Fase 1
0.0(0)
Studied by 16 peopleCard Sorting
1/64
Earn XP
Description and Tags
Last updated 5:58 AM on 9/7/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
65 Terms
1
New cards
Science
• Knowledge that covers general truths
or the operation of general laws,
especially when acquired and tested by
the scientific method.
• Fields of study that attempt to
comprehend the nature of the
universe.
or the operation of general laws,
especially when acquired and tested by
the scientific method.
• Fields of study that attempt to
comprehend the nature of the
universe.
2
New cards
Natural science
• Astronomy
• Biology
• Chemistry
• Earth science
• Physics
• Biology
• Chemistry
• Earth science
• Physics
3
New cards
Life sciences
Biology
Anthropology
Ecology
Neuroscience
Biophysics
Biochemistry
Anthropology
Ecology
Neuroscience
Biophysics
Biochemistry
4
New cards
Physical sciences
Astronomy
Geology
Physics
Chemistry
Biophysics
Biochemistry
Geology
Physics
Chemistry
Biophysics
Biochemistry
5
New cards
Scientific reasoning
Knowledge
• Inductive reasoning
• Deductive reasoning
• Inductive reasoning
• Deductive reasoning
6
New cards
Descriptive science
• Observe
• Explore
• discover
• Explore
• discover
7
New cards
Hypothesis-based science
• Specific question/problem
• Testable solution
• Testable solution
8
New cards
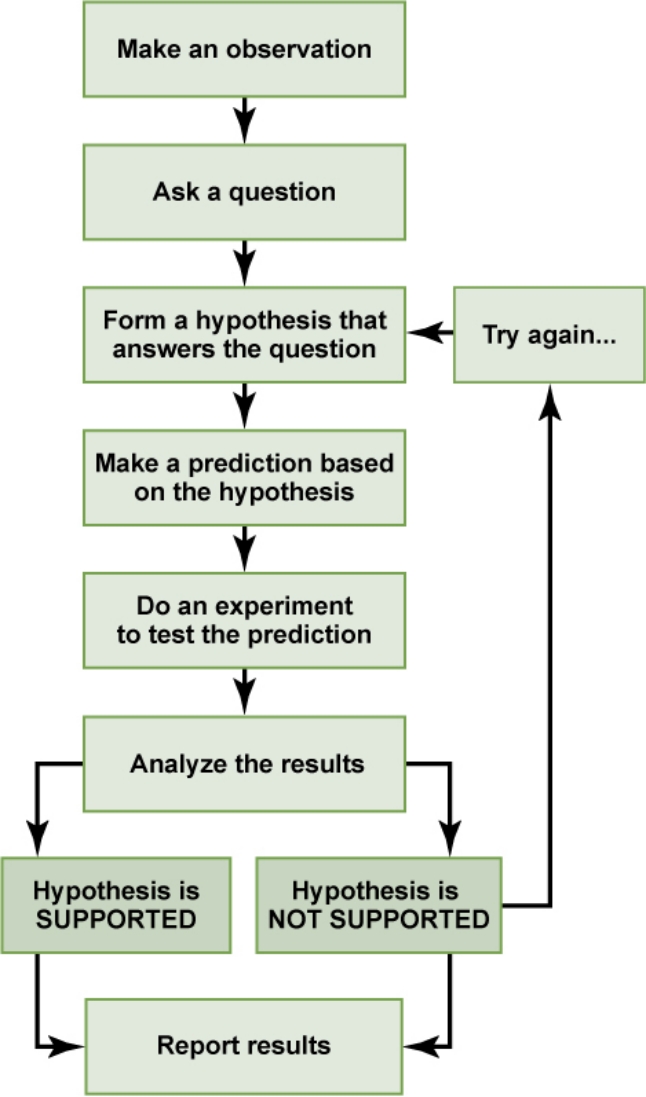
Scientific method
A method of investigation involving observation and theory to test scientific hypotheses. Made by Sir Francis Bacon (1561-1626)
9
New cards
Testing a Hypothesis
• Testable
• Falsifiable
• Science does not claim to “prove”
anything
• openness to disproving ideas
• Experiments
• Variables
• Control groups
• Falsifiable
• Science does not claim to “prove”
anything
• openness to disproving ideas
• Experiments
• Variables
• Control groups
10
New cards
Basic science
To pursue science for the
sake of simply gaining knowledge
sake of simply gaining knowledge
11
New cards
Applied science
Scientific knowledge that we can apply it to solving a specific problem or to bettering our lives
12
New cards
Properties of life
• Order
• Sensitivity or response to stimuli
• Reproduction
• Growth and development
• Regulation
• Homeostasis
• Energy processing
• Sensitivity or response to stimuli
• Reproduction
• Growth and development
• Regulation
• Homeostasis
• Energy processing
13
New cards
Order
Organisms are highly organized, coordinated structures that consist of one or more cells. In single-celled inside each cell, atoms comprise molecules, these in turn comprise cell organelles and other cellular inclusions. In multicellular organisms, similar cells form tissues. Tissues, in turn, collaborate to create organs (body structures with a distinct function). Organs work together to form organ systems.
