organic mechanisms
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
chlorination of alkanes conditions and equations
UV light
Cl-Cl -> .Cl + .Cl
.Cl + H-CH3 ------> Cl-H + .CH3
.CH3 + Cl - Cl -------> CH3Cl + .Cl
.Cl + .CH3 ------> CH3Cl
.Cl + .Cl -----> Cl2
.CH3 + .CH3 -----> C2H6
what reactions do halogenoalkanes undergo
nucleophilic substitution
elimination
nucleophilic substitutions of halogenoalkanes with :OH-
potassium/sodium hydroxide
warm and aqueous
nucleophilic substitutions of halogenoalkanes with :CN-
aqueous, ethanolic and warm
potassium cyanide
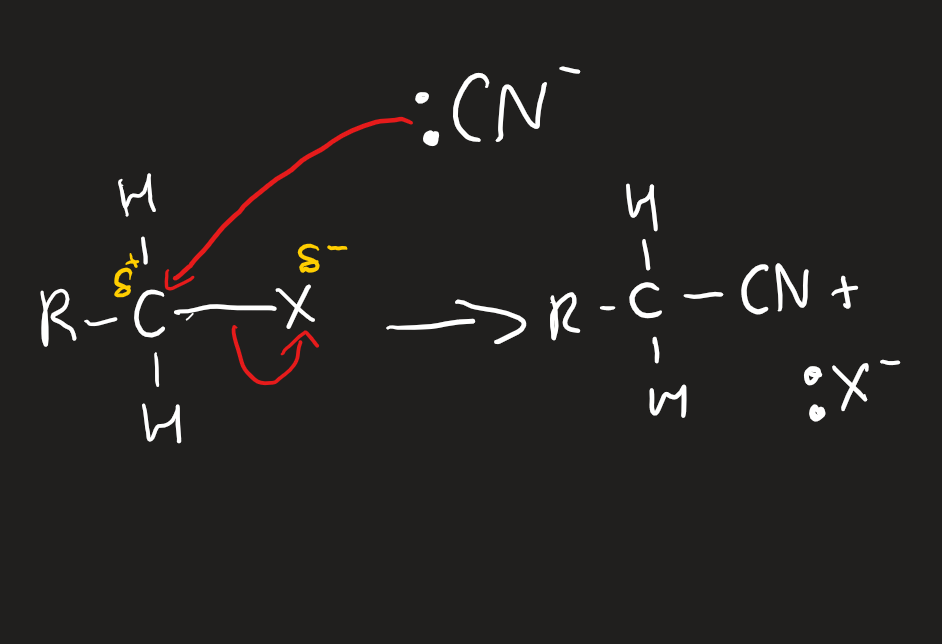
