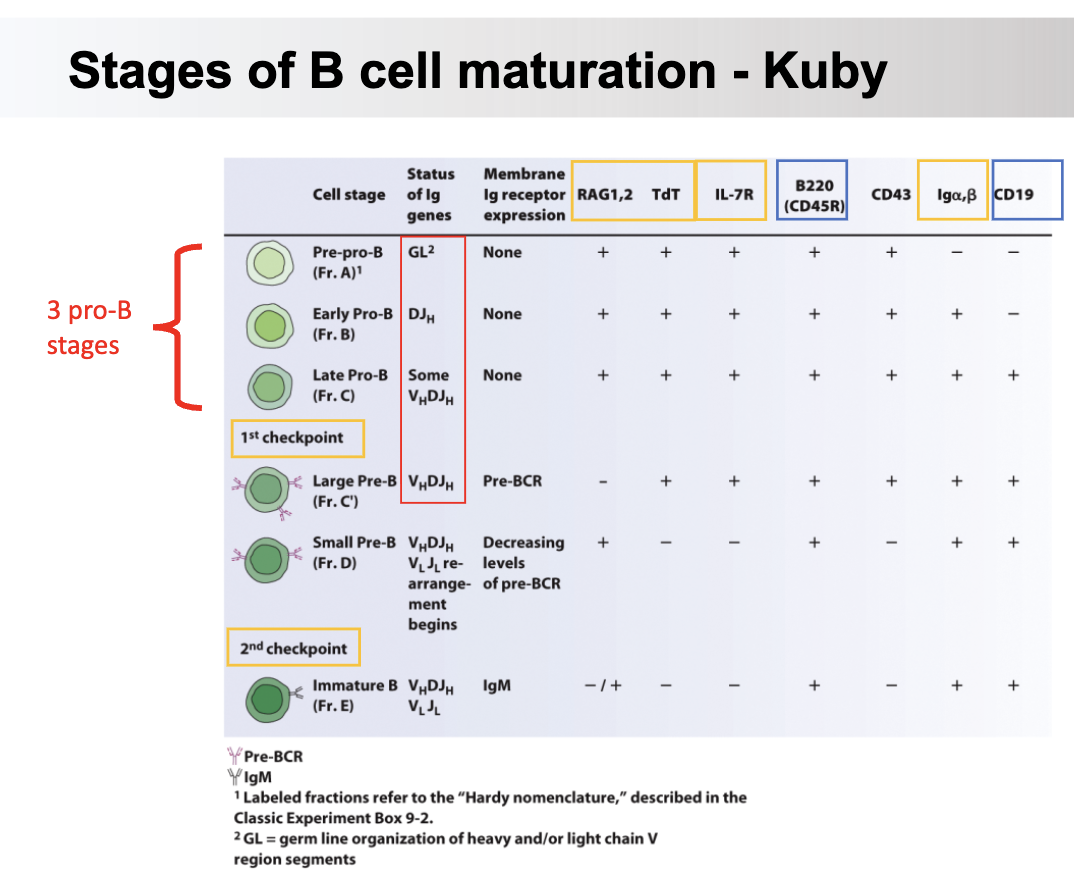
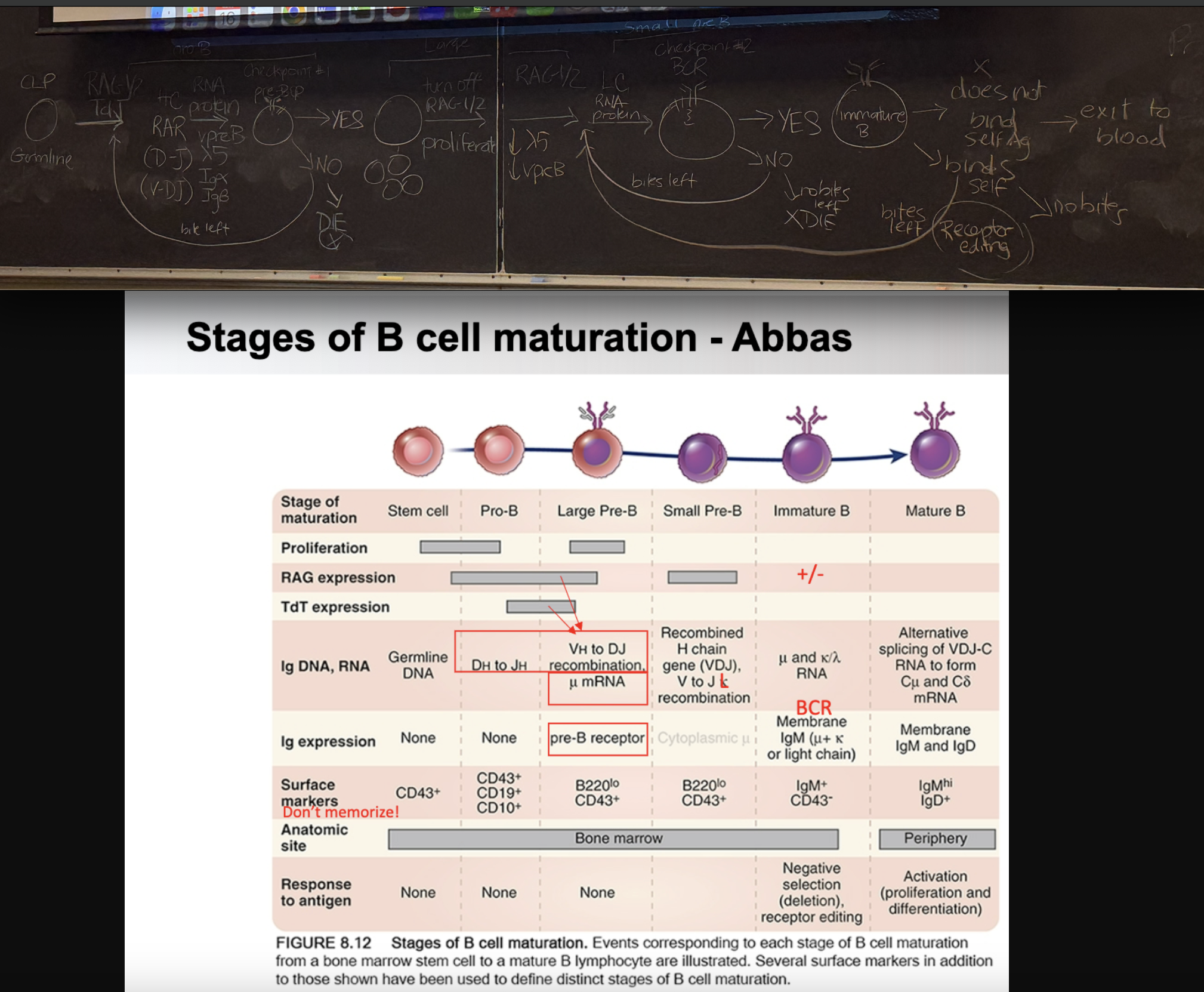
STEPWISE:
PRO B
CLP is genomically like _.
Turn on _ and _ —> _ (D-J) (V-DJ)
Turn on surrogate light chain (vpreB, lambda5, Igalpha/beta). —> _
LARGE PRE BCHECKPOINT #1:
If NO —> go back to step _ (try rearrangement again - if fails twice = DEATH).
If YES —> turn OFF _ (don’t want DSB during cell replication), and undergo _ —>
SMALL PRE BTurn ON _, express LC RNA and proteins
CHECKPOINT #2:
Turn OFF _ and _, forming —>
If signals YES —> _
If signals NO —> go back to step _ (if fails again = DEATH)If doesn’t bind self, exits to _.
If binds self, undergoes _, goes back to step _.
Ag
MHC
POSITIVE
B-cell development begins in the bone marrow and is completed in the periphery
Negative selection must occur, BUT ….
NO requirement for _ processing
NO need for _ restriction
NO _ SELECTION per se
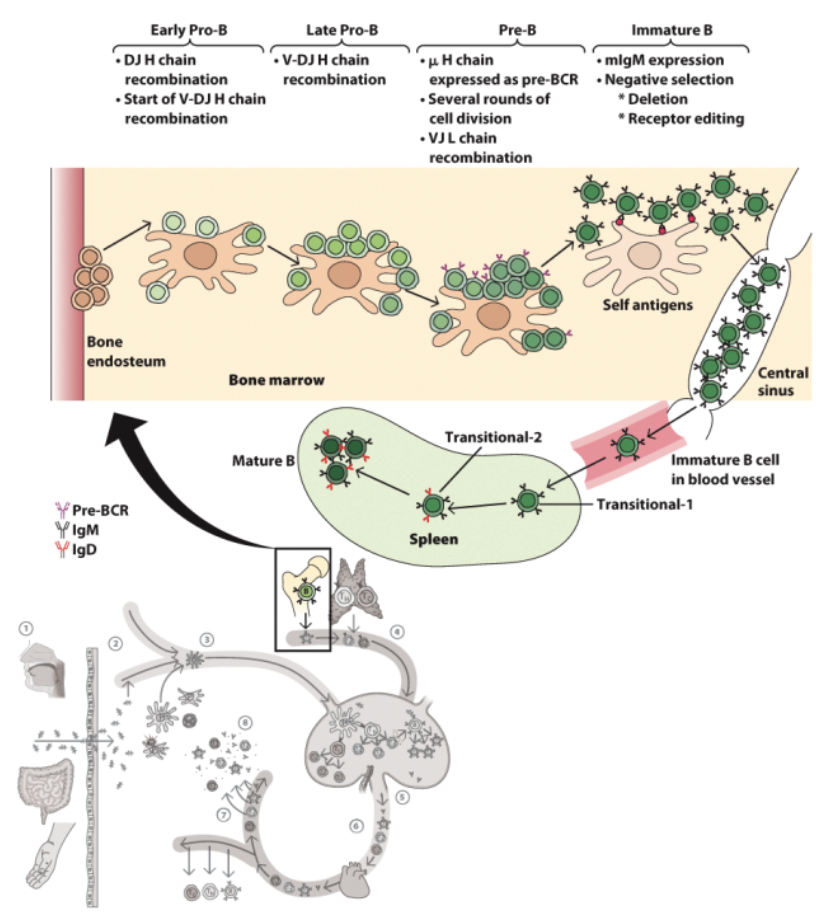
Immature B
self-antigens
B cell tolerance mechanisms: starts in BM. **Many, but not all, self-reactive B cells are deleted within the bone marrow
_ cells are “exquisitely sensitive to tolerance induction”
BCR is tested against _—three possible outcomes
Surface
Soluble
Checking for Self Antigen in B Cell Tolerance:
BM: Check for _ Ag
Spleen: Check for _ Ag
Clonal deletion
Death of strongly autoreactive cells by apoptosis
Receptor editing
Reactivation of light-chain recombination machinery
Anergy
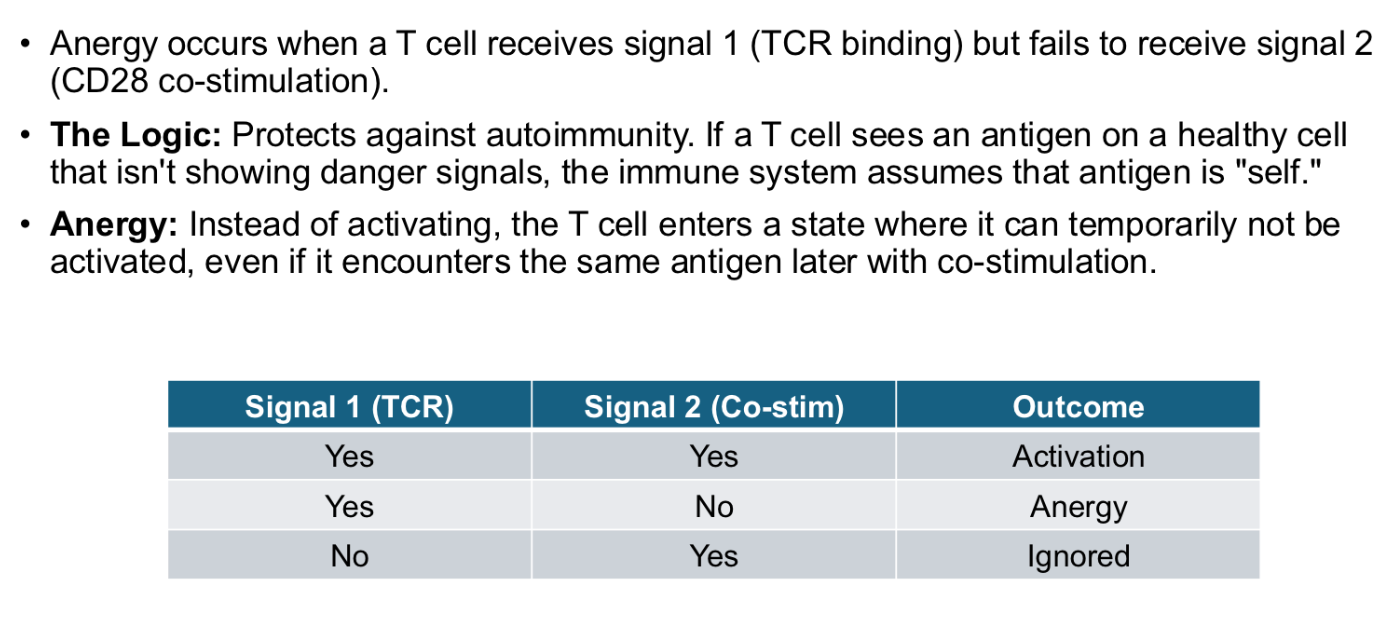
a state of immune system unresponsiveness where lymphocytes, particularly T cells, become functionally inactive upon encountering an antigen without necessary co-stimulatory signals. (even self-antigen stimuli)
functionally immature
B cells exported from the BM are ___
Transitional
screening
T1 and T2 subsets of immune responses (these are NOT T cells!)
T = _
Differ in gene expression as they progress through the spleen for further maturation.
The T1 subset is still undergoing _, possessing the possibility of a negative selection event.