14
New cards
Sensitivity or Response to Stimuli
Organisms respond to diverse stimuli. Movement toward a stimulus is a positive response, while movement away from a stimulus is a negative response.
15
New cards
Reproduction
• Single-celled organisms reproduce by first duplicating their DNA,the possibility for organisms to adapt to a changing environment.
• Multicellular organisms often produce specialized reproductive germline, gamete, oocyte, and sperm cells. After fertilization (the fusion of an oocyte and a sperm cell), a new individual develops.
When reproduction occurs, DNA containing genes are passed along to an organism’s offspring. These genes ensure that the offspring will belong to the same
species and will have similar characteristics, such as size and shape.
• Multicellular organisms often produce specialized reproductive germline, gamete, oocyte, and sperm cells. After fertilization (the fusion of an oocyte and a sperm cell), a new individual develops.
When reproduction occurs, DNA containing genes are passed along to an organism’s offspring. These genes ensure that the offspring will belong to the same
species and will have similar characteristics, such as size and shape.
16
New cards
Adaptation
It is a consequence of evolution by natural selection. All adaptations enhance the reproductive potential of the individuals exhibiting them, including their ability to survive to reproduce. Adaptations are not constant. As an environment changes, natural selection causes the characteristics of the individuals in a population to track those changes.
17
New cards
Growth and development
Organisms grow and develop as a result of genes providing specific instructions that will direct cellular growth and development. This ensures that a species’ young will grow up to exhibit many of the same characteristics as its parents.
18
New cards
Regulation
Multiple regulatory mechanisms to coordinate internal functions,
respond to stimuli, and cope with environmental stresses.
respond to stimuli, and cope with environmental stresses.
19
New cards
Homeostasis
In order to function properly, cells require appropriate conditions such as proper temperature, pH, and appropriate concentration of diverse chemicals. These conditions may, however, change from one moment to the next. Organisms are able to maintain internal conditions within a narrow range almost constantly, despite environmental changes, through homeostasis
20
New cards
Energy processing
All organisms use a source of energy for their metabolic activities. Some organisms capture energy from the sun and convert it into chemical energy in food. Others use chemical energy in molecules they take in as food.
21
New cards
Evolution
The possibility for organisms to adapt to a changing environment. An organism that evolves characteristics fit for the environment will have greater reproductive success, subject to the forces of natural selection.
22
New cards
Organization levels
- Atom
- Molecules
- Organelles
- Cell
- Tissues
- Organs
- Organ system
- Organisms
- Population
- Community
- Ecosystem
- Biosphere
- Molecules
- Organelles
- Cell
- Tissues
- Organs
- Organ system
- Organisms
- Population
- Community
- Ecosystem
- Biosphere
23
New cards
Atoms
The atom is the smallest and most fundamental unit of matter. It consists of a nucleus surrounded by electrons. Atoms form molecules.
24
New cards
Molecules
A molecule is a chemical structure consisting of at least two atoms held together by one or more chemical bonds.
Many molecules that are biologically important are macromolecules, large molecules that are typically formed by polymerization.
Many molecules that are biologically important are macromolecules, large molecules that are typically formed by polymerization.
25
New cards
Organelles
Organelles are small structures that exist within cells. Examples of organelles include mitochondria and chloroplasts.
26
New cards
Cells
The cell itself is the smallest fundamental unit of structure and
function in living organisms. Some organisms consist of a single cell and others are multicellular.
Scientists classify cells as prokaryotic or eukaryotic.
function in living organisms. Some organisms consist of a single cell and others are multicellular.
Scientists classify cells as prokaryotic or eukaryotic.
27
New cards
Prokaryotic cells
Prokaryotes are single-celled or colonial organisms that do not have membrane-bound nuclei.
