Unit 4: Chemical Reactions
Types of Reactions
- Synthesis reaction: when elements or simple compounds are combined to form a single, more complex compound. A+B → AB
- Decomposition: opposite of synthesis-occurs usually in the presence of heat. AB → A + B
- Acid-base: when an acid (H+) reacts with a base (OH-) to form water and a salt. HCl (aq) + NaOH (aq) → NaCI (aq) + H2O (l)
- Oxidation-reduction (redox): results in change of oxidation state (only for some). Cu 2+ (aq) + 2 e- → Cu (s)
- Hydrocarbon combustion: carbon + hydrogen (and sometimes oxygen) is ignited and produces CO2 and water, sometimes other compounds as well depending on the elements present. C4H10 + O2 → CO2 + H2O
- Precipitation: two aqueous solutions mixing to create a solid precipitate. A (aq) + B (aq) → C (aq) + D (s)
- Some solubility rules:
- Compounds with alkali metals and ammonium are always soluble.
- Compounds with nitrate are always soluble.
Chemical Equations
Balancing Chemical Equations and Calculations
- Stoichiometry steps:
- Covert given quantity to moles → use equation coefficients to determine limiting reactant → use balanced equation to determine how much product generated → convert moles to desired unit.
Percent error
- % error = (|experimental value - expected value| / expected value) x 100%
Combustion Analysis
- Because of the law of conservation of mass, when a hydrocarbon is combusted, all of the carbon in the hydrocarbon will create CO2 and all of the hydrogen will end up as H2O.
Gravimetric Analysis
- Gravimetric analysis is used to determine an unknown element in a precipitate reaction.
- A 4.33 g sample of an unknown alkali hydroxide compound is dissolved completely in water. A sufficient solution of copper (IL) nitrate is added to the hydroxide solution such that it will fully precipitate copper (Il) hydroxide via the following reaction: Cu 2+ (aq) + 2 OH- (aq) → Cu (OH)2 (s). After the precipitate is filtered and dried, its mass is found to be 3.81 g. Is the original alkali hydroxide sample most likely LiOH, NaOH. or KOH?
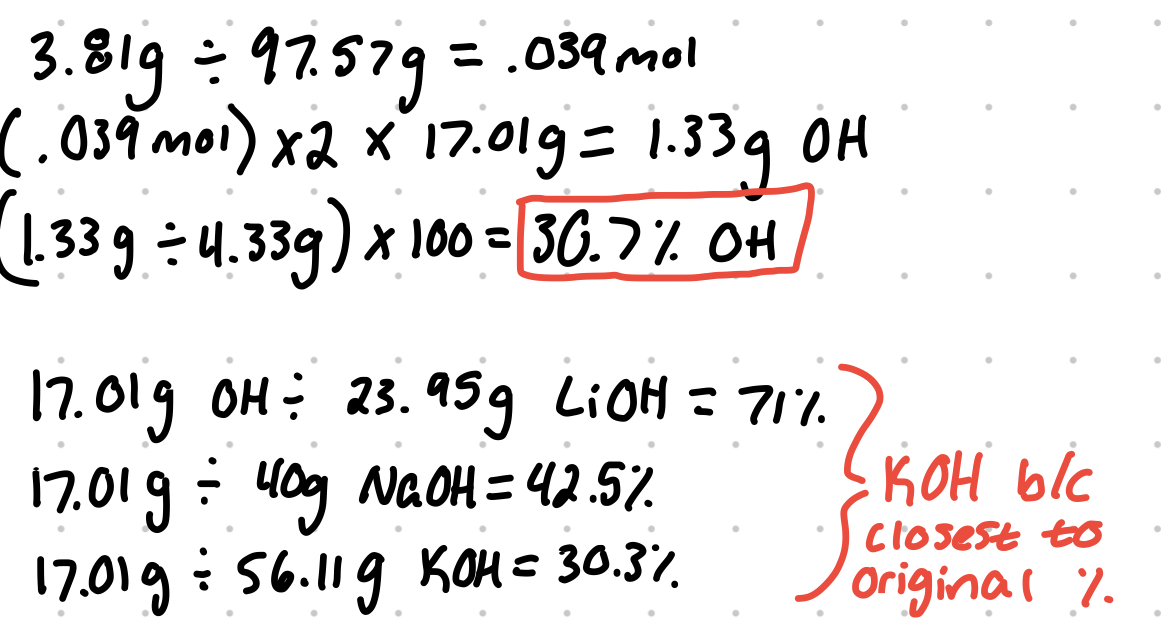
Oxidation States
- In chemical reactions, electrons are transferred between reactants and can be determined with oxidation states, which have several rules.
- Any neutral atom not bonded has an oxidation state of zero.
- A singular atom has an oxidation state equal to it's charge.
- In most compounds, oxygen is -2. One exception is H2O2, where oxygen is -1
- When bonded to a nonmetal, hydrogen is +1. When bonded to a metal, hydrogen is -1.
- When oxygen is not present, the most electronegative element has a state equal to it's most common charge.
- The combined oxidation states must be equal to the charge of that compound
Oxidation-Reduction Reactions
- In redox reactions, electrons are swapped between reactants and the oxidation states of the reactants are changed.
- In Fe + 2 HCL → FeCl2 + H2, iron's state goes from 0 to +2 and H goes from +1 to 0.
- Fe was oxidized
- It can be written as a half reaction
- Fe → Fe 2+ + 2e- oxidation
- 2 H+ + 2e- → H2 reduction
Redox Titrations
- To determine the concentration of an unknown solution, a chemical added until usually a color change occurs.
- If KMnO4 is added to a solution with MnO4 (which is a deep purple), the Mn is reduced and a color change occurs. If KMnO4 is added to a colorless solution that can be oxidized, it will turn pink.
Acids and Bases
- An acid is a substance capable of donating a proton (H+) and a base is considered a substance capable of accepting a proton.
- HC2H3O2 + H2O ⇌ C2H3O2- + H3O-
- — HC2H3O2 and H3O are acids while H2O and C2H3O2 are bases
- The H+ ion is the acid and the one without is the base. These are called conjugate pairs
- HC2H3O2 and C2H3O2-
- H2O and H3O-
- However, water can sometimes act as an acid, this is called amphoteric