Ch 21
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
enols
ketone and enols are tautomers, position of equilibrium favors ketone
enolates
ambident nucleophiles because they possess two nucleophilic sites
more reactive than enols
acid-base equilibrium favors weaker acid (higher pKa)
keto-enol tautomerization
alpha carbon can act as a carbon nucleophile under acidic or basic conditions
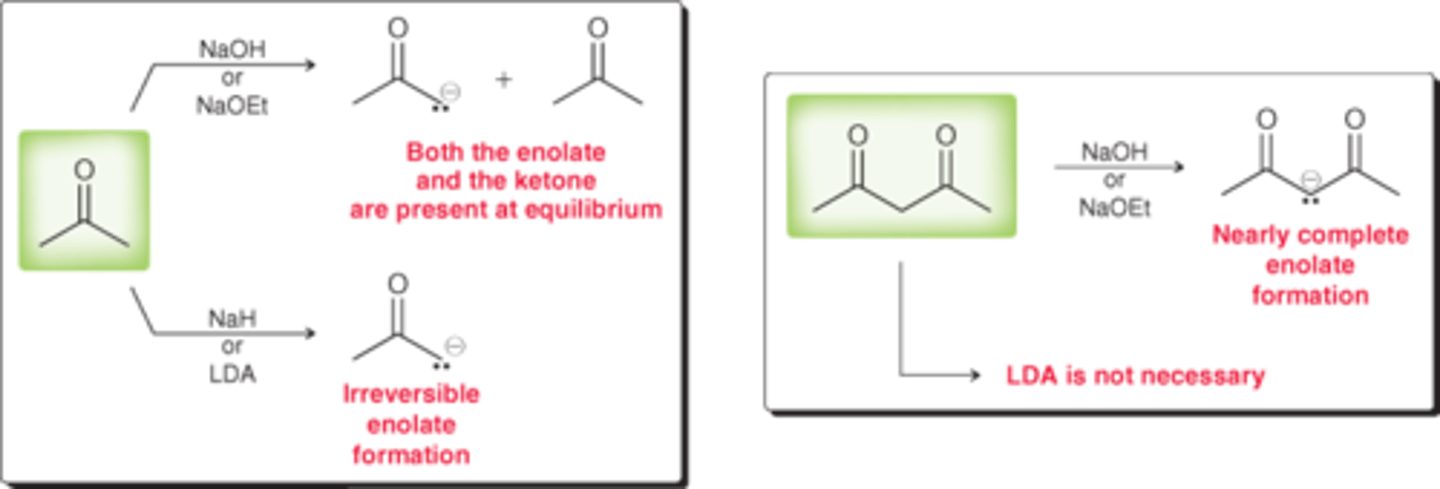
choosing a base for enolate formation
alkoxide ion; equilibrium where alkoxide ion and enolate ion are both present