28
New cards
Eukaryote
The cells of eukaryotes do have membrane-bound organelles and a membrane-bound nucleus.
29
New cards
Tissues
Groups of similar cells carrying out similar or related functions.
30
New cards
Organs
Collections of tissues grouped together perfoming a common function.
31
New cards
Organ system
Higher level of organization that consists of functionally related organs.
32
New cards
Organisms
Individual living entities. (Single-cell eukaryotes and prokaryotes are also organism, usually called microorganisms)
33
New cards
Population
All the individuals of a species living within a specific area
34
New cards
Community
The sum of population inhabiting a particular area
35
New cards
Ecosystem
Consists of all the living things in a particular area together with the abiotic (nonliving parts of the environment)
36
New cards
Biosphere
The collection of all ecosystems, and it represents the zones of life on Earth. It includes land, water and even the atmosphere to a certain extent.
37
New cards
Taxonomy
A classification of organisms into groups based on similarities of structure or origin
- Arrangment law
- Hierarchical syste,
- Arrangment law
- Hierarchical syste,
38
New cards
Linnaean system
-Domain
-Kingdom
-Phylum
-Class
-Order
-Family
-Genus
-Species
-Subspecies
-Kingdom
-Phylum
-Class
-Order
-Family
-Genus
-Species
-Subspecies

39
New cards
Binomial nomeclature
- Genus + species
40
New cards
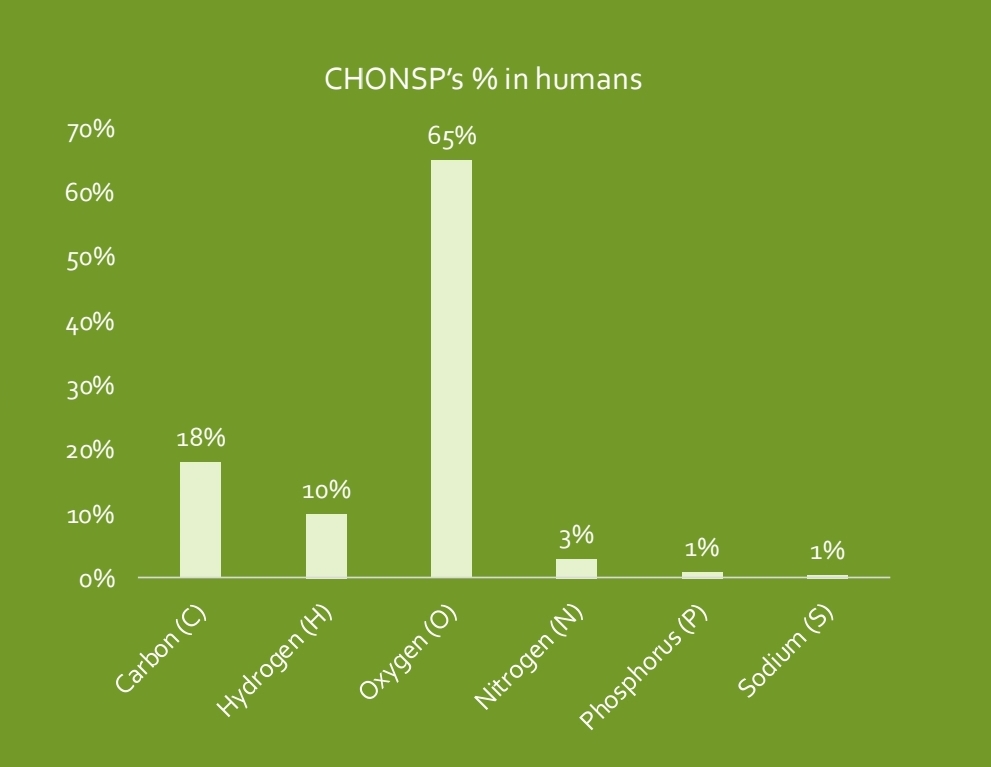
The essential elements
90% of an organism's matter is made by the essential elements (O, C, H and N) which are rare in nonliving stuff
41
New cards
Inorganic compounds
Substance that lacks carbon–hydrogen bonds
- Many contain H
- Few contain
Essential for life are: Water, salts, acids and bases
- Many contain H
- Few contain
Essential for life are: Water, salts, acids and bases
42
New cards
Water and it's properties
It makes up about 60-70% of adults' body weight, it is found within and between the cells
Water properties:
- Polarity
- Different matter states
- High heat capacity
- Vaporization
- Is a solvent
- Cohesiveness and adhesiveness
Water properties:
- Polarity
- Different matter states
- High heat capacity
- Vaporization
- Is a solvent
- Cohesiveness and adhesiveness
43
New cards
Water polarity
One of water’s important properties is that it is composed of polar molecules: the hydrogen and oxygen within water molecules
(H2O) form polar covalent bonds.