This outcome is lost as the cells transition to T2 stage
AIRE
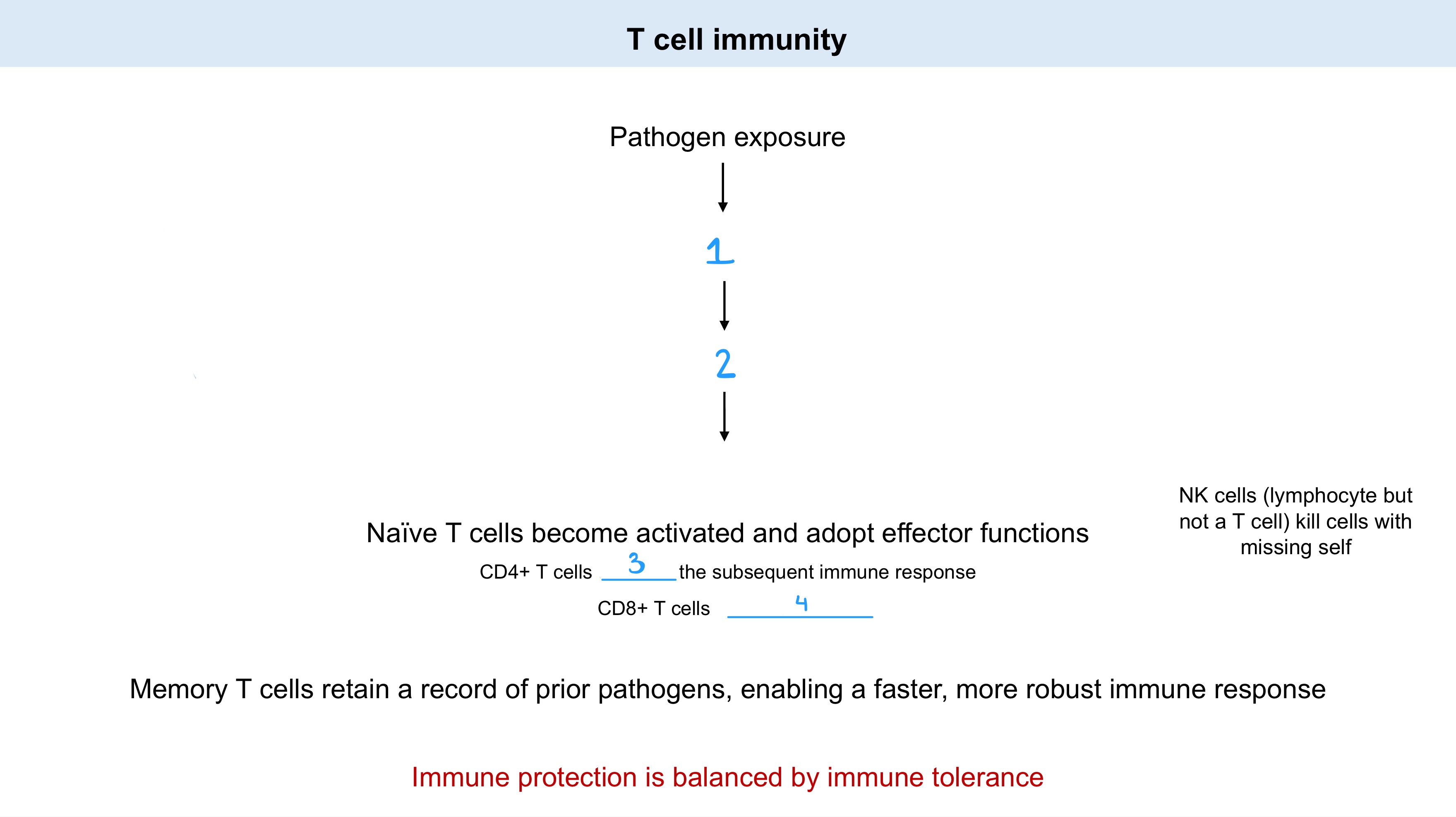
T cells originate from the bone marrow and undergo positive and negative selection in the thymus
Medullary thymic epithelial cells (mTECs) produce __
Result are CD4+ T cells and CD8+ T cells
secondary lymphoid tissues
Mature T cells circulate between thenblood and lymphatic system and reside within __
• E.g. spleen and lymph nodes
lymph nodes
Fill in blank
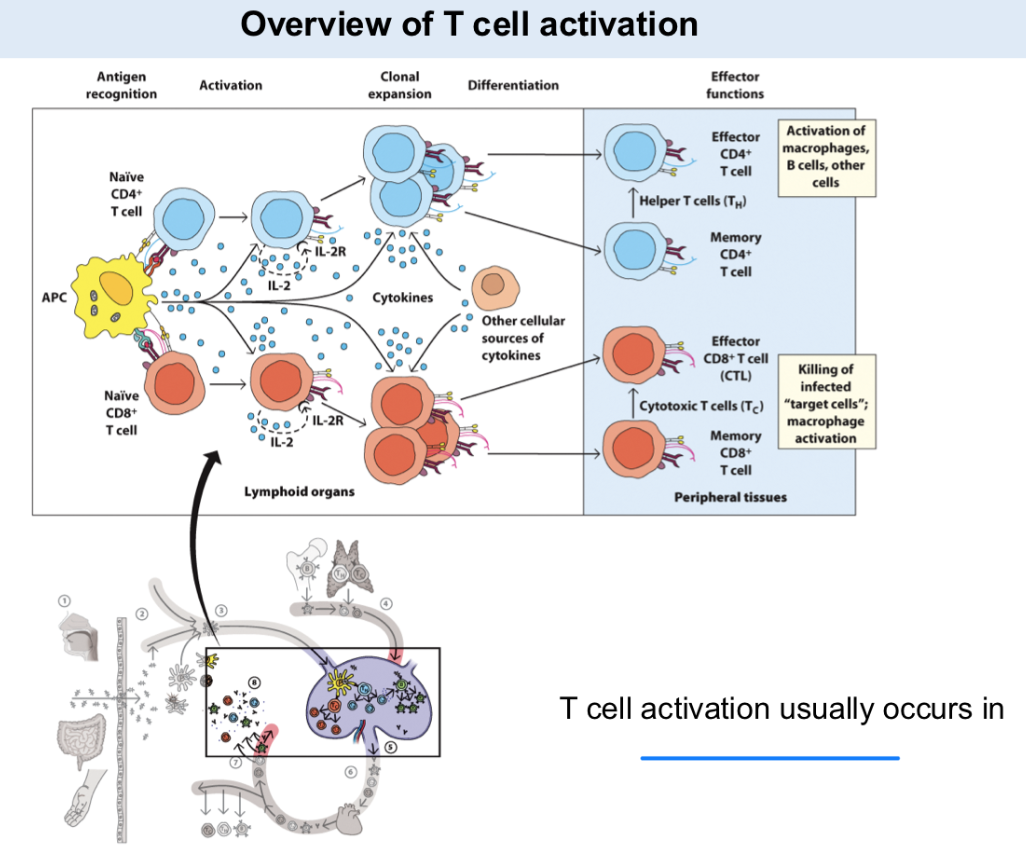
TCR recognition of antigen-MHC
Contact with costimulatory ligands
Cytokines directing T cell differentiation into distinct effector cell types
T cell activation is mediated by three signals:
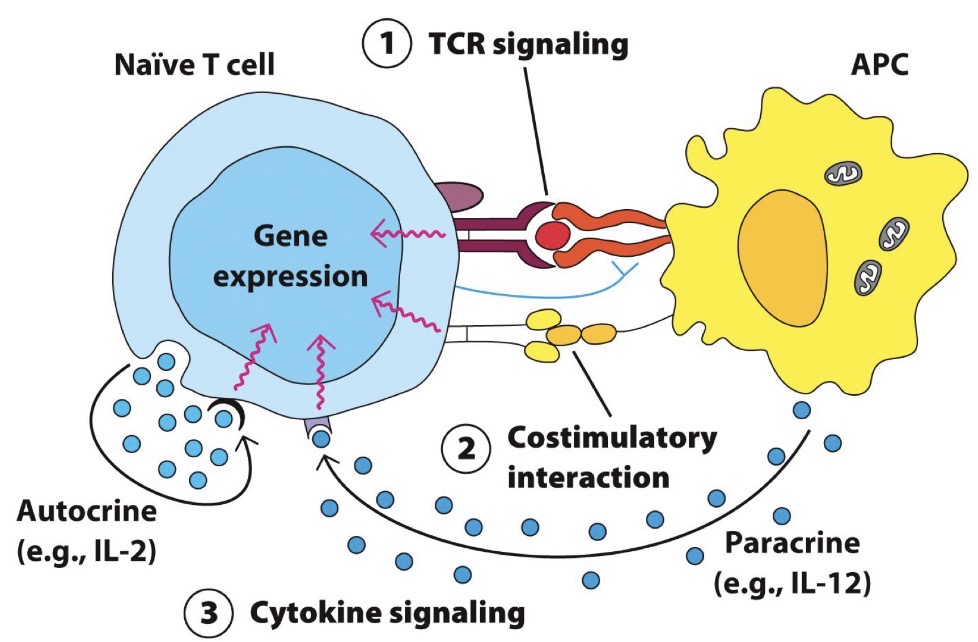
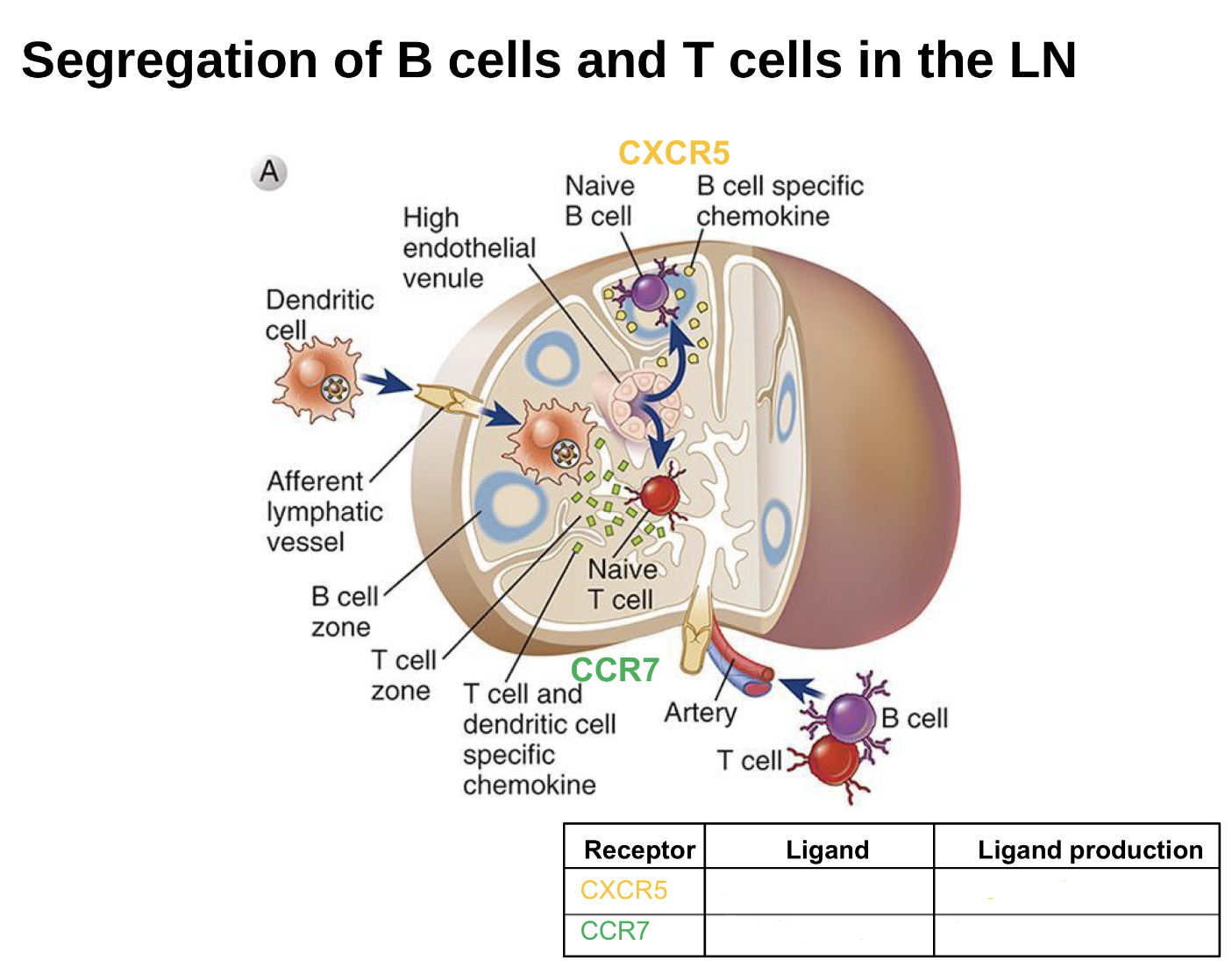
CXCL13 ; B-cell follicles
CCL19, CCL21 ; T-cell zone
Fill in table blanks
innate
overreaction
danger
Why does a purified antigen not evoke immune response, only with adjuvent it does?
immune system doesn’t just react to “foreignness.”
It reacts to foreignness plus danger signals.Immune system (esp. DCs) needs _ immune activation signals (called PAMPs or DAMPs) to fully switch on.
The body constantly encounters harmless proteins (food, self proteins, environmental particles). To avoid _, the immune system is set to default tolerance unless danger is detected.
Adjuvant solves this problem by mimicking _.
peptide-MHC complex ; APC
APC ; TCR
CD28
ANERGICAPC ; Th1, Th2, Th17
T cell activation is mediated by three signals:
TCR recognition of antigen-MHC.
TCR binds to _ on _.
Ensures only T cells specific to particular pathogen are activated.
CD4/CD8 coreceptors stabilize TCR-MHC interaction.Contact with costimulatory ligands.
Interaction btwn costimulatory molecules on _ and _.
_ on T cell binds to CD80 or CD86 on APC.
Promotes T cell survival, prolif, and IL-2 prod.
IF SIGNAL 1 OCCURS WITHOUT SIGNAL 2, THE T CELL BECOMES _.Cytokines directing T cell differentiation into distinct effector cell types.
Cytokines released by _ and surrounding environment bind to TCR. Directs differentiation of T cells into specific effector subsets (ex. ___) and promotes rapid expansion.

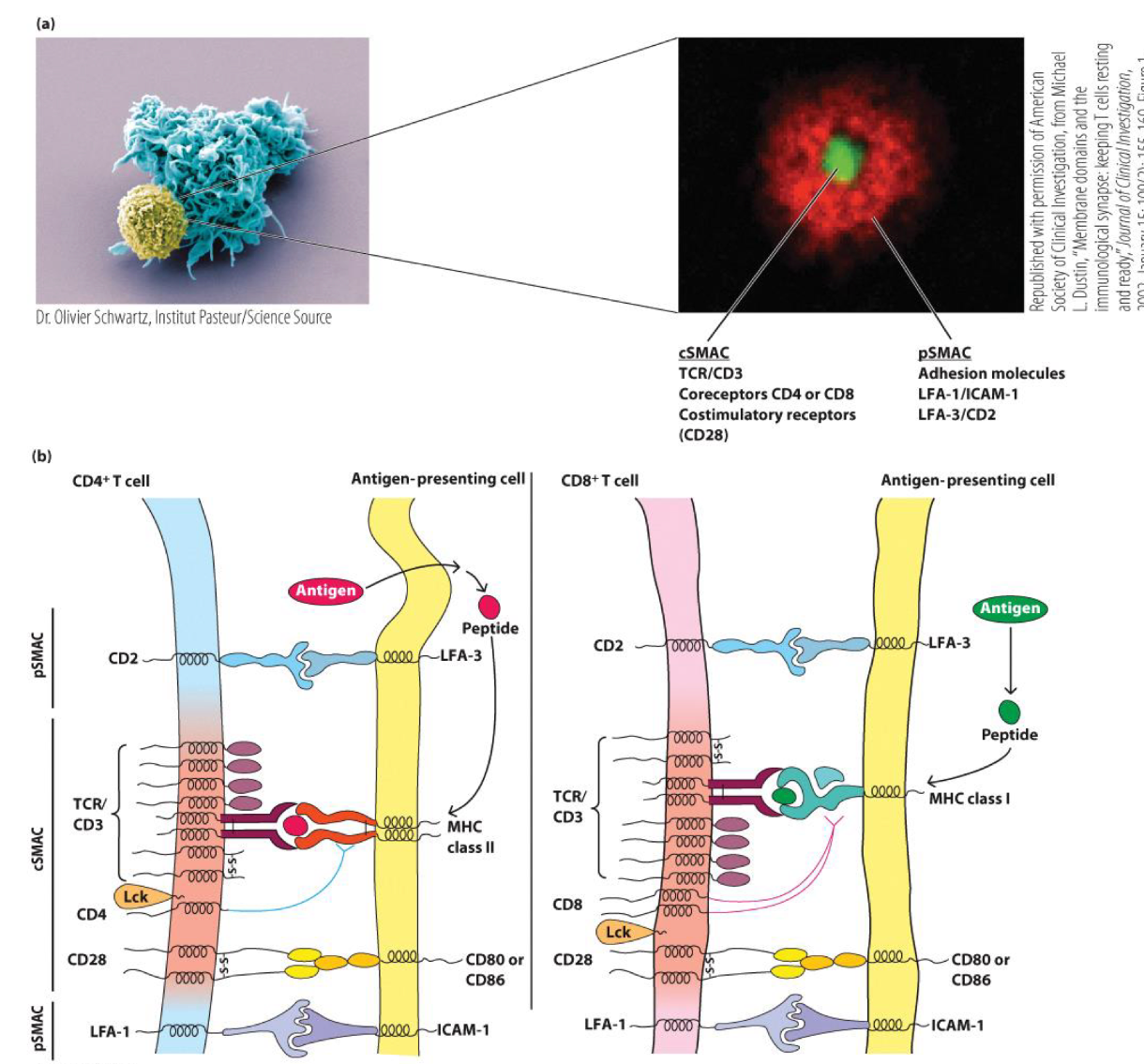
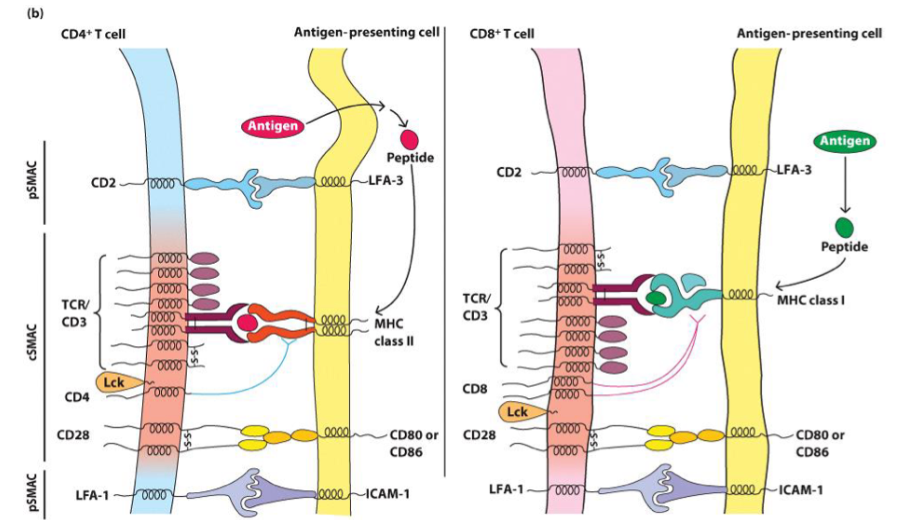
cSMAC (central supramolecular activating complex)
__
TCR/MHC-peptide and co-receptor complex.
Costimulatory molecules.
Adhesion molecules/bound ligands peripherally localize.