sodium hydride; completely converts ketone into enolate
LDA; irreversible enolate formation
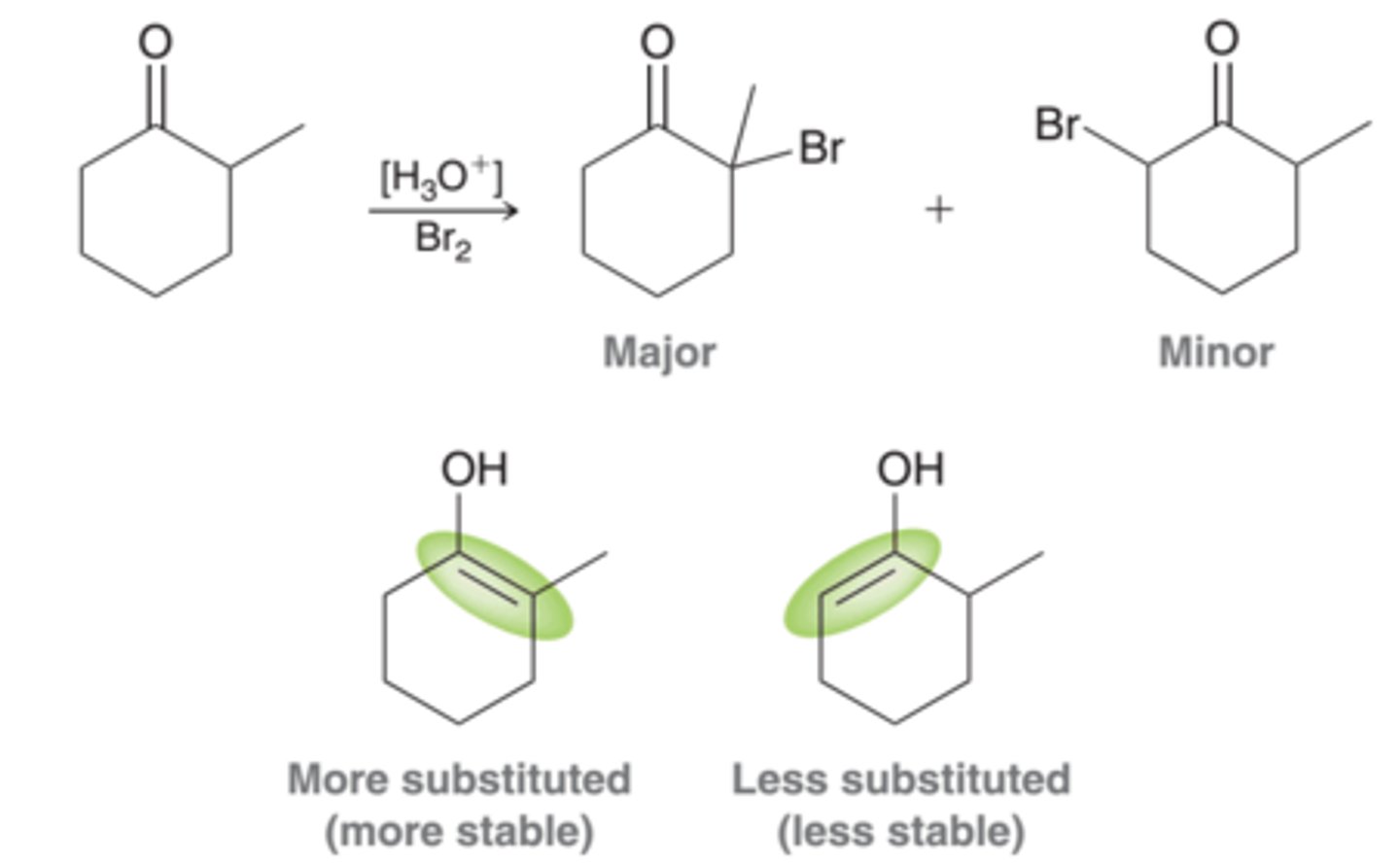
alpha halogenation in acidic conditions
ketones and aldehydes undergo halogenation at alpha position, occurs at more substituted position
can undergo elimination with variety of bases like pyridine, Li2CO3, or KTbuOk
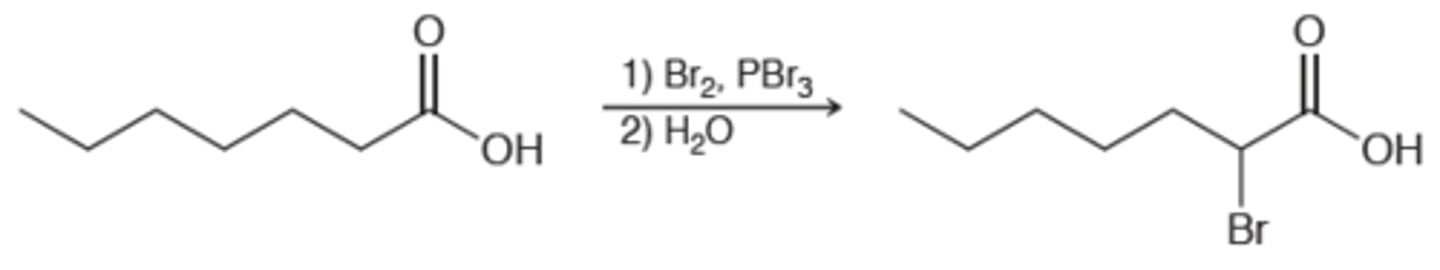
Hell-Volhard-Zelinksy reaction
alpha bromination of carboxylic acids with PBr3
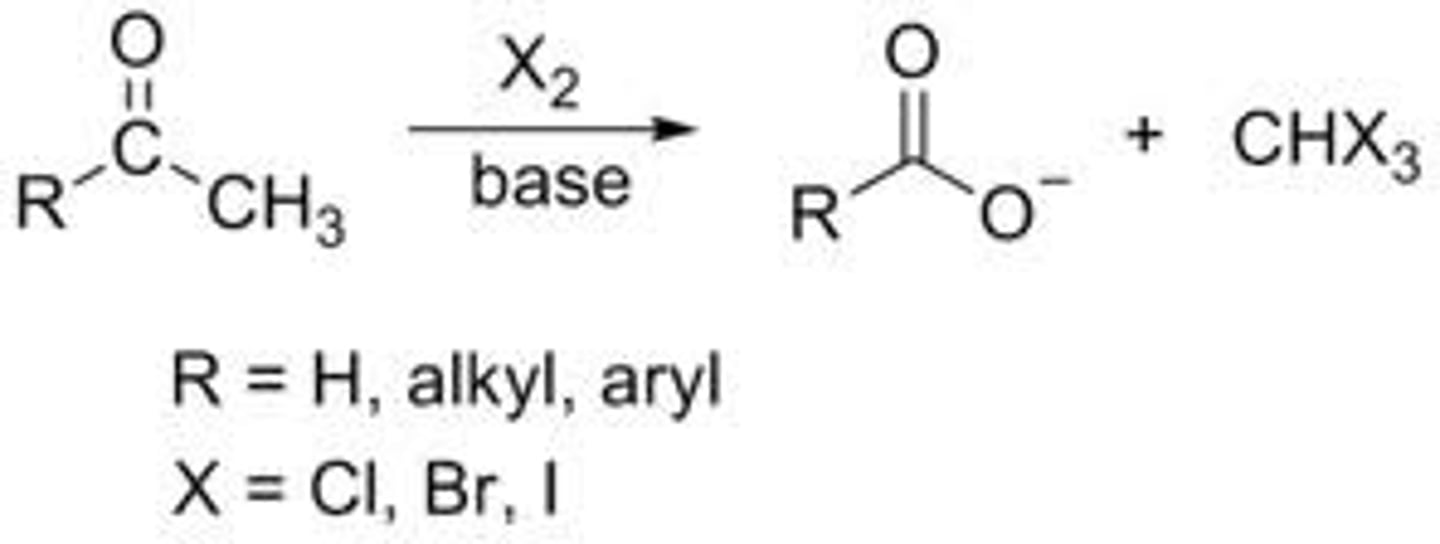
haloform reaction
