IB Chemistry HL review
1/105
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
106 Terms
Relative atomic mass
weighted mass relative to one twelfth of the mass of a carbon-12
Relative Molecular Mass
mass of a molecule calculated through the summation of all the relative atomic masses of its constituent atoms.
Mass number
number of protons and neutrons added together
atomic number
equal to the number of protons in the nucleus of the atom
Isotopes of an element
Isotopes of atoms of the same element contain the same number of protons but differ in neutrons
first ionization energy
minimum energy required to remove a mole of electrons from a mole of gaseous atoms to form a mole of cations in the gaseous state.
X -> X+ and e-
Electronegativity
is the ability of an atom to attract a pair of electrons.
Ligand
neutral molecule or negative ion that donates a pair of electrons to form a coordination complex.
(lewis bases)
Exothermic reaction
releases heat from the formation of energetically more stable products than the reactants. (negative ∆H)
Endothermic reaction
absorbs heat from the surroundings as a result of forming products energetically less stable than the reactants.
Standard Enthalpy of a reaction (∆H)
Heat transferred during a reaction using molar quantites carried out under STP. (1 atm, 298K, and all substances pure in their standard states)
Average bond enthalpy
is the energy required to break a mole of covalent bonds in the reactant, all reactants and products being in the gaseous state.
X--Y -> X(g) + Y(g)
Standard state
the standard state of an element is its most stable state under stp.
Standard enthalpy of formation (∆Hf)
Enthalpy change when one mole of a compound/molecule is formed from its constituent parts.
Standard enthalpy of combustion (∆Hc)
Enthalpy change when one mole of a compound is combusted under STP.
Electron affinity
enthalpy change occuring when a gaseous atom gains one electron to form a negative ion.
*First electron affinity might be endothermic
X(g) + e- > X- (g)
Rate of reaction
is the increase in concentration of one of the products per unit time.
Activation energy
minimum amount of energy required for a chemical reaction to take place
Bronsted Lowry acid-base
Proton donator - Acid
Proton acceptor - Base
Lewis acid-base
substance that can accept a pair of electrons
Substance that can donate a pair of electrons
Oxidation
loss of electrons
Reduction
gain of electrons
Oxidizing/Reducing agent
substance that easily reduces - aiding in oxidation
substance that easily oxidizes - aiding in reduction
Standard Electrode potential
electrode potential relative to a standard hydrogen half-cell, measured under STP ( 1atm, 298K, all solutions at 1M)
Iodine number
is the mass of iodine in grams required to react with 100g of a substance.
Continuous spectrum
shows an unbroken sequence of frequencies or wavelengths, such as the spectrum of visible light.
line spectrum
is a spectrum that only contains certain discrete frequencies of light.
Precision
all taken measurements are close to eachother
A graph with pressure (y) and 1/volume looks like?
straight line because they are inversely proportional
1.0 mol of any gas occupies?
22.4 dm3 at standard conditions
An ideal gas is a gas that does not have?
molecular volume, or inter-particle forces
1 atm equals how many pascals?
1.01325 x 10^5 pascals
The Mass spectrometer steps?
Vaporization, ionization, acceleration, deflection, detection.
continuous vs line spectrum?
continuous contains all the wavelengths/energy, while a line spectrum contains only specific energies/wavelengths.
Lyman Series is?
the ultraviolet series, all transitions end at 1
Balmer series is?
visible light - all transitions end at n=2
Paschen Series
infrared, all transitions end at n=3
Reactions of Alkali metals with water?
alkali metals with water form hydrogen gas and the corresponding alkali (NaOH)
Iodine, Bromine, and Chlorine are what colors?
purple, orangy/brown, and yellow
A complex ion is?
a transition metal, surrounded by ligands
pure covalent bonds have electronegativity values of?