Lck
TCR-MHC
CD3
ZAP-70
LAT
PLCy
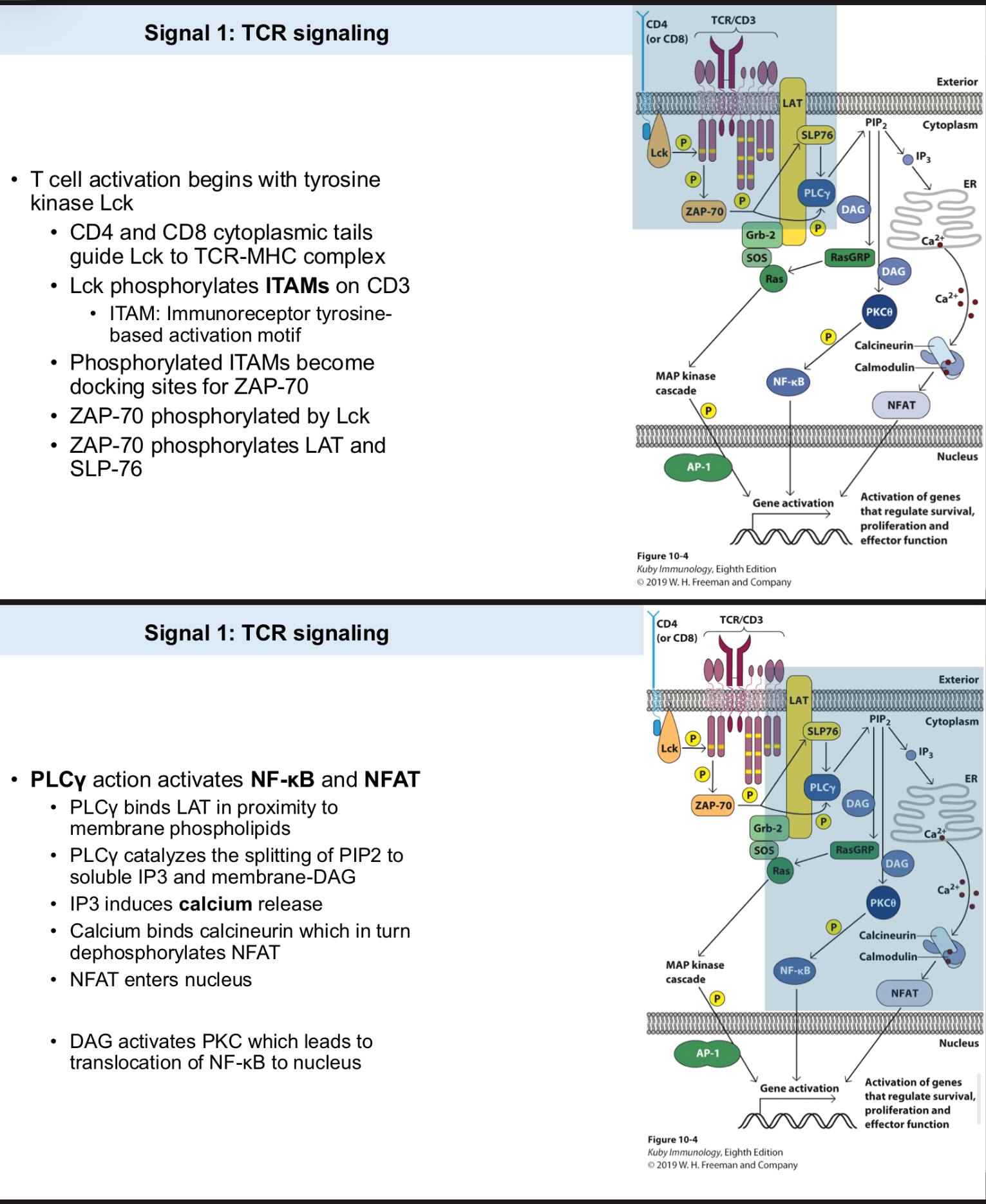
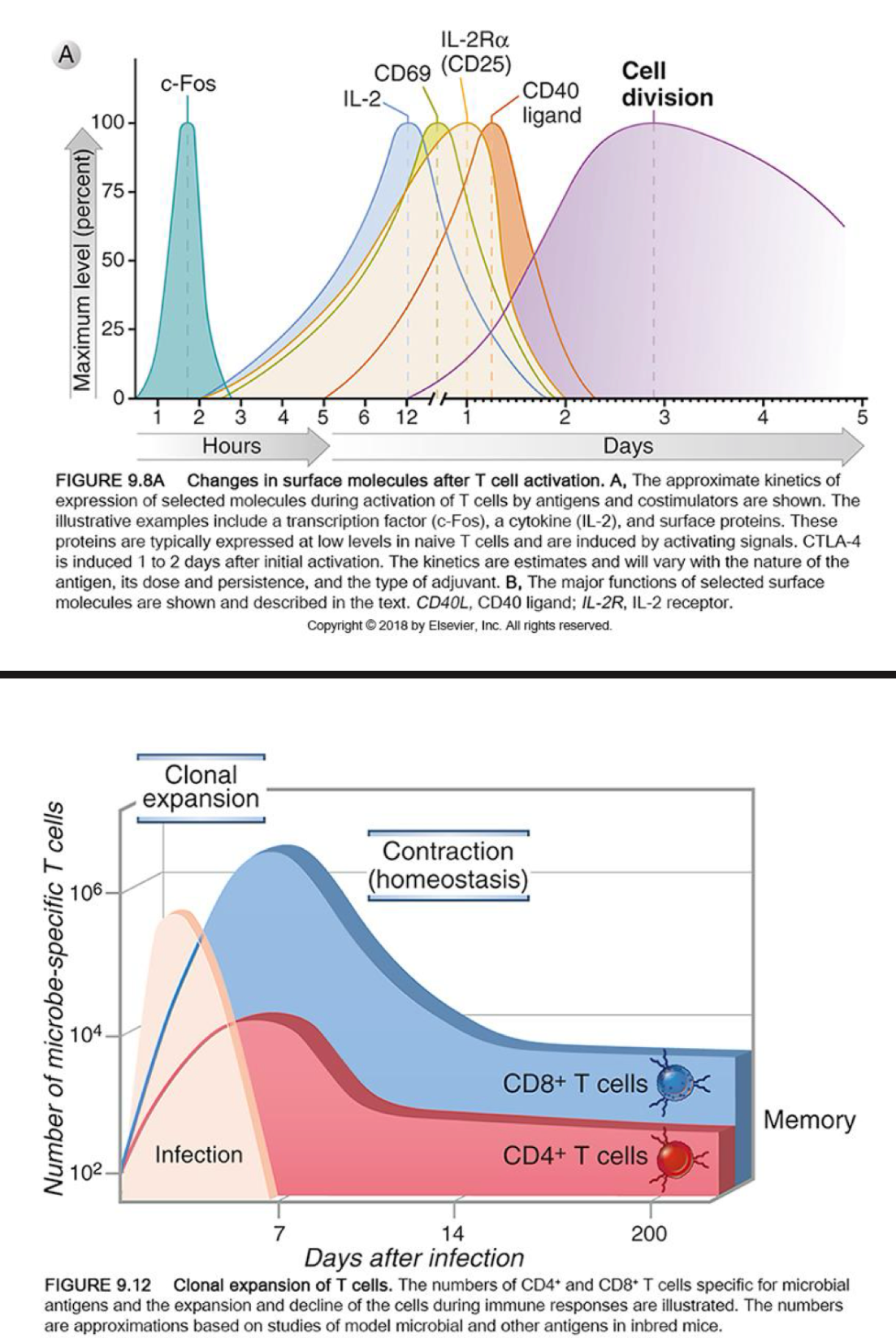
T cell activation begins with tyrosine kinase _
CD4 and CD8 cytoplasmic tails guide Lck to _ complex.
Lck phosphorylates ITAMs on _.
ITAM: Immunoreceptor tyrosine-based activation motif.
Phosphorylated ITAMs become docking sites for _.
ZAP-70 phosphorylated by Lck.
ZAP-70 phosphorylates _ and SLP-76.
_ action activates NF-kB and NFAT
PLCy binds LAT in proximity to membrane phospholipids.
PLCy catalyzes the splitting of PIP2 —> IP3 and membrane-DAG
IP3 —> Ca2+ release
Calcium binds calcineurin —> dephosphorylates NFAT
NFAT enters nucleus
DAG activates PKC —> translocation of NF-kB to nucleus
RAS-ERK signaling & activation of AP-1 transcription factor.
Ras pathway triggers MAPK activation
MAPK cascade phosphorylates in sequence, RAF, MEK, ERK
ERK activates AP-1
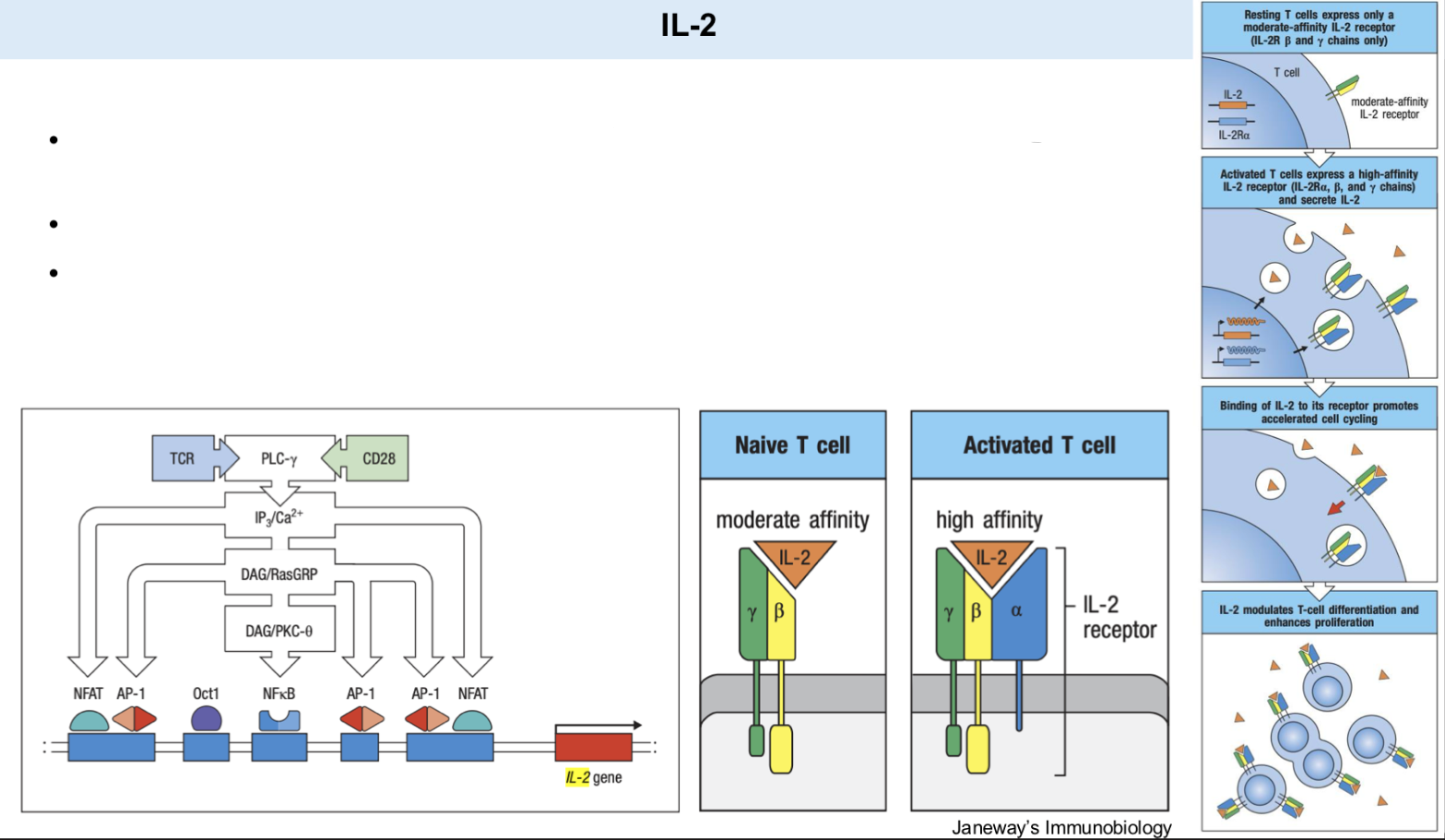
IL-2 ; IL-2R
T cell
• TCR activation leads to transcription of the cytokine _ as well as its high affinity receptor _ (CD25).
• Autocrine and paracrine signaling.
• IL-2 signaling is necessary for _ proliferation.
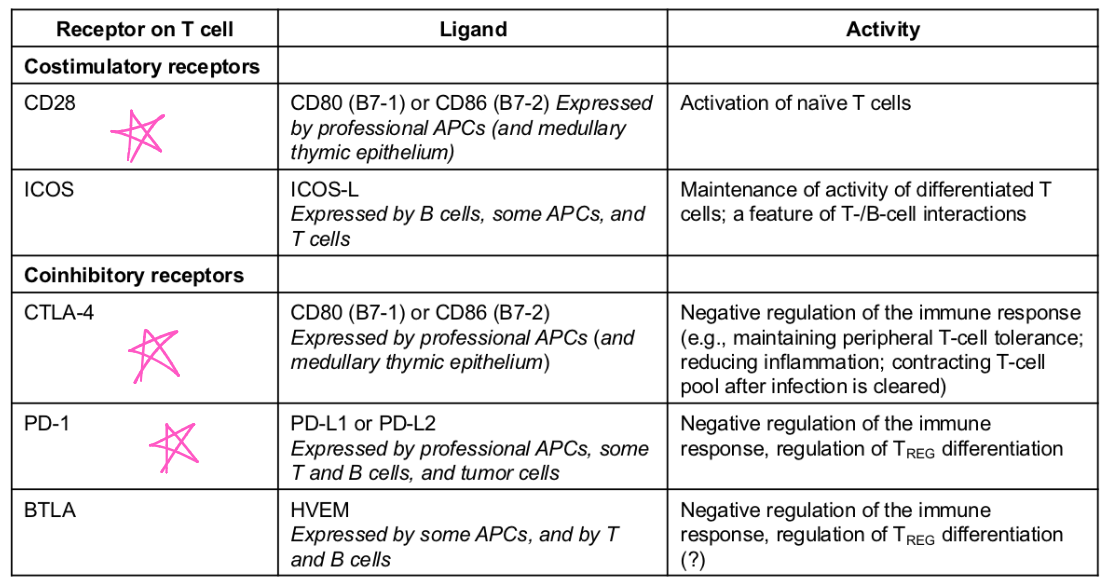
Costimulatory
__ signals are required for T cell activation and proliferation.
Signal 1 gives the cell a target.
(TCR + MHC)Signal 2 gives the context that APC has detected danger.
(CD28 on T cell + CD80/86 on APC)
CD28
CTLA-4, PD-1, BTLA
Costimulatory signals are required for T cell activation and proliferation.
Positive costim. receptors —> activation
Example(s): _Negative costim. receptors —> OFF
Example(s):
prosurvival
IL-2 and IL-2R
clonal cells
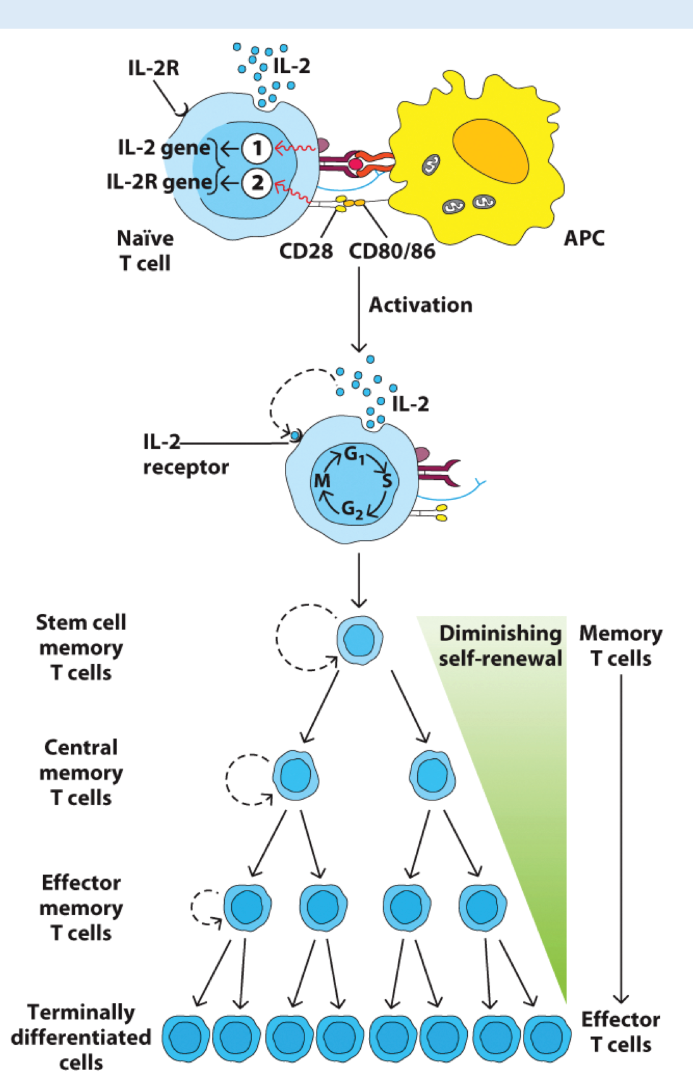
Clonal Expansion
Initial activation signals 1 and 2 induce
Upregulation of _ genes, e.g., Bcl-2.
Transcription of _ and _ genes
Autocrine and paracrine signaling.
Potent cytokine and growth factor crucial for the proliferation, survival, and differentiation of T cells
Outcome is activation and robust proliferation.
Production of memory cells and effector _.
T cell
Timeframe of _ activation
Superantigen
outside
a type of microbial protein (usually from bacteria or viruses) that causes an abnormally large and nonspecific activation of T cells, leading to an intense and potentially dangerous immune response.
