orgo prelim 2 halides n stuff?
1/8
Earn XP
Description and Tags
halide? man why'd he do that
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
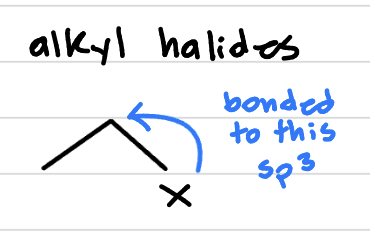
alkyl halide, is it reactive?
halogen right on a carbon, all single bonds. very reactive!
allyl halides, is it reactive?
halogen on a carbon that is Adjacent to a double bond. very reactive!

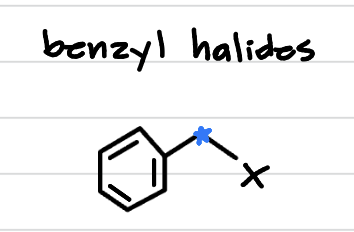
benzyl halide, is it reactive?
halogen on a carbon attached to a benzyl ring. very reactive!
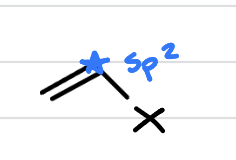
vinyl halide, is it reactive?
halogen right on an sp2 carbon (has its own double bond). unreactive!
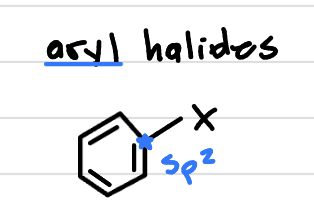
aryl halides, is it reactive?
halogen right on the benzyl ring (right next to a double bond). unreactive!
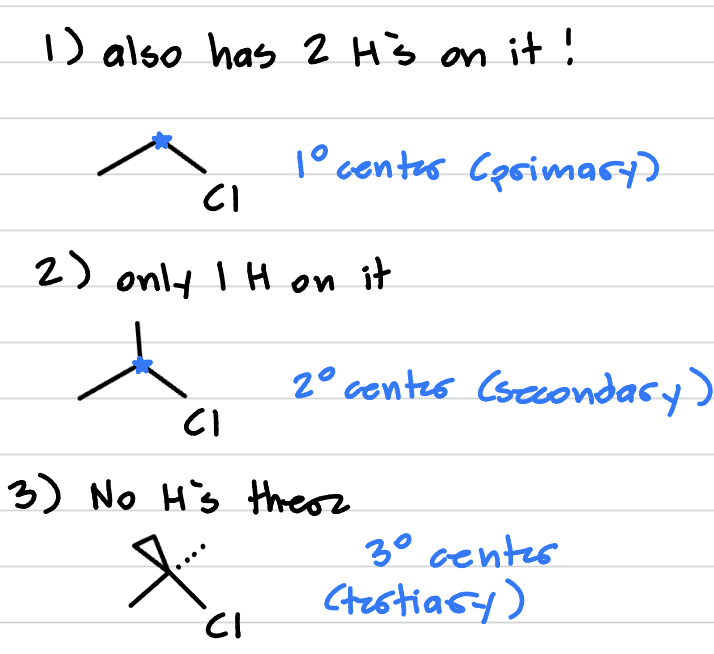
1° vs 2° vs 3° halides
1° (primary): has only 1 other carbon on it + the halogen
2° (secondary): has 2 other carbons on it + the halogen
3° (tertiary): has 3 other carbons on it + the halogen
do halides like being nucleophiles or electrophiles?
ALWAYS elecrophiles. they would rather die than be nucleophiles and go positive
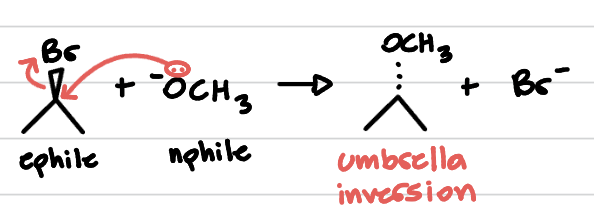
SN2 reaction mechanism and just. final outcome. solvents too!
UMBRELLA INVERSION!
basically, nphile comes and knocks LG off and replaces him but in the opposite direction.
concerted, so no CBCT intermediate
important: a bulky nphile (if u must) w/ smaller ephile (u MUST!!)
good LG’s are weak bases! and tosylate.
solvents: we love polar aprotic! (DMF, DMSO, HMPH, acetonitrite!) no OH or NH groups.
do NOT bring polar protic solvents around here.
SN1/E1 reactions