We call a polar substance that interacts readily with or dissolves in water hydrophilic. In contrast, nonpolar molecules such as oils and fats do not interact well with water, we call such nonpolar compounds hydrophobic.
(H2O) form polar covalent bonds.
We call a polar substance that interacts readily with or dissolves in water hydrophilic. In contrast, nonpolar molecules such as oils and fats do not interact well with water, we call such nonpolar compounds hydrophobic.
44
New cards
Water's states
As water molecules make hydrogen bonds with each other, water takes on some unique chemical characteristics compared to other
liquids. In liquid water, hydrogen bonds constantly form and break as the water molecules slide past each other. When the heat rises as water boils,
the water molecules' higher kinetic energy causes the hydrogen bonds to break completely and allows water molecules to escape into the air as gas (steam or water vapor). Alternatively, when water temperature reduces and water freezes, the water molecules form a crystalline structure maintained by hydrogen bonding.
liquids. In liquid water, hydrogen bonds constantly form and break as the water molecules slide past each other. When the heat rises as water boils,
the water molecules' higher kinetic energy causes the hydrogen bonds to break completely and allows water molecules to escape into the air as gas (steam or water vapor). Alternatively, when water temperature reduces and water freezes, the water molecules form a crystalline structure maintained by hydrogen bonding.
45
New cards
Water's high heat capacity
Water’s high heat capacity is a property that hydrogen bonding among water molecules causes. We define specific heat as the amount of heat one gram of a substance must absorb or lose to
change its temperature by one degree Celsius. For water, this amount is one calorie.
change its temperature by one degree Celsius. For water, this amount is one calorie.
46
New cards
Water's heat of vaporization
Water also has a high heat of vaporization, the amount of energy required to change one gram of a liquid substance to a gas.
47
New cards
Water's solvent properties
Since water is a polar molecule with slightly positive and slightly negative charges, ions and polar molecules can readily dissolve
in it. Therefore, we refer to water as a solvent.
in it. Therefore, we refer to water as a solvent.
48
New cards
Water’s Cohesive and Adhesive Properties
In cohesion, water molecules are attracted to each other (because of hydrogen bonding), keeping the molecules together at the liquid-gas (water-air) interface, although there is no more room in the glass. Cohesion allows for surface tension.
These cohesive forces are related to water’s property of adhesion, or the attraction between water molecules and other
molecules.
These cohesive forces are related to water’s property of adhesion, or the attraction between water molecules and other
molecules.
49
New cards
pH
The pH of a solution indicates its acidity or basicity. The pH test measures hydrogen ions' concentration in a given solution.
50
New cards
Acid
A substance that increases hydrogen ions' (H+) concentration in a solution, usually by having one of its hydrogen atoms dissociate.
51
New cards
Base
A base provides either hydroxide ions (OH–) or other negatively charged ions that combine with hydrogen ions, reducing their concentration in the solution and thereby raising the pH.
52
New cards
pH scale
The pH scale is, as we previously mentioned, an inverse logarithm and ranges from 0 to 14. Anything below 7.0 is acidic, and anything above 7.0 is alkaline. Extremes in pH in either direction from
7.0 are usually inhospitable to life.
7.0 are usually inhospitable to life.
53
New cards
Biomolecules
These molecules always contain carbon
Four main groups:
• Carbohydrates
• Lipids
• Proteins
• Nucleic acids
Four main groups:
• Carbohydrates
• Lipids
• Proteins
• Nucleic acids
54
New cards
Biomolecules: dehydration
Bonds are formed and energy is required
55
New cards
Biomolecules: Hydrolysis
H2O breaks a covalent bond and energy is releases. Enzymes speed up these processes.
56
New cards
Carbohydrates
They consist entirely of carbons and hydrogen.