Binds directly to MHC class II
(_ the normal peptide-binding groove)

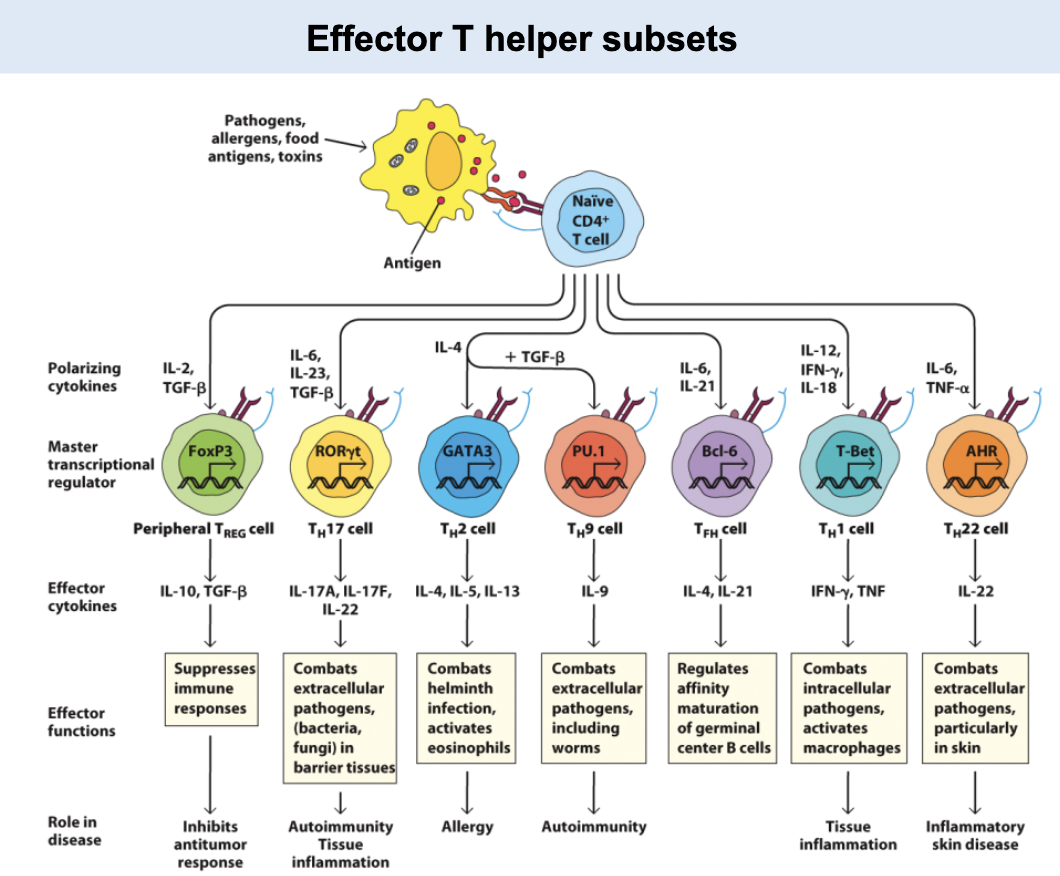
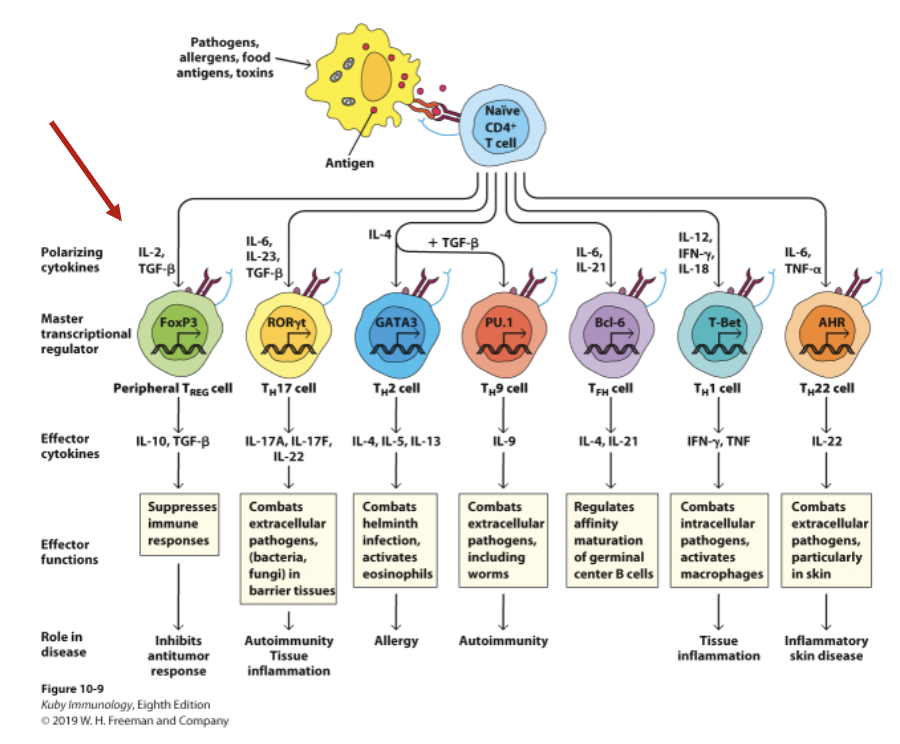
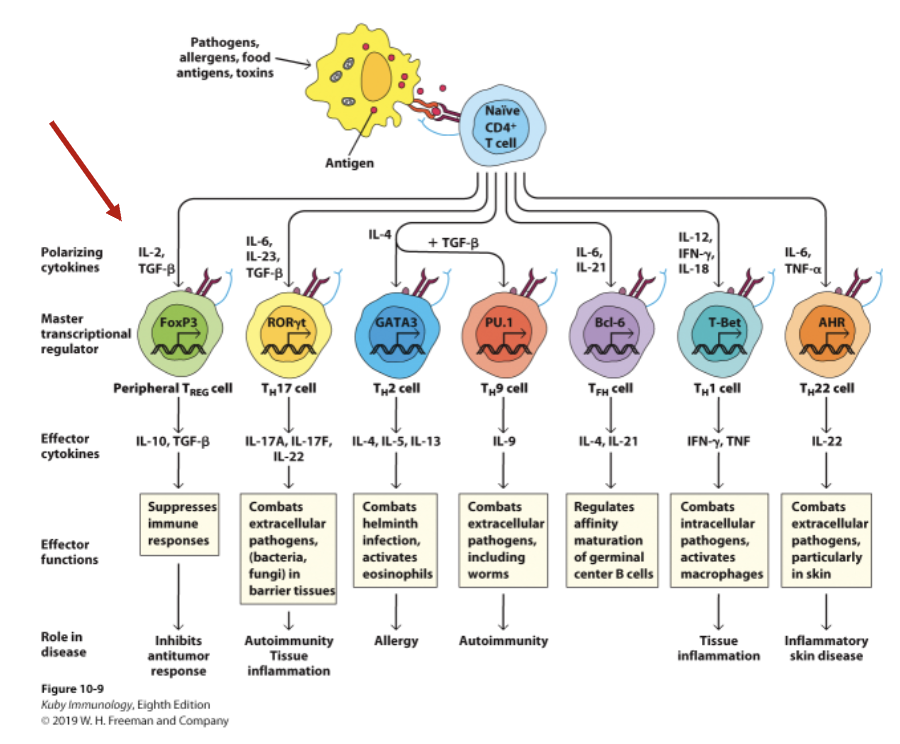
Effector T helper
_ subsets are distinguished by these properties:
Distinct polarizing cytokine set that induces expression of a master gene regulator.
A signature set of effector cytokines produced by that subset.
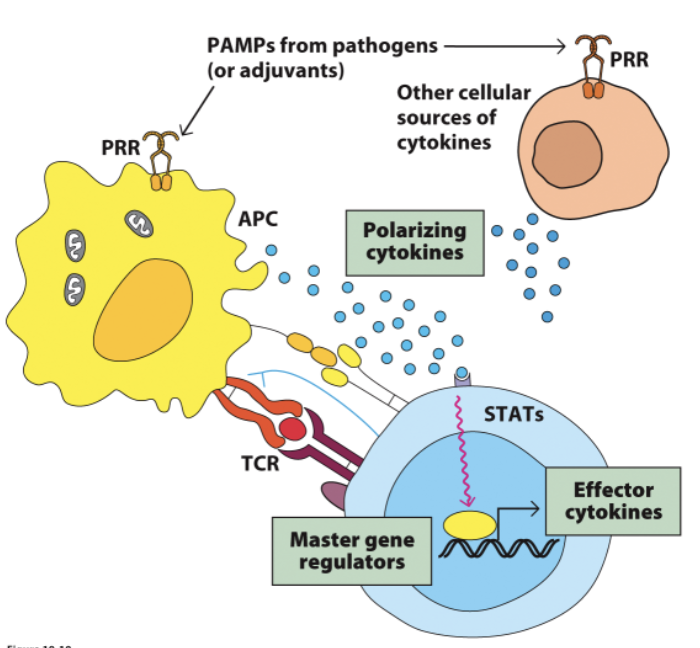
TH1
TH2
TH17
TREGs
TFH
Helper T cells can be divided into at least five distinct subsets
_ regulate immunity to intracellular bacteria and viruses.
_ regulate immunity to parasites.
_ regulate immunity to extracellular bacteria and fungi.
_ are inhibitory in terminating immune responses and inhibiting autoimmunity.
_ regulate humoral immunity (B cells).
TH2
TH1 and _ response/ indicators
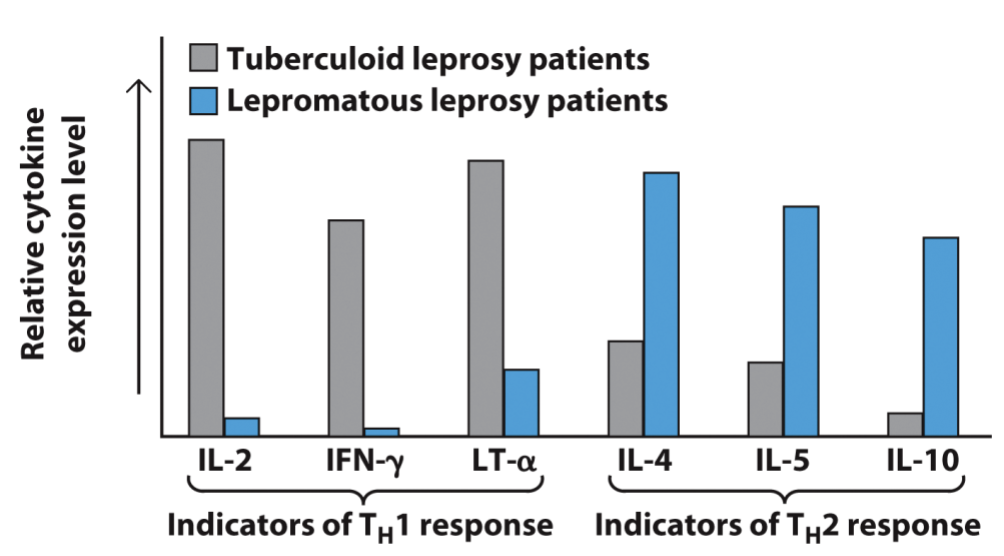
antigen clearance
IL-2
apoptosis signaling
T cell contraction is the phase after an immune response where 90-95% of effector T cells die by apoptosis, leaving a
small, stable population of memory cells.
It is primarily mediated by…
_ (withdrawal of stimulating signals).
diminished _ availability
__ __ via Bim and Fas-FasL interactions
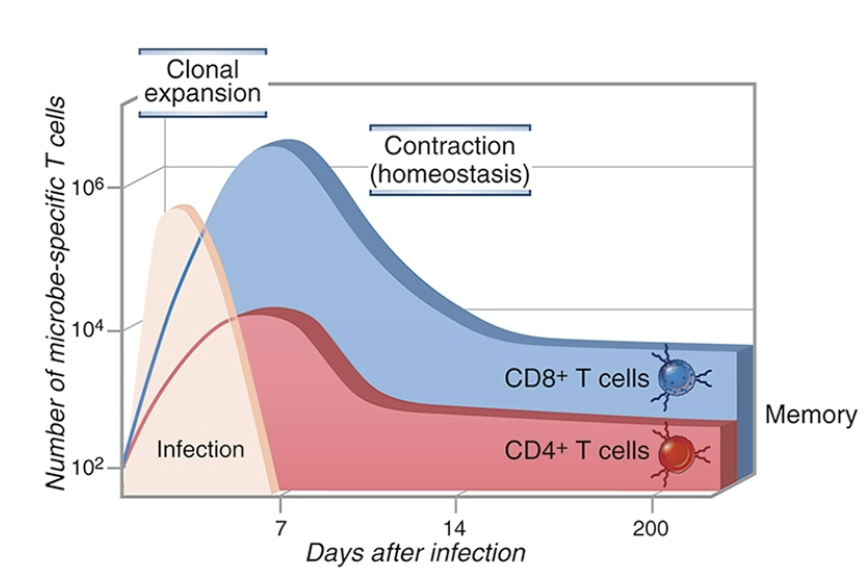
Memory T cells
Definition: Long-lived, previously activated T cells that provide rapid, enhanced protection against pathogens from initial infection or
vaccination.
Persistence: They reside in lymphoid and peripheral tissues, self-renewing for life without constant antigen stimulation.
Rapid Response: Upon re-encountering an antigen, they quickly produce high numbers of effector cells, minimizing the immune response lag time.
TEM (effector memory T cells)
TRM (permanent residents of previously infected tissue )
Memory T cells are distinguished by their location and commitment to effector function
They circulate in the blood and non-lymphoid tissues. They are ready to act immediately, providing quick protection by killing infected cells or releasing signals to help other immune cells.
They do not circulate. Instead, they stay behind in specific barrier tissues (like skin, lungs, or gut) where the original infection occurred, providing immediate, localized protection against reentry.