alpha halogenation in basic conditions of ketones
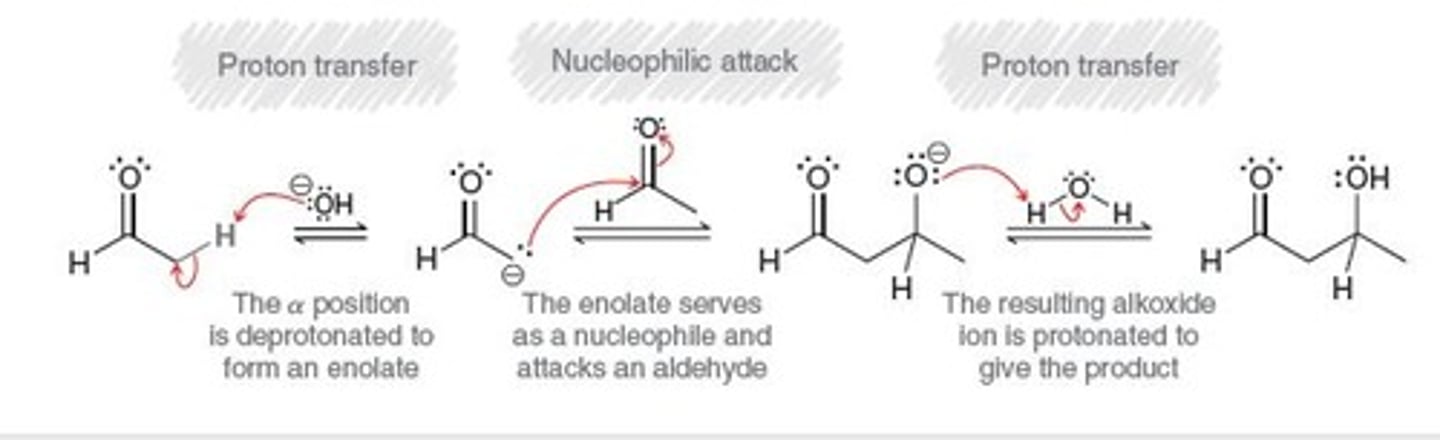
aldol addition
product exhibits both aldehydic and hydroxyl group
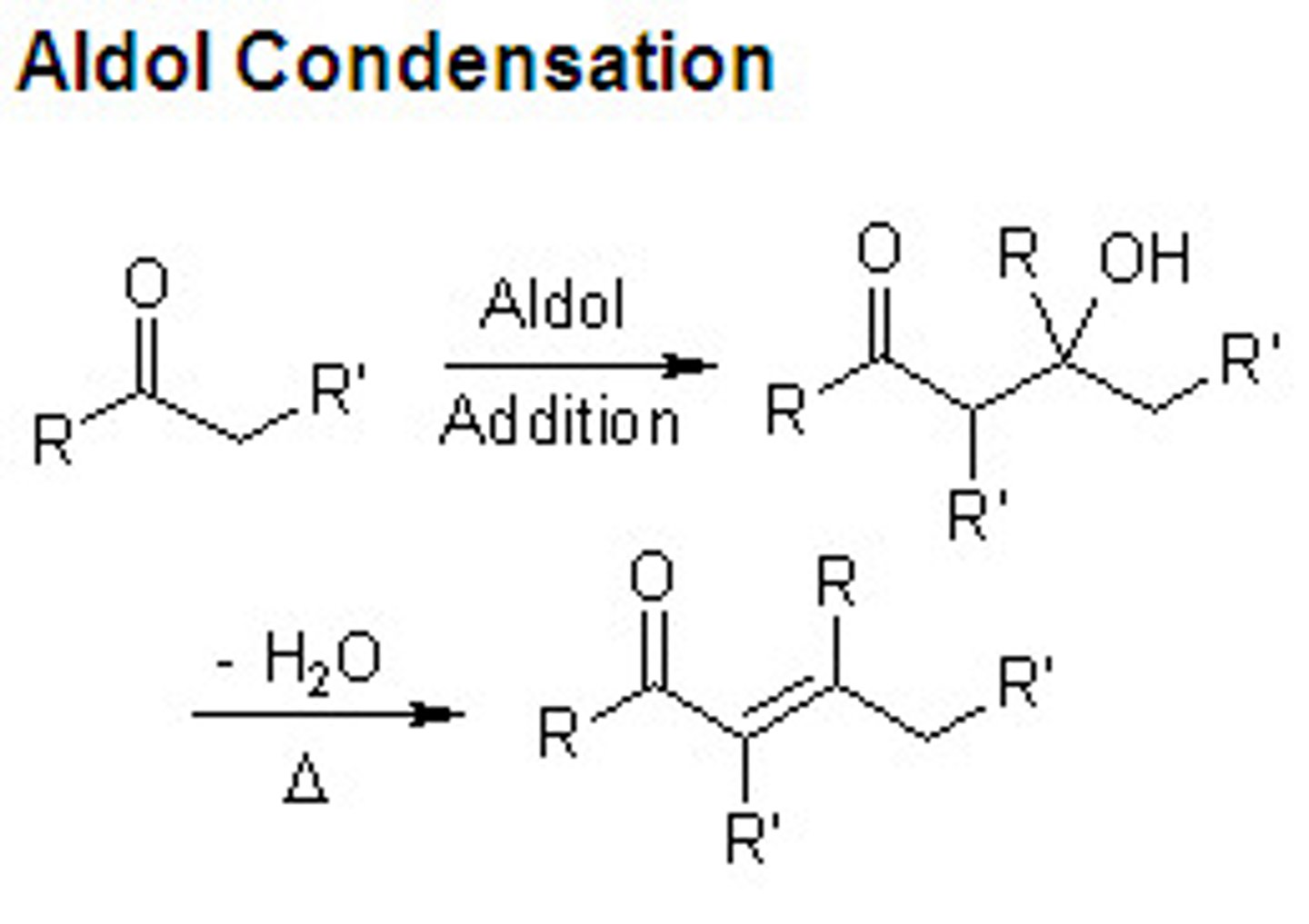
aldol condensation
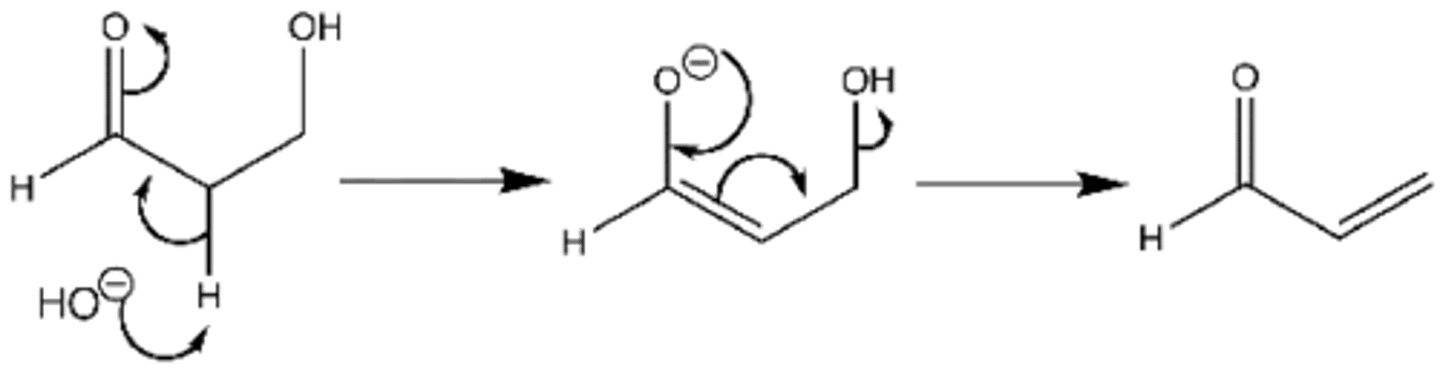
when heated in acidic or basic conditions, product of an aldol addition reaction undergoes elimination to produce unsaturation between the alpha and beta positions
dehydration product is irreversible, called a thermodynamic sink
product is an a,B-unsaturated ketone/aldehyde
condensation
