Chemistry - Energy Changes
0.0(0)
Studied by 0 peopleCard Sorting
1/49
There's no tags or description
Looks like no tags are added yet.
Last updated 5:05 PM on 4/4/26
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
1
New cards
What is the mass-mr-moles equation
Mass = mr x moles
2
New cards
How to find a concentration in mol/dm3
Concentration = moles/volume
3
New cards
From cm3 to dm3
Divide by 1000
4
New cards
How to go from mol/dm3 to g/dm3
Conc(g) = conc(mol) x mr
5
New cards
The bonds between reactants must be made/broken in a reaction
Broken
6
New cards
Breaking bonds is endo/exothermic
Endothermic
7
New cards
The bonds of a product must be made/broken in a reaction
made
8
New cards
Making bonds is endo/exothermic
exothermic
9
New cards
How to calculate energy change in a reaction
Draw out the equation with lines (eg. O=O)
Look up each type of bond in the data sheet
Multiply energy by the number of that bond
Do this for every bond
Add reactants. Add products
Reactant energy - product energy
Look up each type of bond in the data sheet
Multiply energy by the number of that bond
Do this for every bond
Add reactants. Add products
Reactant energy - product energy
10
New cards
If the energy given out when forming/breaking bonds is is greater than the energy needed to form/break bonds, then the sign of the answer will be postive/negative, and the reaction will be exothermic
Forming, break, negative, exothermic
11
New cards
If the energy given out when forming/breaking bonds is is less than the energy needed to form/break bonds, then the sign of the answer will be postive/negative, and the reaction will be endothermic
Forming, break, positive, endothermic
12
New cards
What is the first straight bit of the energy profile diagram
The energy held in the bonds of a reactant
13
New cards
What is the steep bit going up on an energy profile diagram
The energy given to a reaction e.g. by a flam
14
New cards
What is the steep bit going down on an energy profile diagram
The reaction is occuring
15
New cards
What is the last straight bit on the energy profile diagram
The energy held between the bonds of products
16
New cards
Energy profile diagram: if the products bit is higher than the reactants bit, then…
The reaction is endothermic
17
New cards
Energy profile diagram: if the products bit is lower than the reactants bit, then…
The reaction is exothermic
18
New cards
What is a limiting agent in a reaction
The reaction that there is the least of. This is because, when this is used up, the reaction ends, even if there is still some of the other reactant(s)
19
New cards
How to calculate the energy change in a reaction
Q=mcΔt
Energy = mass x sp.heatcap x temp change
*if there is no mass given, use volume in ml, assuming density is 1
Then
Change in energy = energy/moles of limiting agent
Energy = mass x sp.heatcap x temp change
*if there is no mass given, use volume in ml, assuming density is 1
Then
Change in energy = energy/moles of limiting agent
20
New cards
How to make a cell
Connecting two different electrodes with a wire and placing them in an electrolyte (often a voltmeter as well to measure potential difference)
21
New cards
What is an electrolyte
A liquid through which charged particles can flow
22
New cards
Why are the electrodes made of metal
So they can conduct electricity
23
New cards
What is a battery
A number of cells connected in series to create a greater voltage
24
New cards
Factors affecting the voltage of a cell
Difference in reactivity between the two metals (greater difference = greater reactivity)
Type and concentration of the electrolyte
Conditions like temperature
Type and concentration of the electrolyte
Conditions like temperature
25
New cards
How do rechargeable batteries work? Where might you find them?
The chemical reactions in them can be reversed when they are supplied with an external electrical current. They are found in laptops and phones
26
New cards
How do non-rechargeable batteries work? Where might you find them?
The reaction stops when one of the reactants has been used up, which means the battery runs out. You may find them in smoke alarms and tv remotes (alkaline batteries)
27
New cards
In a cell the BLANK reacts with the BLANK
Electrodes, electrolyte
28
New cards
State the reactivity series
Potassium, sodium, lithium, calcium, magnesium, aluminium, carbon, zinc, iron, nickel, tin, lead, hydrogen, copper, mercury, silver, gold, platinum
Police Sergeant Little Charlie MAZINTL (he) Caught Me Stealing Gold Plate
When you’re stuck on carbon don’t forget, it's between the start and the end of the alphabet
Hydrogen is between lead and copper
Police Sergeant Little Charlie MAZINTL (he) Caught Me Stealing Gold Plate
When you’re stuck on carbon don’t forget, it's between the start and the end of the alphabet
Hydrogen is between lead and copper
29
New cards
What gives a bigger voltage: Magnesium + copper cell or a magnesium + zinc cell and why?