no reg T cells
Read case study

Central tolerance
deletion of autoreactive T cells (lymphocytes) during maturation/before they mature
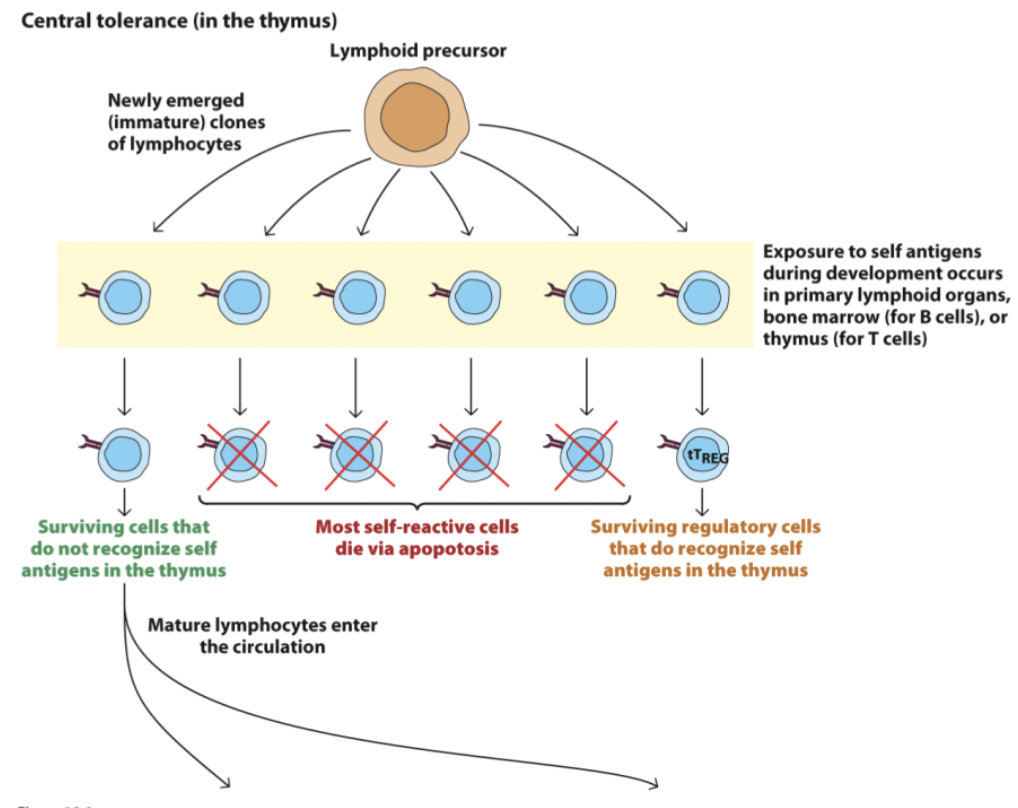
Peripheral tolerance
occurs when mature lymphocytes that escaped central deletion are suppressed ; active generation of inhibitory lymphocytes
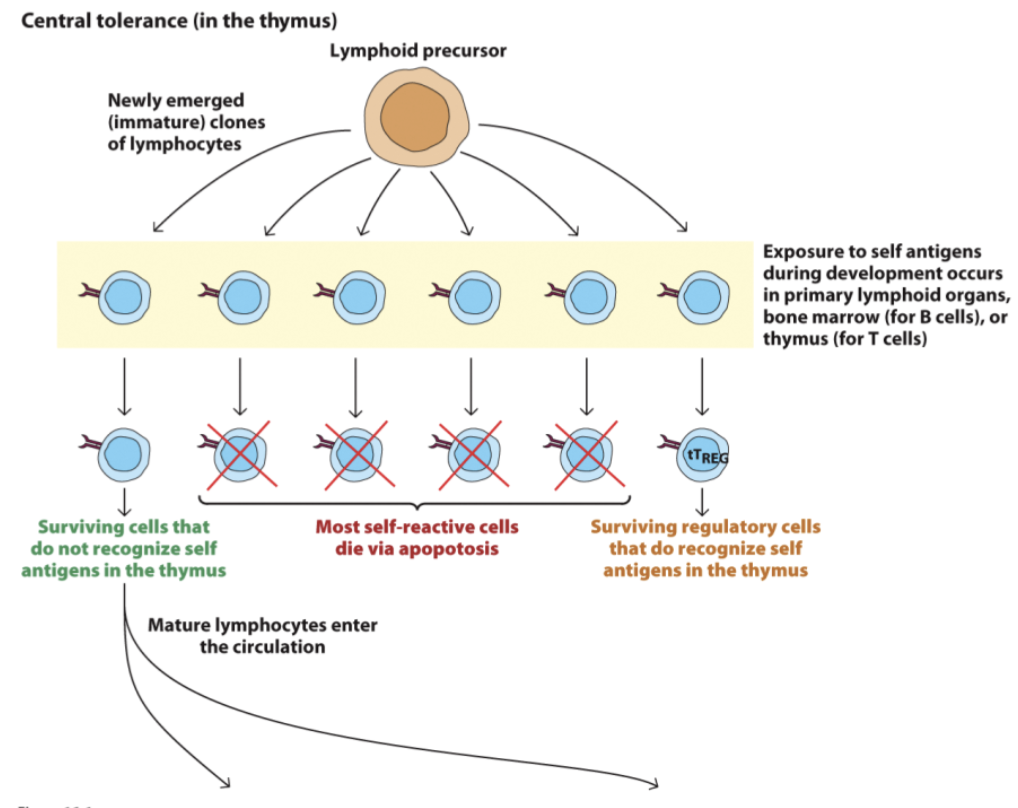
Failure of tolerance
Immune system attacks healthy tissues or benign entities, resulting in disease
TREG
_ cells negatively regulate immune responses:
Belong to a subset of CD4 T cells characterized by the expression of Foxp3 transcription factor
Function to:
Deplete the local area of stimulating cytokines
Produce inhibiting cytokines
Inhibits the activity of APCs, T cells, etc
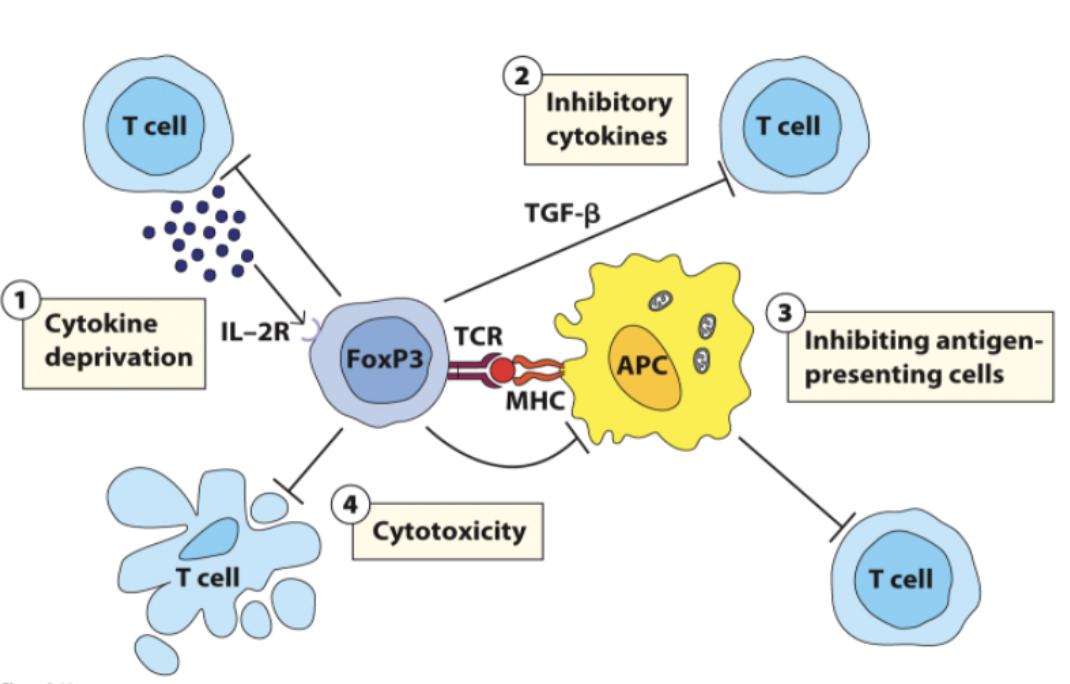
tTREG
Type of TREG:
Can be generated in the thymus (_ cells) in response to high affinity to self-peptide
Still engage Ag-MHC class II complexes through TCR , Downregulate responses when they do so
pTREG
Type of TREG:
Can also be generated in the periphery following Ag induction (_ cells) in the presence of retinoic acid, TGFβ, and IL-2
Still engage Ag-MHC class II complexes through TCR, Downregulate responses when they do so
IL-2 receptor (CD25)
Tregs constitutively express the high-affinity _, allowing them to consume local IL-2 and deprive other T cells, preventing their activation
In comparison, other T cells inducibly express the high-affinity IL-2 receptor
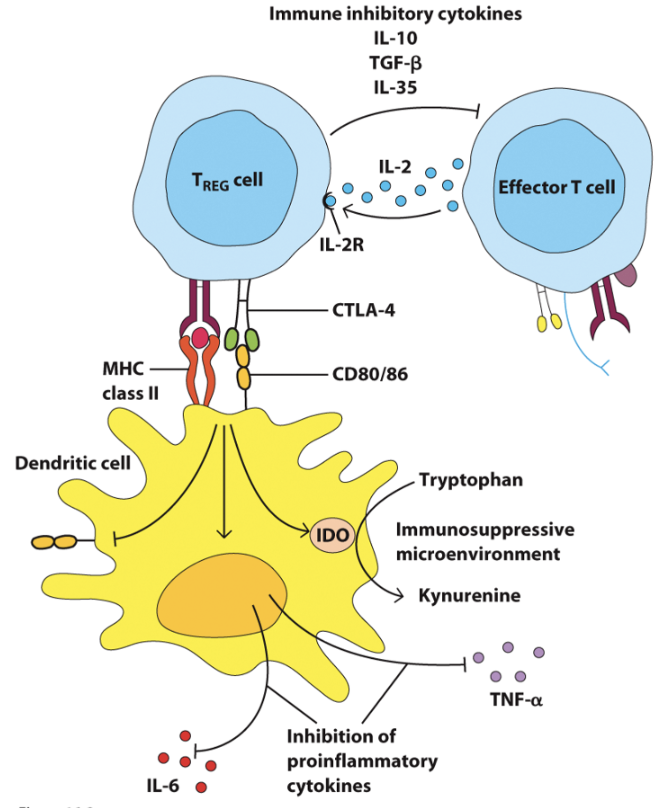
IL-10
Tregs produce broadly inhibitory cytokines such as _
CTLA-4
Tregs constitutively express _, which binds to CD80/CD86 on APCs with higher affinity than CD28, effectively depriving effector T cells of necessary costimulatory signals.
What is the term to describe what happens to cells that receive signal 1 (TCR stim) but not co-stim?
This is a form of competitive inhibition
CTLA-4 ; CD80/CD86
PD-1
Negative receptors - help turn activation off
_ (CD152) competes for binding to _
Induced within 24 hours after activation, peaks 2–3 days post-stimulation
Binds to CD80 (B7-1)/CD86 (B7-2) with higher affinity than CD28 but shuts down signaling pathways (“putting the brakes on”)
Program death-1, CD279
_ binds to its ligands PD-L1/PD-L2, which inhibits T cell activation, proliferation, and dephosphorylaties TCR machinery
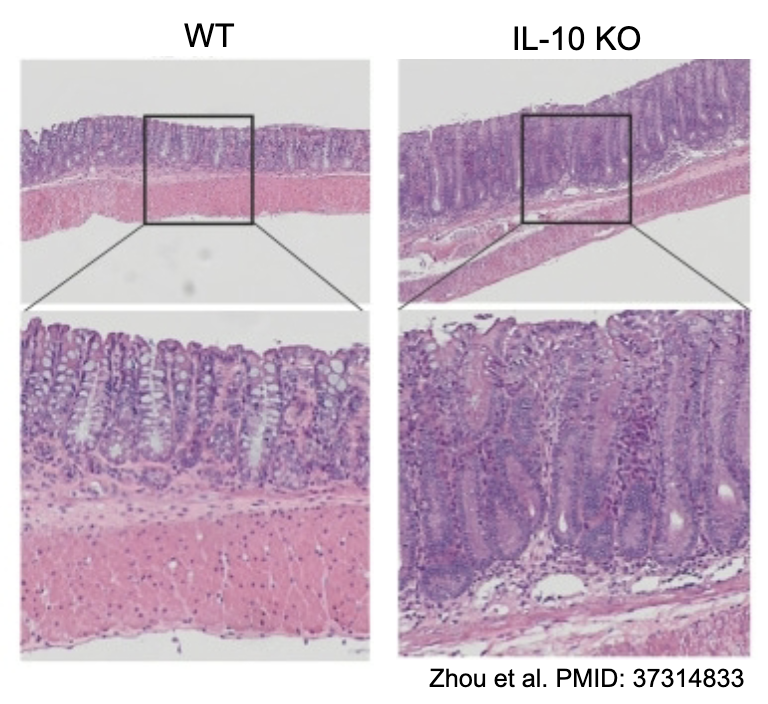
IL-10
_ is a broadly inhibitory cytokine essential for maintaining self-tolerance and immune homeostasis
Inhibits proinflammatory cytokine production (IL-6, IL-12) by APCs.
Suppresses Th1 and Th17 cell differentiation.
Promotes Treg differentiation.
What about this cytokine makes it effective across so many different cell types?
Link to excess inflammation: Deficiency in IL-10 can lead to spontaneous colitis and increased Th17 pathology.
Tregs
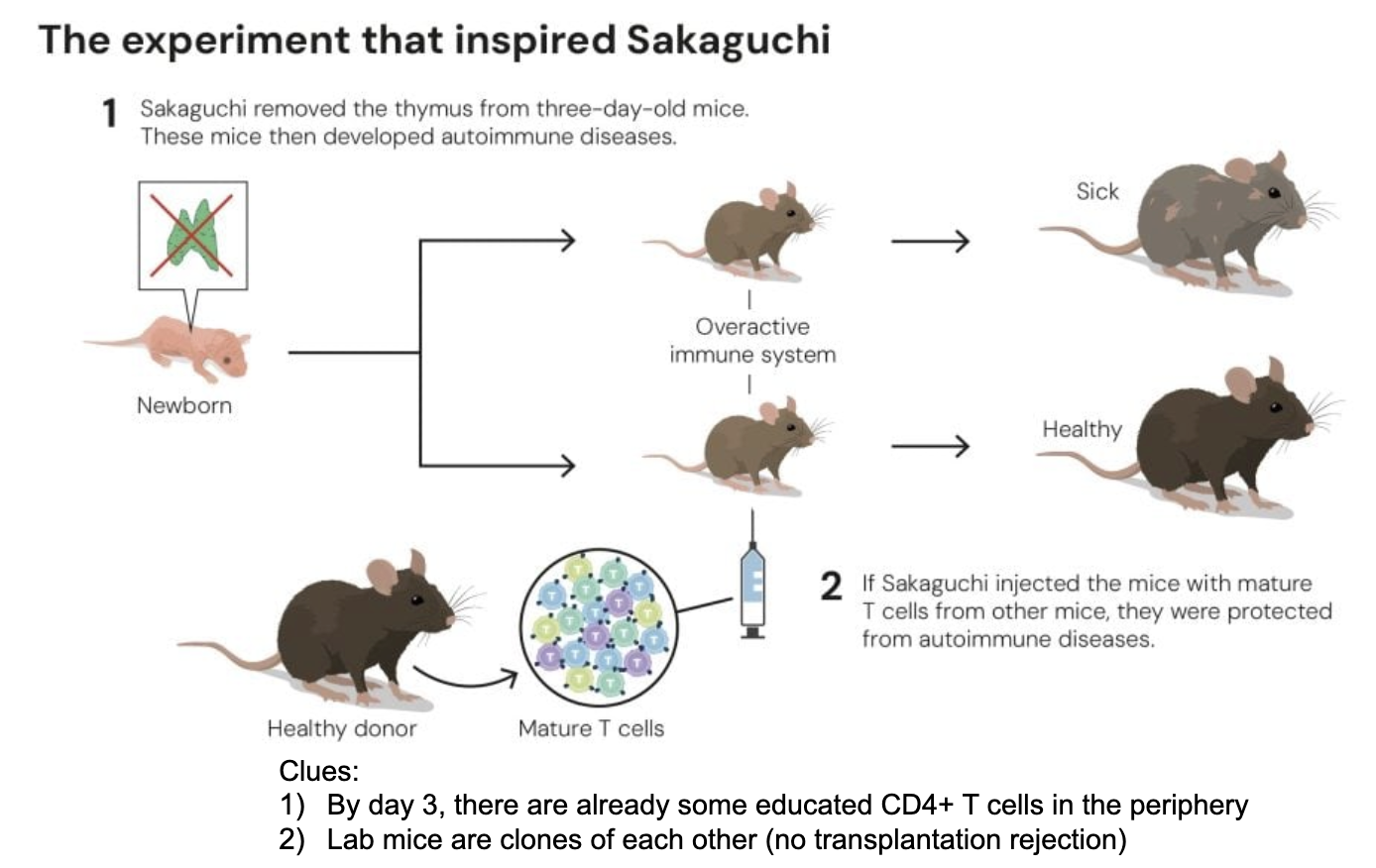
Shimon Sakaguchi’s pivotal 1995 experiments discovered _ by showing that removing the thymus from 3-day-old mice caused autoimmune diseases, which could be prevented by injecting CD4+CD25+ T cells from healthy adults.
This demonstrated that _^_, maturing in the thymus, prevent self-attacking immune responses.
ok
Other regulatory types

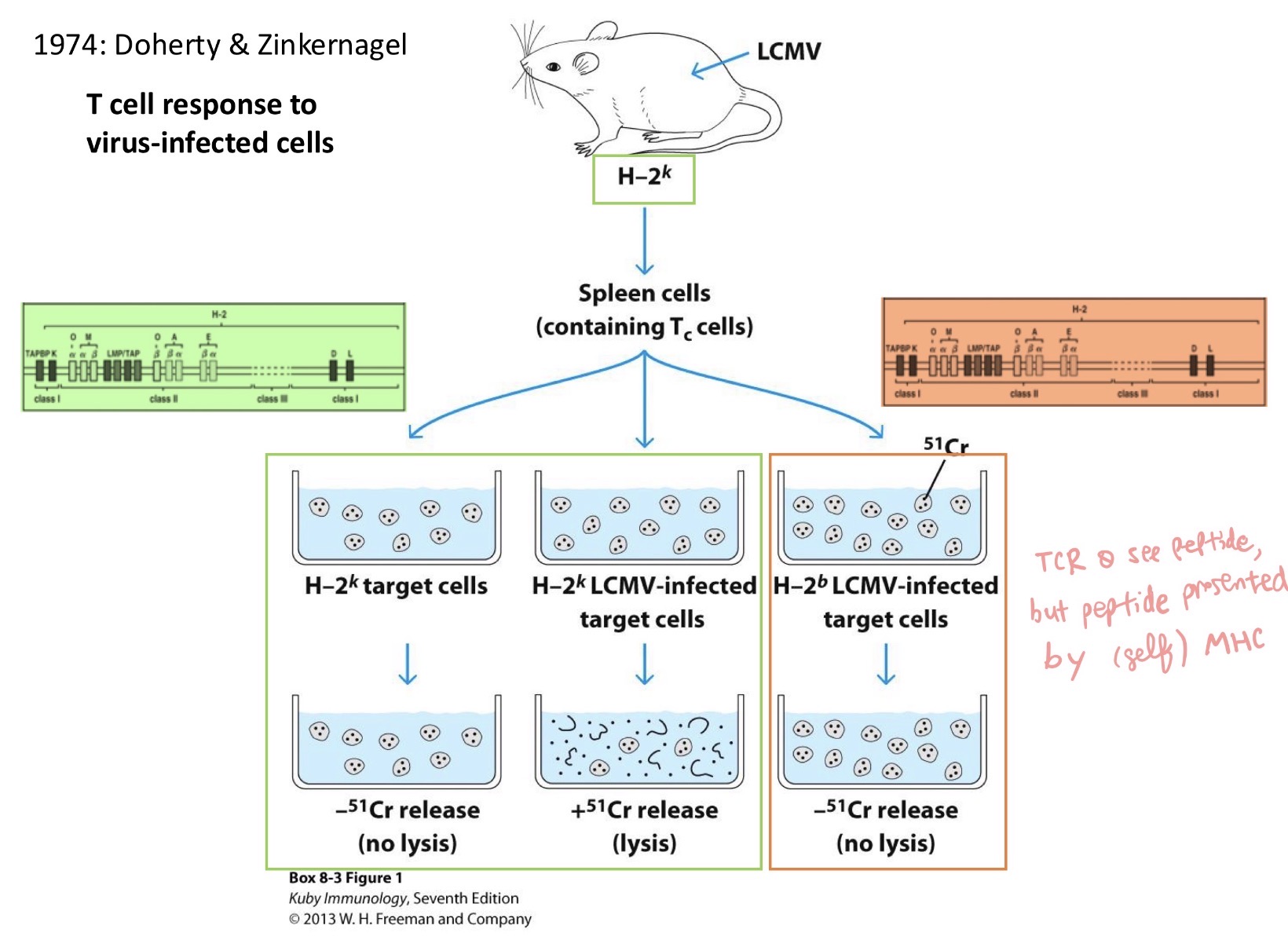
TCR doesn't recognize entire peptide/antigen, but peptide presented by SELF MHC (not someone else's MHC)
What does this experiment show?