any reaction in which two molecules undergo addition accompanied by loss of a small molecule such as water, carbon dioxide, or nitrogen gas
E1cB Reaction
A unimolecular elimination reaction in which a proton is first removed to give a carbanion intermediate, which then expels the leaving group in a separate step
cb = conjugate base
1 = first order reaction
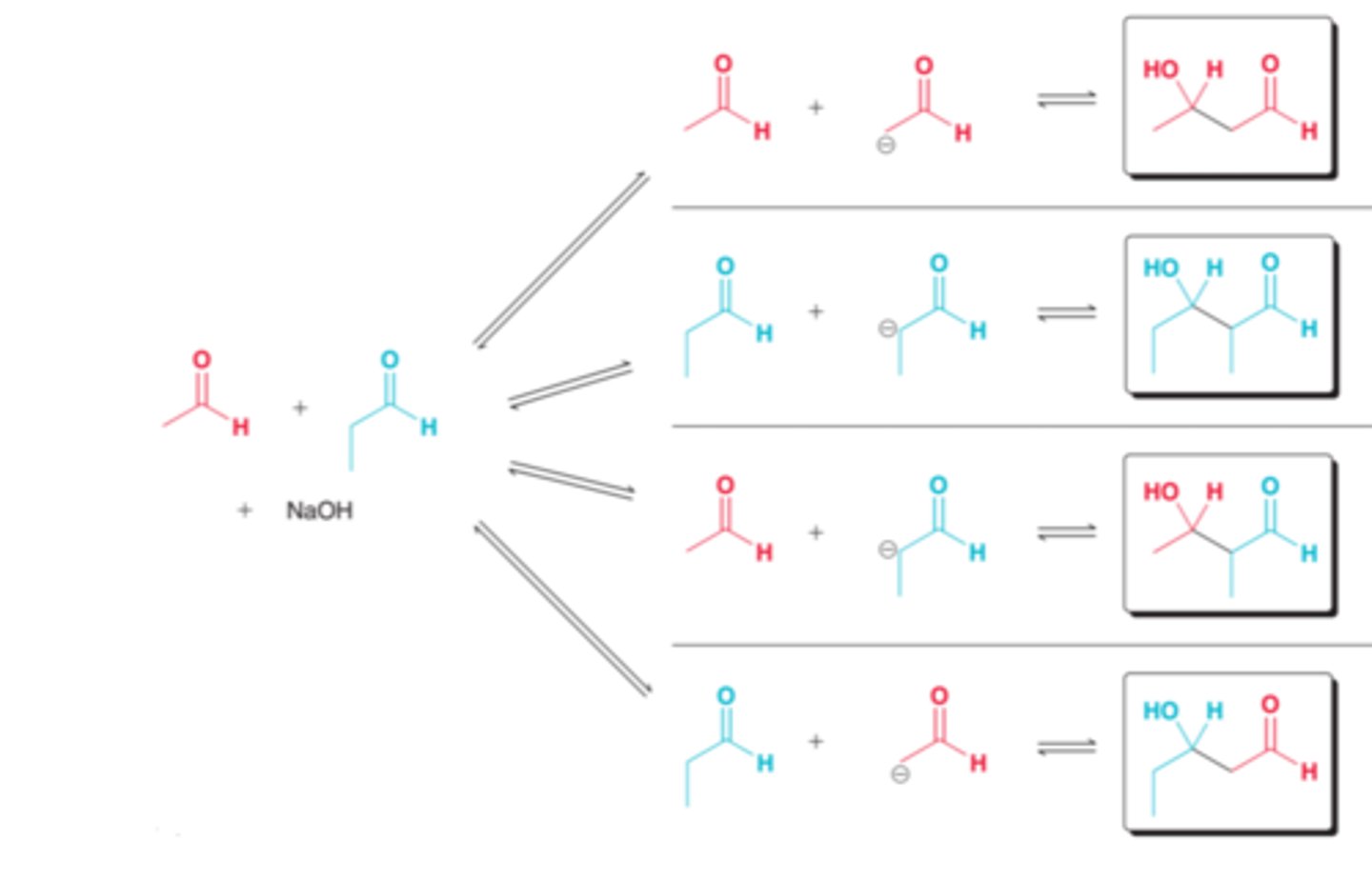
crossed aldol reactions
occurs between two different partners
only aldehydes with no alpha protons can act as electrophile
intramolecular aldol reactions
