Chemistry Year 1
1/191
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
192 Terms
What are the Trends in Atomic Radius, Reactivity, Ionisation Energy, Boiling Point and Electronegativity down group 7?
Atomic Radius - The atomic radius increases down the group due to additional electron shells.
Reactivity - The reactivity decreases down the group due to additional electron shielding weakening the attraction from the nucleus to accept an electron.
Ionisation Energy - The ionisation energy decreases down the group due to greater atomic radius and increased electron shielding.
Boiling Point - The boiling point increases down the group due to increasing van der Waals forces holding the molecule together.
Electronegativity - The electronegativity decreases down the group due to increased atomic radius and electron shielding.
What is Oxidising Power and the trend down group 7?
Oxidising Power - The ability of a oxidising agent to accept electrons in a reaction and be reduced.
Oxidising Power/Ability decreases down the group due to their ability to attract electrons decreasing from electron shielding and atomic radius. This means that a halide will displace and halogen below it in the periodic table in a displacement reaction: Cl2(aq) + 2NaBr(aq) → Br2(aq) + 2NaCl(aq)
What is Reducing Power and the trend for halide ions down group 7?
Reducing Power - The ability of a reducing agent to donate electrons in a reaction and be oxidised.
Reducing Power/Ability increases down the group for halide ions. This is due it the ability to lose electrons increasing from electron shielding and atomic radius. This trend can be seen in the reactions of solid sodium halides with concentrated sulfuric acid.
Describe the reactions of Halide Ions with Concentrated Sulfuric Acid?
Fluoride and Chloride Ions:
NaF + H2SO4 → NaHSO4 + HF
NaCl + H2SO4 → NaHSO4 + HCl
Bromide Ions:
NaBr + H2SO4 → NaHSO4 + HBr
2HBr + H2SO4 → Br2 + SO2 + 2H2O
Iodide Ions:
NaI + H2SO4 → NaHSO4 + HI
2HI + H2SO4 → I2 + SO2 + 2H2O
6HI + SO2 → H2S + 3I2 + 2H2O
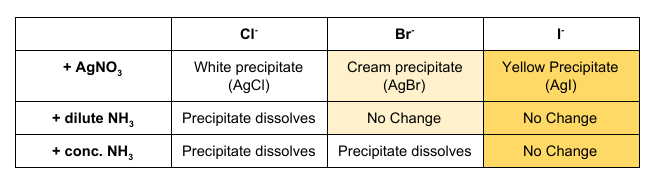
How can Halide Ions be distinguished using Silver Nitrate and Ammonia?
Acidified Silver Nitrate is used to test for halide ions as they react to form coloured precipitates. As the precipitate may not be clear to distinguish, different concentrations of ammonia can be added. Silver Nitrate is acidified to remove any ions that may interfere with the reaction.
See Table for Results.
How does Chlorine react with water and why is it used?
Chlorine reacts with water in a disproportionation reaction as chlorine is both oxidised and reduced: Cl2 + H2O → ClO- + Cl- + 2H+
Chlorine is used in small quantities to kill bacteria and sterilise water. Even though chlorine can be toxic, the benefits still outweigh the risks of using chlorine.
In the presence of UV light, chlorine decomposes with water, where chlorine is reduced in this reaction: 2Cl2 + 2H2O → 4HCl + O2
How can Chlorine be used in Bleach Production?
Chlorine can be mixed with cold, aqueous sodium hydroxide to produce sodium hypochlorite, a key ingredient in bleach: 2NaOH + Cl2 → NaClO + NaCl + H2O
This reaction is a disproportionation reaction.
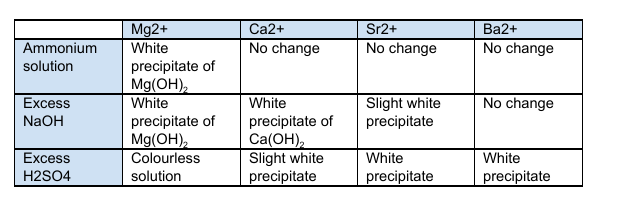
Required Practical 4 - Carrying out simple test-tube reactions to identify cations and anions?
Cations (Group 2 metals or Ammonia):
Group 2 Metals
Add 10 drops of your unknown solution, and 10 drops of dilute sodium hydroxide into a test tube
Stir the test tube and record any observations
Continue to add sodium hydroxide dropwise and continue to record observations
Once completed, dispose of any contents in a bowl of water and repeat
The dilute sodium hydroxide could be substituted with dilute sulphuric acid
The results are in the table attached
Ammonium Ions
Place 10 drops of your unknown solution in a test tube with 10 drops of sodium hydroxide and shake
Heat the mixture in a water bath gently and test the fumes produced
It should turn damp red litmus paper to blue
Anions (Halides, Sulfates, Hydroxides, Carbonates)
Hydroxides
Test a 1cm depth of your solution with damp red litmus paper
It should turn the litmus paper blue if it is a hydroxide
Carbonates
Add an equal volume of dilute HCl to your unknown solution and transfer the gas produced to a tube of lime water
The limewater will go cloudy if it is a carbonate
Sulfates
Add an equal volume of dilute HCl and barium chloride to your unknown solution
Once reacted add a bit more HCl to test if the precipitate dissolves
If the precipitate remains, sulphate or hydrogen sulphate ions are present
Halides
Silver Nitrate Test
What are the trends in Ionisation Energy, Atomic Radius, Reactivity and Melting Points down group 2?
Ionisation Energy - The first ionisation energy decreases down group 2 due to a greater atomic radius and increased electron shielding.
Atomic Radius - The atomic radius increased down group 2 due to additional electron shells.
Reactivity - Reactivity increases down group 2 due to increased electron shielding making the outer electrons easier to lose.
Melting Point - Melting Point decreases down group 2 due to the ions getting larger meaning the attractive forces are weaker as they have to travel a further distance.
What are the Reactions of Group 2 Elements with Water?
The group 2 metals increasingly react down the group with water in a redox reaction to produce a metal hydroxide and hydrogen. The metal hydroxides form as an alkaline solution.
Example Reaction: M(s) + 2H2O(l) → M(OH)2(aq) + H2(g), Where M is any group 2 metal
Magnesium reacts very slowly with water, but rapidly with steam as it provides the reaction with extra energy. It also causes magnesium to burn with a bright white flame to form magnesium oxide, a white powder: Mg(s) + H2O(g) → MgO(s) + H2(g)
What is the Solubility of Group 2 Hydroxides?
The solubility of group 2 hydroxides increases down the group. The hydroxides are all white solids. Magnesium hydroxide is sparingly soluble and Barium hydroxide is the most soluble.
As magnesium hydroxide is sparingly soluble, it is used in medicine (milk of magnesia) as an antacid as it is alkaline, and can neutralise acids.
Calcium hydroxide is used in agriculture to treat acidic soil and neutralise it due to its alkalinity.
What is the Solubility of Group 2 Sulfates?
The solubility of group 2 sulfates decreases down the group. Magnesium Sulfate is the most soluble and Barium Sulfate is insoluble.
As Barium Sulfate is insoluble, it is used in medicine as barium meals which allow internal tissue and organs to be imaged. It is toxic if it enters the blood steam but as it is insoluble it cannot be absorbed into the blood.
What are some other Uses of group 2 compounds?
Metal Extraction - Magnesium is used to extract titanium from titanium chloride via a displacement reaction: TiCl4 + 2Mg → 2MgCl2 + Ti
Flue Gas Removal - Calcium Oxide reacts with sulfur dioxide to remove it from factory pollutants and prevent it from reaching the atmosphere. It forms calcium sulfite and water: CaO + 2H2O + SO2 → CaSO3 + 2H2O
Testing Sulfate Ions - Acidified Barium Chloride solution is used as a test for sulfate ions as it reacts to form barium sulfate, a white precipitate when sulfate ions are present. It is acidified to remove interfering ions that cause a false positive. Ba2+ + SO42- → BaSO4(s)
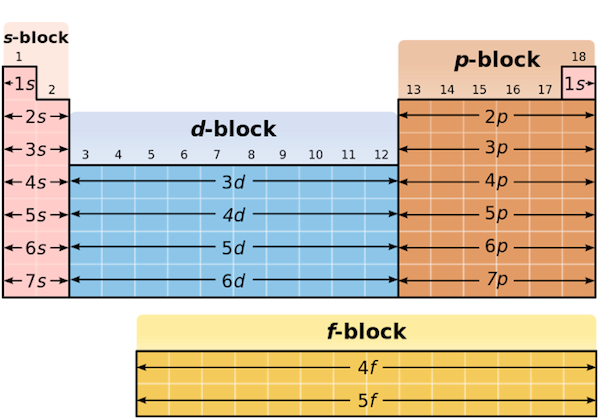
How are Elements classifies in the Periodic Table?
They are arranged due to proton number. Elements in a period have the same number of electron shells. Elements in a group have the same number of outer electrons. Elements are also classified into s-, p-, d- and f- blocks to show electron configuration.
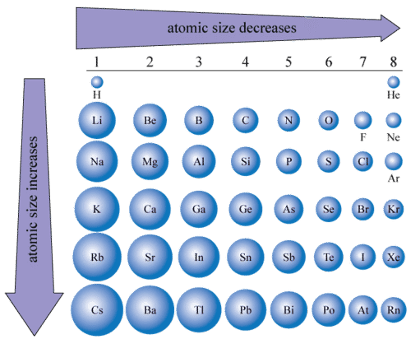
Explain the trend in Atomic Radius across a Period and down a Group?