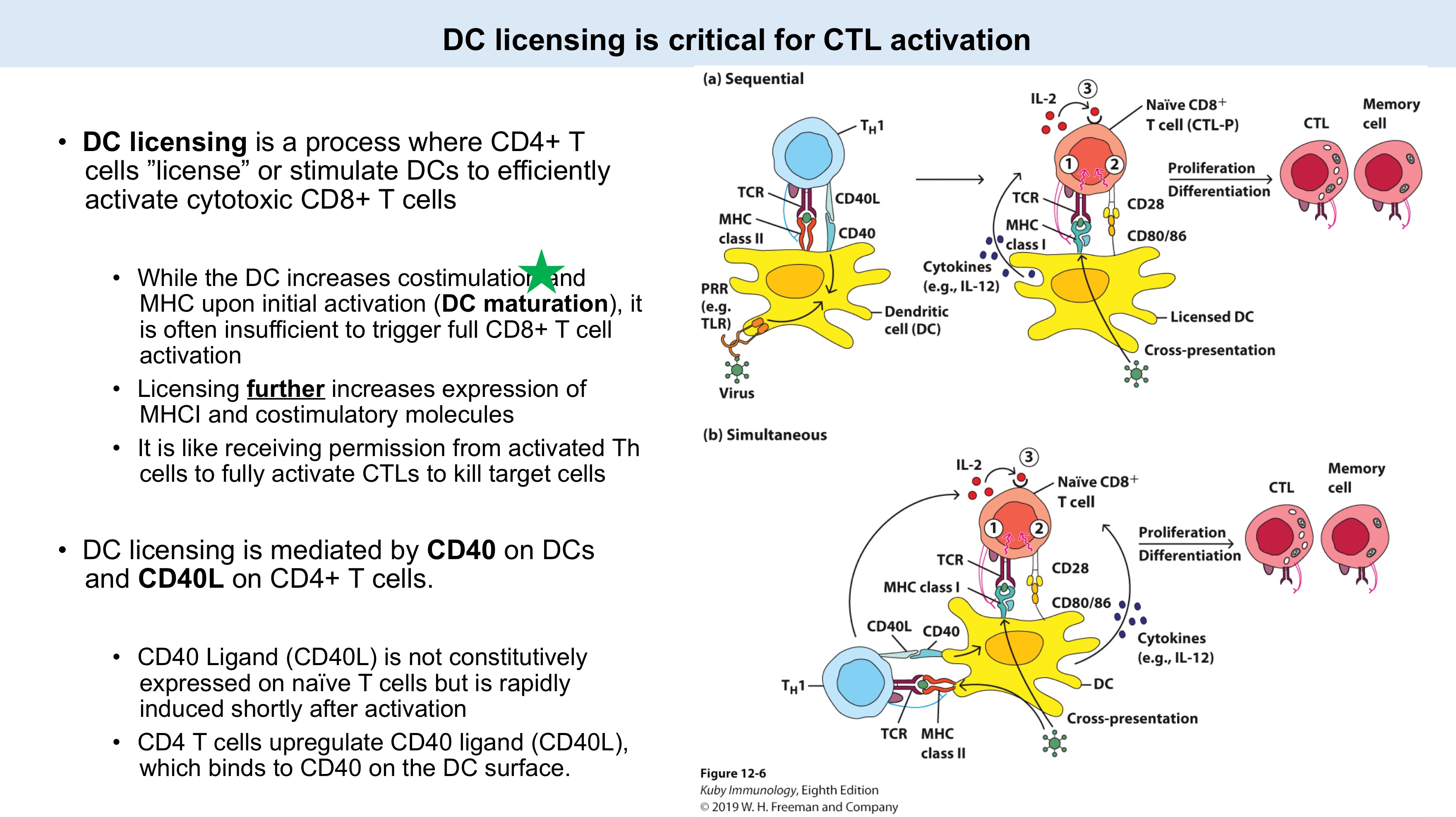
DC licensing
DC maturation
MHCI ; costimulatory
Th
CD40 Ligand (CD40L)
DC
__ is a process where CD4+ T cells ”license” or stimulate DCs to efficiently activate cytotoxic CD8+ T cells.
While the DC increases costimulation and MHC upon initial activation (__), it is often insufficient to trigger full CD8+ T cell activation.
Licensing further increases expression of _ and _ molecules.
It is like receiving permission from activated _ cells to fully activate CTLs to kill target cells.
DC licensing is mediated by CD40 on DCs and CD40L on CD4+ T cells.
_ is not constitutively expressed on naïve T cells but is rapidly induced shortly after activation.
CD4 T cells upregulate CD40 ligand (CD40L), which binds to CD40 on the _ surface.
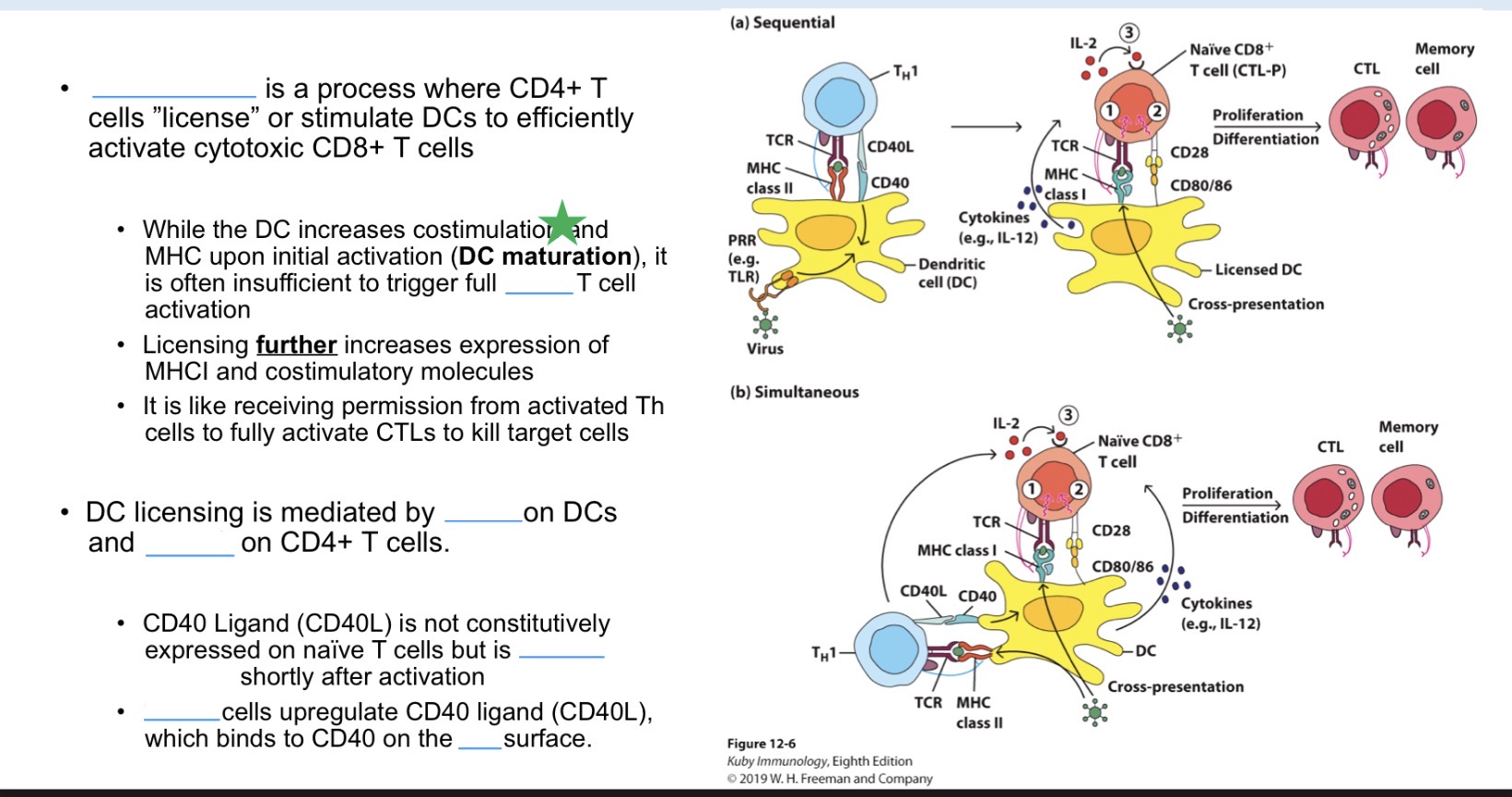
done
Explain this graph
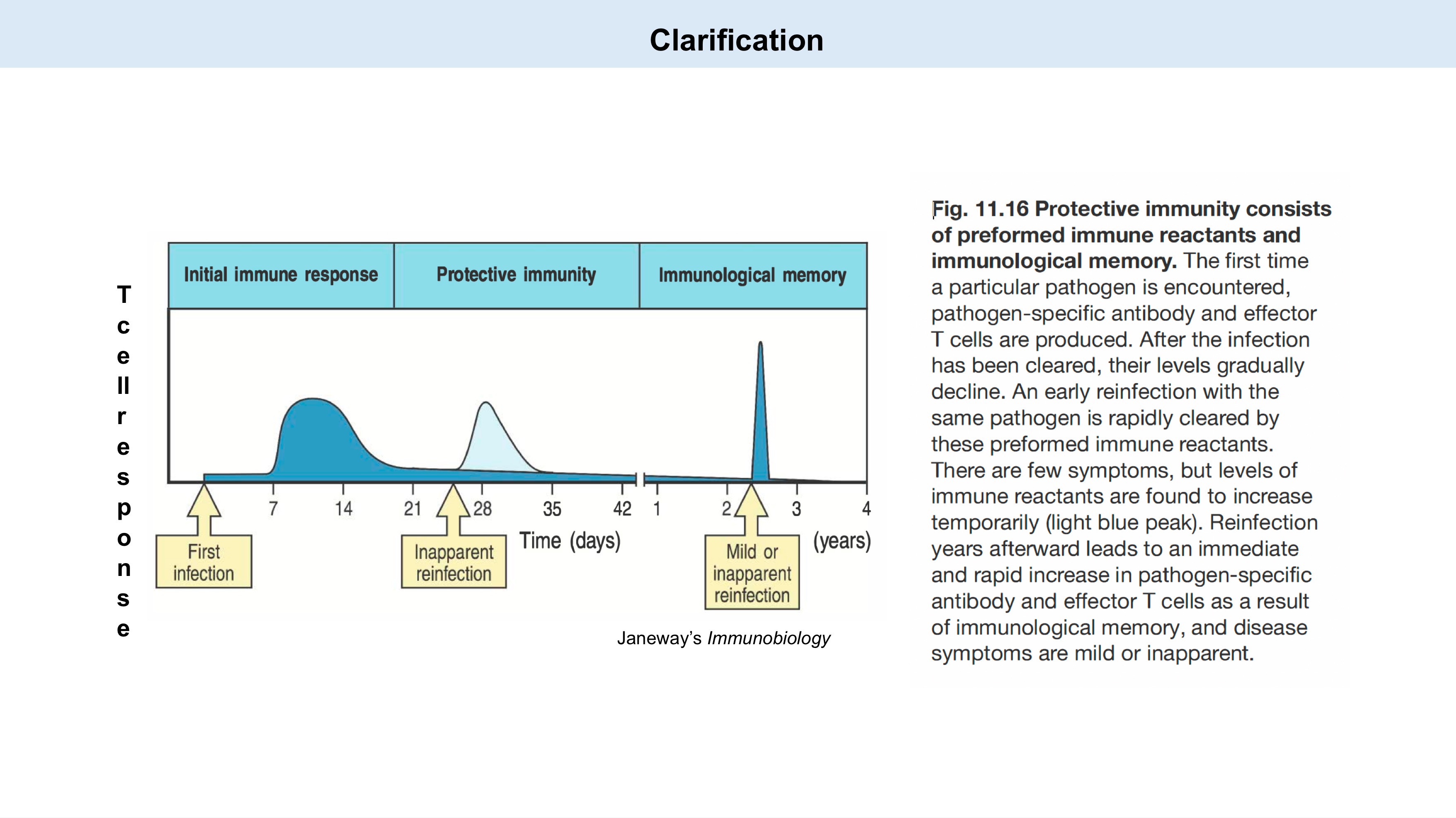
cell-surface
fluorescent
naïve T cells ; effector memory
cytokines
Flow cytometry allows for the identification of immune cell populations, subsets, and functions by detecting _ proteins and is used in the clinic as well as in research.
It uses fluidics, optics (lasers and filters), and electronics to detect _ signals on cells.
Can help to identify CD4+ helper T cells, CD8+ cytotoxic T cells, and Tregs as well, & can aid in distinguishing between _ (CD62L+, CD44-) and _ (CD62L-, CD44+) cells.
Intracellular staining allows for the detection of _ (IFNγ, TNF, IL-2) to assess T cell function rather than just presence.
CD4+ T helper
_ cells coordinate the immune response
They recognize antigens via MHCII on specialized cells, then secrete cytokines to activate other immune cells
(e.g. B cells, macrophages, DCs).
CD8+ T cytotoxic
_ cells directly destroy threats.
They patrol the body, recognize antigens via MHCI, and kill infected cells directly, typically through apoptosis.
all nucleated cells VS professional APCs
endogenous (infection, cancer) VS exogenous
thymus (DP —> SP, thymic selection)
What cells express MHCI vs MHCII?
What peptides (endogenous/exogenous) are presented on MHCI vs MHCII?
When does a CD4+ or CD8+ T cell acquire these co-receptors?
naïve CD8+ T cells ; lymph node
MHC I
CD28-CD80/CD86
IL-2
CTL Activation:
CD8+ T cell effectors are generated from _ in the _
Signal 1 — TCR binds peptide presented by APC on _
Signal 2 — Costimulatory signal transmitted by _ interaction btwn T cell and APC
Signal 3 — Provided by _, inducing prolif. and differentiation into CTL form
periphery
MHC I
granules (perforin/granzymes) ; apoptosis
Following activation, CTLs go to _ to scan cells to see if healthy or diseased.
Infected cells present foreign peptides on their surface using _ molecules
When a CTL’s TCR binds to this peptide-MHC I complex, it activates T cell to release _ that force target cell to undergo _
TCR ; tight junction ; lytic
Fas-FasL
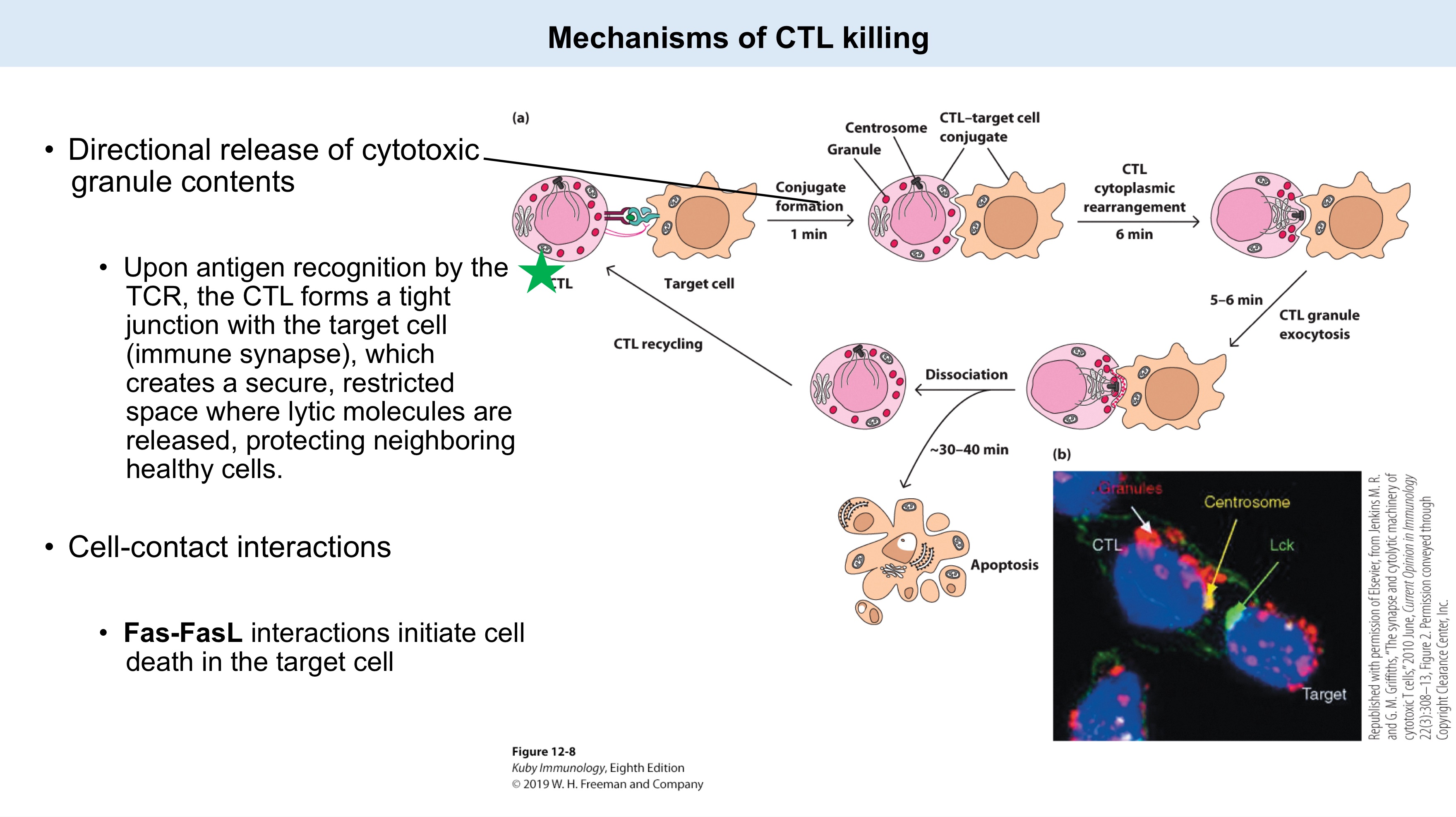
Mechanisms of CTL killing:
Directional release of cytotoxic granule contents
Upon antigen recognition by the _, the CTL forms a _ with the target cell (immune synapse), which creates a secure, restricted space where _ molecules are released, protecting healthy neighboring cells.