These are found in grains, fruits, and vegetables
• Keyword: ENERGY
These are found in grains, fruits, and vegetables
• Keyword: ENERGY
57
New cards
Types of carbohydrates
Monosaccharide: Single sugar (simple sugars)
Disaccharide: Two sugar molecules linked
Polysaccharide: Many sugar molecules linked
Disaccharide: Two sugar molecules linked
Polysaccharide: Many sugar molecules linked
58
New cards
Lipids
They are non-polar
• Hydrophobic = Insulation
• Long-term use energy
Building blocks
• Hormones
• Cellular membranes
Are divided in fats, oils, waxes, phospholipids,
steroids
• Hydrophobic = Insulation
• Long-term use energy
Building blocks
• Hormones
• Cellular membranes
Are divided in fats, oils, waxes, phospholipids,
steroids
59
New cards
Proteins
These are the most structurally diverse and the most functionally diverse biomolecules
Enzymes:
• Breakdown.
• Rearrangement.
• Synthesis.
Hormones:
• Regulation.
Enzymes:
• Breakdown.
• Rearrangement.
• Synthesis.
Hormones:
• Regulation.
60
New cards
Nucleic acids
The most important macromolecules, it is the cell's blueprint
- DNA
- RNA
- DNA
- RNA
61
New cards
Nucleotides
1. Nitrogenous base
2. Pentose
3. Phosphate group
2. Pentose
3. Phosphate group
62
New cards
Nitrogenous bases
• (A)denine
• (G)uanine
• (C)ytosine
• (T)hymine
• DNA only
• (U)racil
• RNA only
• (G)uanine
• (C)ytosine
• (T)hymine
• DNA only
• (U)racil
• RNA only
63
New cards
DNA double-helix structure
Each pair of nitrgenous bases is binded by hydrogen bonds. Strands hold antiparallel orientation
64
New cards
RNA
Single-stranded (usually)
• Ribonucleotides
• Ribose
• A nitrogenous base
• Phosphate group
• Ribonucleotides
• Ribose
• A nitrogenous base
• Phosphate group
65
New cards
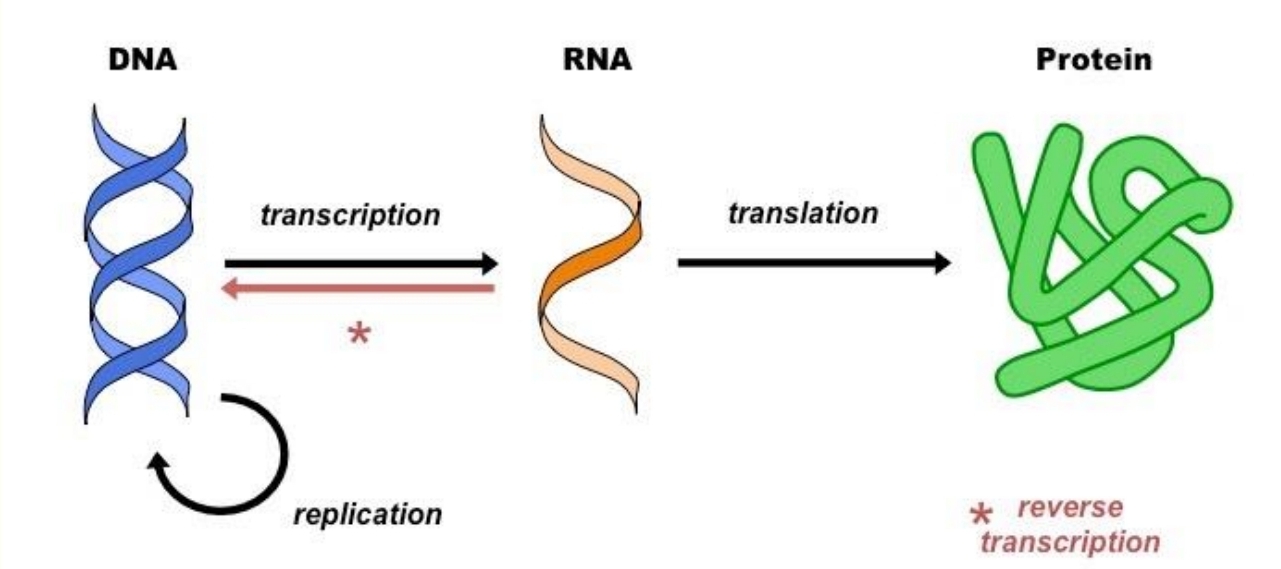
The Central Dogma of Life
Transcription:
• DNA dictates the structure of mRNA.
Translation:
• RNA dictates the protein's structure.
• DNA dictates the structure of mRNA.
Translation:
• RNA dictates the protein's structure.