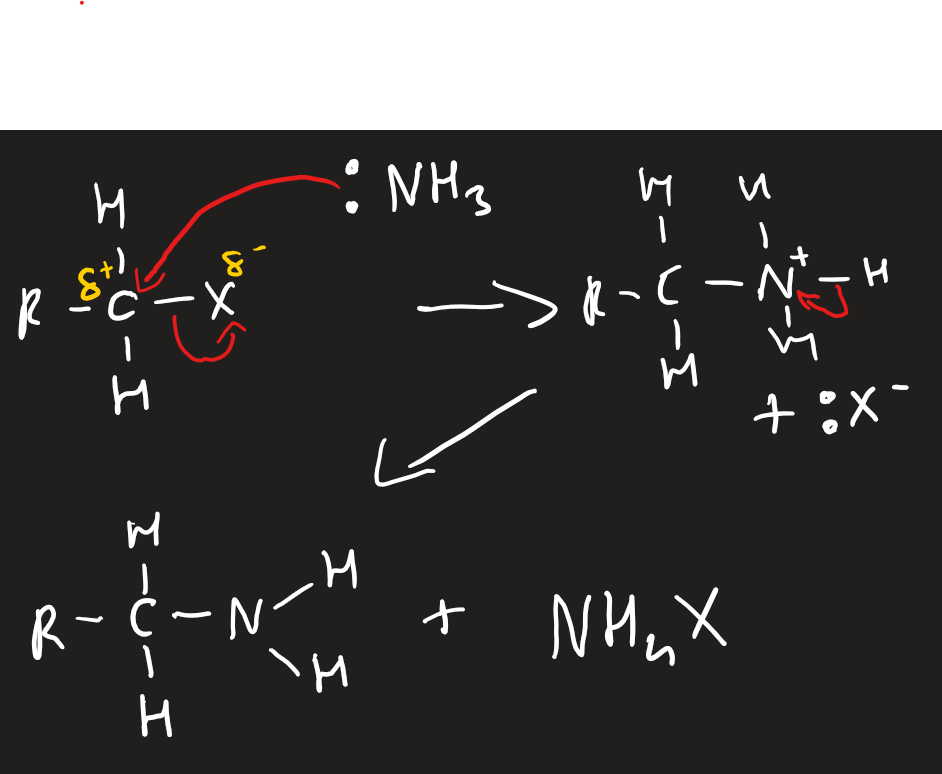
nucleophilic substitutions of halogenoalkanes with :NH3
excess concentrated ammonia dissolved in ethanol at pressure in a sealed container
need two ammonias per reaction to make an ammonium halide as well as amine
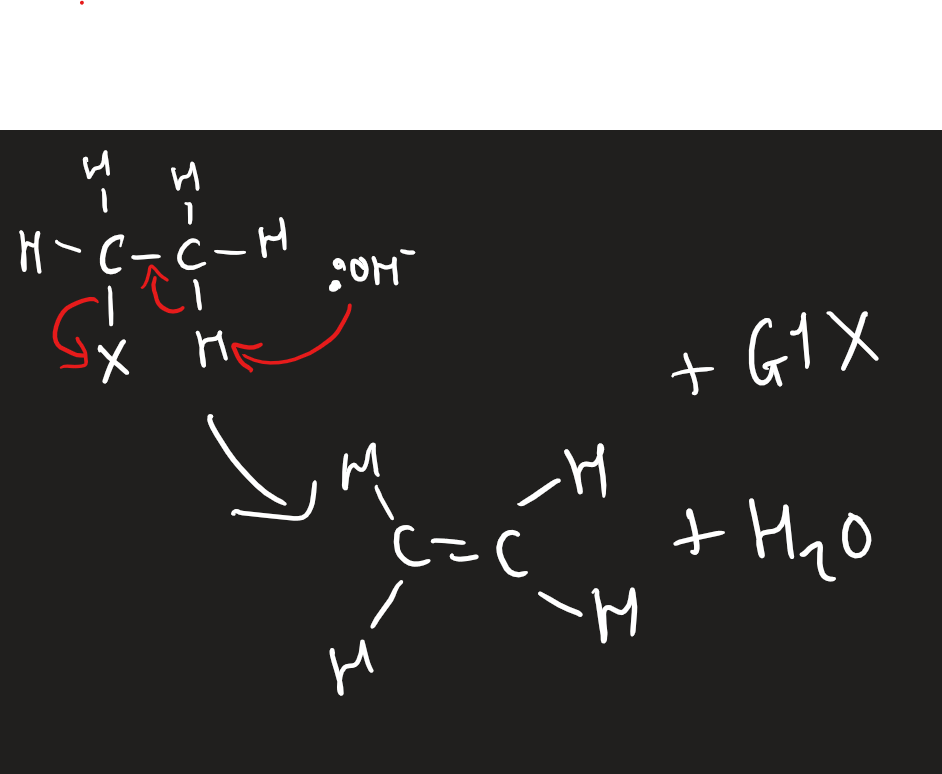
elimination of halogenoalkanes with :OH-
hot and ethanolic
potassium/sodium hydroxide
what reactions do alkenes undergo
electrophilic addition
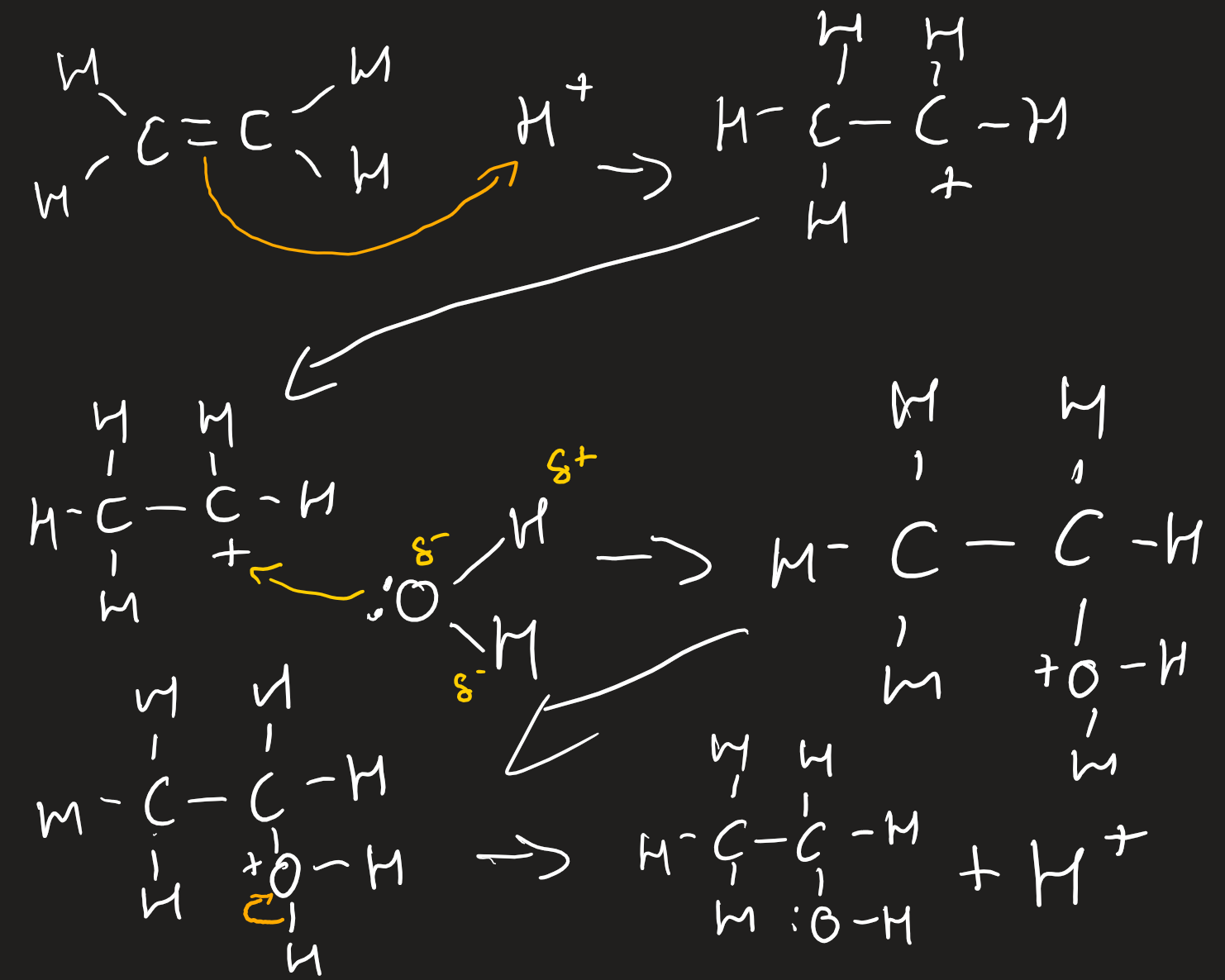
electrophilic addition of alkenes with hydrogen halides
make sure to think about
Major product formed via more stable carbocation