compounds that possess two carbonyl groups can undergo intramolecular aldol reactions; forms five- and six-membered rings
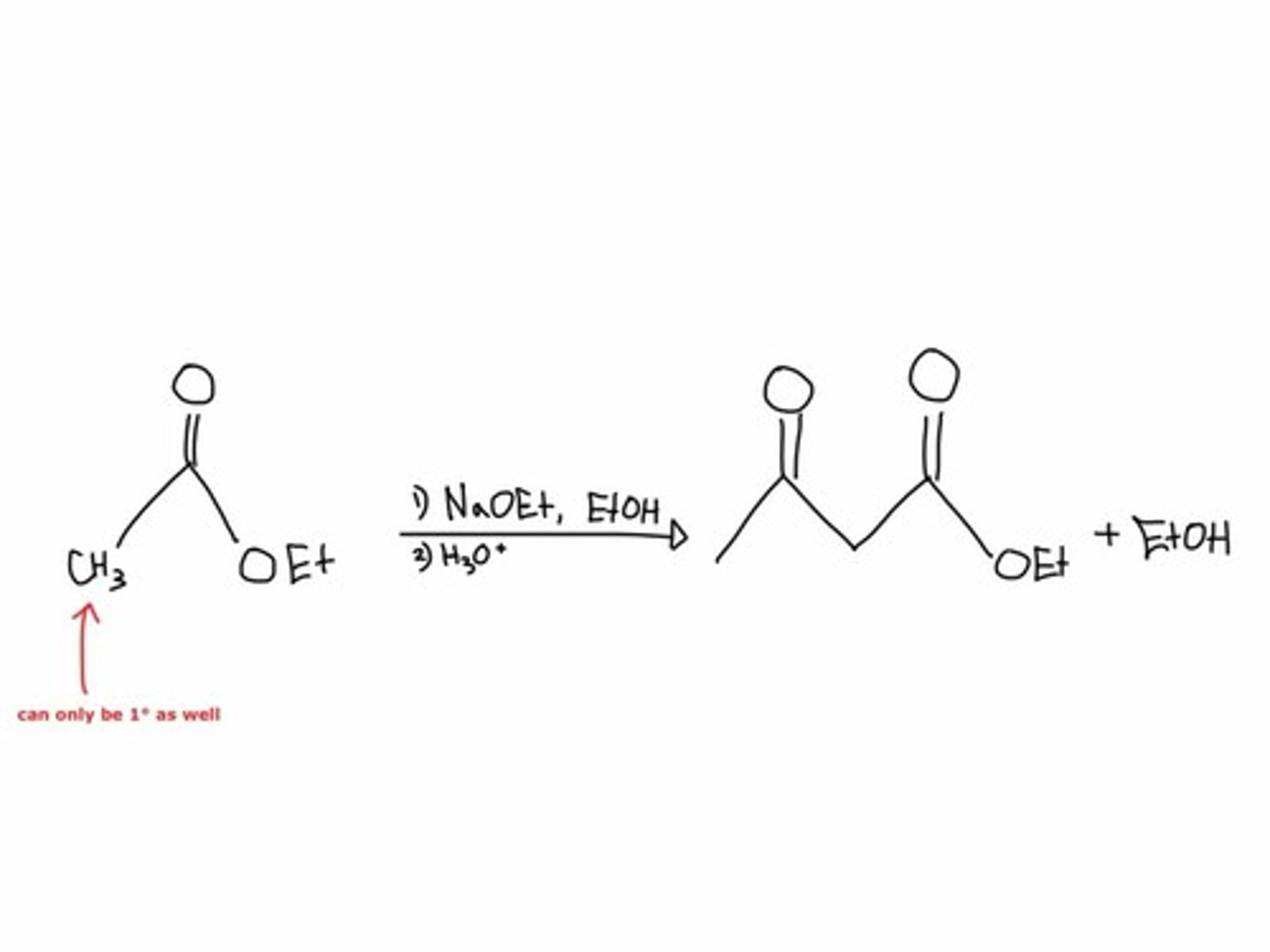
Claisen condensation
enolate ion of one ester acts as nucleophile attacking another ester
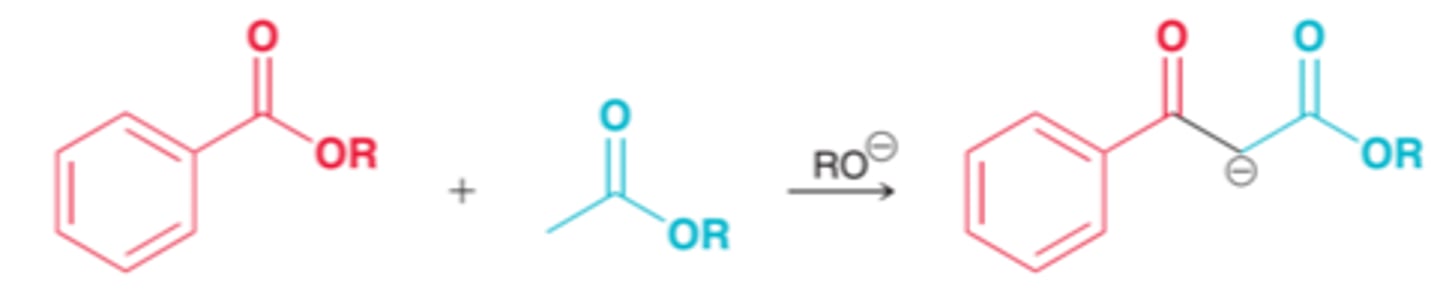
crossed claisen condensation
claisen condensation reaction that occurs between two different partners
Dieckmann Cyclization
intramolecular claisen condensation; forms five- and six- membered rings
alkylation via enolate ions
