Understanding Metallic Bonding and Lattice Structure
1/18
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
19 Terms
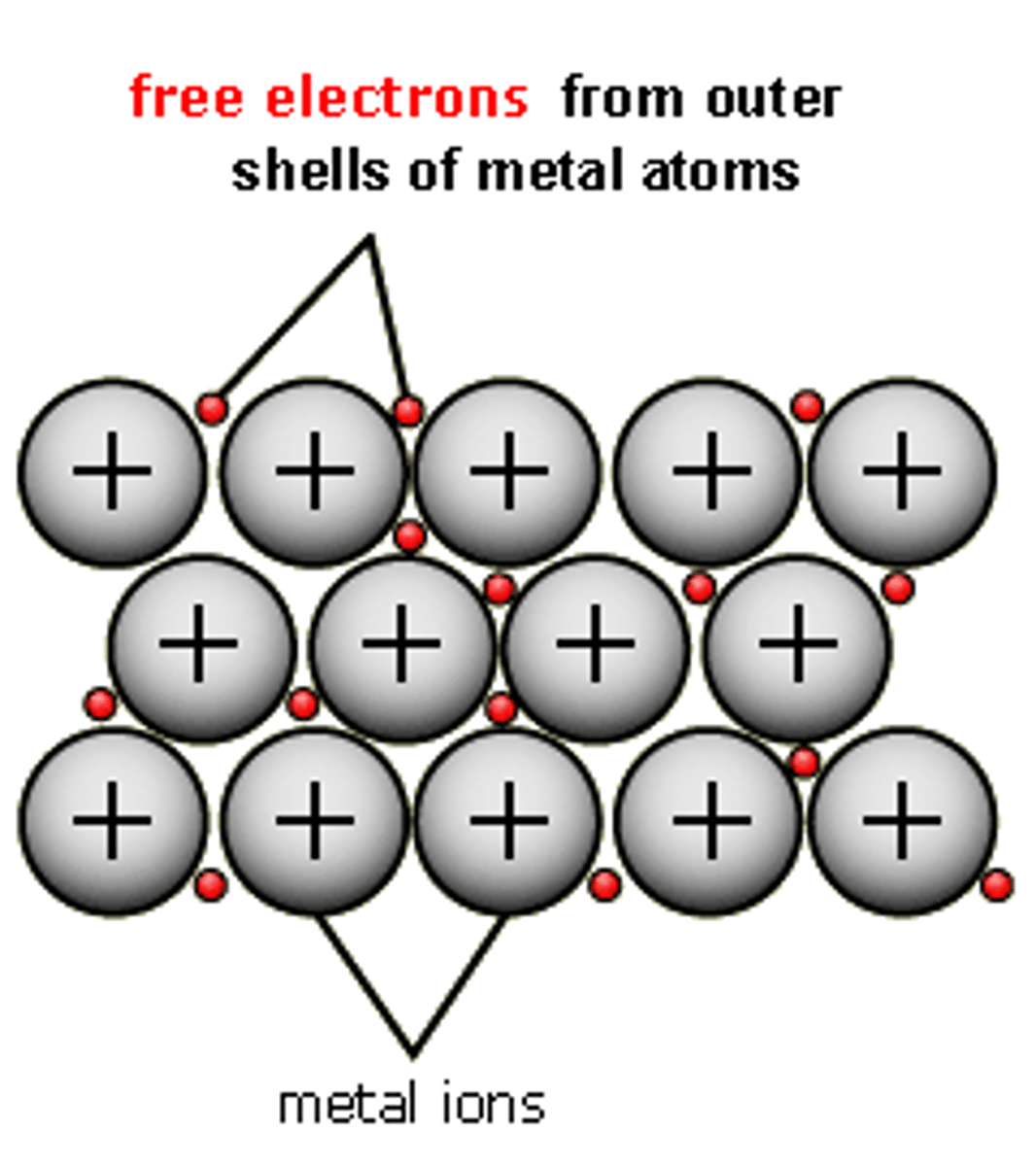
Metal Ions
Positively charged atoms formed by electron loss.
Delocalised Electrons
During metallic bonding, metal atoms lose their outer shell electrons into a delocalised "sea" of free-flowing electrons.
Metallic Lattice
Regular 3D arrangement of metal ions and electrons.
How should a metallic lattice be drawn?
Three down by five across (5x3) with precise number of electrons and ions as well as labels. Charge must also be drawn into the diagram.
Hardness
When a metal is scratched, the layers of the lattice are parted. The resistance of the metal to scratching is how resistant the the electrons and ions are to parting based on how attractive the electrons and ions are to each other.
High melting point
A relatively large amount of energy is required to disrupt the metallic bonds holding the lattice together.
High boiling point
A large amount of energy is required to overcome the metallic bonds holding the lattice together.
Mohs Scale
A scale that ranks minerals based on their ability to scratch one another, ranging from talc (1) to diamond (10).
Ductility/malleability
The ability of a material to be stretched into a wire, deformed without breaking, or beaten into a sheet. The reason why a lattice is not brittle is delocalised electrons would move and cushion the ions, compensating for the external force. Otherwise, the ions would touch each other and repel, causing cracks, a feature of a brittle material.
Elasticity
The ability of a material to return to its original shape after being deformed by an external force.
Compressive Strength
The capacity of a material to withstand axial loads that tend to compress it.
Tensile Strength
The maximum amount of tensile (stretching) stress that a material can withstand before failure.
Ionic Size
Smaller ions pack closer, enhancing bond strength.
Ionic Charge
Higher charge increases attractive force in bonds.
How is a lattice formed?
Metals lose their electrons to form ions. When more than a few metal atoms come together, they release their outer shell electrons into a delocalised sea of electrons.
The electrostatic attraction between metal ions to the delocalised electrons holds the ions and electrons in a 3D regular arrangement of particles called a lattice.
Why are metals good conductors of electricity?
For a substance to conduct electricity, a substance needs free-moving charged particles. The delocalised electrons are both free moving and charged which allows metals to conduct electricity.
How does heat energy affect a metal lattice?
Heat energy applied to a metal lattice causes the ions and delocalised electrons to gain kinetic energy, which spreads exponentially to the rest of the lattice. The rate at which this kinetic energy is transferred is dependant on a range of factors, including but are not limited to, density, hardness, and electrostatic attraction.
Why are metals not brittle?
The reason why a lattice is not brittle is delocalised electrons would move and cushion the ions, compensating for the external force. Otherwise, the ions would touch each other and repel, causing cracks, a feature of a brittle material.
What trend does the reactivity of metals with acid follow?
It is the same trend as with oxygen and water.