State which one is the more stable and that the major came from that
Due to positive inductive effect of three alkyl/two alkyl groups rather than one
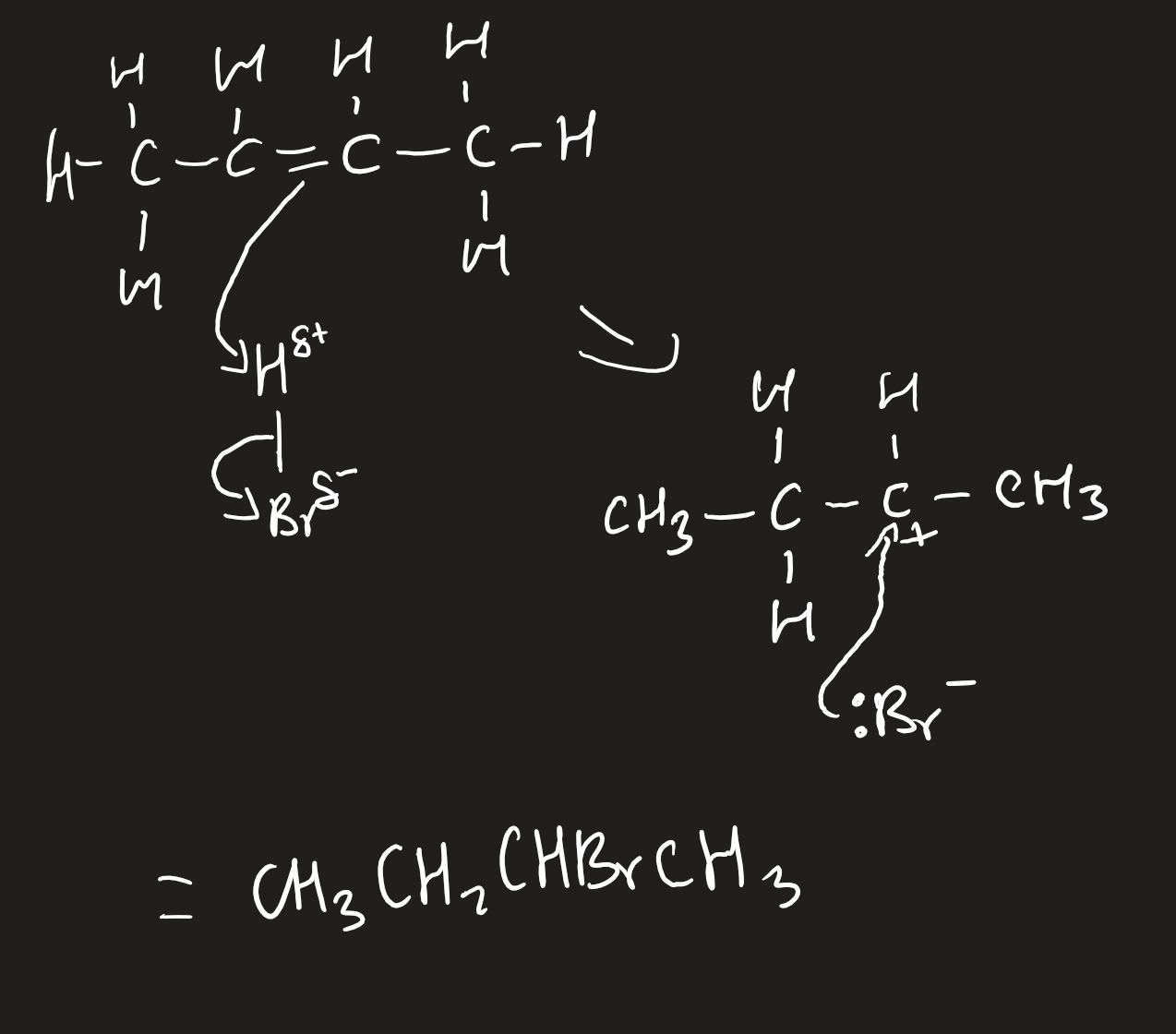
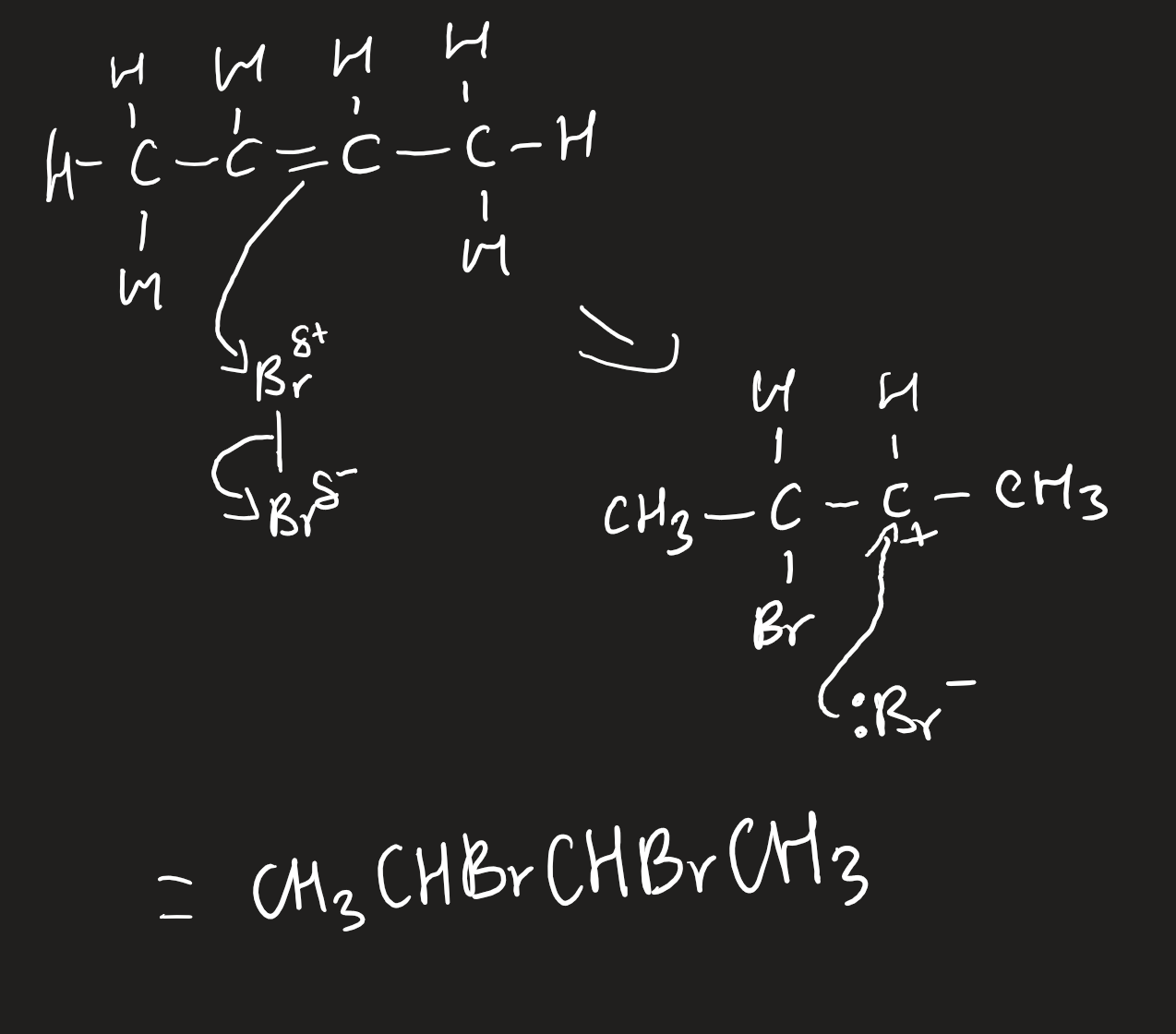
electrophilic addition of alkenes with halogens
aqueous
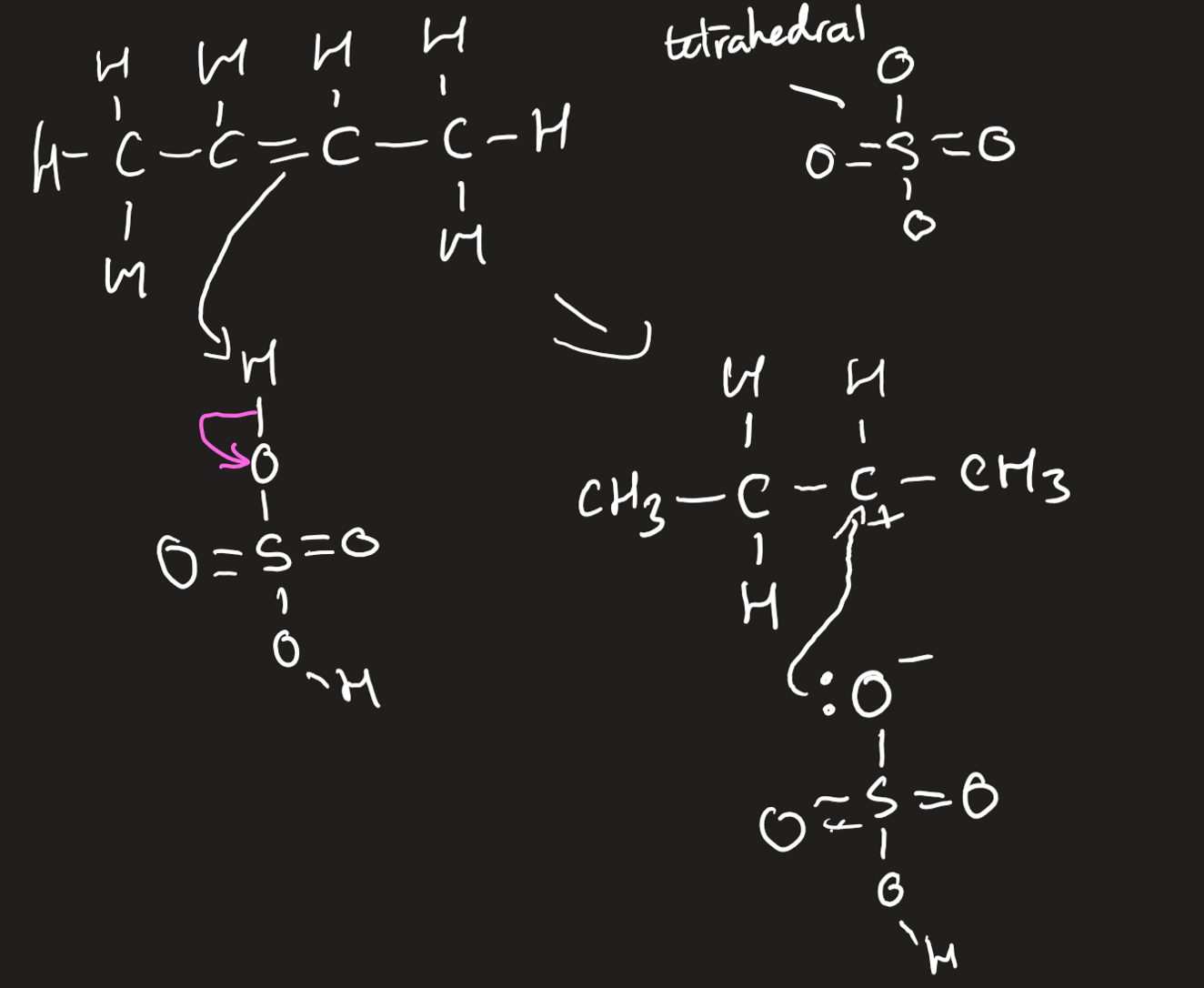
electrophilic addition of alkenes with concentrated sulfuric acid
must be concentrated and cold
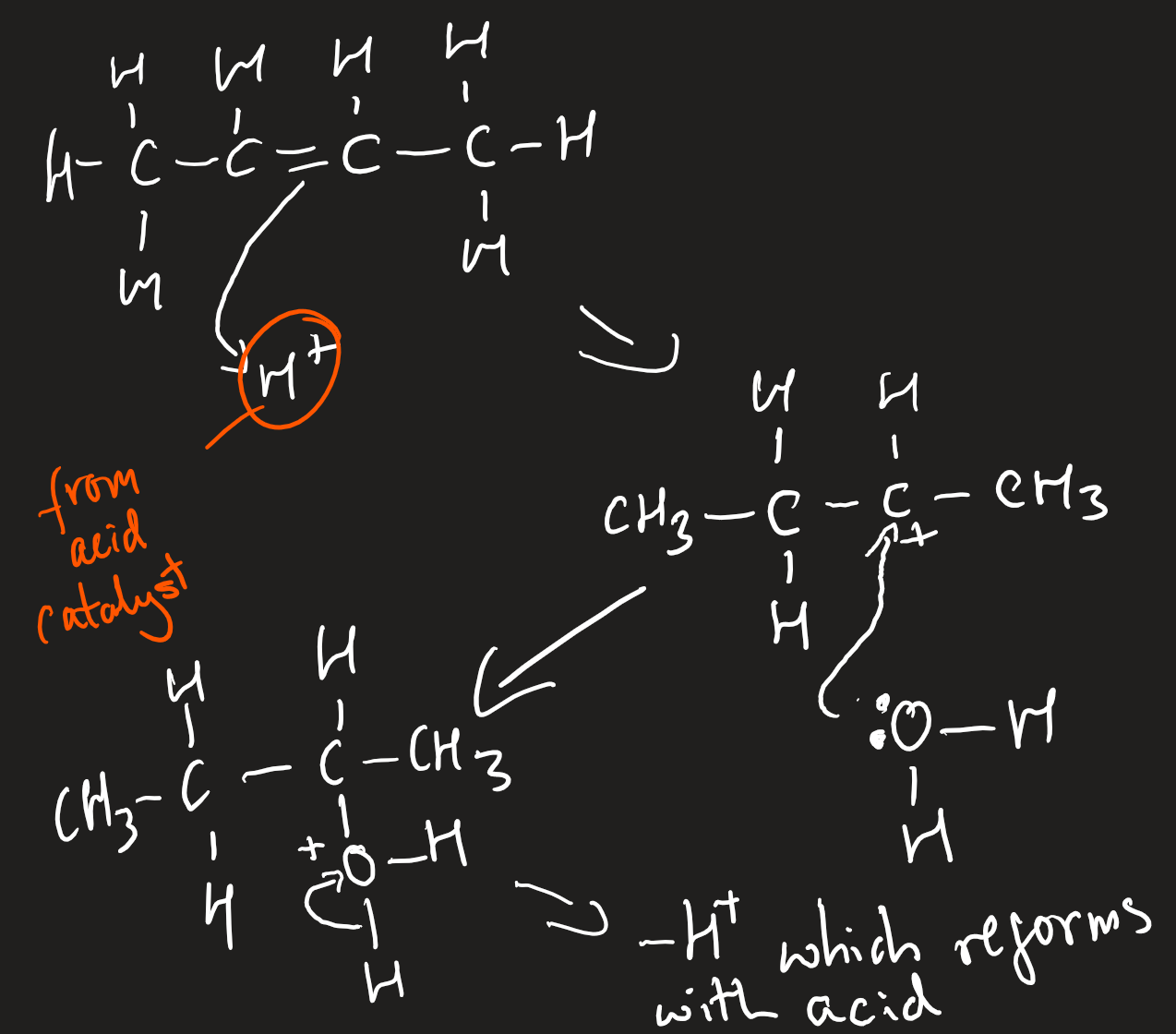
electrophilic addition of alkenes with water
steam with strong sulfuric/phosphoric acid catalyst
300*C and 70atm
alcohol formed
what reactions do alcohols undergo