less than .4
ionic bonds have electronegativity values of
greater than 1.7
2 bond pairs 0 lone pairs
linear - bond angle 180
3 bond pairs and 0 lone pairs
trigonal planar - 120
4 bond pairs and 0 lone pairs
tetrahedral 109.5
3 bond pairs and 1 lone pair
trigonal pyramidal 107
2 bond pairs and 2 lone pairs
bent/ v-shaped 104.5
Graphite
Sp2 hybridization - high conductivity (4th electron is found delocalized in the pi bond) - 2 dimensional - very soft (three sheets are weakly attracted to each other by weak van der waal's forces)
Diamond
Sp3 hybridization - no conductivity - very hard - 3 dimensional
Fullerene
Intermediate conductivity - 2 dimensional - soft
if there are 5 bond pairs it is a type of trigonal bipyramidal
classic example (PCl5) - 90 and 120 degree angle
4 bond pairs 1 lone pair
see-saw 90degree and 120
3 bond pairs 2 lone pair
T-Shaped with 90
2 bond pairs 3 lone pairs
linear - 180
6 bond pairs means it is octahedral
classic example (SF6)
6 and zero
octahedral 90
5 bond pairs and 1 lone pair
square pyramidal 90 90
4 bond pairs and 2 lone pairs
square planar 90
Hydrogen bonding occurs only in
Nitrogen, oxygen, or fluroine
sp3 hybridization
all single bonds, tetrahedral, 109.5
sp2
double bond, planar triangle 120
sp
triple bonds or 2 double bonds - linear - 180
a single bond has how many sigma/pi bonds?
1 and zero
a double bond has how many sigma/pi bonds?
1 sigma, 1 pi bond
a triple bond has how many sigma/pi bonds?
1 sigma 2 pi bonds
Resonance structures are?
just structures with the double bond in all locations
Delocalization occurs when?
there is a presence of unhybridized p-orbitals on all of the atoms in the structures.
1 liter of water weighs?
1kg
Arrhenius equation k, A, equals?
rate constant, and prefactor
slope equals?
-Ea/R
where R = universal gas constant
Which enthalpy formula to use?
If given formation enthalpy, use products - reactants.
If given combustion enthalpy or bond enthalpies, use reactants - products. This is only to find the Enthalpy of the reaction. Do not switch signs for any of them.
Entropy signs are?
positive when entropy increases, negative when entropy decreases. (don't get confused)
Entropy can increase by?
Mising, increase in the number of particles, change of state, creation of a gas.
Hydrogen emission spectrum?
series of lines, electrons transitioning energy levels then falling back down. First transition causes UV light, second visible light, and third infrared light.
Differences between an electrolytic and voltaic cell?
non-spontaneous - spontaneous, anode/cathode opposite, requires energy - creates energy, etcs.
When asked to label a voltaic cell remember to? (ionic bridge)
show the movement of both cations and anions
Why are alcohols worse than alkanes as far as fuel is concerned?
Alcohols are partially oxidized, and have stronger intermolecular forces of attraction to hold them together. Making it less favorable to combust than alkanes.
when determining lattice enthalpy strength?
Look at the oxidation states of each atom in each molecule. E.G. MgO or NaF? Which has the greatest enthalpy value?
dynamic equilibrium exists when?
closed system and there are no changes in properties such as concentration.
Kc is only affected by?
temperature - not pressure or anything else.
Any "define enthalpy change" is a definition question haha
Enthalpy of formation: enthalpy change when one mole of a product is formed from its constituent atoms under standard conditions - all under STP.
Protein hydrolysis? reagents, conditions, bond broken?
Reagent- Acid, Conditions - High temperature, Bond broken - peptide bond.