Across a Period:
Atomic Radius decreases. This is due to an increased nuclear charge of the elements with the same number of electron shells. This means that there is a greater attraction from the nucleus so the outer electrons are pulled in closer, reducing the atomic radius.
Down a Group:
Atomic Radius Increases. This is due to an increasing amount of electron shells. This increases the distance between the outer electrons and the nucleus, reducing the attraction. More shells also increase nuclear shielding by blocking the attractive forces, reducing the nuclear attraction further, increasing the atomic radius.
Explain the trend in Ionisation Energy across a Period and down a Group?
Across a Period:
Ionisation Energy generally increases. This is due to the decreasing atomic radius and increasing nuclear charge, causing electrons to be attracted more to the nucleus, so more energy is required to overcome the attraction and remove the electron.
Down a Group:
Ionisation Energy decreases. This is due to the nuclear attraction between the nucleus and outer electrons reducing and the increasing amounts of shielding means that less energy is required to remove an outer electron.
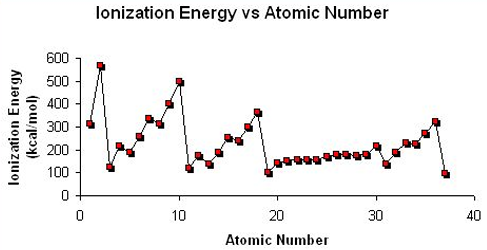
Explain the trend in Ionisation Energy across Period 3?
Ionisation Energy generally increases along Period 3. This is due to the decreasing atomic radius and increasing nuclear charge, causing electrons to be attracted more to the nucleus, so more energy is required to overcome the attraction and remove the electron.
There is a drop in First Ionisation Energy between groups 2 and 3 as it is now removing a p-block electron which is in a higher energy level compared to the s-electrons so less energy is required to remove it.
There is a drop in First Ionisation Energy between groups 5 and 6 as electrons are now being added to half-filled orbitals. This means that due to two electrons being in the same orbital, there will be electron-electron repulsion form the two negative charges, so it is easier to lose.
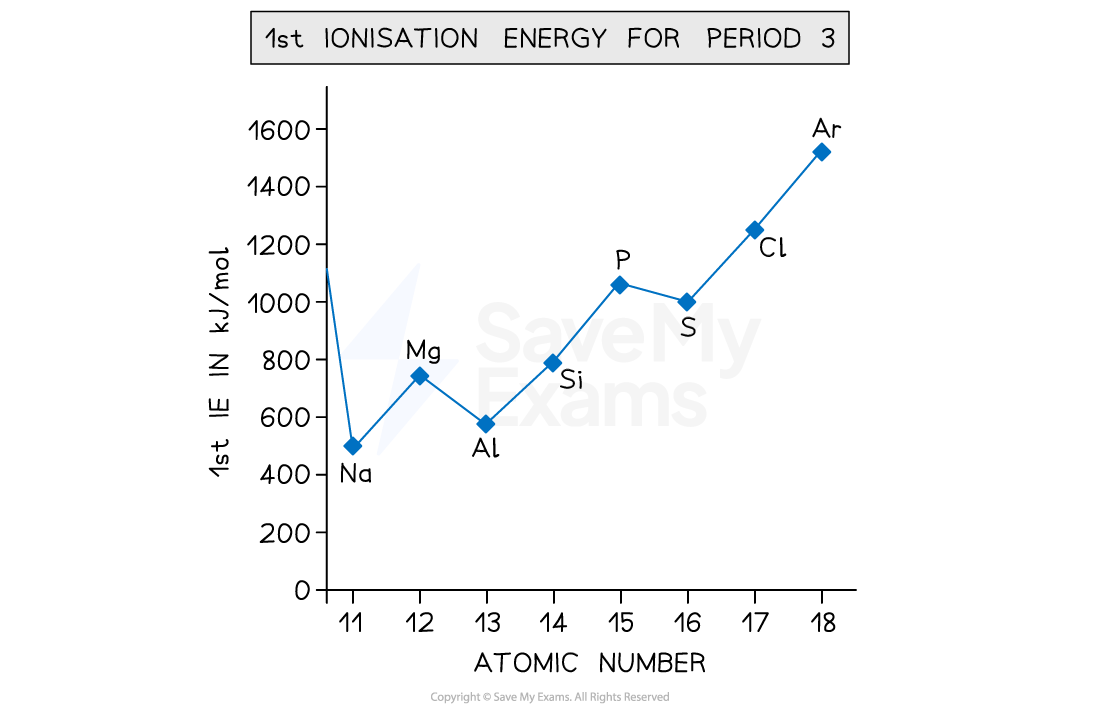
Explain the trend in Melting Point across Period 3?
Sodium - Aluminium:
These 3 elements all have metallic bonding. This means that their melting point increases based on charge. As your go along the period, the charge on the ion increases. This means that there are more electrons released so join the sea of delocalised electrons. As a result, the electrostatic forces increase so the metallic bond becomes stronger.
Silicon:
Silicon has a macromolecular structure. This means that it contains very strong covalent bonds which require lots of energy to overcome giving it the highest melting point.
Phosphorus - Chlorine:
These 3 elements are all simple covalent molecules (P4, S8, Cl2) held together by weak van der Waals forces. As these don’t require much energy to overcome their melting points are lower. The molecules with more electrons have more van der Waals forces so it has a higher melting point, making the order S8 > P4 > Cl2.
Argon:
Argon is a noble gas that exists as an individual atom due to its full outer shell. This makes it very stable but with very weak van der Waals forces. As a result, it has the lowest melting point causing it to exist as a gas at room temperature.
Describe bonding in Carbon Compounds?
In all stable carbon compound, carbon forms four covalent bonds and has eight electrons in its outer shell. These can be used to form rings of carbon or chains.
It can do this by forming 4 single bonds (Methane), forming two single bonds and one double bond (Ethene) or by forming one single bond and one triple bond (Ethyne)
The carbon-carbon bond is relatively strong and non-polar.
What are the different ways of writing and representing Organic Compounds?
Empirical Formula - The simplest whole number ratio of atoms of each element in a compound
Molecular Formula - The true number of atoms of each element in a compound
General Formula - All members of a homologous organic series follow its general formula (CnH2n+2)
Structural Formula - A representation of the unique arrangement of atoms in a molecule (CH3CH3). Branches in the carbon chain are shown in brackets (CH3CH(CH3)CH3)
Displayed formula - A graphic representation showing every atom and bond in a molecule
Skeletal Formula - A simplified graphic representation that shows only the bonds and any non-carbon atoms in a compound. The vertices are carbon atoms and hydrogen is assumed to be bonded unless stated otherwise (See Image)
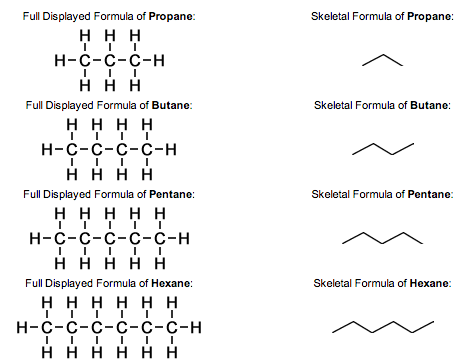
What is a Homologous Series?
A set of organic compounds with the same functional group and general formula but different carbon chain length. They react similarly and each member differs by CH2.
The length of the chain affects properties like melting point, boiling point and solubility.
Melting and Boiling points increase by a small amount as the number of carbon atoms in the chain increases due to the intermolecular forces increasing.
Chain branching also reduces melting point because the molecules pack together less well.
What is a Functional Group?
An atom or a group of atoms in an organic molecule which is responsible for the characteristic reactions of that molecule.
What are Reaction Mechanisms?
The series of simple steps that lead from reactants to products in a chemical reaction.
Curly arrows are used to show the movement of electrons. The arrow usually starts from a lone pair of electrons or from a covalent bond to a positively charged area of a molecule to form a new bond.
Free Radicals can also be shown when a covalent bond breaks away so that one electron goes to each atom in the original bond, resulting in the unpaired electrons making the atom extremely reactive.
What are the steps to naming a Organic Compound?
Root - The systematic name has a root which tells us the longest unbranched hydrocarbon chain or ring. Meth = 1, Eth = 2, Prop = 3, But = 4, Pent = 5, Hex = 6.
Suffix - The suffix indicates the functional group of the compound. Year 1 suffixes shown at bottom.
Chain/Position Isomers - The labelling of a side chain or functional group relative to the main chain. The number indicates the position of the functional group or branch, starting from either end to the nearest functional group. The substituents are listed in alphabetical order with their location numbers.
If you have more than one substituent you can use prefixes like di- , tri- and tetra-.
What is an Isomer?
Isomers are molecules that have the same molecular formula but have a different structural arrangement of atoms.