oxidation and elimination
how is ethanol formed from ethene
70atm
phosphoric acid catalyst
300*C
ethanol from fermentation
35*C, anaerobic, yeast
ethene from ethanol = elimination
hot
conc sulfuric acid
180*C
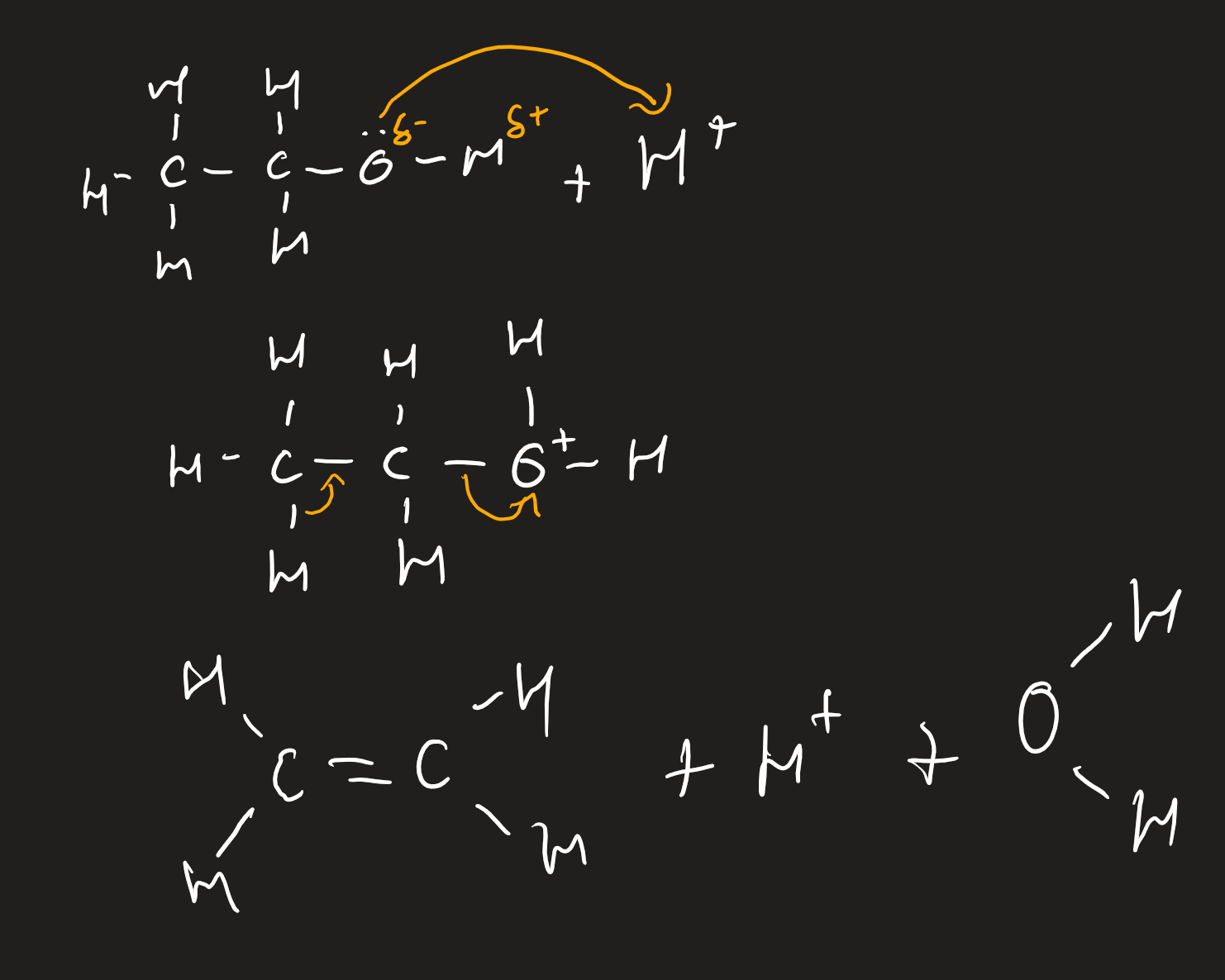
oxidation
acidified potassium dichromate (VI)
with sulfuric acid catalyst
heat to boiling point to go to aldehyde and immediately distil off
reflex - heating to boiling point for prolonged period of time for carboxylic acid
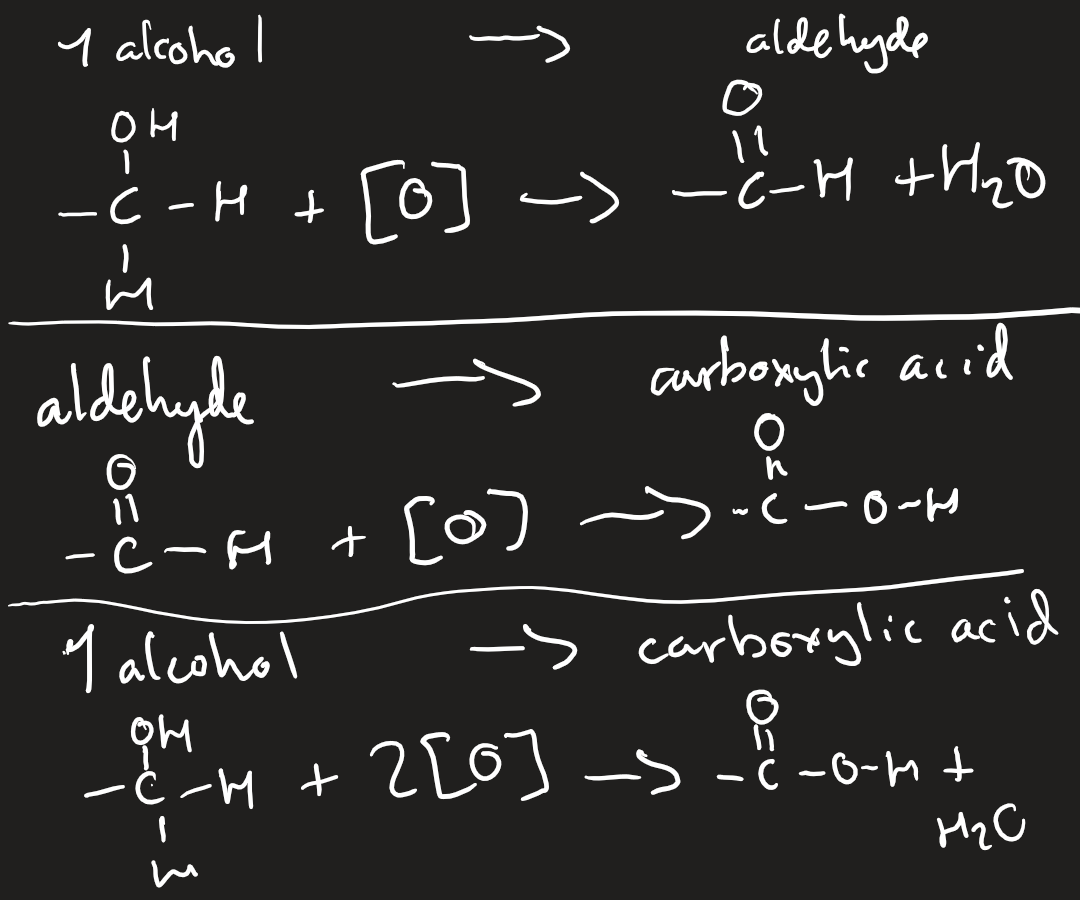
also Tollens and Fehlings are oxidation
Tollens - silver mirror formed on warming with aldehyde