Cell-contact interactions
_ interactions initiate cell death in target cell
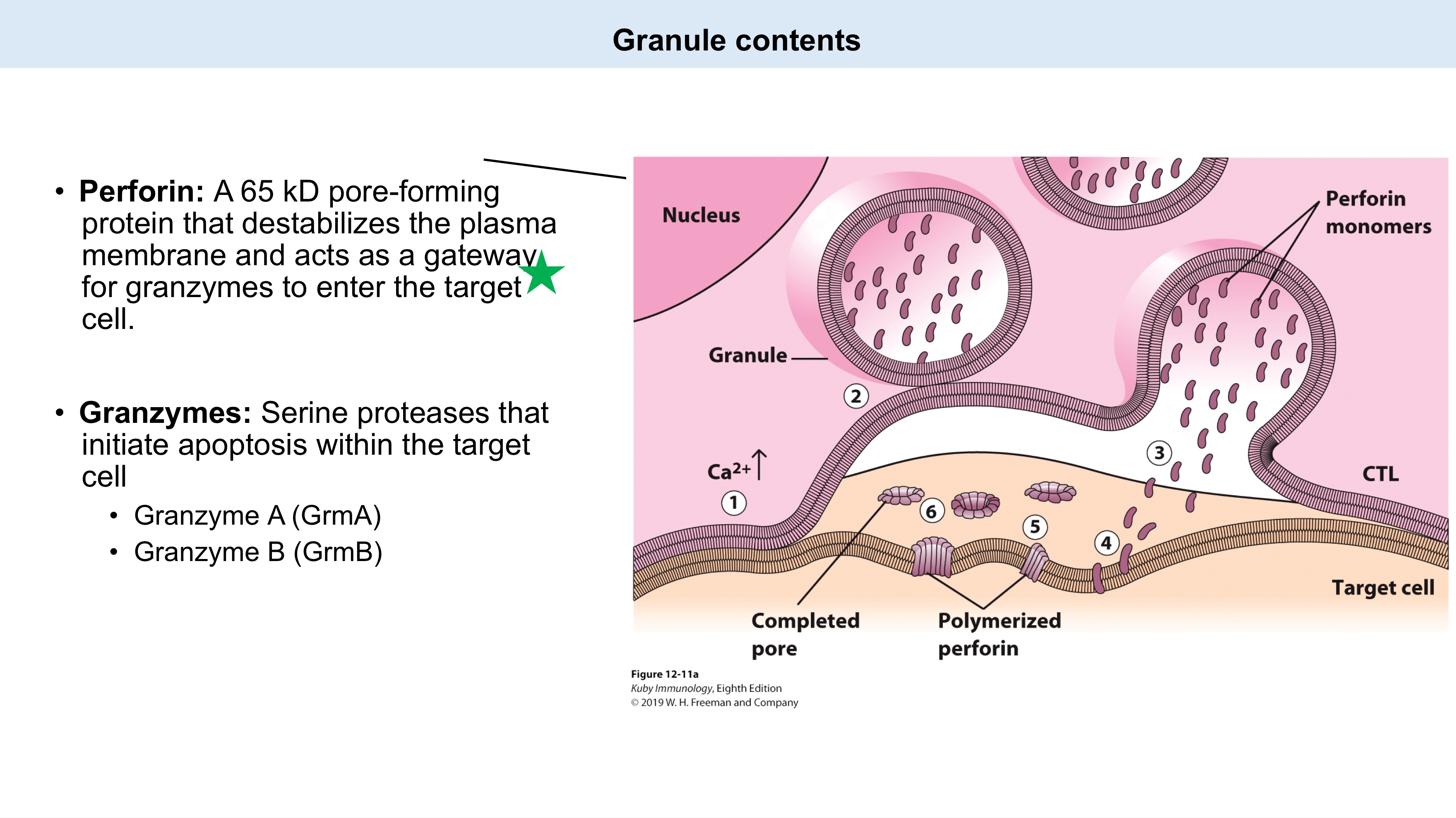
Perforin
Granzymes ; Serine
Granule contents of CTL killing
_: A 65 kD pore-forming protein that destabilizes the plasma membrane and acts as a gateway for granzymes to enter the target cell.
_: _ proteases that initiate apoptosis within the target cell
GrmA
GrmB
Caspase-3
DNA fragmentation ; cytoskeletal breakdown
mitochondrial
phagocytes
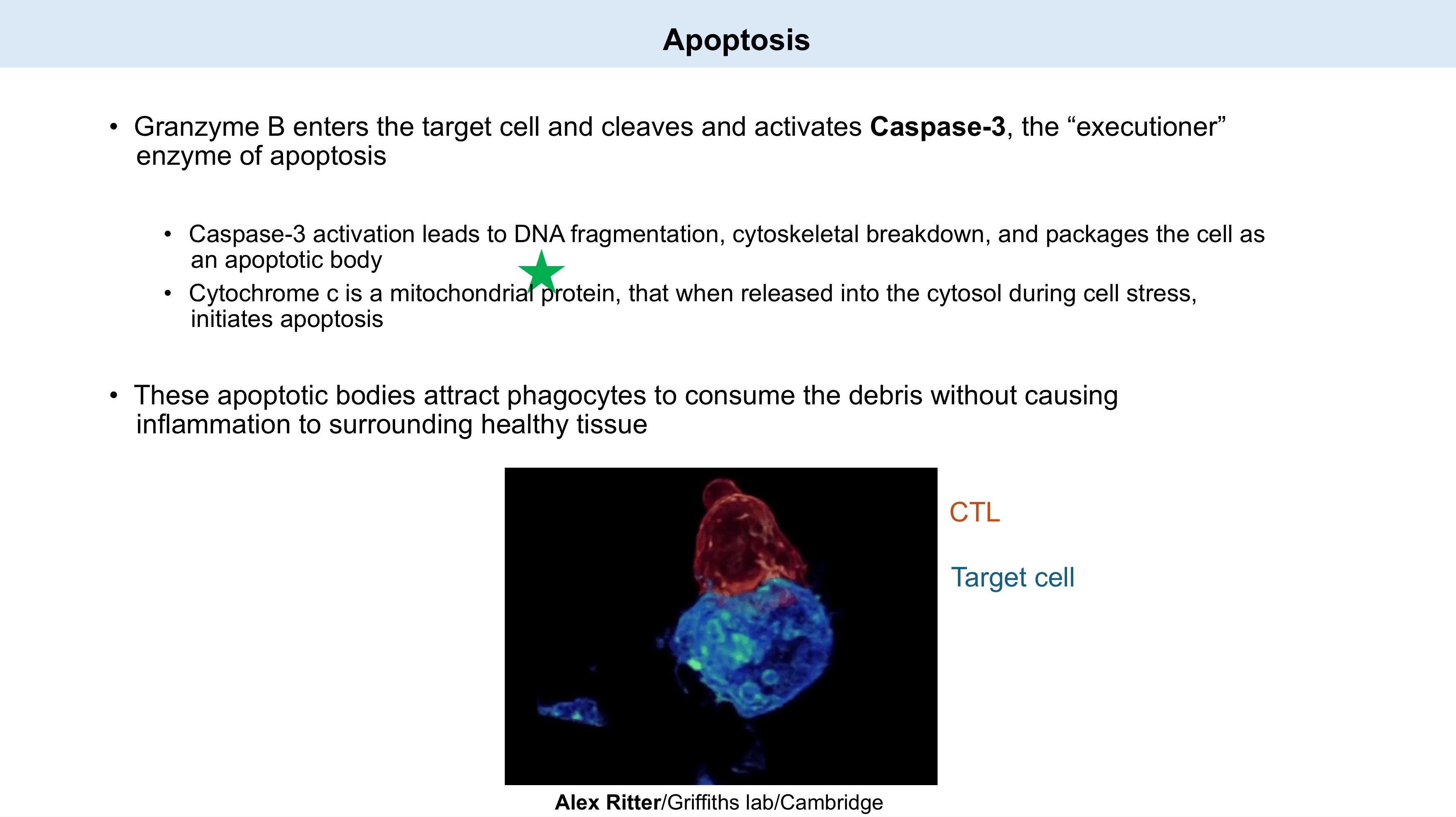
Apoptosis by GrmB:
Granzyme B enters the target cell and cleaves and activates _, the "executioner" enzyme of apoptosis.
Activation of this leads to _, _, and packages the cell as an apoptotic body.
Cytochrome c is a _ protein, that when released into the cytosol during cell stress, initiates apoptosis.
These apoptotic bodies attract _ to consume the debris without causing inflammation to the surrounding healthy tissue.
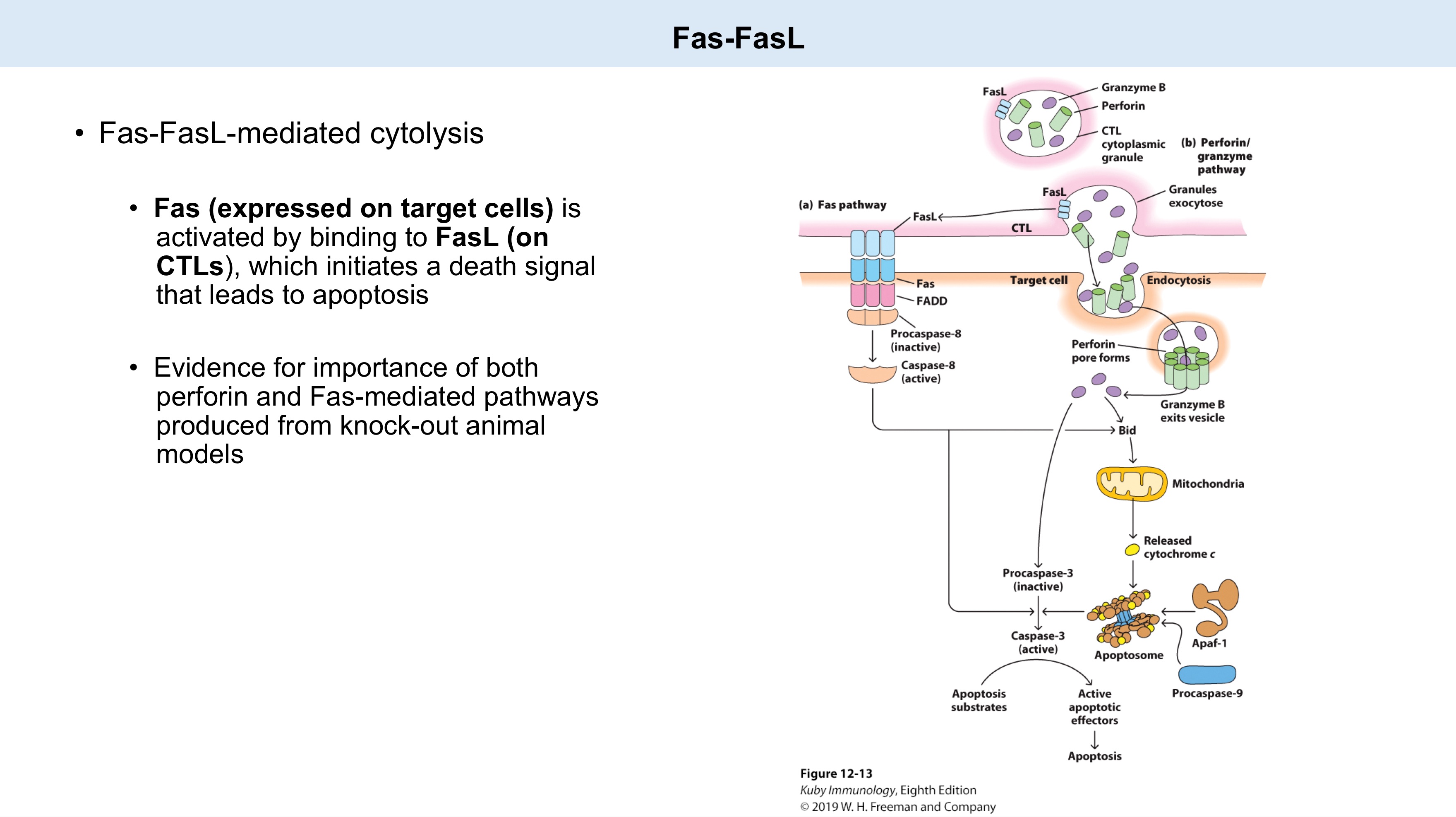
target ; CTLs
perforin
Fas-FasL-mediated cytolysis
Fas (expressed on _ cells) is activated by binding to FasL (on _), which initiates a death signal that leads to apoptosis.
Evidence for the importance of both _ and Fas-mediated pathways produced from knock-out animal models.
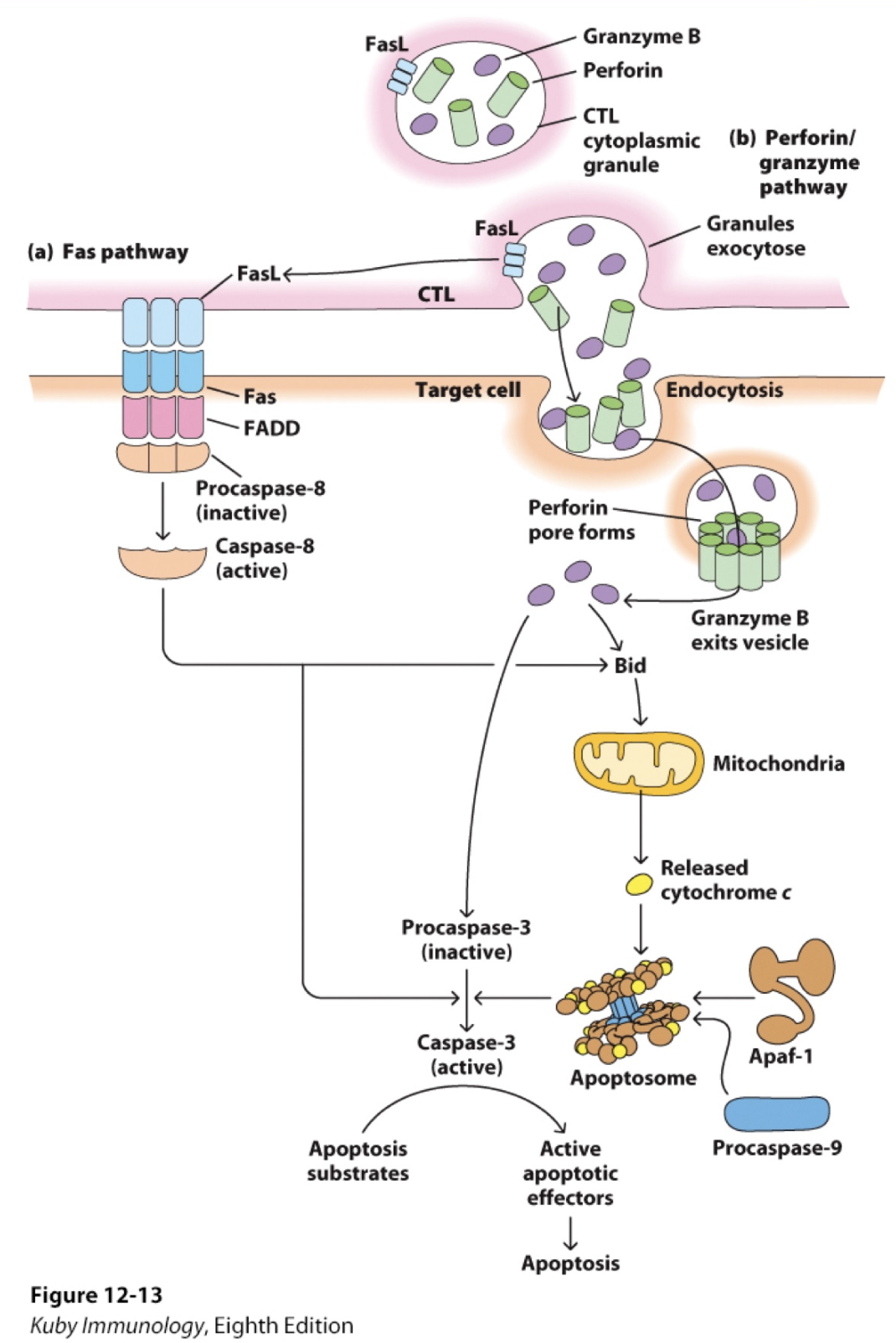
cross-presentation ; DCs ; MHC I
co-stimulation
Conundrum...
To become activated, a naive T cell needs to see its specific antigen presented by a professional APC (usually a DC).
If a tumor or virus does not directly infect a DC, how does a CD8+ T cell become activated to fight it?
Through _, where _ engulf dead/dying tumor or infected cells, process the foreign antigens, and present them on _ molecules to T cells.
DCs also receive necessary _, upregulating activation markers (CD80/86) to fully prime the T cells in lymph nodes.