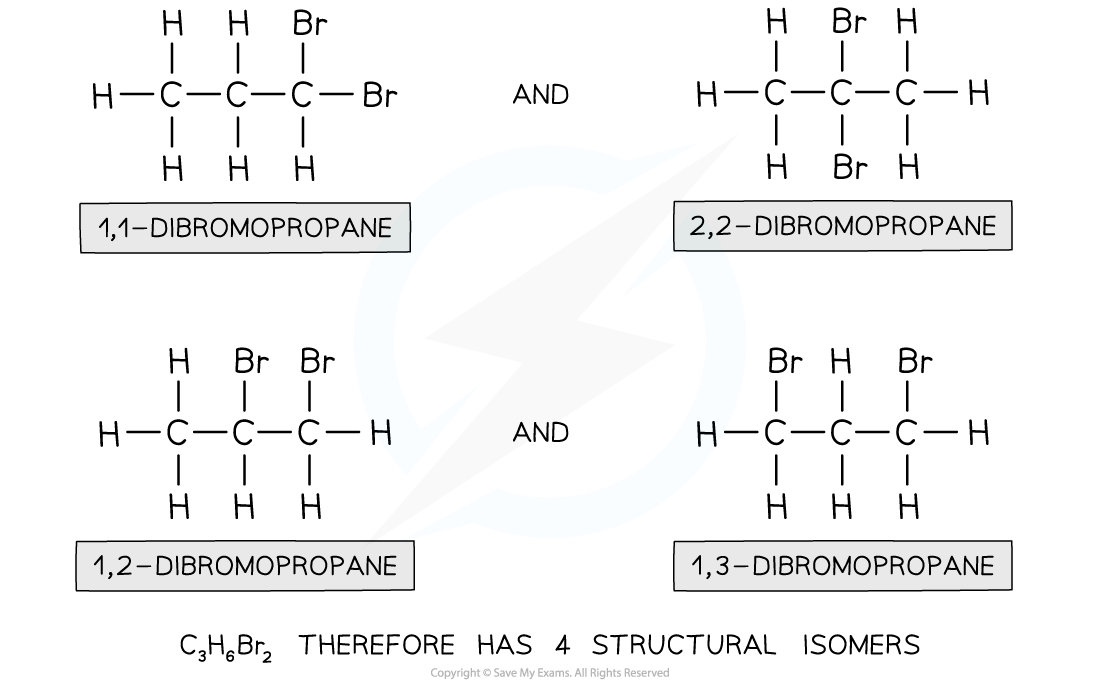
What are Structural Isomers?
A structural Isomer is a isomer with the same molecular formula but different structural formula. There are three sub-divisions: Positional, Functional and Chain Isomerism.
Positional Isomerism - Isomers with the same functional groups attached to the main chain at different points. C3H7Cl could represent 1-Chloropropane or 2-Chloropropane.
Functional Group Isomerism - Isomers that contain different functional groups. C2H6O could represent Ethanol or Methoxymethane (A Ether).
Chain Isomerism - Isomers that have a different arrangement of the hydrocarbon cain. C4H9OH could represent Butan-1-ol or 2-methyl-propan-1-ol.
What is Stereoisomerism?
Stereoisomerism is where molecules have the same molecular formula and the same structure but a different position of atoms in space. The two types are E-Z Isomerism and Optical Isomerism.
E-Z Isomerism - A type of stereoisomerism which tells us about the positions of substituents at either side of a carbon-carbon double bond. Two substituents on the same side begin with Z- and on the opposite sides begin with E- .
Substituted groups joined by a single bind can rotate around it so there are no isomers, but there is no rotation around a double bond so Z- and E- isomers are separate compounds.
Optical Isomerism is not needed in Year 1.
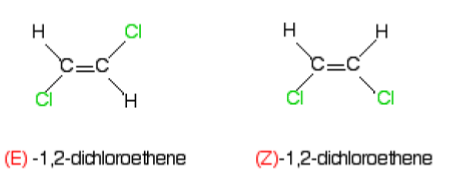
What are the Cahn-Ingold-Prelog (CIP) Priority Rules?
The CIP priority rules determine whether a compound has E or Z Isomerism. The first atom which is directly bonded to the carbon atom with the highest atomic number is given priority over the other atom bonded to it. If both the atoms with the highest atomic number are on the same side (Both top or bottom) then it is Z-, if not it is E-.
What are Alkanes?
Alkanes are Saturated Hydrocarbons meaning they contain olny carbon-carbon and carbon-hydrogen single bonds. They are among the least reactive organic compounds and are used for fuel and lubricants due to their stability.
General Formula - CnH2n+2
Functional Group - None
The C-C angle is approximately 109.5 degrees in alkanes.
Methane, Ethane and Propane have no isomers and after that the amount of isomers increases with the chain length.
What are the Physical Properties of Alkanes?
Polarity - Alkanes are almost non-polar because the electronegativities between carbon and hydrogen are so similar. As as result, there is only van der Waals forces between molecules.
Boiling Points - The increasing intermolecular forces with increasing molecule size cause an increase in boiling point as chain length increases.
Solubility - Alkanes are insoluble in water as the water molecules are held together by hydrogen bonds which are much stronger than the van der Waals forces that act between alkane molecules. However, alkanes do mix with other relatively non-polar liquids.
Reactions - They are relatively unreactive. They don’t react with acids, bases, oxidising agents and reducing agents. They do burn and react with halogens under suitable conditions.
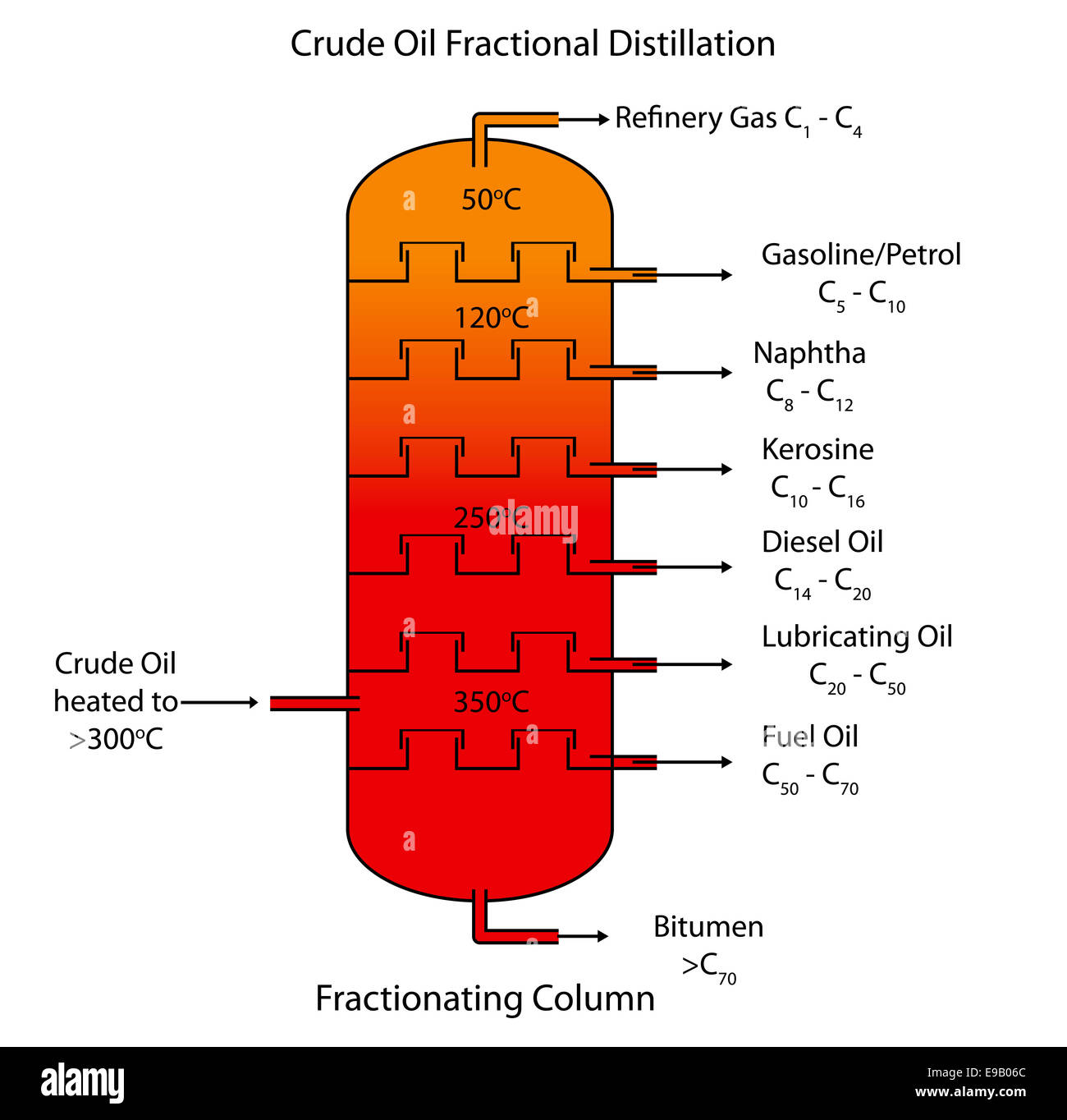
What is Fractional Distillation and Crude Oil?
Crude Oil is a mixture of different hydrocarbons that can be separated out into different molecules by Fractional Distillation
Fractional Distillation:
The Crude Oil is vaporised into the fractionating column
Inside there is a negative temperature gradient meaning it is cooler at the top and hotter at the bottom
The fractions are separated as longer chain hydrocarbons have higher boiling points so they condense into liquids first nearer to the bottom and shorter chain hydrocarbons condense near the top.