Fehlings - on warming with aldehyde, blue solution to brick-red precipitate
what reactions do aldehydes and ketones
nucleophilic addition and reduction
reduction of aldehydes and ketones equation
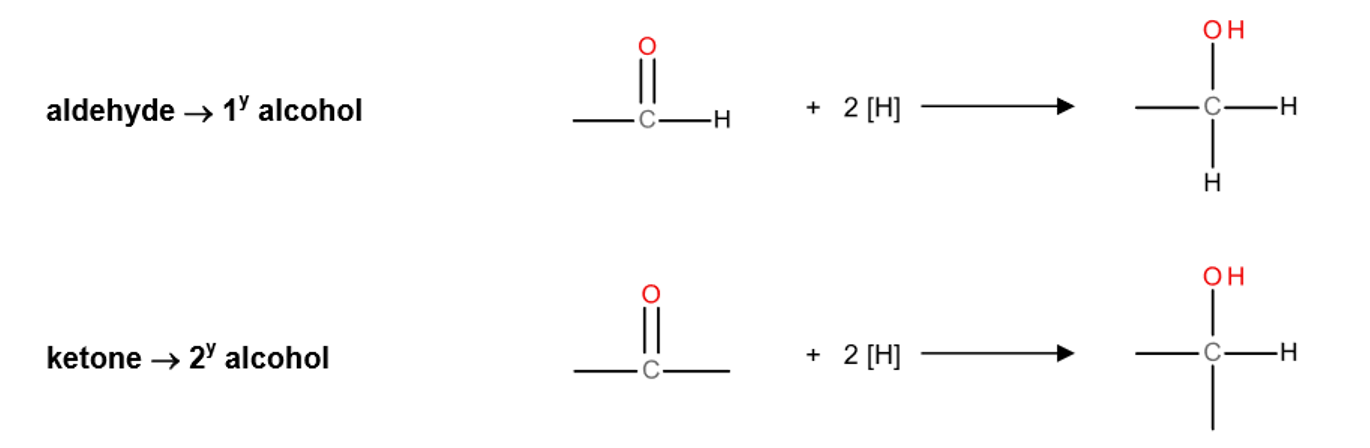
nucleophilic addition of aldehydes and ketones
aqueous with NaBH4
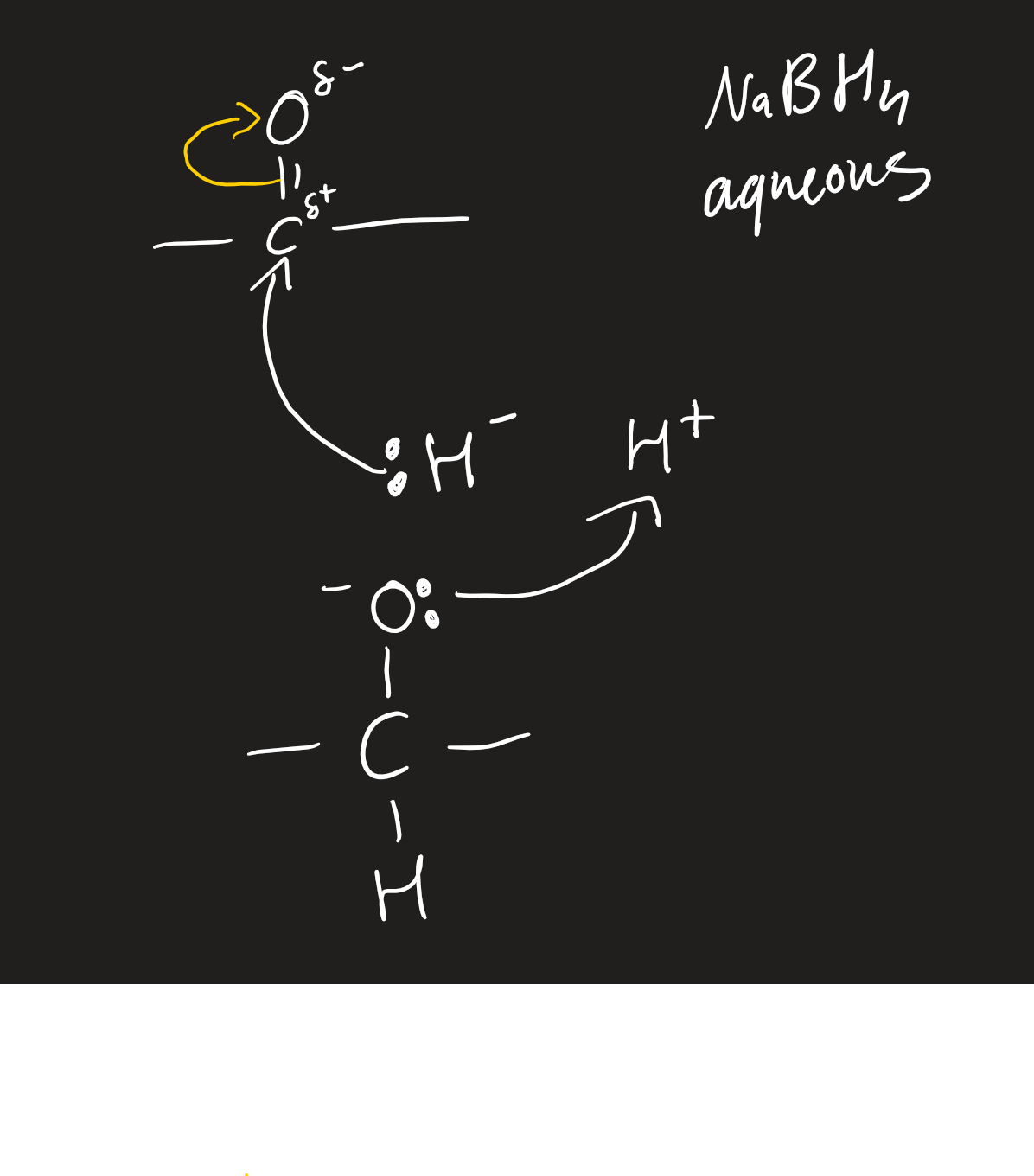
nucleophilic addition of aldehydes and ketones
sodium/potassium cyanide and dilute hydrochloric acid
aqueous
produce hydroxynitriles
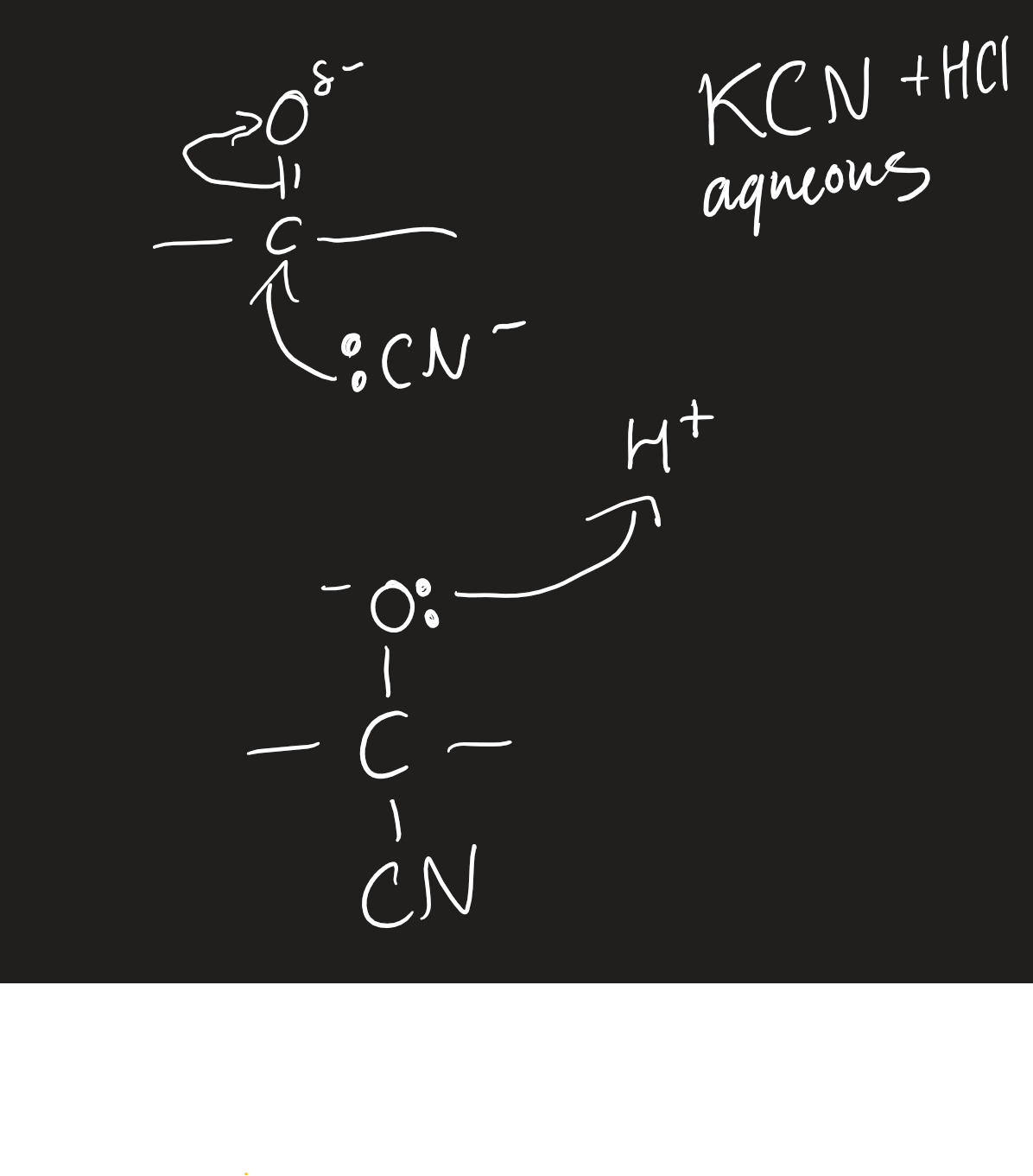
what reactions do carboxylic acids and derivatives undergo
esterification and nucleophilic addition-elimination reactions