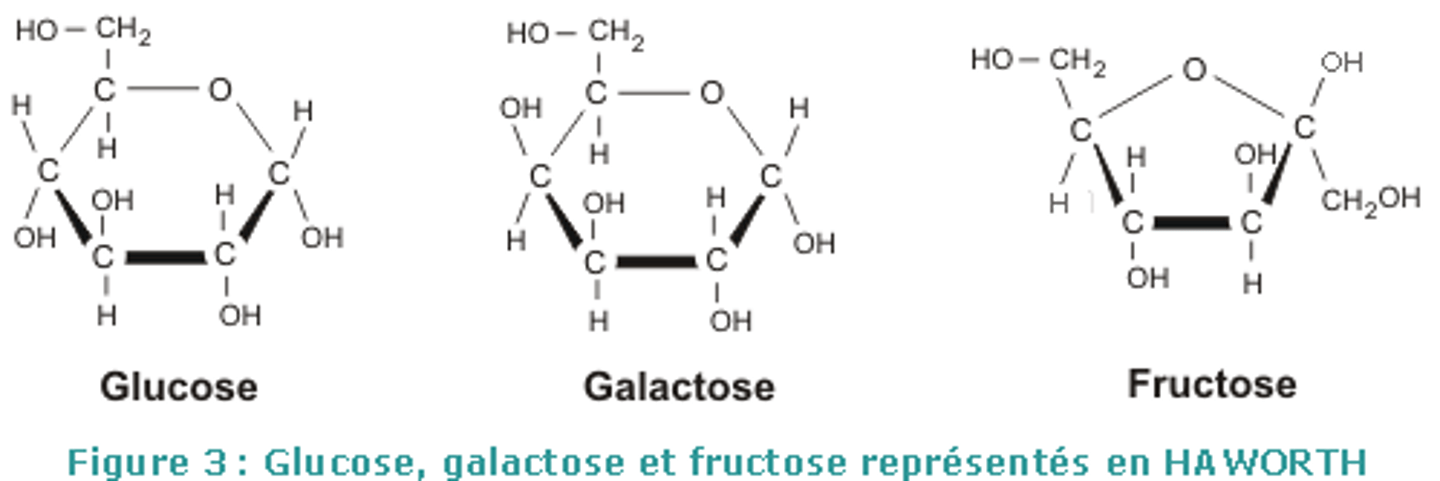
Differences between glucose, galactose, and fructose?
Isolectric point is?
the pH point at which the protein experiences no net charges. (Amine group produces a more positive charge, and therefore more basic pH while the opposite is true for the carboxyl terminal)
Amylose and Amylopectin differences?
both starches but differences in solubility and chain structure.
Differences between alpha and beta pleated proteins?
These secondary structures differ in their hydrogen bonding. Alpha sheets have hydrogen bonding within the protein chain, while hydrogen bonding occurs between other protein chains in beta pleated sheets.
fats and oils differences? similarities?
solid, liquid
both consist of fatty acids and glycerol
galactose is trans, glucose is cis
...
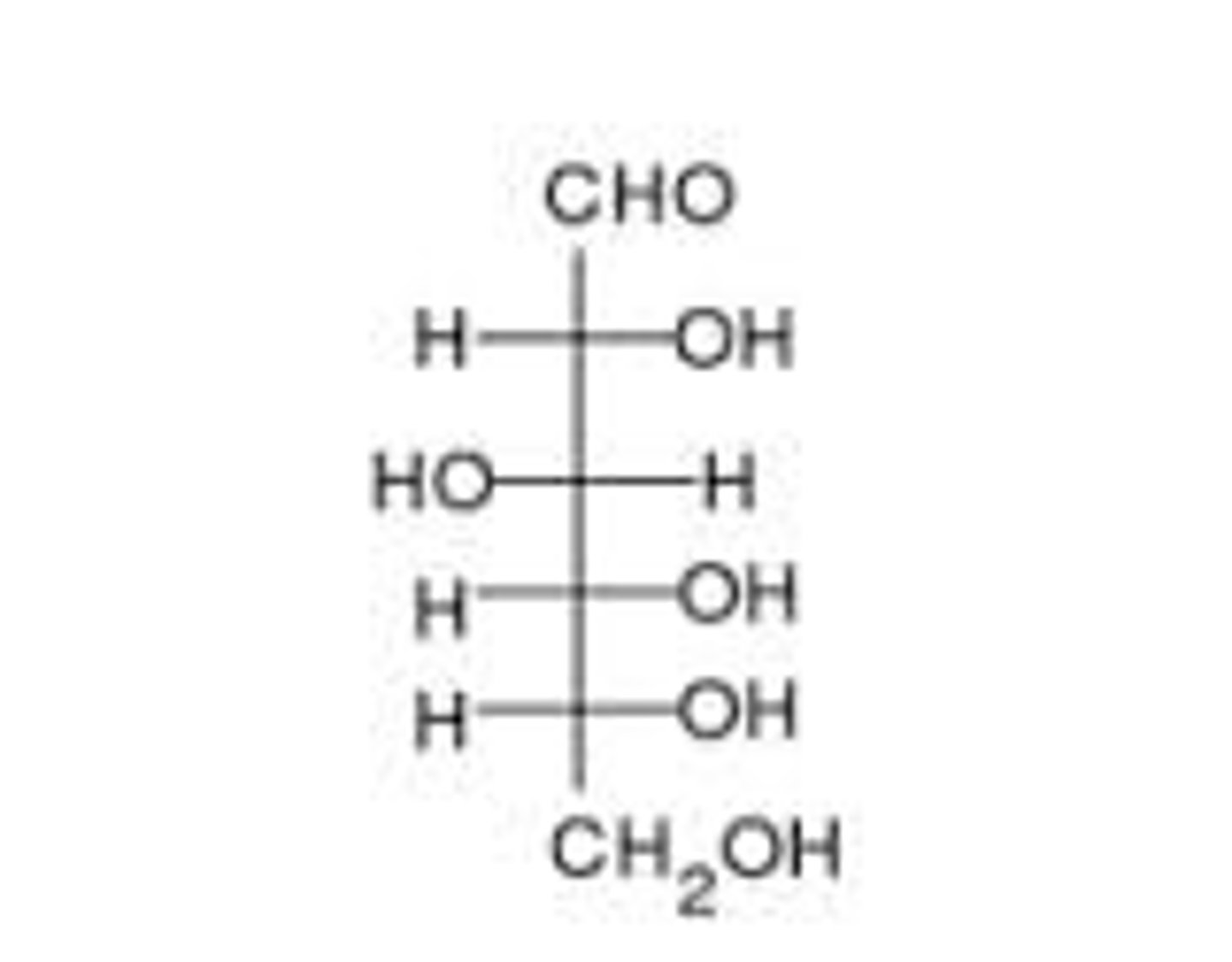
Straight chain structure of glucose?
Two naturally occuring fatty acids?
stearic acid, oleic acid
Two essential fatty acids?
omega 3, omega 6 fatty acids, and linoleic acid
effects of non-competitive inhibitor on km and Vmax?
Km remains unchanged because the active site isn't blocked, but the Vmax decreases because it is achieved in less time.
effects of competitive inhibitor on Km and Vmax?
Km increases - more substrate needed to reach Vmax. Vmax remains the same but it takes longer to get there due to the competitive inhibitor blocking the active sites and causing the reaction to not occur as quickly.
Beta-lactam ring importance?
it is the 90 degree bond angle structure consisting of 3 carbons and 1 nitrogen responsible for reacting with bacteria and causing their cell walls to burst.
Chirality
refers to asymmetry about one atom in optical isomers
overprescription of antibacterials?
kills good bacteria, develops resistence
How to make molecules more soluble in water? (easier to administer)
Make the molecule ionic. If the dominating functional group is a carboxylic acid - react with a base to make the salt. If the dominating group is an amine, react with HCl to make the
Combinatorial Chemistry
various amino acids are attached to resin beads
these products are mixed and split resulting in all combinations of dipeptides
these combinations of dipeptides are then again mixed and split, resulting in many thousands of combinations of peptides.
Enzyme and inorganic catalyst difference?
enzymes are easily denatured by high temperatures, inorganic compounds are not. Enzymes are specific, inorganic compounds are not. etc.
Vitamin A deficiency?
blindness