The products of Fractional Distillation are:
Refinery Gas - BP ~20, Fuels for Vehicles
Petrol - BP ~40-110, Fuel for Cars
Naphtha - BP ~110-180, making Chemicals and Plastics
Kerosene (Paraffin) - BP ~180-250, Jet Fuel
Diesel - BP ~250-350, Fuel for Diesel Engines
Fuel Oil - BP ~350-450, Fuel for Ships
Bitumen - BP ~450+ / Doesn’t Vaporise, Road Surfacing
What is Cracking?
Many of the shorter chain hydrocarbons like naphtha are in more demand than the longer chain hydrocarbons which you get more of in fractional distillation.
Cracking is used to break down these longer chain hydrocarbons into shorter more useful chains, as well also producing alkenes as another product which are much more reactive than alkanes.
The main two ways to carry out Cracking are Thermal Cracking and Catalytic Cracking.
What is Thermal Cracking?
A cracking process which involves heating alkanes to high temperatures of ~1200 K and under high pressure of ~7000 kPa.
The carbon-carbon bond then breaks in such a way that initially one electron from the pair goes to each carbon atom, resulting in two carbon atoms with an unpaired electron (A Free Radical).
As there are then not enough hydrogen atoms to produce two alkanes, one or more alkenes are produced.
Thermal Cracking produces a high proportion of alkenes and shorter chain hydrocarbons, making it effective for increasing the yield of useful products.
What is Catalytic Cracking?
A cracking process that takes place at a lower temperature of ~720k, a lower pressure compared to Thermal Cracking (But above atmospheric pressure) and uses a zeolite catalyst.
Zeolites have a honeycomb structure with an enormous surface area and are also acidic.
This form of cracking is used mainly to produce motor fuels and the products are mostly alkanes, cycloalkanes and aromantic compounds. The products are separated by fractional distillation.
Describe the two types of Combustion of Alkanes?
Combustion - The shorter chain alkanes burn completely in a plentiful supply of oxygen, producing carbon dioxide and water. Combustion reactions give out heat and have negative enthalpies. The more carbons present, the greater the heat output.
Incomplete Combustion - In a limited supply of oxygen, carbon monoxide is formed and with even less oxygen, carbon (soot) is produced.
What are some Pollutants and their problems?
Carbon Monoxide - A poisonous gas produced by incomplete combustion.
Nitrogen Oxides (NO, NO2, N2O4) - Oxides produced at high temperatures in engines, contributing to smog formation, acid rain and respiratory problems.
Sulfur Dioxide - Produced from sulfur impurities in crude oil and contribute to acid rain.
Carbon Particles/Particulates - Small solid particles that exacerbate asthma and cause cancer.
Unburnt Hydrocarbons - Compounds that escape combustion and enter the atmosphere, contributing to photochemical smog and many health problems.
Carbon Dioxide - A greenhouse gas formed from burning hydrocarbons and is linked to global warming and climate change.
What is Flue gas Desulfurisation?
Sulfuric acid is produced in the air when fuels containing sulfur are burned and the sulfur dioxide reacts with oxygen and water vapour. SO2 + ½ O2 + H2O → H2SO4
These gases given out by power stations are called Flue Gases, so the process of removing sulfur is flue gas desulfurisation.
One method is to spray a slurry of calcium oxide and water into the flue gas to form calcium solfite which can then be oxidised to form calcium sulfate (Gypsum). Gypsum is a saleable product used to make builder’s plaster.
CaO +2H2O +SO2 +1/2 O2 → CaSO4 . 2H2O
This can also be be done with Calcium Carbonate:
CaCo3 + ½ O2 + SO2 → CaSO4 + CO2
What are Catalytic Converters?
They are a honeycomb made of a ceramic material coated with platinum and rhodium metals (They have a large surface area) that act as catalysts to convert harmful emissions into less harmful gases:
2CO +2NO → N2 + 2CO2
These reactions take place on the surface of the catalyst, on the layer of metals.
How do Halogenoalkanes form and what are Free Radicals?
Alkanes react with halogens in the presence of UV light to produce Halogenoalkanes. The UV breaks down the halogen bonds producing reactive intermediates called free radicals.
Free Radicals - A chemical species with an unpaired electron that is usually highly reactive.
These Free Radicals attack the alkanes resulting in a series of reactions: Initiation, Propagation and Termination.
What is Initiation?
The first step of Free Radicals Substitution where the chlorine-chlorine bond is broke to form two chlorine atoms.
The chlorine molecule absorbs the energy of a single quantum of UV light which is enough to break the bond.
Since both atoms are the same, the bond breaks homolytically, meaning one electron goes to each chlorine. These chlorine atoms are free radicals.
There is not enough energy to break the carbon-hydrogen bonds producing reactive in a quantum of UV light.
What is Propogation?
A two stage reaction where the halogen radicals react with alkanes in a chain reaction, where the radical acts as a catalyst.
Step 1: A halogen radicals react with takes a hydrogen from the alkane to form a stable compound: Cl• + CH4 → HCl + •CH3
Step 2: The alkane radical produced now reacts with a halogen molecule, forming a halogen radical and a new stable molecule: •CH3 + Cl2 → CH3Cl +Cl•
The effect of these two steps is to produce a hydrogen halide, a haloalkane and a new halogen radical. These steps can take place 1000s of times before termination.
What is Termination?
The final step in Free Radical Substitution where the free radicals are removed by reacting together, stopping the reaction. However, radicals are more likely to meet a molecule instead of another radical so this step takes a long time.
What are Halogenoalkanes?
Halogenoalkanes are organic compounds where on or more halogen atoms replace the hydrogen atoms in a alkane. The general formula is CnH2n+1X, where X is the halogen. Their functional group is the C-X bond.
The C-X bond in halogenoalkanes is polar as the halogens are more electronegative than carbon (C is more positive, X is more negative). As you go down the halogen group, the bonds get less polar so the bond strength decreases.
What are Primary, Secondary and Tertiary Halogenoalkanes?
Primary - The halogen the carbon is attached to only has one carbon bonded to it.
Secondary - The halogen the carbon is attached to has two carbons attached to it.
Tertiary - The halogen the carbon is attached to has three carbons attached to it.
What are the properties of Halogenoalkanes?
Solubility - The polar C-X bonds are not polar enough to make the halogenoalkanes soluble in water. The main forces are dipole-dipole and van der Waals forces.
Boiling Point - The boiling point increases with chain length and down the halogen group. These are due to there being a larger molecule so increased van der Waals forces. Similarly to other groups, branching lowers the melting point.
What is the reactivity of Halogenoalkanes like?
When they react, the C-X bond usually breaks, with how long it takes being dependent on the C-X bond polarity and enthalpy.
Polarity - The polarity of the C-X bond would suggest that C-F is the most reactive as it is the most polar and C-I is the least polar.
Enthalpy - The bonds get weaker as they go down the group meaning C-F is the strongest bond and the least reactive, and C-I is the weakest bond and the most reactive.
Experiments confirm that C-I is the most reactive meaning that enthalpy is more important of a factor than polarity.
What is a Nucleophile and some common examples?
A nucleophile is a negative ion or a negatively charged molecule (due to a negative atom) that is able to donate a pair of electron and take part in an organic reaction by attacking an electron-deficient area in another reactant.
A nucleophile can do this as it has a lone pair of electrons situated on an electronegative atom which it can donate to form a new bond.
Common Examples:
-OH (Lone pair on the O)
NH3 (Lone pair on the N)
-CN (Lone pair on the C)
They replace the halogen in a halogenoalkane in a reaction called nucleophilic substitution.
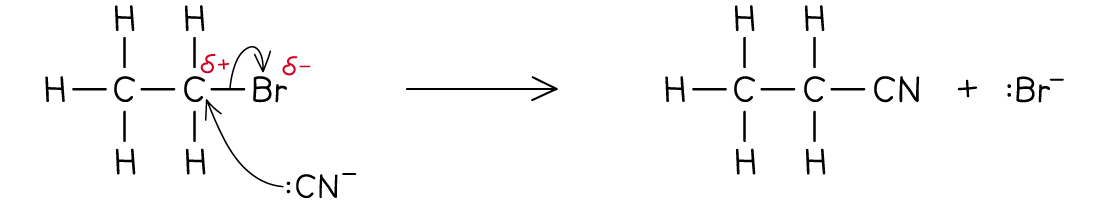
What is Nucleophilic Substitution?
A reaction where a nucleophile attacks a compound and replaces the leaving group, usually a halogen.
Leaving Group - An atom or group of atoms that is ejected from the starting compound, often taking an electron pair to form a negative ion.
A substitution between bromoethane and a cyanide ion is shown in the image.
What are some examples of Nucleophilic Substitution.
Halogenoalkanes with cyanide ions - Forms a nitrile and a halogen ion, the main compound also now has an extra carbon atom.
Halogenoalkanes with hydroxide ions - Forms a alcohol and a halogen ion
Halogenoalkanes with Ammonia - Forms an amine, a halogen ion and a Ammonia ions (NH4+)
Ammonia does not have a negative charge so it is a neutral nucleophile. This means that a positive ion will be left on the main compound so a proton must be lost by reacting with a second ammonia molecule forming NH4+ (See Image:)

What is a Base?