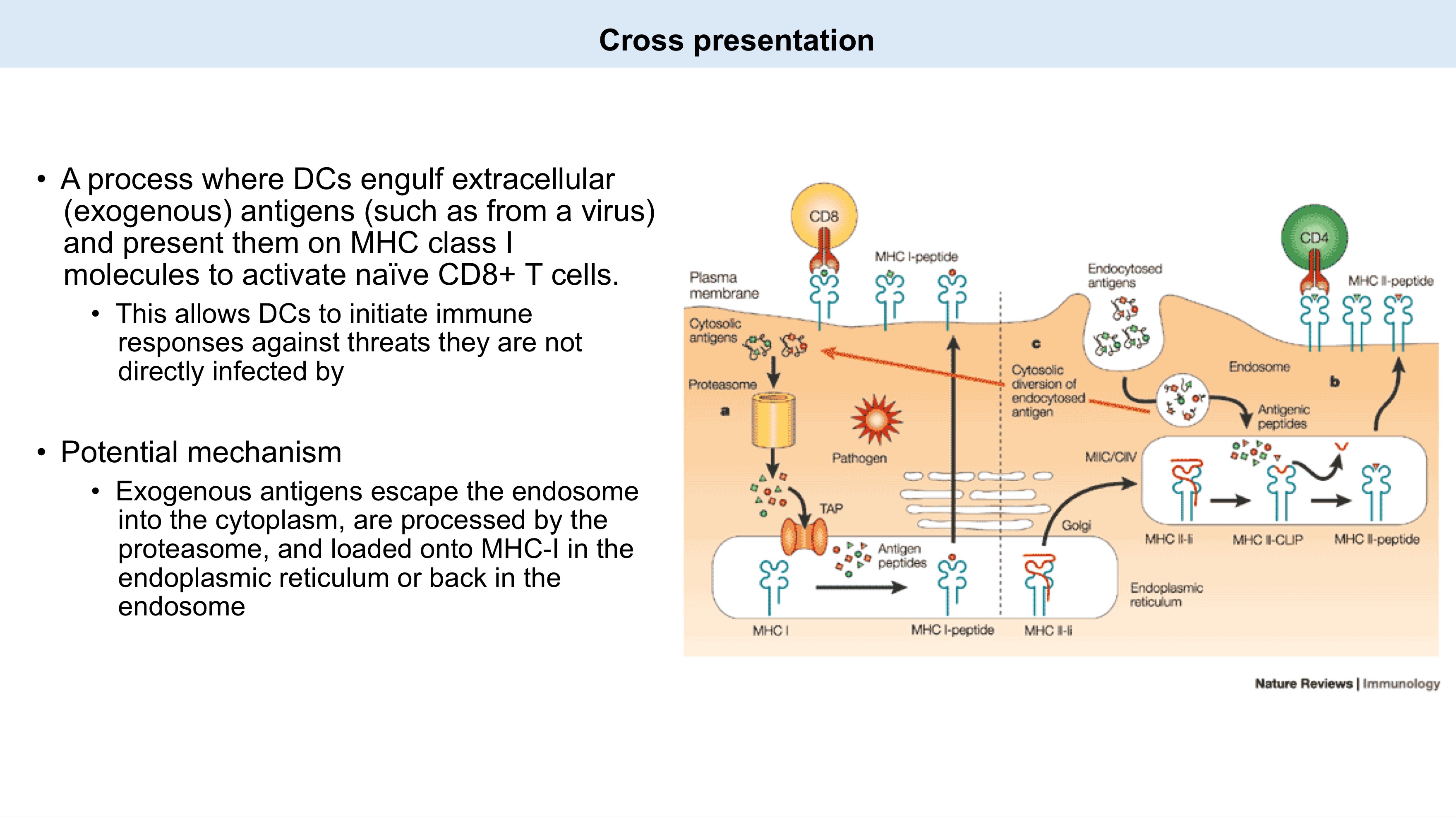
Periphery
(Found patrolling the entire body to eliminate infected or abnormal cells, with their location shifting based on their activation state)
Where are CTLs/activated CD8+ T found?
CD8+ T ; effector
fail to respond
IL-2
killing
PD-1
CTLA-4
CTL exhaustion:
A state of dysfunction in _ cells caused by chronic antigen exposure (e.g., cancer or persistent infections), characterized by the progressive loss of _ functions.
CTLs _ despite seeing necessary activation signals.
Loss of effector functions
Decreased _ production
Decreased _
Inhibitory receptor expression
_
_
Allows for viral infections and tumors to persist.
Reversing exhaustion is promising therapeutic potential in cancer.
Checkpoint immunotherapy (e.g. anti-PD-1, anti-CTLA-4)
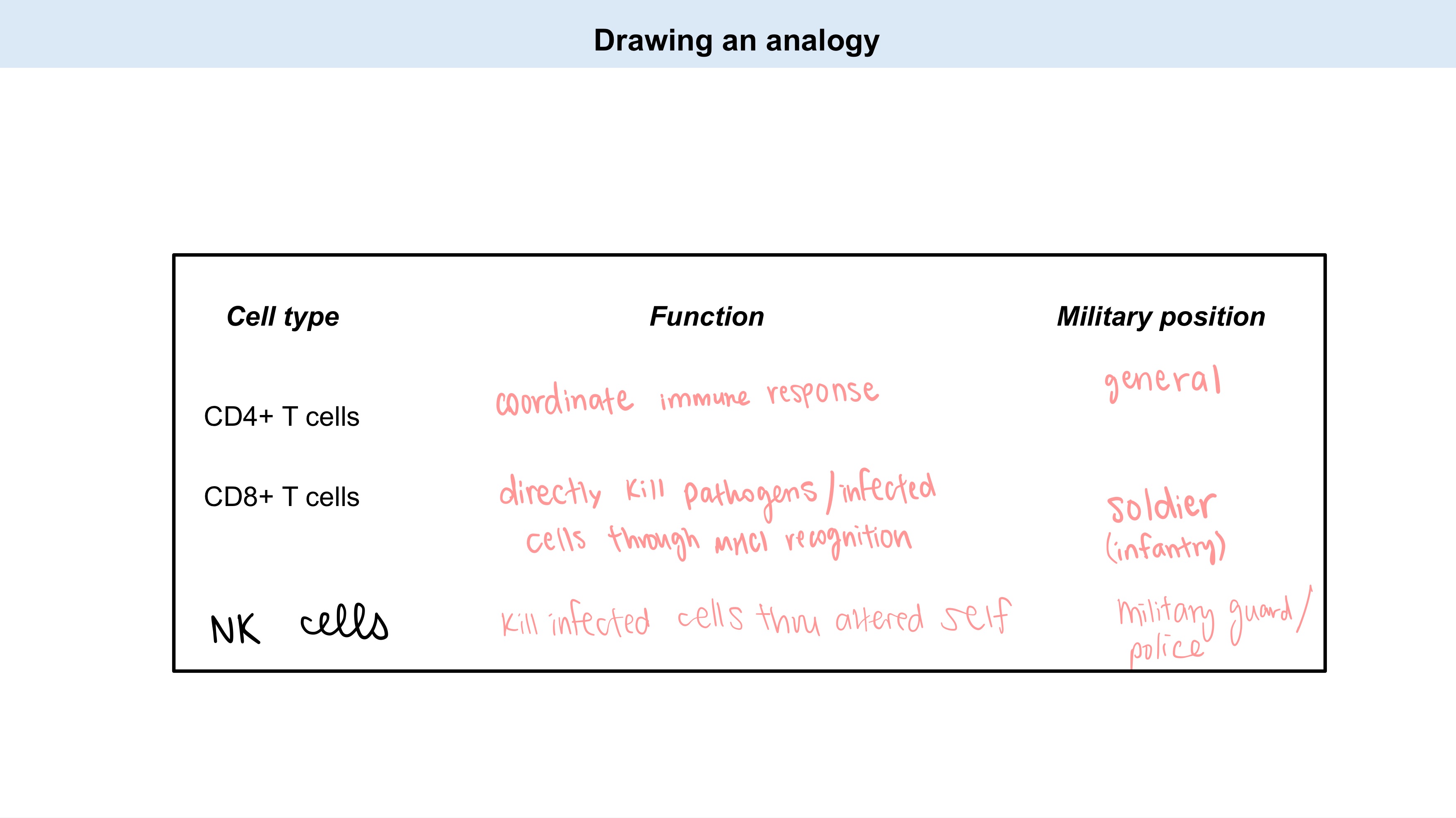
coordinate immune response
directly kill pathogens /infected cells through MHC I recognition
kill infected cells through altered self
Cell type | Function | Military Position |
CD4+ T cells | general | |
CD8+ T cells | _ | soldier |
NK cells | _ | military guard |
Fill in FUNCTION!
NO!
(NK cells dom’t need MHC - can kill without)
Is downregulating MHC-I a good immune evasion strategy for pathogens?
absence of MHC class I
common lymphoid progenitors (CLPs)
TCR
Thymus
Inhibitory
Activating
earlier
5-10
Ag
Natural killer (NK) cells
Natural killer cells recognize and kill infected cells and tumor cells by their _
NK cells are derived from _ in the bone marrow
Are considered lymphoid cells but NOT T cells (as they don't express _)
_ not required for NK development
Do not undergo receptor gene rearrangements
Defining trait is the expression of a set of activating and inhibiting NK receptors
These receptors are used to determine whether to kill a target or not
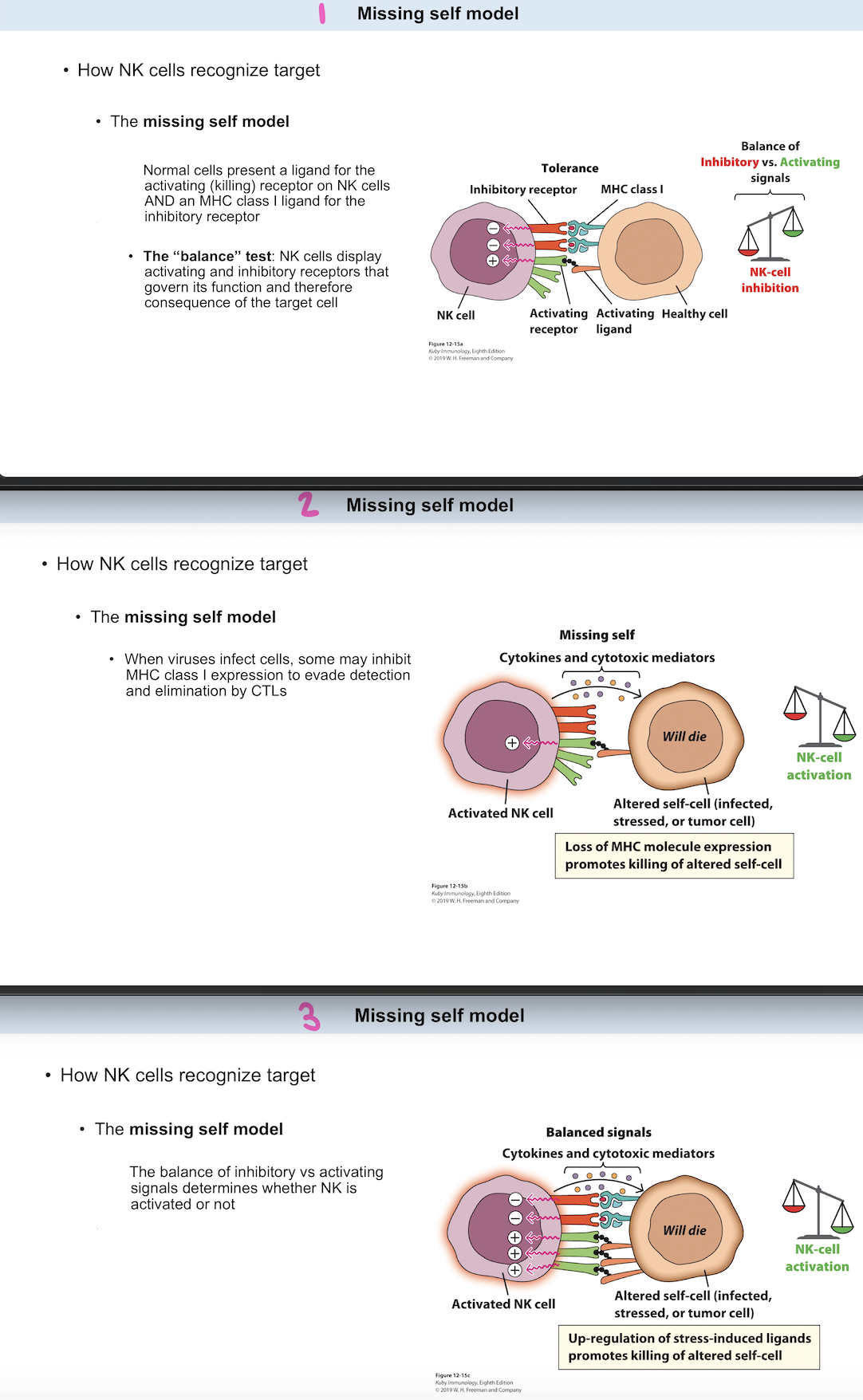
_ receptors: recognize self-MHC-I, which provides an "off' signal
_ receptors: recognize ligands induced by stress, infection, or DNA damage
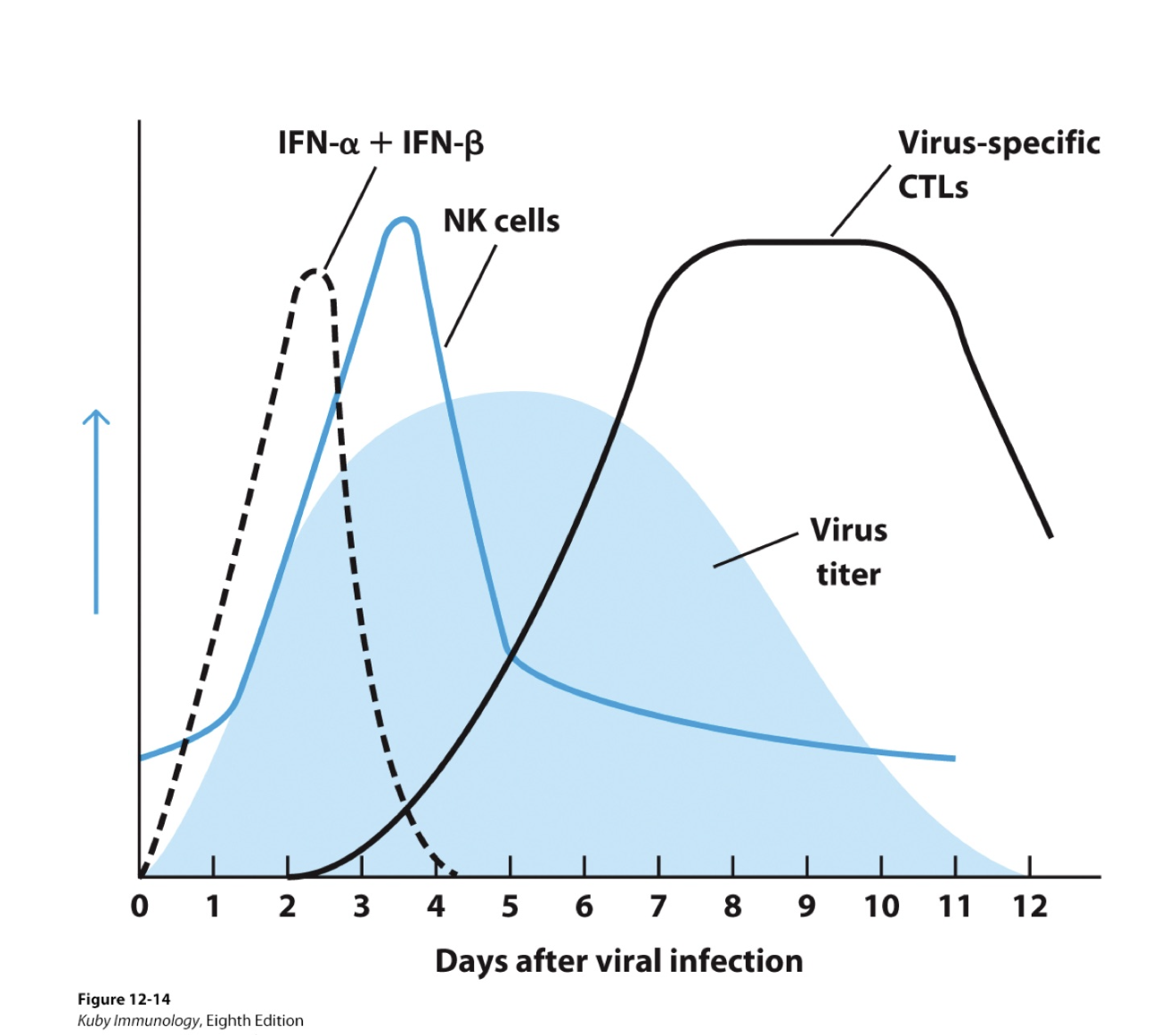
NK cells proliferate _ in infection than CTLs
NK cells make up _% of circulating lymphocytes
60-80% T cells
10-20% B cells
Virus titer
NK cells lack specific _ receptors
NK cells recognize and kill pathogen-infected cells and abnormal tumor cells
The missing self model
The "balance" test
How NK cells recognize target:
_
Normal cells present a ligand for the activating (killing) receptor on NK cells AND an MHC class I ligand for the inhibitory receptor.
When viruses infect cells, some may inhibit MHC class I expression to evade detection and elimination by CTLs.
The balance of inhibitory vs activating signals determines whether NK is activated or not.
_
NK cells display activating and inhibitory receptors that govern its function and therefore consequence of the target cell
bone marrow
functional ("licensed") ; MHC-I
exhibit restraint
NK cell maturation
NK cells education and licensing occur in the _ during maturation.
Immature NK cells become _ by interacting with _, allowing them to distinguish self from non-self
This gives the "license" only to those NK cells that can _ when encountering a healthy, normal cell.
apoptosis
CTLs
perforins/granzymes
memory
Cell-mediated effector responses:
NK cells induce _ of their targets
Once activating signal molecules are engaged, NK cells use mechanisms very similar to _ to induce target cell death
Release of _ at the junction between two cells
Some evidence suggests NK cells can generate a _ response
Altered function: Increased proliferation, greater degranulation for a duration of time
Detection by patrolling APCs
APCs process and present antigen to naïve T cells
organize
kill infected cells
Fill in blanks!