alkylate alpha position with formation of an enolate followed by treatment with alkyl halide; sn2
use LDA for less substituted product (kinetic enolate), NaH for more-substituted product (thermodynamic enolate)
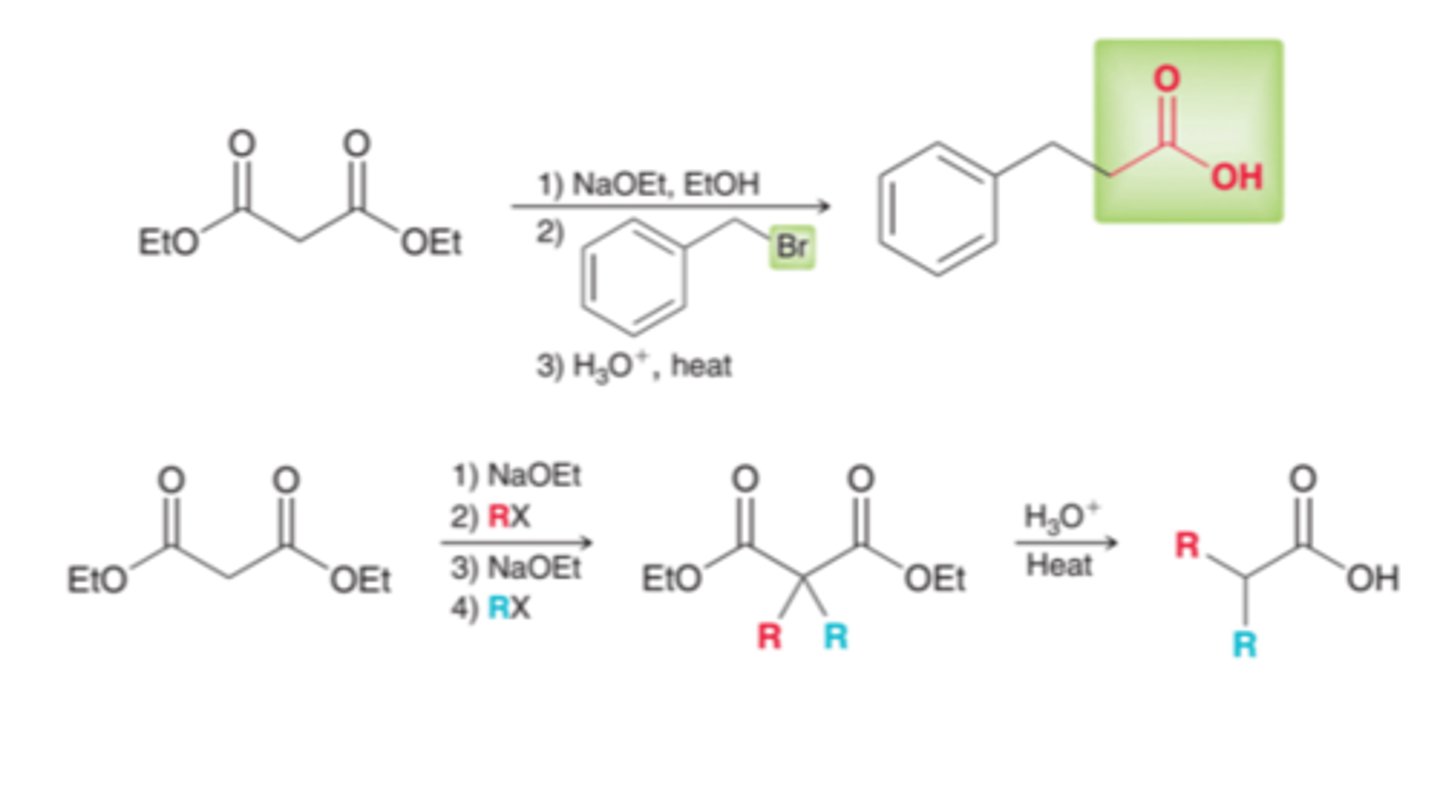
Malonic ester synthesis
enables the transformation of a halide into a carboxylic acid with the introduction of two new carbon atoms
done with diethyl malonate
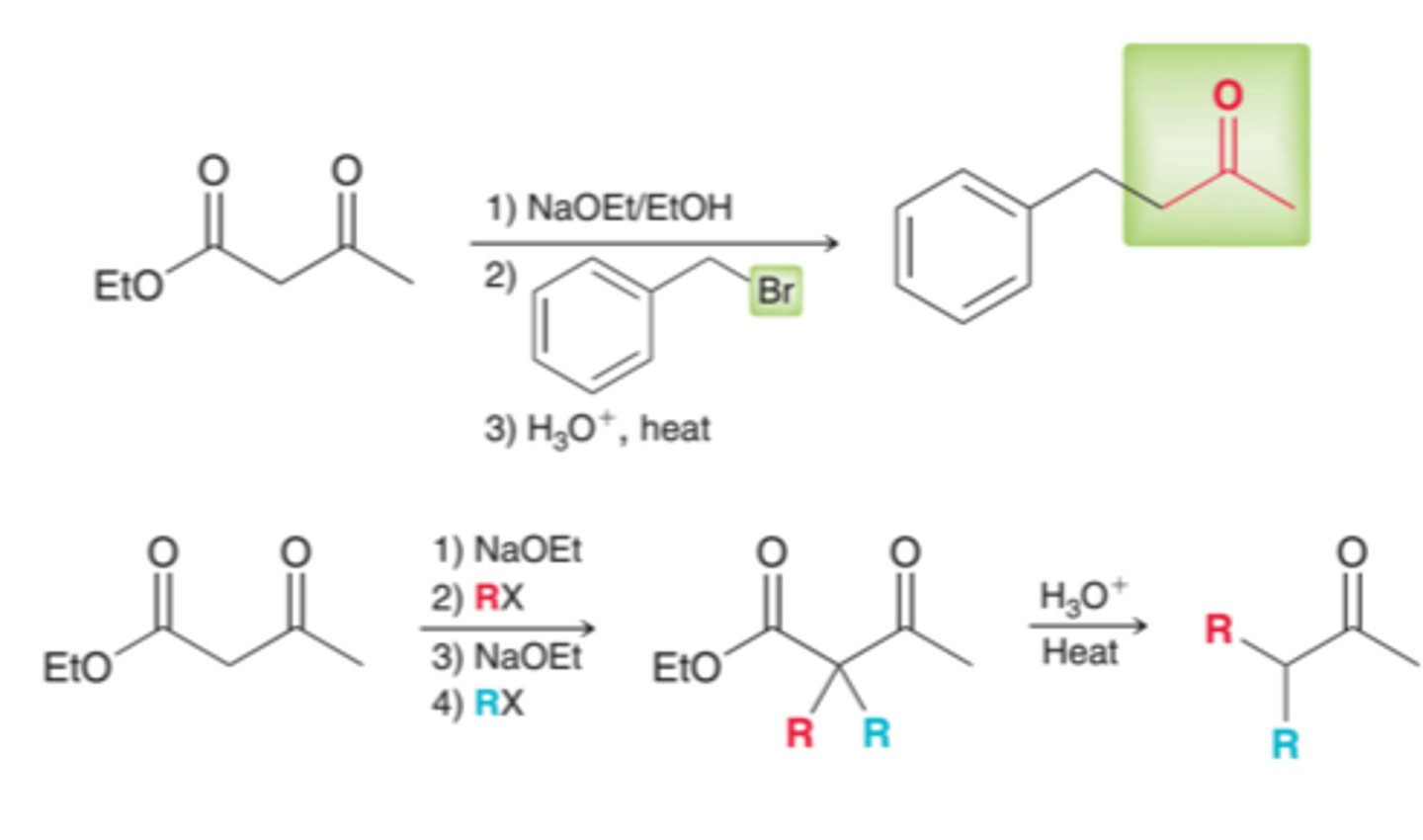
acetoacetic ester synthesis
converts an alkyl halide into a methyl ketone with the introduction of three new carbon atoms