A base is a substance that can accept hydrogen ions (protons) and are useful in elimination reactions.
What are Elimination Reactions in Halogenoalkanes?
A reaction where a molecule loses a hydrogen halide, leaving a double bond in its place so it can form an alkene. These reactions usually occur with a base.
Under the right conditions, -OH can act as a base, eliminating a hydrogen halide.
The conditions needed for a elimination reaction are a hydroxide (usually Sodium or Potassium) is dissolved in ethanol and mixed with the halogenoalkane. The mixture is then heated, producing ethene, which can be tested by collecting the gas and burning it and seeing if it decolourises bromine solution.
What is the mechanism of a Elimination Reaction?
If the base is a hydroxide ion, then a alkene, water molecule and halogen ion is formed (See Image)
If the base is a sodium/potassium hydroxide molecule, then a alkene, water molecule and potassium halide molecule is formed.

When is a reaction a Substitution or Elimination?
A hydroxide ion will react with halogenoalkanes as a nucleophile or as a base depending on the conditions. this depends on two factors: The type of Halogenoalkane (Primary, Secondary, Tertiary) and the Reaction Conditions (Aqueous or Ethanolic Solution)
Reaction Conditions:
Hydroxide Ions at room temperature, dissolved in water (aq), favour substitution.
Hydroxide ions at high temperature, dissolved in ethanol, favour elimination.
Type of Halogenoalkane:
Primary/Secondary Halogenoalkanes react by substitution.
Secondary/Tertiary Halogenoalkanes react by elimination.
What are Chlorofluorocarbons (CFCs)?
They are halogenoalkanes containing both chlorine and fluorine atoms but not hydrogen (CCl3F). They are very unreactive under normal conditions .
Short chain CFCs are gases and were used as refrigerants and aerosol propellants. Longer chain CFCs are used as dry cleaning solvents.
How do CFCs affect the Atmosphere and the Ozone Layer?
Ozone is a molecule O3 that decomposes to oxygen, and at ground level can cause smog and respiratory issues.
The Ozone Layer is important as it protects the Earth from exposure to too many UV rays by absorbing a significant amount of ultraviolet radiation from the sun.
CFCs form chlorine free radicals when the C-Cl bond breaks down homolytically in the presence of UV radiation which then attack ozone molecules: Cl• +O3 → ClO• + O2
The resulting free radicals also attack ozone and regenerate Cl•: ClO• + O3 → 2O2 + Cl•
By adding these two equations together (2O3 → 3O2) you can see that chlorine radicals act as a catalyst here and are not destroyed.
CFCs are now being phased out with new CFC free solvents to prevent them entering the atmosphere.
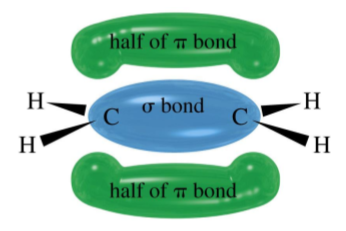
What is the Structure of an Alkene?
Alkenes are unsaturated hydrocarbons that contain at least one carbon-carbon double bond (C=C). They have the general formula CnH2n.
Alkenes are planar molecules with no rotation around the double bond due to the π-orbital restricting the rotation of the bonded atoms and creating a cloud of high electron density.
What types of Isomers can be present in Alkenes?
Position Isomers - Isomers with the double carbon bond in different positions.
Geometrical Isomers/E-Z - Isomers that differ in space around the carbon double bond due to restricted rotation.
What are the Physical Properties of Alkenes?
They have very similar properties to alkanes as the double carbon bond does not greatly affect properties like MP/BP. van der Waals forces are the only intermolecular force present. They are insoluble in water and MP/BP increase with carbon chain length.
How do Alkenes React?
Alkenes are more reactive than alkanes with the C=C bond being an electron-rich area which can be attached by electrophiles.
Electrophile - An electron-deficient atom, ion or molecule that attacks areas of high electron density. An electron pair acceptor.
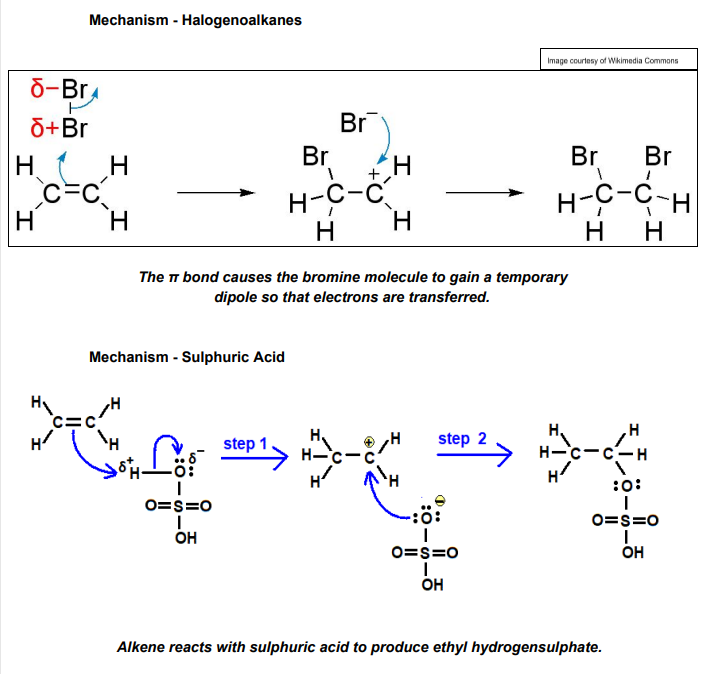
What are Electrophilic Addition Reactions?
Electrophilic Addition is when a electrophile attacks the alkene and forms a bond using the electrons in the π bond. This forms a carbocation where there is an intermediate/positive charge attached to one of the carbons.
What are some mechanisms for Electrophilic Addition Reactions?
Ethene + Hydrogen Bromide
Propene + Sulfuric Acid
Butene + Bromine
What is the Positive Inductive Effect?
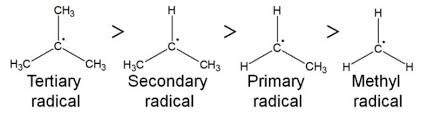
The Positive Inductive Effect is the electron-donating effect of alkyl groups that stabilizes positive charges in a carbocation.
A primary, secondary or tertiary carbocation forms where the intermediate attaches to a carbon atom with the most carbons bonded to it.
Primary Carbocation - The intermediate attaches to a carbon bonded to one other carbon
Secondary Carbocation - The intermediate attaches to a carbon bonded to two other carbons
Tertiary Carbocation - The intermediate attaches to a carbon bonded to three other carbons, the most stable carbocation.
What are Major and Minor Products?
Major Products - The products in greater yield due to stability.
Minor Products - The products in lesser yield due to lower stability.
What is the test for the Double Carbon Bond?
The addition reaction of adding Bromine solution to an alkene which results in the decolourisation of the solution, from brown to colourless, as the products are colourless.
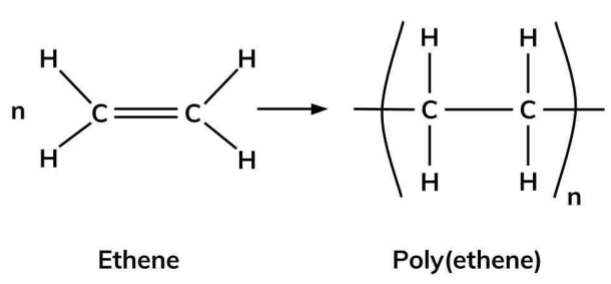
What is Addition Polymerisation?
A chemical reaction where monomers combine together by splitting the double carbon bond to form a long chain polymer (See Image).
What is PVC and Plasticisers?
PVC is a polymer, also known as poly(chloroethene) or polyvinyl chloride, that is used for things like drainpipes and electrical insulation.
Plasticisers are substances added to polymers such as PVC to increase flexibility and workability, which is how PVC gains its properties for its many uses.
How can the Reaction Conditions be altered to give different property Polymers?
Low Density Polymers - Polymers produced at high temperatures and high pressures causes them to have more branches and therefore lower intermolecular forces, so they are less dense and more flexible. Useful for making plastic bags and insulation.
High Density Polymers - Polymers produced at low temperatures and pressures causes them to have very little branching and therefore higher intermolecular forces, so they are more dense and rigid. Useful for containers and pipes.
Why are Polymers not Biodegradable?
Polymers are very unreactive molecules, due to the long chain saturated alkane structure, with non-polar C-C and C-H bonds. This makes them resistant to biological agents like enzymes, so they persist in the environment for along time.
What are some Solutions to Pollution by plastics?
Mechanical Recycling - The process of collecting and reprocessing plastic wasted into new products.
Feedstock Recycling - The process of heating and breaking down plastics into there original monomers and using them for new plastics.
What is the Structure of an Alcohol?
Alcohols are organic compounds with the functional group -OH and the general formula CnH2n+1. They are very reactive. They are named with the suffix ol (Ethanol, butan-1-ol, propan-2-ol), unless another functional group is present then the prefix hydroxy- is used.
The C-O-H bond angle is ~105o.
What are Primary, Secondary and Tertiary alcohols?
Primary Alcohols - The carbon atom with the -OH group attached has one R group attached to it.
Secondary Alcohols - The carbon atom with the -OH group attached has two R groups attached to it.
