orgo 1 exam 2 flashcards
1/26
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
27 Terms
LG ranking
Cl, Br, I, Ots, OMs, OTF, positive LG - weak base=good LG
draw reaction chart
correct?
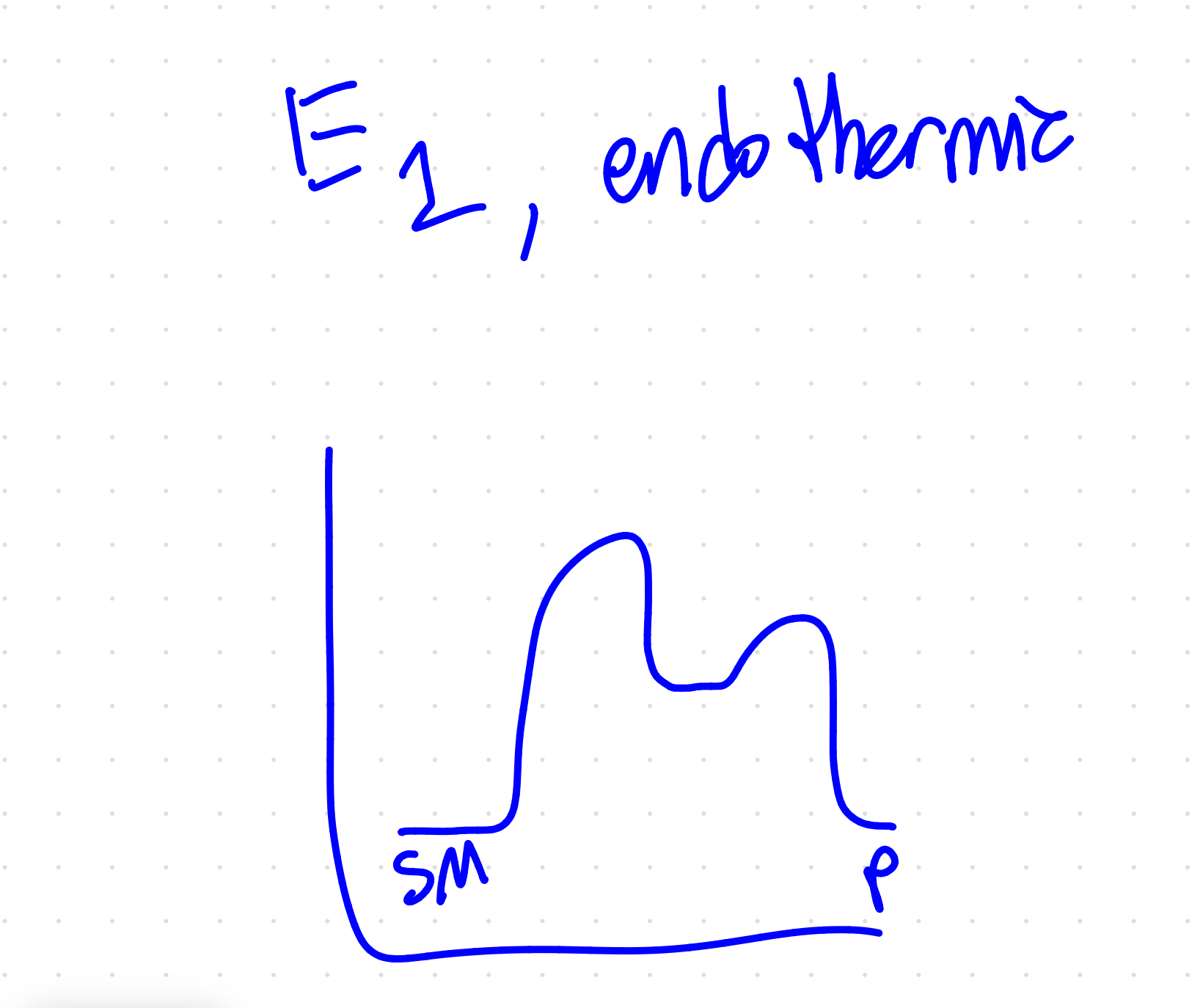
does heat favor sn1 or e1
e1
Strong base, weak nucleophile
Kot-bu, LDA
strong base, strong nuc
NaOH, NaNH2
weak base, strong nucleophile
X-, NaCN, NaSH, N3
weak base, weak nuc
H2O, MeOH, EtOH, amides
Polar protic solvents and what to they favor
H2O, ROH -favor 1