Magnesium and copper as there is a greater difference in reactivity
30
New cards
The BLANK the reaction between the electrolyte and the electrodes, the greater the voltage
Faster
31
New cards
If 3 cells connected in series have a voltage of 1V each, how big is the voltage of the battery
3V
32
New cards
What does a fuel cell need to produce electricity
Oxygen and fuel
33
New cards
What does a hydrogen oxygen fuel cell use? What does it produce
Needs: hydrogen and oxygen
Produces: water
Produces: water
34
New cards
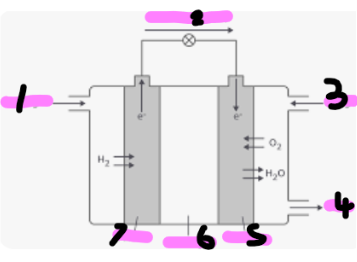
Hydrogen
Electron flow
Oxygen
Water
Cathode
Electrolyte
anode
Electron flow
Oxygen
Water
Cathode
Electrolyte
anode
35
New cards
Is the anode positive or negative
Negative (andy is a really pessimistic guy)
36
New cards
Is the cathode positive or negative
Positive (cathy is very optimistic)
37
New cards
What are the electrodes made of
Porous carbon and a catalyst
38
New cards
How does a hydrogen fuel cell work with equations (full description0
Hydrogen enters from the left side and is oxidised by the anode (this means it loses its electrons
Hydrogen is now H+, a positive ion, (H2 = 2H+ + 2e-)
The electrons pass through the wire to the cathode
Hydrogen ions move through the electrolyte to the cathode
The oxygen gains 2 electrons and reacts with hydrogen (O2 + 4H+ + 4e- → 2H2O)
Water leaves the fuel cell via the outlet
Hydrogen is now H+, a positive ion, (H2 = 2H+ + 2e-)
The electrons pass through the wire to the cathode
Hydrogen ions move through the electrolyte to the cathode
The oxygen gains 2 electrons and reacts with hydrogen (O2 + 4H+ + 4e- → 2H2O)
Water leaves the fuel cell via the outlet
39
New cards
What are the two equations in a hydrogen fuel cell called (hint: this name describes both of them)
Half equations
40
New cards
In a fuel cell, hydrogen goes through BLANK
Oxidation
41
New cards
In a fuel cell, oxygen goes through BLANK
Reduction
42
New cards
How is electrical energy created in a fuel cell
Through the electrons moving through the wire at the top
43
New cards
Describe how a fuel cell works (exam words)
The fuel is oxidised, which sets up a potential difference across the cell, where the anode and the cathode have different charges. This drives the electrons to move through the wire, generating electrical energy
44
New cards
Where would hydrogen fuel cells mostly be used
Car batteries
45
New cards
Pros of hydrogen fuel cells
Only uses hydrogen and oxygen, which are abundant
Only produces water, (no toxic waste products like how normal car batteries produce CO2)
Simple device which is not very polluting to dispose of and lasts longer than normal batteries
Only produces water, (no toxic waste products like how normal car batteries produce CO2)
Simple device which is not very polluting to dispose of and lasts longer than normal batteries
46
New cards
Cons of hydrogen fuel cells
Hydrogen is a gas, which means that is takes up a lot more space to store than fossil fuels
It is explosive when it reacts with air, so is dangerous to store
Making hydrogen fuel requires a lot of energy in itself, and this is normally obtained from fossil fuels
It is explosive when it reacts with air, so is dangerous to store
Making hydrogen fuel requires a lot of energy in itself, and this is normally obtained from fossil fuels
47
New cards
Pros of alkaline cells
Cheap to make and easy to use
48
New cards
Cons of alkaline cells
Often end up in landfill after only one use
49
New cards
Pros of rechargeable cells
Can be used over and over again
50
New cards
Cons of rechargeable cells
Expensive to make and can take a long time to recharge