Tertiary Alcohols - The carbon atoms with the -OH group attached has three R groups attached to it.
What are the Physical Properties of Alochols?
The -OH group forms hydrogen bonds, giving alcohols higher boiling points and melting points than other hydrocarbons of similar molecular mass. The -OH group of alcohols can also bond to water molecules, but the main hydrocarbon chain cannot as it is non-polar. This means that short chain alcohols are soluble, as the hydrogen bonds predominate, but the longer chain alcohols are insoluble in water as the non-polar long chain dominates.
How are Alcohols produced by Hydration of Alkenes?
An alkene reacts with a water molecule, under the presence of an acid catalyst to form an alcohol. Phosphoric acid is commonly used as the catalyst with a high temperature and a high pressure.
This process has a very high percentage yield as the alcohol is the only product, so it is favoured by industry.
See Image for Mechanism
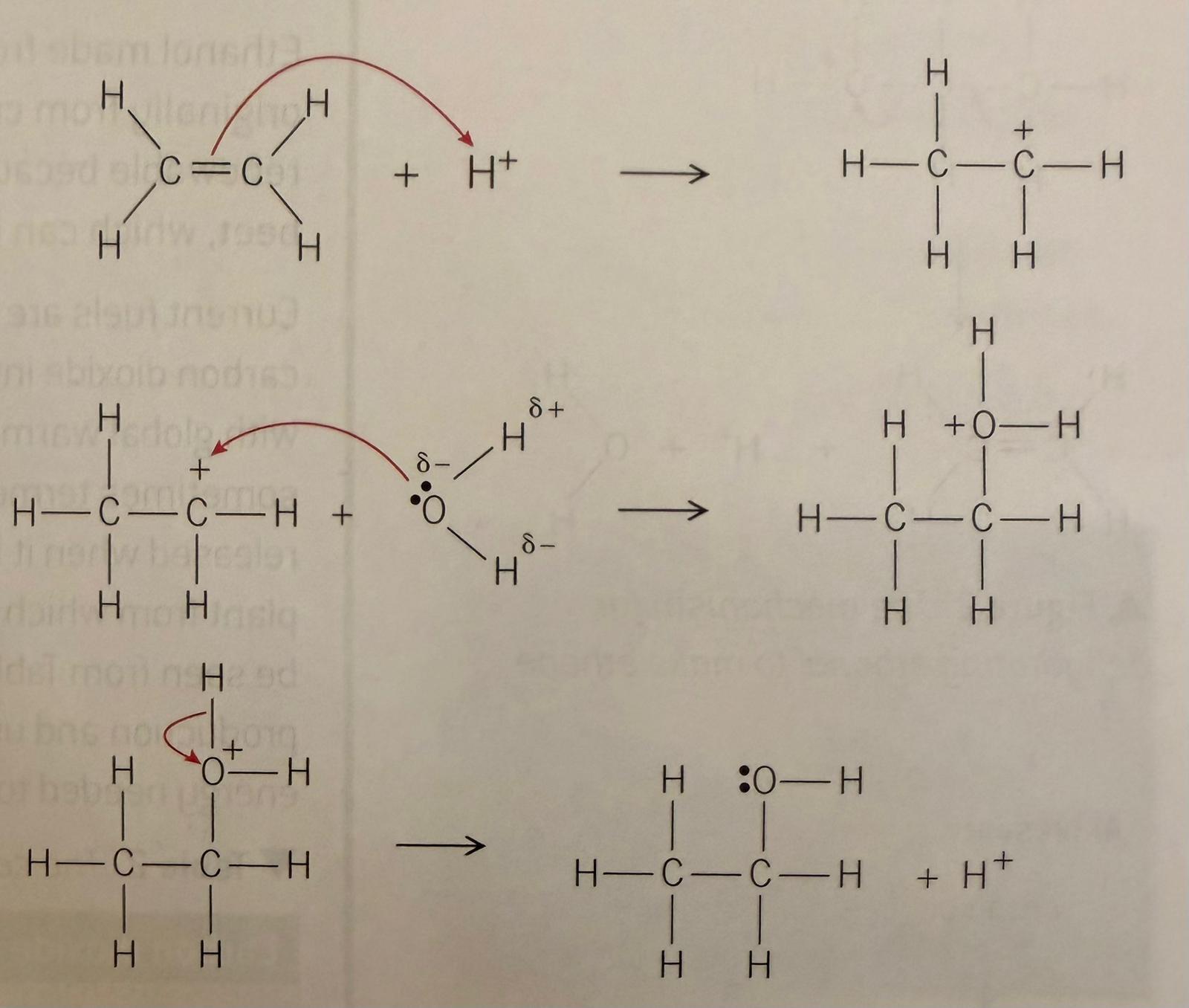
How is Ethanol produced by Fermentation?
Enzymes break down starch from crops into sugars, and then fermented by yeast to make ethanol and carbon dioxide. Its conditions are a compromise temperature of ~35oC (Enzyme ideal conditions), with no oxygen present (Prevents oxidation to make Ethanoic Acid) and the yeast acts as a catalyst.
The ethanol produced is dilute with a solution only containing about 15% ethanol, as any higher kills the yeast. It can then be distilled from the mixture by fractional distillation as it has a lower boiling point than water.
As it is produced in batches it is a lot slower than hydration which is continuous, and it has a lower percentage yield.
What is a Biofuel and how is Fermentation carbon neutral?
Biofuel - A duel that is derived from living matter. Ethanol is a biofuel in fermentation.
Carbon Neutral - Something is described as Carbon-Neutral if the production and use has no net increase in the amount of Carbon Dioxide in the atmosphere. The use of ethanol from fermentation is carbon-neutral (See Image).
Issues with Biofuel: Deforestation/Loss of habitat, Land and water use, Increased food prices as food used for fuel.
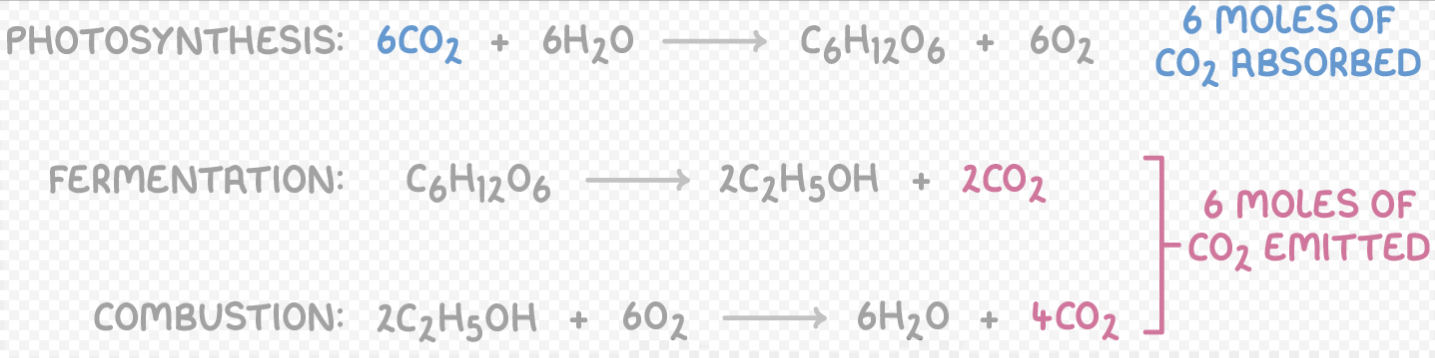
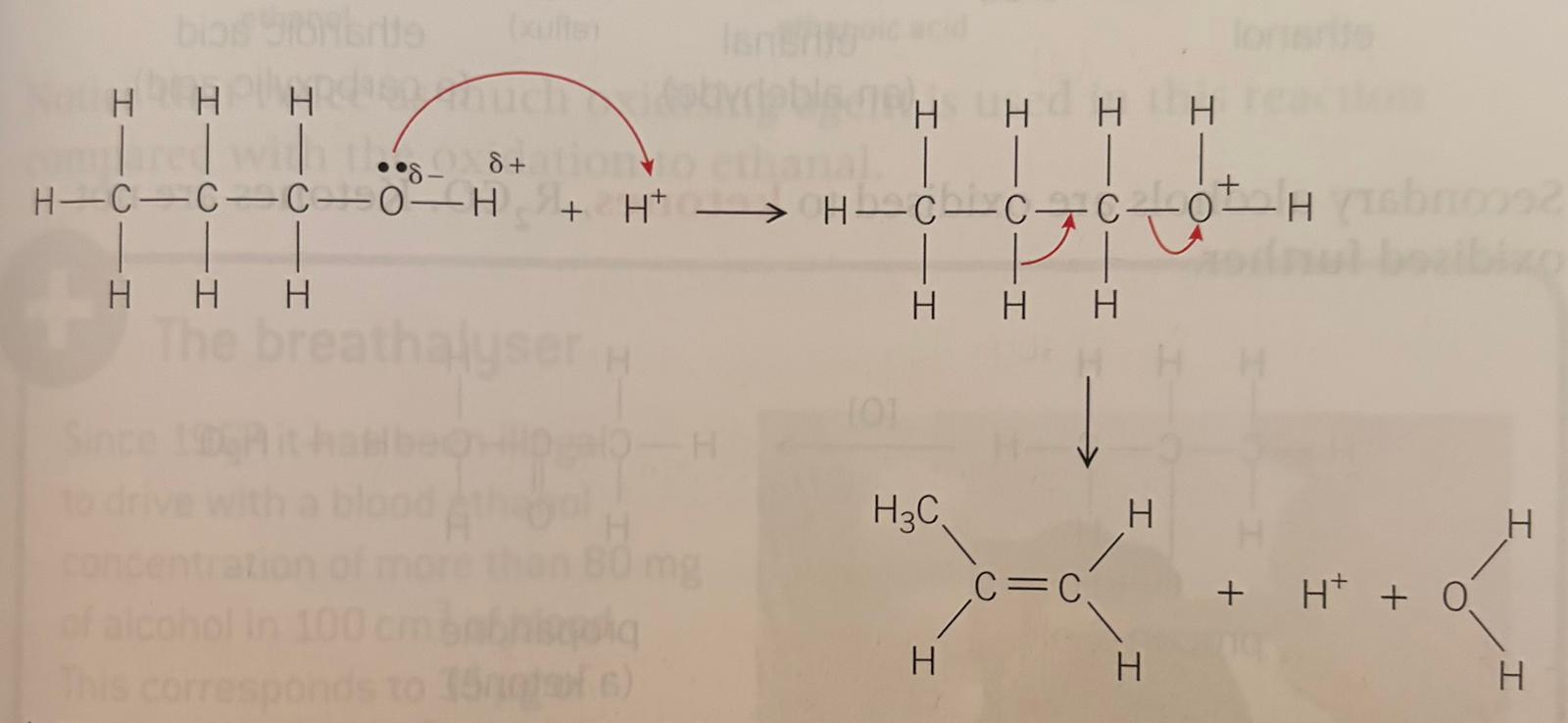
What are Elimination Reactions and the Dehydration Mechanism?