stabilize formation of C+, makes nucleophile worse
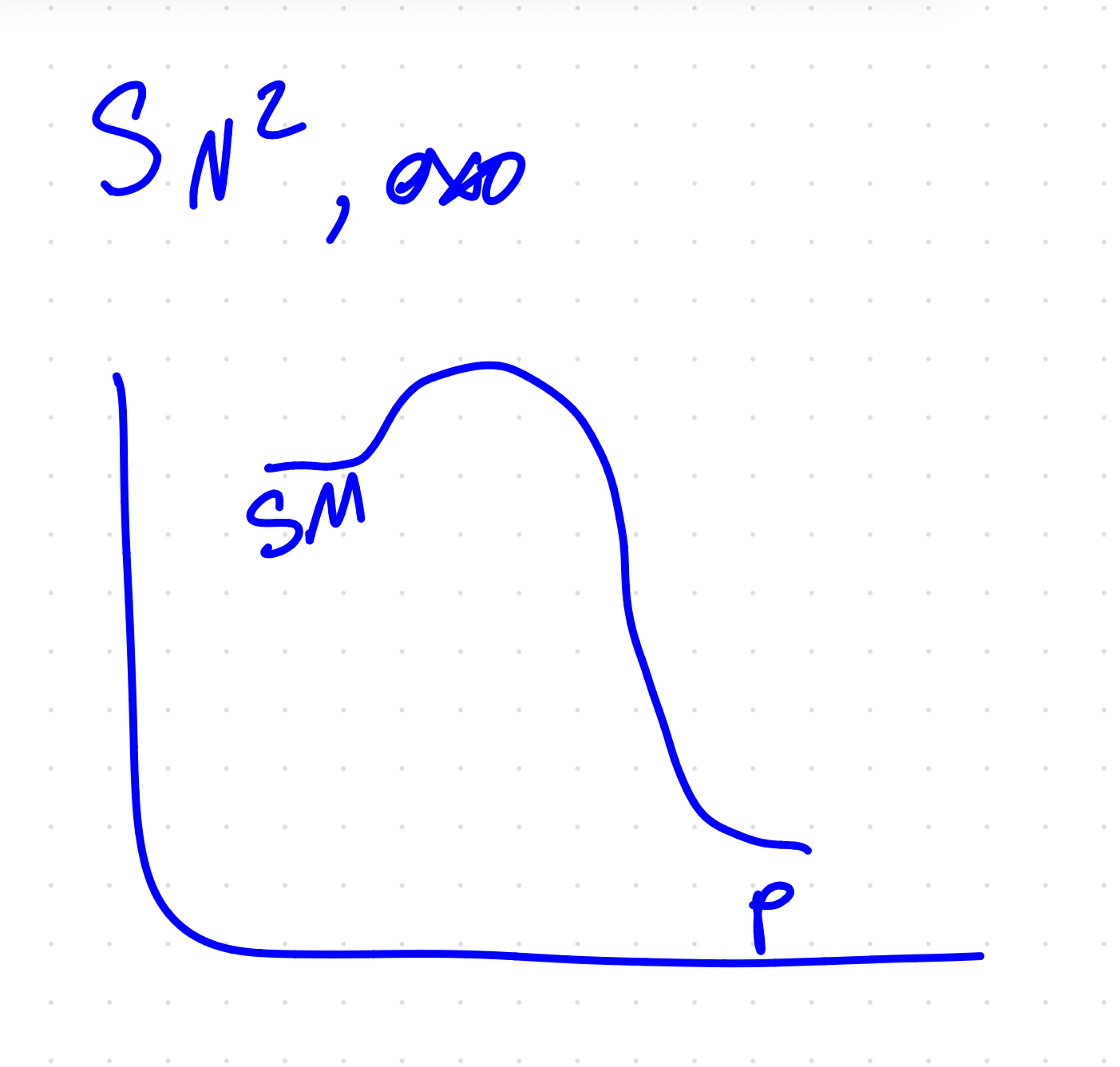
polar Aprotic solvents and what do they favor
acetone, DMSO, DMF, THF - favor 2
C+ wont form, makes nucleophile better
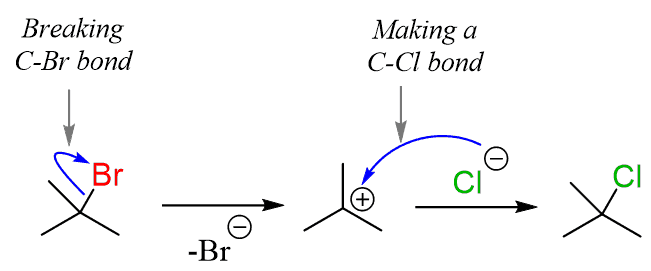
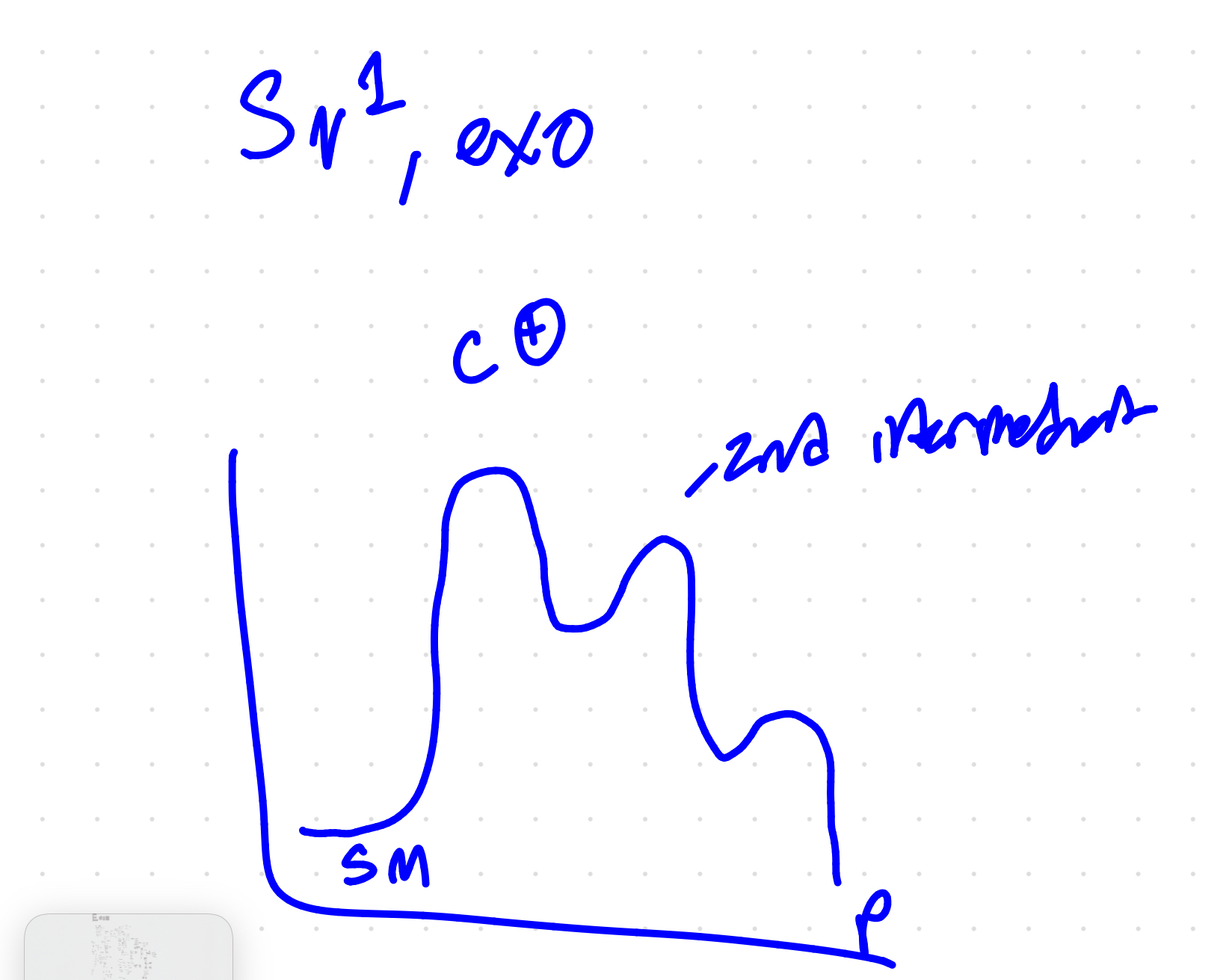
sn1 mechanism with carbocation intermediate
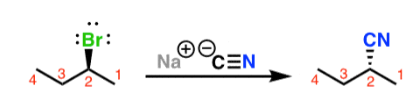
sn2 mechanism
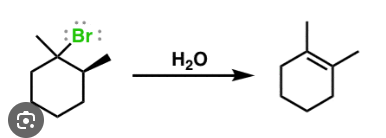
with carbocation intermediate
E1 mechanism