esterification
strong acid catalyst - sulfuric/hydrochloric
heat/warm
reversible reaction so hydrolysis in acidic conditions is the same
hydrolysis of esters
aqueous base (NaOH)
warm
alcohol and salt of acid produced
not reversible
biodiesel
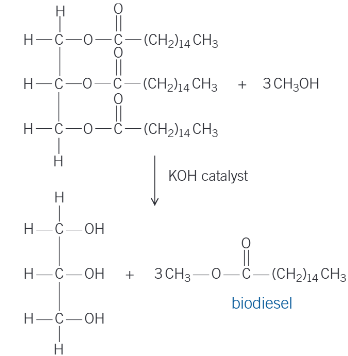
nucleophilic addition-elimination reactions
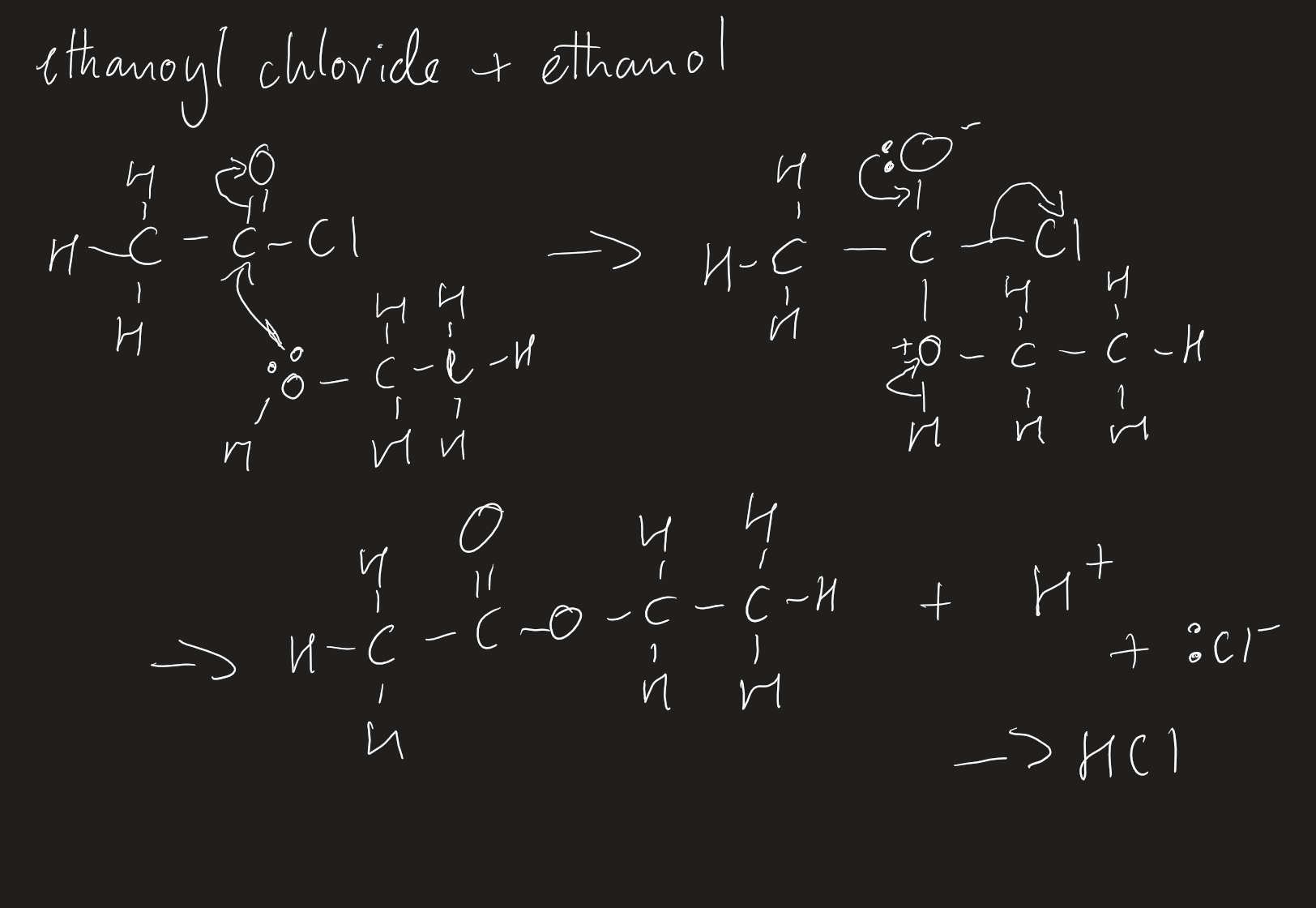
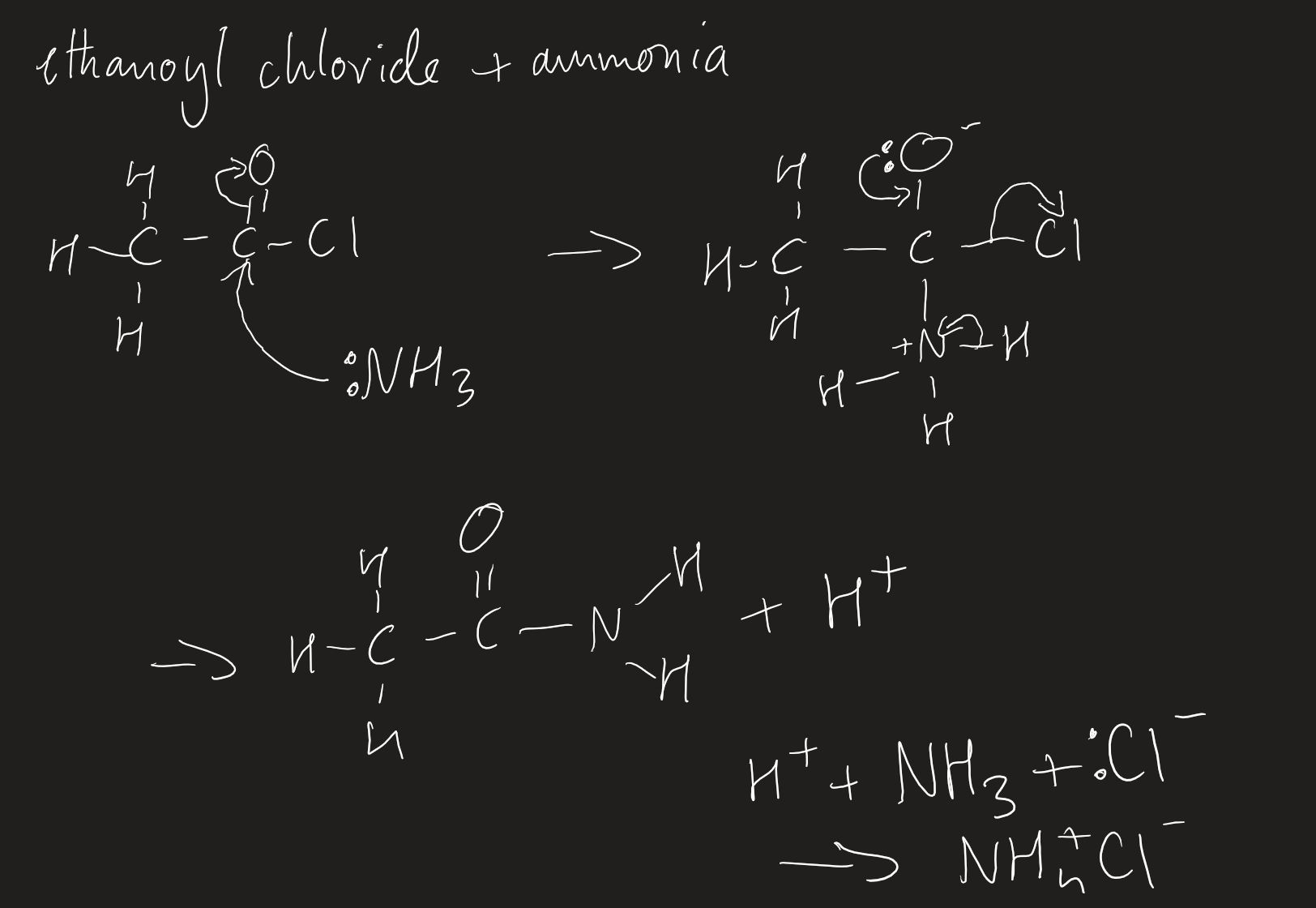
products with acid anhydrides and acyl chlorides
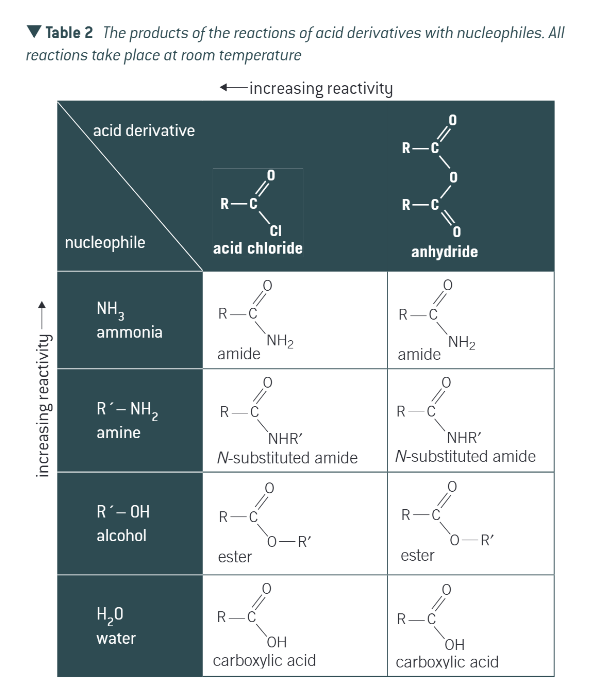
with water and alcohol
no special conditions
with ammonia and amine
two molecules of ammonia/amine needed