Conjugate addition
Strongly basic nucleophiles react at the carbonyl carbon
Less basic nucleophiles react at the beta-carbon (1,4-addition)
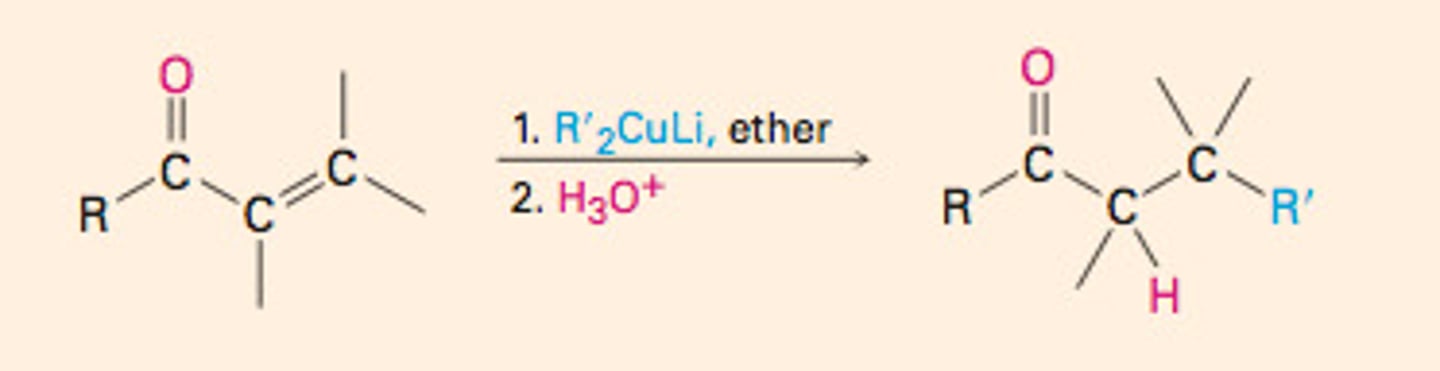
thermodynamic control
most stable product obtained from reaction (1,4 product)
Kinetic control
products are reflective of the path that reacts fastest (1,2 product)
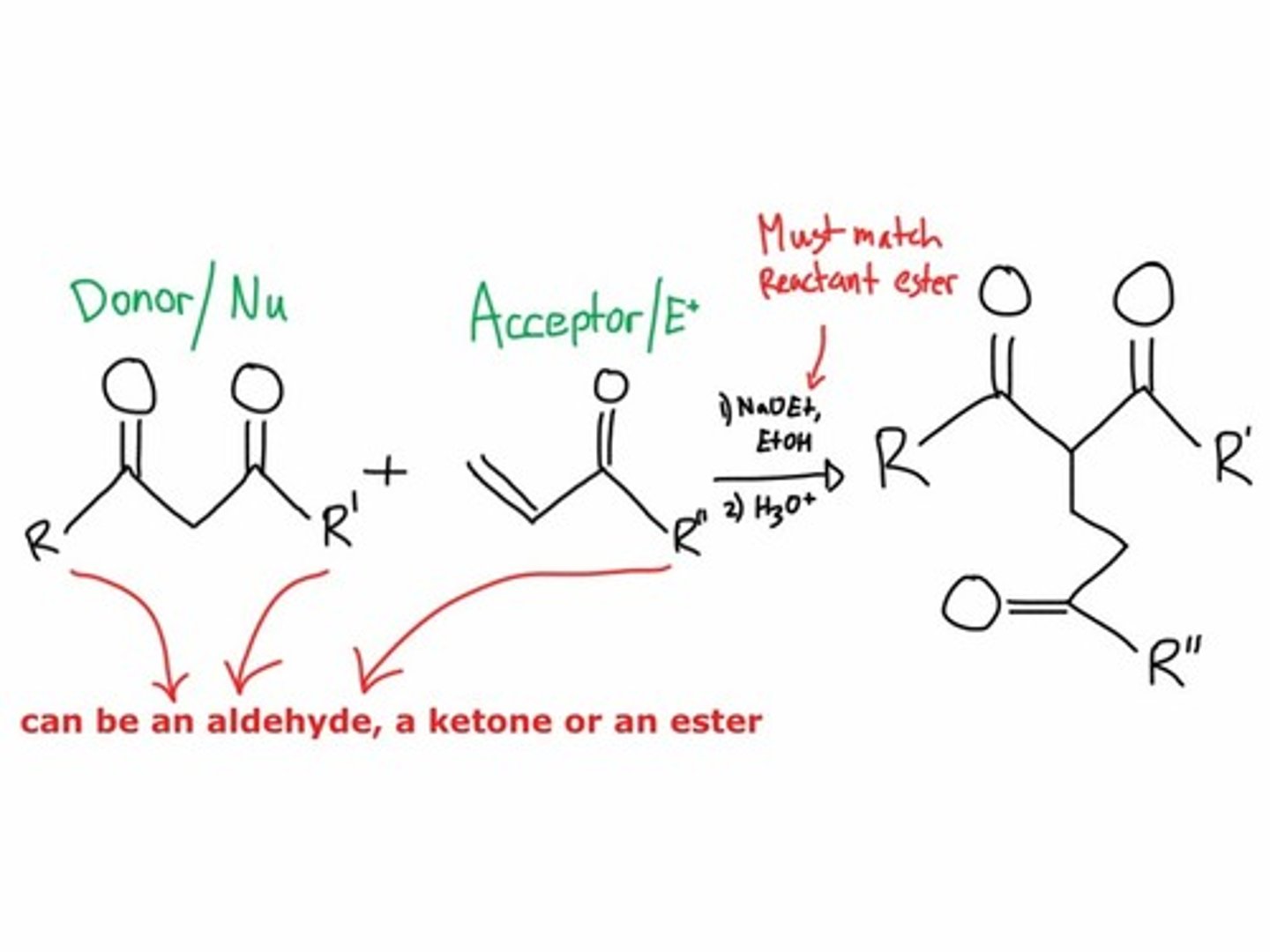
Michael reaction
1,4 nucleophilic addition of enolate anion or enamine to a,b-unsaturated carbonyl (done over EtO- and EtOH)
