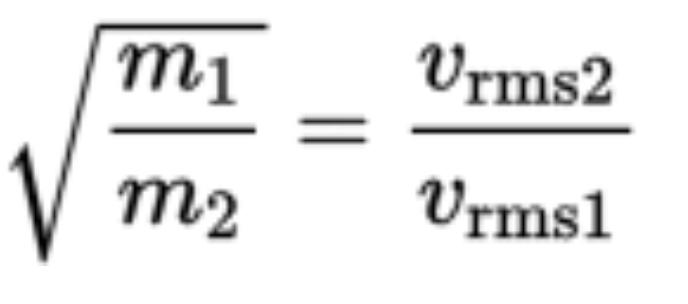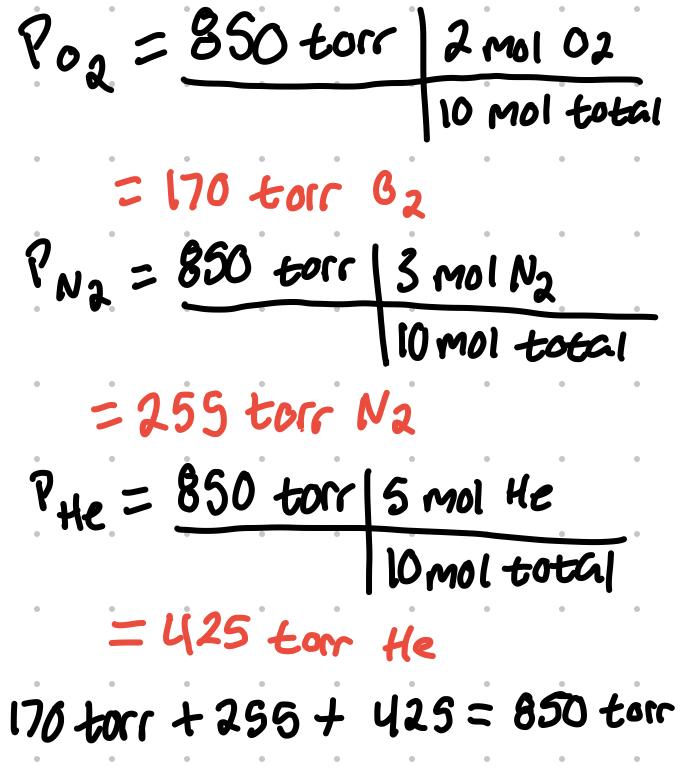Chapter 6: Gasses
- The ideal gas law describes how pressure, volume, temperature, and moles of gas change.
- Kinetic molecular theory describes the behavior of all gasses and why they behave the way they do
The Gas Laws
- Gasses have four properties
- Temperature (T)
- Pressure (P)
- Volume (V)
- Moles of gas (n)
- Each gas law holds two properties constant while one of the properties is changed
Boyle’s Law
- Boyle’s law is the inverse pressure-volume relationship
- If a sample of gas starts with initial conditions of pressure and volume and an experiment is done ONLY affecting pressure and volume, you get the equation
- PiVi=PfVf
- i = initial
- f = final
Charles’s Law
- Charles’s law is the direct relationship of temperature and volume
- (Vi)/(Ti) = (Vf)/(Tf)
- Absolute zero is the lowest possible temperature.
- -273 celsius or 0 Kelvin
Gay-Lussac’s Law
- Gay-Lussac’s law is the direct relationship of pressure and temperature
- (Pi)/(Ti)=(Pf)/(Tf)
Avogadro’s Principle
- Also known as Avogadro’s Law, it describes that equal numbers of molecules/atoms contain equal volumes of gases under identical conditions of temperature and pressure
- (ni)/(Vi)=(nf)/(Vf)
Ideal Gas Law
- Combining the previous gas laws, the ideal gas law is obtained
- PV = nRT
- R is the universal gas constant. In this equation, R is .08206 L·atm/mol·K
- Example
- A gas occupies 250 mL, and its pressure is 550 mmHg at 25°C.
- If the gas is expanded to 450 mL, what is the pressure of the gas now?
- What temperature is needed to increase the pressure of the gas to exactly 1 atmosphere and 250 mL?
- How many moles of gas are in this sample?
- The sample is an element and has a mass of 0.525g. What is it?
Standard Temperature and Pressure (STP)
- If a gas is stated to be at STP, it will be at 1 atm and 273 kelvin
Molar Mass, Density, and Molar Volume
- Molar mass can be determined if P, V, g, and T are known
- PV = (g/molar mass)RT
- Density can be determined if P, T, and molar mass are known
- P(molar mass) = (g/V)RT
- Molar volume can be determined by rearranging the ideal gas law equation
- (V/n) = (RT/P) or
- (V/n) = 22.4 L/mol if at STP
Kinetic Molecular Theory
- Kinetic molecular theory describes gasses at the particle level.
- Gasses consist of molecules or atoms in continuous random motion
- Collision between molecules/atoms are elastic
- Volume taken up by gaseous molecules is negligibly small
- The attractive and repulsive forces between gaseous molecules is negligible
- Average kinetic energy of gaseous molecules is directly proportional to the Kelvin temperature of the gas
- Pressure is determined by the velocity of gas particles colliding with container walls. Changing temperature changes the force of collision in addition to the frequency.
- If the volume of a container is decrease, the particles will collide with the wall more frequently, and pressure will increase
- By increasing temperature, the average kinetic energy is increase so the particle velocity is increased, and the pressure will increase since the collisions are stronger
- Graham’s law of effusion compares the rate of effusion of two gasses and says the rates are inversely related to the square root of the mass of the gas particles
- Effusion through a pinhole in a vacuum requires a gas to hit the pinhole just right in order to escape. More collisions mean a higher rate of effusion, or a higher likelihood that it will escape.
Average Kinetic Energies and Velocities
- Average kinetic energy is sometimes higher or lower than estimated.
- KE = (.5)mv^2
Real Gasses
- The ideal gas law does not work well at very high pressures or very low temperatures
- Gases close to the condensation point will deviate slightly because it breaks two gas assumptions: gasses have no volume and have no repulsive/attractive forces
- An ideal gas must follow the assumptions stated earlier.
Dalton’s Law of Partial Pressures
- Dalton’s law of partial pressures says that if two gasses are mixed together, they will act independently of each other.
- Total pressure is the sum of all partial pressure of gasses in a container
- Example
- A mixture of gasses contain 2 mol of O2, 3 mol of N2, and 5 mol of He. Total pressure is 850 torr. What is the partial pressure of each gas?
Experiments Involving Gases
- Pneumatic troughs are used to collect gases produced in a reaction vessel.
- To find the gas collected in pressure,
- Pgas = Patm - Pwater