what reactions do aromatics undergo
electrophilic substitutions
electrophilic substitution with nitronium ion
50*C, concentrated nitric and sulfuric acid
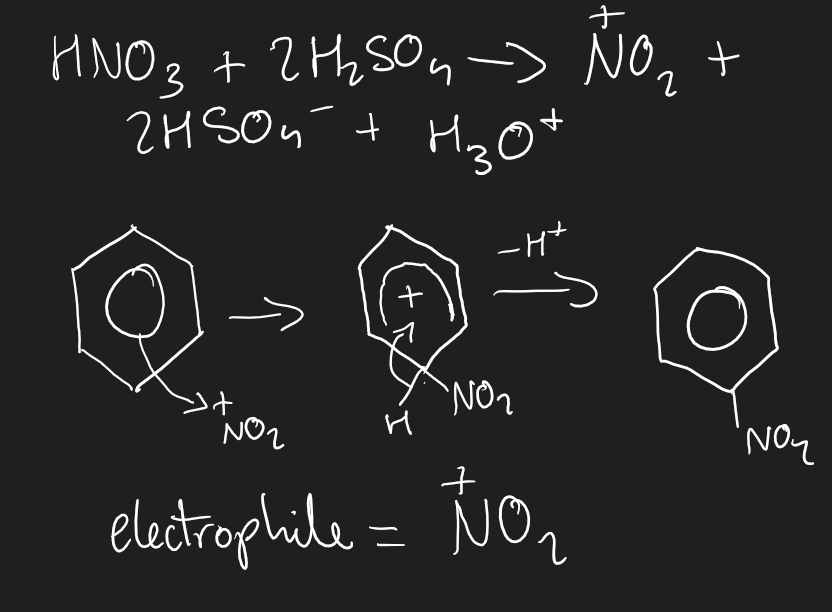
electrophilic substitution with acylium ion
acid anhydride/acyl chloride with AlCl3
aluminium chloride is catalyst
anhydrous to prevent reaction of AlCl3
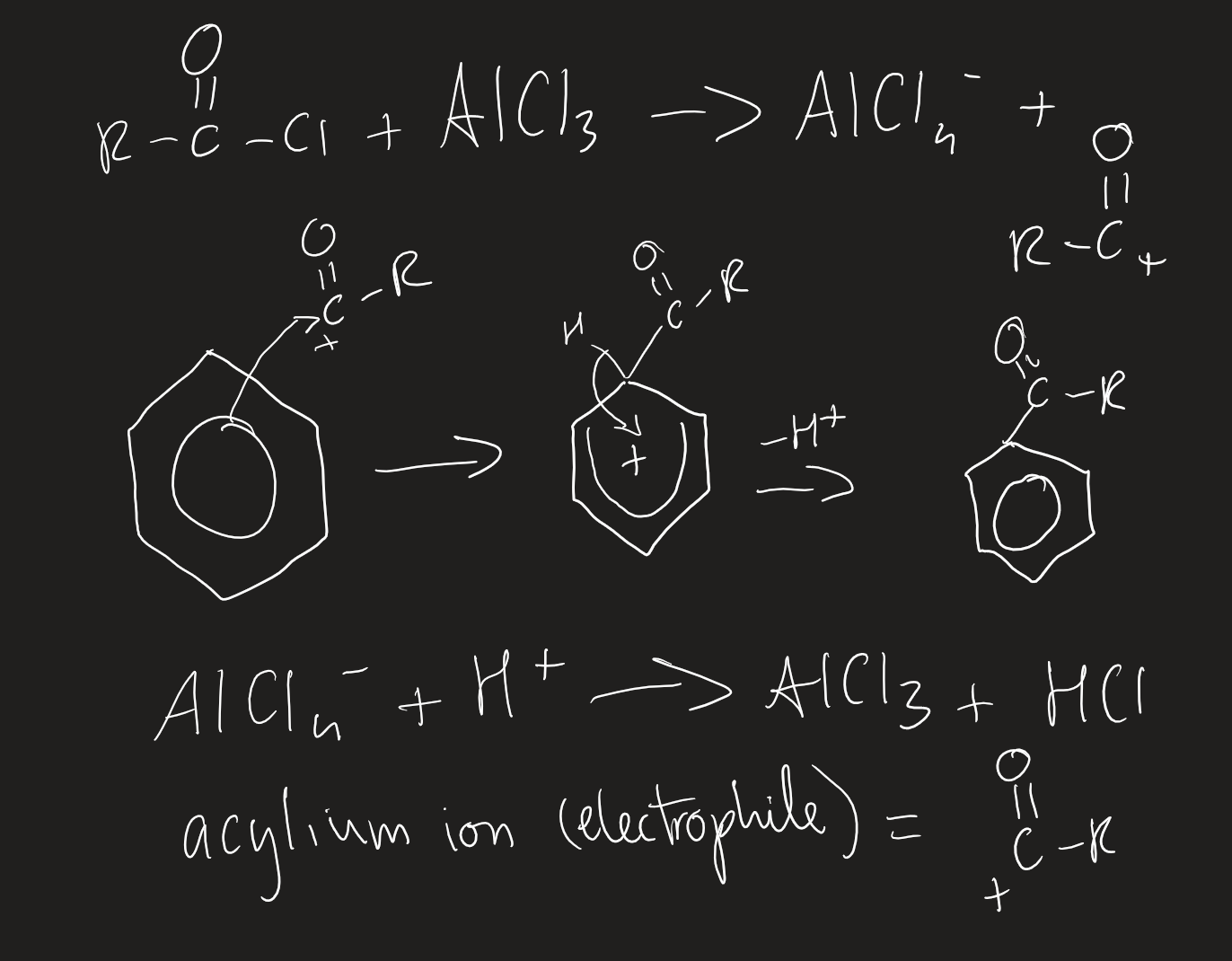
mechanism with acid anhydride
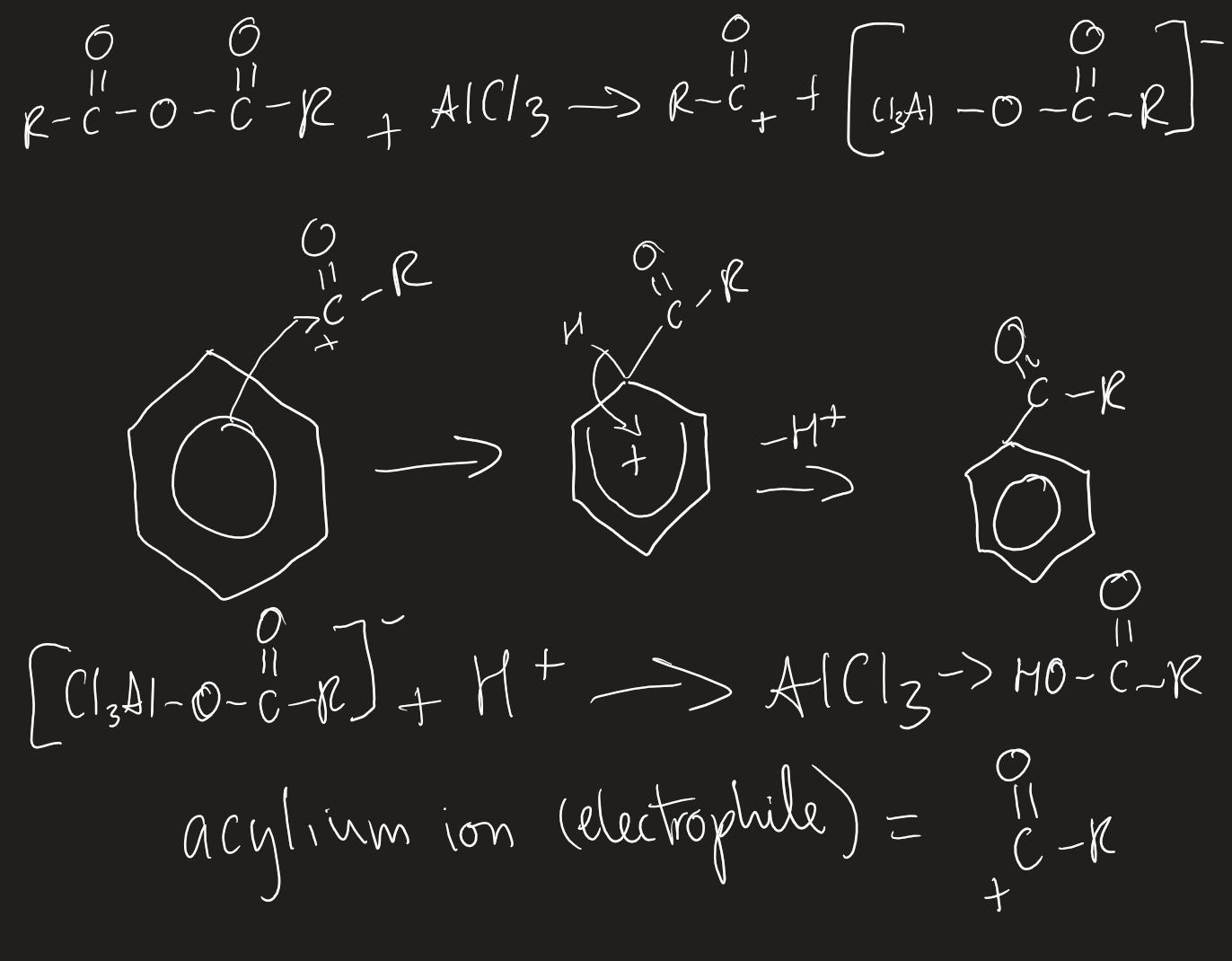
what reactions make amines and what reactions can amines/ammonia do
nucleophilic substitution with halogenoalkanes
reduction of nitriles