stork enamine synthesis
effective Michael donors
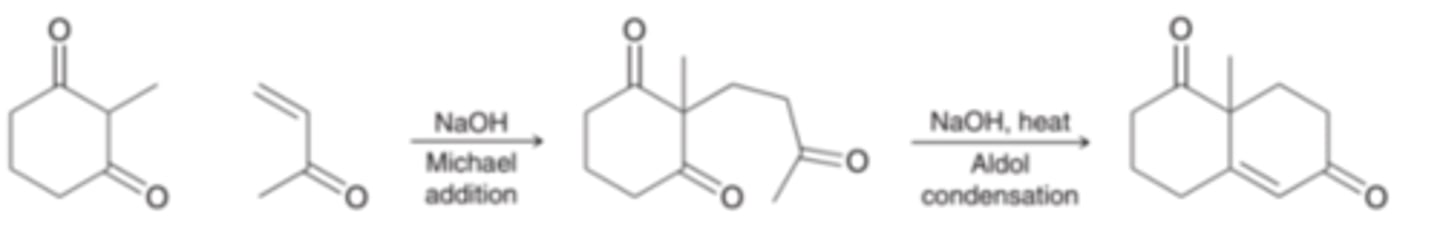
Robinson Annulation Reaction
forms a ring, Michael addition is followed by an intramolecular aldol condensation
Synthesis strategies
Stork enamine synthesis produces 1,5-difunctionalized compounds
aldol addition reactions and claisen condensation reactions produce 1,3-difunctionalized compounds
alkylation of alpha and beta positions
instead of quenching an enolate ion in a Michael addition, treat with an alkyl halide to get alkylation of the alpha position