Reactions where a small molecule leaves a parent molecule, for alcohols a water molecule leaves.
Dehydration:
Alcohols can be dehydrated with excess hot concentrated sulfuric acid at a high temperature or by passing their vapours over heated aluminium oxide.
For mechanism See Image.
This reaction means that addition polymers can be produced without the need for Crude Oil.
Describe the Oxidation of Primary Alcohols?
Primary Alcohols (1o) can be heated gently in the presence of potassium dichromate solution, acidified with dilute sulfuric acid (K2Cr2O7 + H2SO4) to form an aldehyde. The aldehyde product is then distilled off.
[O] = Oxidising Agent = K2Cr2O7
The alcohol could be refluxed with twice the oxidising agent to form a carboxylic acid.
As the Alcohol/Aldehyde is oxidised the Orange Cr2O72- ions are reduced to green Cr3+ ions.
![<p>Primary Alcohols (1<sup>o</sup>) can be heated gently in the presence of potassium dichromate solution, acidified with dilute sulfuric acid (K<sub>2</sub>Cr<sub>2</sub>O<sub>7</sub> + H<sub>2</sub>SO<sub>4</sub>) to form an aldehyde. The aldehyde product is then distilled off.</p><p>[O] = Oxidising Agent = K<sub>2</sub>Cr<sub>2</sub>O<sub>7</sub></p><p>The alcohol could be refluxed with twice the oxidising agent to form a carboxylic acid.</p><p>As the Alcohol/Aldehyde is oxidised the Orange Cr<sub>2</sub>O<sub>7</sub><sup>2-</sup> ions are reduced to green Cr<sup>3+</sup> ions.</p>](https://assets.knowt.com/user-attachments/122759da-c241-4600-985d-49bbec5628cb.png)
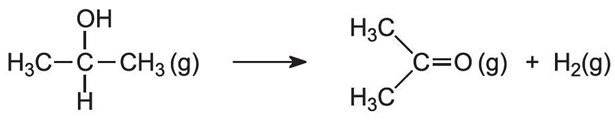
Describe the oxidation of Secondary Alcohols and Tertiary Alcohols?
Secondary Alcohols (2o) can be heated gently in the presence of acidified potassium dichromate to produce ketones only. It does not need to be distilled off as it cannot be further oxidised.
Tertiary Alcohols (3o) are not easily oxidised as you would have to break C-C bonds compared to before where your breaking only C-H bonds.
How can you test for Aldehydes and Ketones?
Tollens’ Test - Tollens’ Reagent is a gentle oxidising agent that is an aqueous alkaline solution of silver nitrate. When warmed in water bath with an aldehyde, the Ag+ Ions can be reduced to silver atoms, a silver mirror forms.
Fehling’s Test - Fehling’s Reagent is a gentle oxidising agent that contains blue copper (II) ions which oxidise aldehydes but not ketones. When warmed with an aldehyde, a brick red precipitate forms as Copper (I) Oxide forms.
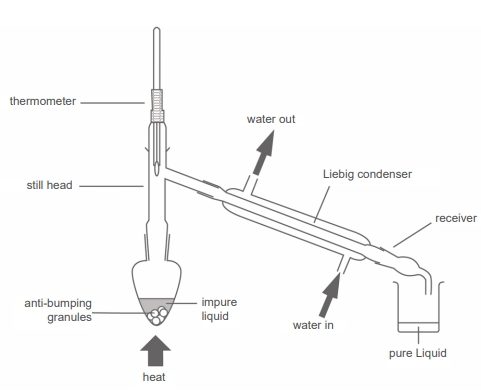
RP 5: Distillation of a product from a reaction
Preparation of cyclohexene by the dehydration and distillation of cyclohexanol:
Distillation Part:
Pour 25cm3 of cyclohexanol into a pear shaped flask of know mass and measure the new mass on a weighting scale, then calculating the mass of cyclohexanol
Use a dropping pipette to add 8cm3 of concentrated Phosphoric acid to the flask and some anti-bumping granules
Heat the solution gently with the apparatus shown, collecting any liquid formed at around ± 2oC of its BP
A cooled collection vessel is essential so that all the product is collected
Dehydration Part:
Pour the distillate collected into a separating funnel and add 50cm3 saturated sodium chloride solution
Shake the mixture so the two layers separate and run off the lower layer so you can transfer the upper layer which contains the crude cyclohexene into a conical flask
Add a few lumps of anhydrous calcium chloride to remove any water, stopper the flask and shake until the liquid is clear
Decant the liquid into a clean, dry sample container that has been weighed
Reweigh the container and calculate the mass of dry cyclohexene produced
You can test it using bromine water
How do you test for these Functional Groups?
Alkenes - Add bromine water to test for unsaturated hydrocarbons - Orange to colourless if alkene present
Carboxylic Acid - Reacts with Sodium Carbonate to produce CO2 - Bubble the gas made through limewater and if turns cloudy its a Carboxylic Acid
Aldehyde (Fehling’s) - Add to your solution and heat in water bath - Turns from blue to brick red precipitate if Aldehyde present.
Aldehyde (Tollens’) - Add to your solution and heat in water bath - A silver mirror forms if Aldehyde Present
Alcohols - Add acidified Potassium Dichromate to your solution - If primary/secondary alcohol it turns green - If tertiary alcohol remains orange
What is Mass Spectrometry?
An analytical technique used to identify compounds and determine their molecular formula.
High Resolution MR - A sensitive form of Mass Spectrometry that allows the Mr of a substance to be measured to 4 dp. Precise atomic masses are given and can be used to calculate the molecular formula of a compound being tested.
How do Isotopes affect Mass Spectrometry and their calculations?
Isotopes cause multiple molecular ion peaks in a mass spectrum. The ratio between abundances will be the same as the ratio between peaks.
Example 1: CH3Br - Bromine atoms are 50% 79Br and 50% 81Br
Therefore CH3Br where Br = 79 has a mass of 94 with an abundance of 50% - CH3Br where Br = 81 has a mass of 96 with an abundance of 50%
Example 2: CCl4 - Chlorine atoms are 75% 35Cl and 25% 37Cl
CCl4 Where all Chlorine are 35 has a mass of 152 and abundance of (0.75 × 0.75 × 0.75 × 0.75 = 31.6% x1 = 31.6%)
CCl4 Where Chlorine atoms have masses 35,35,35,37 and abundance of [ (0.75 × 0.75 × 0.75 × 0.25 = 10.55) x 4 = 42.2% - It is multiplied by 4 as the 37 mass chlorine could be the 1st, 2nd, 3rd, or 4th atom.
And so on… (Uses Binomial Probabilities/ Pascal so it goes 1,4,6,4,1)
What is Infrared Spectroscopy and how does it work?
It is an analytical method that uses Infrared Radiation (IR) to determine the different functional groups in a compound.
A pair of atoms joined by a chemical bond is always vibrating, with stronger bonds vibrating faster (higher frequency) and heavier atoms vibrating slower (lower frequency). Each bond has a unique frequency that is in the IR region of the electromagnetic spectrum. Any particular bond can only absorb radiation of the same frequency as the natural frequency of that bond.
A beam of Infrared Radiation containing a spread of frequencies is passed through a sample. The radiation that merges will be missing the frequencies that particular bonds absorbed as they vibrate at that frequency. The instrument plots a graph of the intensity of the radiation emerging from the sample against frequency, which is expressed as a wavenumber / cm-1
How can you read Infrared Spectrums?
Where there are peaks (Look like dips) in the graph, that frequency has been absorbed. you can then compare it to the wavenumber values of certain bonds to identify a compound.