reduction of nitro compounds
amines and ammonia can undergo nucleophilic substitution repeatedly with halogenoalkanes and nucleophilic addition-elimination reactions with acyl chlorides and acid anhydrides
nucleophilic substitution with halogenoalkanes
same as the above one from that topic
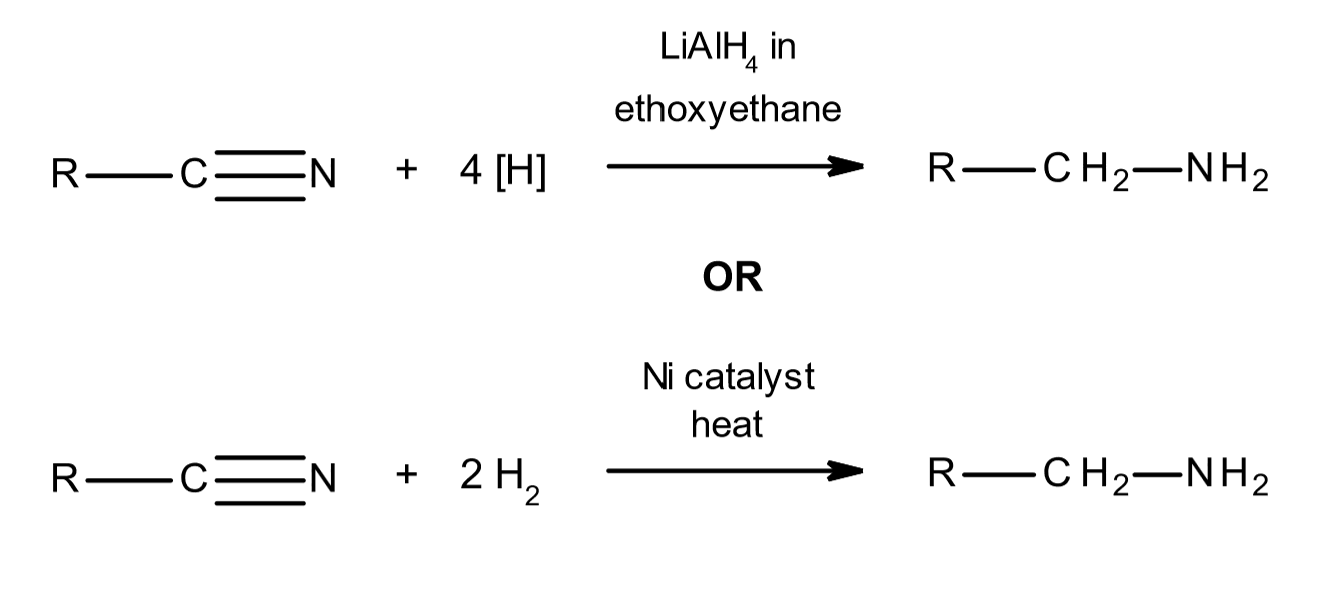
reduction with nitriles
first step is nucleophilic substitution of halogenoalkane with cyanide ion as above
then react with either:
LiAlH4 in ethoxyethane or nickel catalyst, heat and H2
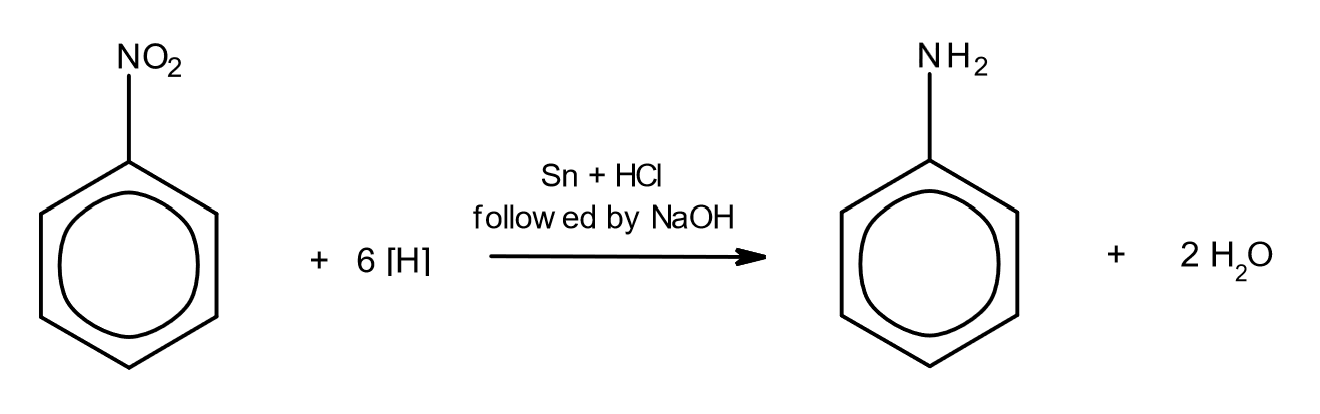
reduction of nitro compounds
react with Sn and HCl, followed by NaOH