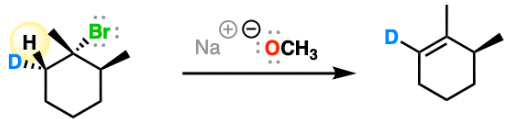
e2 mechanism
- needs strong hindered base, LG must be axial and be anti-peri planar to H
major will be zaitssev unless base is bulky
KOtbu & LDA prefer less substituted
strong base weak nuc
primary LG
secondary LG
Tertiary LG
E2 requirements
strong base strong nuc
primary LG
secondary LG
Tertiary LG
Sn2
E2
E2
weak base strong nuc
primary LG
secondary LG
Tertiary LG
SN2
SN2
Sn1
weak base weak nuc
primary LG
secondary LG
Tertiary LG
no rxn - unless stabilized
Sn1/E1
SN1/E1
neuman projection
top left =
top right =
neuman projection
dash
wedge
chair conformation
wedge =
dash =
wedge = up
dash = down
order of carbocation rearrangement
Ring expansion
hydride shift
1, 3 methyl shift
ring expansion criteria
C+ next to 3,4,5 membered ring
hydride shift
C+ next to tertiary carbon
methyl shift
C+ next to 4 carbon w/ methyl group
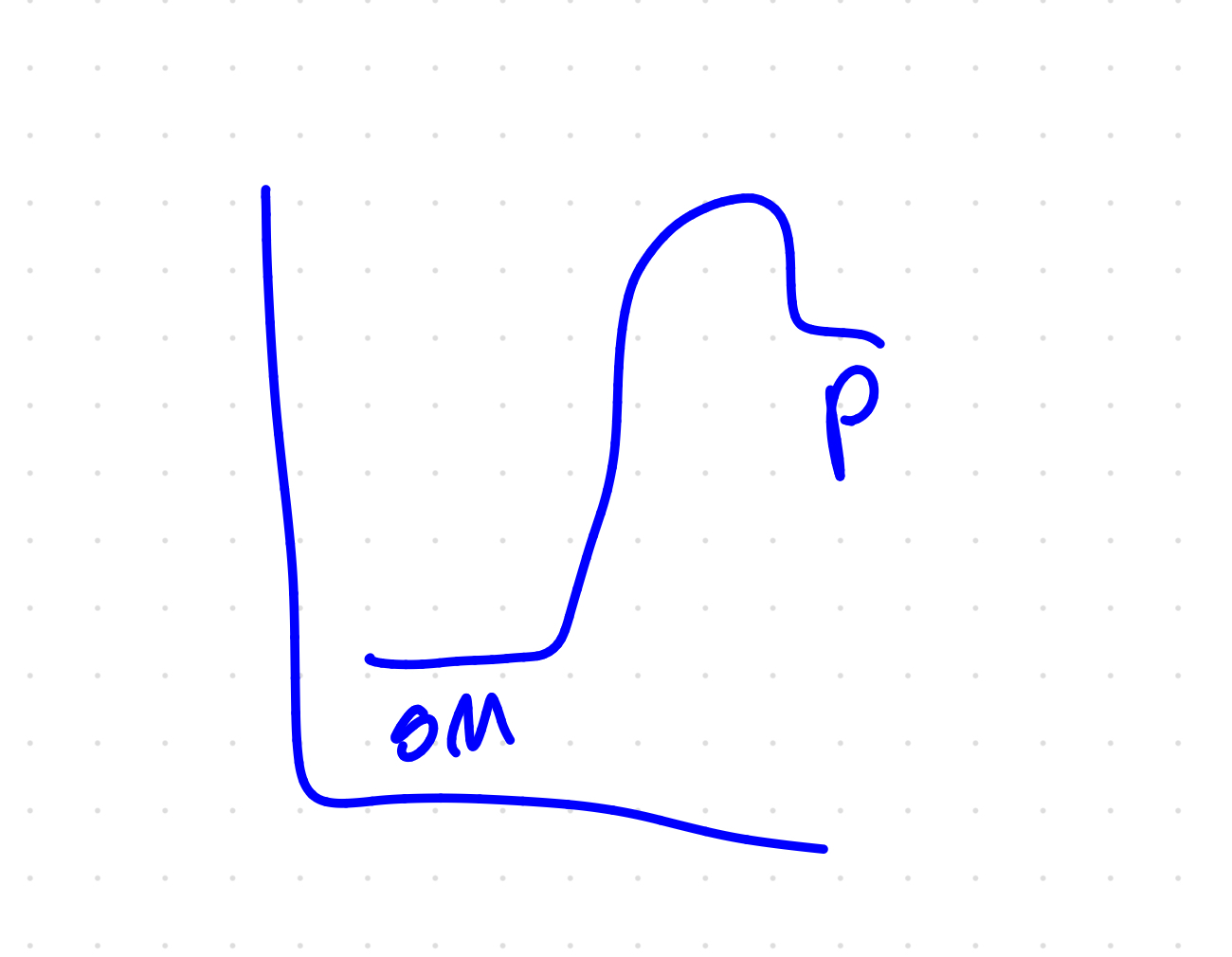
Draw E2 reaction diagram
draw e1 diagram
sn1 diagram
sn2 diagram