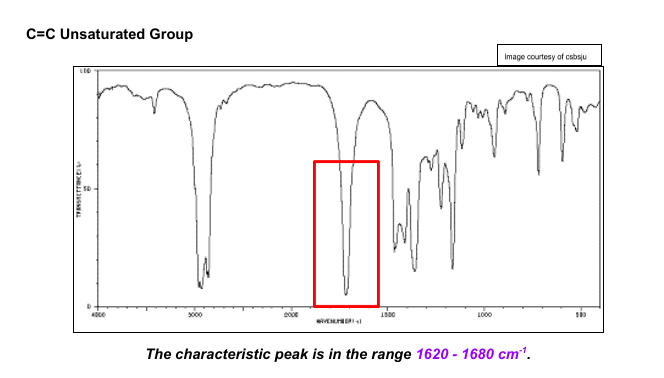
What is the Fingerprint Region?
This is a region in a spectrum below 1550 cm-1 which causes many peaks due to complex vibrations from the whole molecule. The shape of the region is unique for any particular substance so it can be used to identify any sample by comparing it with those on a database of compounds.
How does Infrared Absorption occur in the Atmosphere?
Infrared absorption also occurs in the atmosphere where gases absorb the radiation given off from the surface of the earth that match the frequency at which they vibrate, trapping heat in the atmosphere and increasing the global temperature.
RP 6: Organic Tests
Testing for an Alcohol:
Add a small piece of metallic sodium to an alcohol
It should produce hydrogen which can be confirmed by collecting the gas and adding a lit splint, which should produce a loud pop
Dispose of any excess sodium in a beaker of ethanol as it becomes a fire risk in contact with water
Distinguishing between Primary/Secondary/Tertiary alcohols:
Use a measuring cylinder to add acidified potassium dichromate to your solution
If it is Primary/Secondary it turns green, but if its tertiary it stays orange
Testing for an Aldehyde:
Mix together equal volumes of Fehling’s Solution A and B in a test tube - Should produce a clear dark blue solution
Add 5 drops of this solution to your aldehyde and a few anti bumping granules
Warm the test tube gently in a water bath for a few minutes then leave to stand
A brick red precipitate should then form
You can also use Tollens to test
Testing for an Alkene:
Use a pipette to add 1cm3 of bromine water to 2 drops of the alkene
Shake the test tube vigorously from side to side and it should turn orange.
Testing for a Carboxylic Acid:
Add a spatula of solid sodium carbonate to a test tube of 2cm3 of carboxylic acid
Collect the gas produced and bubble it through limewater, which should turn cloudy
Testing for Halogenoalkanes:
Add 5 drops of your halogenoalkane to 1cm3 of sodium hydroxide and warm your solution at 60oC for a few minutes
Acidify the contents with 2cm3 of dilute nitric acid and add 1cm3 silver nitrate
A silver halide precipitate forms (Cl = White, Br = Cream, I = Yellow)
What are the Historical Ideas of the Atom?
1661 - Robert Boyle proposed that there were some substances that could not be made simpler (Elements)
1803 - John Dalton suggested that elements were composed of indivisible atoms and that atoms of the same element had the same masses and they could not be broken down.
1896 - Henri Becquerel discovered Radioactivity which showed particles could come from inside the atom.
1897 - J J Thomson discovered the electron and proposed the “plum pudding” model.
1911 - Ernest Rutherford and his team found that most of the mass and positive charge was in a tiny central nucleus
1913 - Niels Bohr introduced the idea that the atom consisted of a tiny positive nucleus orbited by electrons in shells which was later used for quantum theory.
1926 - Erwin Schrödinger worked out an equation that used the idea that electrons had some of the properties of waves as well as those particles. This was called quantum mechanics which can be used to predict the behaviour of sub-atomic particles.
1932 - James Chadwick discovered the neutron.
What are the relative masses and charges of the Sub-Atomic Particles?
Electron - M:1/1840 - C:-1
Proton - M:1 - C:+1
Neutron - M:1 - C:0
Protons and Neutrons are often referred as nucleons and are held together in the central atom by the strong nuclear force and is stronger than intermolecular forces so it overcomes the repulsion between protons.
What is the Atomic Number, Mass Number and Isotopes?
Atomic Number (Z) = Number of Protons
Mass Number (A) = Number of Protons + Neutrons
Isotopes - Atoms with the same number of protons and a different number of neutrons. They react chemically in the same way.
What is Relative Atomic Mass/Relative Molecular Mass?
The mean mass of an atom of an element divided by 1/12 the mass of one atom of carbon-12
Ar = Average mass of 1 atom / 1/12 mass of 1 atom of 12C
Mr = Average mass of molecule / 1/12 mass of 1 atom of 12C
What is Carbon Dating?
A method used to determine the age of carbon-based materials using the radioactive isotope carbon-14 which has a half life of 5730 years. This is used to date materials up to 60,000 years old (Which would only contain 0.07% carbon-14 now) but is best for around 2000 years.
What is Mass Spectrometry?
A powerful instrumental method of analysis that is used to: find the abundance and mass of each isotope in an element allowing us to determine its relative atomic mass, find the relative molecular mass of substances made of molecules.
What is Time of Flight (ToF) Mass Spectrometry?
A common form of mass spectrometry where particles of the substance are ionised and accelerated, where their time taken to travel a fixed distance is used to find the mass.
What is the Stage 1 of ToF?
Stage 1 - Ionisation
The sample is ionised using methods like electron impact/ionisation and electrospray ionisation.
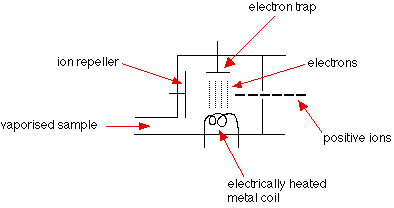
What is Electron Impact/Ionisation?
The sample being analysed is vaporised and then high energy electrons are fired at it. The high energy electrons come from an electron gun (A hot wire filament with a current running through it). This usually knocks off one electron from each particle forming a X+ (Molecular ion)
It is used for elements with low formula mass (Organic or Inorganic molecules). The molecular ion often breaks down into smaller fragments which are also detected in the mass spectrum.
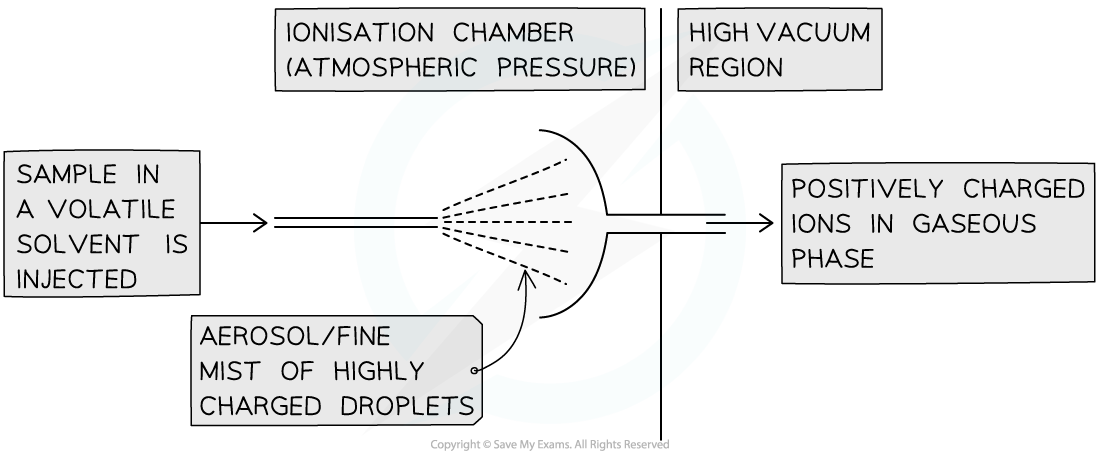
What is Electrospray Ionisation?
The sample is dissolved in a volatile solvent (eg water/methanol) and injected through a fine hypodermic needle to give a fine mist (aerosol). The tip of the needle is attached to the positive terminal of a high-voltage power supply. The particles are ionised by gaining a proton from the solvent as they leave the needle forming XH+ ions). The solvent evaporates away while the XH+ ions are attracted towards a negative plate where they are accelerated.
These ions are known as a protonated molecule as a H+ ion contains a proton and no elctrons.
This technique is used for substances with higher molecular mass including biological molecules such as proteins. This is known as soft ionisation as fragmentation rarely occurs.
What is Stage 2 of ToF?
Stage 2 - Acceleration
The positive ions are accelerated using an electric field so that they all have the same kinetic energy.
KE = ½ M V²
Therefore, the velocity is: V = Root (2KE/m)
Given that all the particles have the same kinetic energy, the velocity of each particle depends on its mass. Lighter particles have a faster velocity and heavier particles have a slower velocity.
What is Stage 3 of ToF?
Stage 3 - Flight Tube
The positive ions travel through a hole in the negatively charged plate into a tube. The time of flight of each particle through this flight tube depends on its velocity which in turn depends on its mass.
The time of flight along the flight tube is given by the equations:
t = d/v where d = Length of tube
Therefore t = d root (m/2KE)
This shows that the time of flight is proportional to the square root of the mass of the ions which explains why lighter ions travel faster and reach the detector in less time.